Abstract
The growth of titanium dioxide nanotubes (TiO2) via anodization process depends on the controlling parameters such as applied potential, anodization time, and electrolyte composition. In the present work, the Taguchi method was applied to evaluate statistically the influence of the anodization parameters on the morphology of anodized TiO2 films. Mixture of ethylene-glycol and glycerol was used as an electrolyte and the settings of the experimental design were parameterized on the basis of four important anodization factors consisting of chemical pretreatment, amount of fluoride, water content and applied potential. Samples were characterized by XRD and FEG-SEM. Based on 4 variables at 3 different settings, full factorial plan requires 34 = 81 tests. In this work the experiment was designed on the basis of an L9 (34) orthogonal array (4 variables, 3 levels, 9 tests). The optimum conditions were found on the basis of smaller-is-better and larger-is-better analyses. The signal-to-noise ratio was employed to find optimal process parameters levels and to analyze the influence of these parameters on the tubular length, internal and external diameters and formation of nanograss on the film surface. Hence, it is clearly shown that the performance of TiO2 nanotubes can be evaluated by the Taguchi method.
Keywords:
Titanium dioxide nanotubes; Taguchi method; Anodization; Parameters
1. Introduction
Titanium dioxide TiO2 based nanostructures are widely used in solar cells11 Roy P, Albu SP, Schmuki P. TiO2 nanotubes in dye-sensitized solar cells: Higher efficiencies by well-defined tube tops. Electrochemistry Communications. 2010;12(7):949-951. DOI: 10.1016/j.elecom.2010.04.029
https://doi.org/10.1016/j.elecom.2010.04...
,22 Roy P, Kim D, Lee, K, Spiecker E, Schmuki P. TiO2 nanotubes and their application in dye-sensitized solar cells. Nanoscale. 2010;2(1):45-59. DOI: 10.1039/B9NR00131J
https://doi.org/10.1039/B9NR00131J...
, hydrogen production33 Li Y, Yu H, Zhang C, Song W, Li G, Shao Z, et al. Effect of water and annealing temperature of anodized TiO2 nanotubes on hydrogen production in photoelectrochemical cell. Electrochimica Acta. 2013;107:313-319. DOI: 10.1016/j.electacta.2013.05.090
https://doi.org/10.1016/j.electacta.2013...
, sensors44 Varghese OK, Gong D, Paulose M, Ong KG, Grimes CA. Hydrogen sensing using titania nanotubes. Sensors Actuators B: Chemical. 2003;93(1-3):338-344. DOI: 10.1016/S0925-4005(03)00222-3
https://doi.org/10.1016/S0925-4005(03)00...
, photocatalysis55 Paramasivam I, Jha H, Liu N, Schmuki P. A Review of Photocatalysis using Self-organized TiO2 Nanotubes and Other Ordered Oxide Nanostructures. Small. 2012;8(20):3073-3103. DOI: 10.1002/smll.201200564
https://doi.org/10.1002/smll.201200564...
6 Zheng L, Xiao X, Li Y, Zhang W. Enhanced photocatalytic activity of TiO2 nanoparticles using WS2/g-C3N4 hybrid as co-catalyst. Transactions of Nonferrous Metals Society of China. 2017;27:1117-1126. DOI: 10.1016/S1003-6326(17)60130-4
https://doi.org/10.1016/S1003-6326(17)60...
-77 Khan S, Teixeira SR, Santos MJL. Controlled thermal nitridation resulting in improved structural and photoelectrochemical properties from Ta3N5 nanotubular photoanodes. RSC Advances. 2015;5(125):103284-103291. DOI: 10.1039/C5RA17227F
https://doi.org/10.1039/C5RA17227F...
, effluent treatment88 Oller I, Malato S, Sánchez-Pérez JA. Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination-A review. Science of The Total Environment. 2011;409(20):4141-4166. DOI: 10.1016/j.scitotenv.2010.08.061
https://doi.org/10.1016/j.scitotenv.2010...
,99 Zhou Q, Huang Y, Xie G. Investigation of the applicability of highly ordered TiO2 nanotube array for enrichment and determination of polychlorinated biphenyls at trace level in environmental water samples. Journal of Chromatography A. 2012;1237:24-29. DOI: 10.1016/j.chroma.2012.02.066
https://doi.org/10.1016/j.chroma.2012.02...
and biomedical applications1010 Tsuchiya H, Macak JM, Müller L, Kunze J, Müller F, Greil P, et al. Hydroxyapatite growth on anodic TiO2 nanotubes. Journal of Biomedical Materials Research. Part A. 2006;77A(3):534-541. DOI: 10.1002/jbm.a.30677
https://doi.org/10.1002/jbm.a.30677...
. TiO2 nanoarchitectured morphologies can be synthesized through various techniques1111 Byranvand MM, Kharat AN, Fatholahi L, Beiranvand ZM. A Review on Synthesis of Nano-TiO2 via Different Methods. Journal of Nanostructures. 2013;3:1-9.,1212 Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews. 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
https://doi.org/10.1021/cr500061m...
. Anodization is one widely used technique that is simple and provides a precise control over the syntheses of nanomorphologies55 Paramasivam I, Jha H, Liu N, Schmuki P. A Review of Photocatalysis using Self-organized TiO2 Nanotubes and Other Ordered Oxide Nanostructures. Small. 2012;8(20):3073-3103. DOI: 10.1002/smll.201200564
https://doi.org/10.1002/smll.201200564...
,1313 Coser E, Bervian A, Khan S, De Léon A, Bussi J, Pianaro SA, et al. Photodegradation of glycerol using nanostructured TiO2 catalyst. Renewable Energies and Power Quality Journal. 2017;15(1):468-472. DOI: 10.24084/repqj15.35
https://doi.org/10.24084/repqj15.35...
,1414 Nischk M, Mazierski P, Gazda M, Zaleska A. Ordered TiO2 nanotubes: The effect of preparation parameters on the photocatalytic activity in air purification process. Applied Catalysis B: Environmental. 2014;144:674-685. DOI: 10.1016/j.apcatb.2013.07.041
https://doi.org/10.1016/j.apcatb.2013.07...
. Based on its large surface area, TiO2 nanotubular morphology synthesized via the anodization process is promising for its potential applications such as in photocatalytic and photovoltaic devices1212 Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews. 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
https://doi.org/10.1021/cr500061m...
,1313 Coser E, Bervian A, Khan S, De Léon A, Bussi J, Pianaro SA, et al. Photodegradation of glycerol using nanostructured TiO2 catalyst. Renewable Energies and Power Quality Journal. 2017;15(1):468-472. DOI: 10.24084/repqj15.35
https://doi.org/10.24084/repqj15.35...
. Anodization in fluorinated electrolytes is one effective method employed to obtain highly ordered nanotube arrays of TiO2 and many other materials with controllable morphologies11 Roy P, Albu SP, Schmuki P. TiO2 nanotubes in dye-sensitized solar cells: Higher efficiencies by well-defined tube tops. Electrochemistry Communications. 2010;12(7):949-951. DOI: 10.1016/j.elecom.2010.04.029
https://doi.org/10.1016/j.elecom.2010.04...
. The uses of organic electrolytes with different concentrations of water and fluoride salts have been widely reported1515 Xue C, Yonezawa T, Nguyen MT, Lu X. Cladding Layer on Well-Defined Double-Wall TiO2 Nanotubes. Langmuir. 2015;31(4):1575-1580. DOI: 10.1021/la504670p
https://doi.org/10.1021/la504670p...
. These electrolytes are less aggressive and offer better efficiency to obtain highly ordered nanotubular arrays1212 Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews. 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
https://doi.org/10.1021/cr500061m...
,1616 Raja KS, Misra M, Paramguru K. Formation of self-ordered nano-tubular structure of anodic oxide layer on titanium. Electrochimica Acta. 2005;51(1):154-165. DOI: 10.1016/j.electacta.2005.04.011
https://doi.org/10.1016/j.electacta.2005...
,1717 Khan S, Zapata MJM, Baptista DL, Gonçalves RV, Fernandes JA, Dupont J, et al. Effect of Oxygen Content on the Photoelectrochemical Activity of Crystallographically Preferred Oriented Porous Ta3N5 Nanotubes. Journal of Physical Chemistry C. 2015;119(34):19906-19914. DOI: 10.1021/acs.jpcc.5b05475
https://doi.org/10.1021/acs.jpcc.5b05475...
.
In the anodization process there are many factors that have significant influences on the morphology of TiO2 nanotubes1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
, e.g. pretreatment of the Ti substrate1919 Albertin KF, Tavares A, Pereyra I. Optimized Ti polishing techniques for enhanced order in TiO2 NT arrays. Applied Surface Science. 2013;284:772-779. DOI: 10.1016/j.apsusc.2013.08.005
https://doi.org/10.1016/j.apsusc.2013.08...
, NH4F concentration2020 Macak JM, Hildebrand H, Marten-Jahns U, Schmuki P. Mechanistic aspects and growth of large diameter self-organized TiO2 nanotubes. Journal of Electroanalytical Chemistry. 2008;621(2):254-266. DOI: 10.1016/j.jelechem.2008.01.005
https://doi.org/10.1016/j.jelechem.2008....
, percentage of H2O contained in the electrolyte2121 Macak JM, Schmuki P. Anodic growth of self-organized anodic TiO2 nanotubes in viscous electrolytes. Electrochimica Acta. 2006;52(3):1258-1264. DOI: 10.1016/j.electacta.2006.07.021
https://doi.org/10.1016/j.electacta.2006...
, anodization potential1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
,2222 Wong YH, Affendy MG, Lau SK, Teh PC, Lee HJ, Tan CY, et al. Effects of anodisation parameters on thin film properties: a review. Materials Science and Technology. 2017;33(6):699-711. DOI: 10.1080/02670836.2016.1193654
https://doi.org/10.1080/02670836.2016.11...
, electrolyte viscosity2323 Ghicov A, Schmuki P. Self-ordering electrochemistry: a review on growth and functionality of TiO2 nanotubes and other self-aligned MOx structures. Chemical Communications. 2009;20:2791-2808. DOI: 10.1039/b822726h
https://doi.org/10.1039/b822726h...
, anodization temperature2424 Xie ZB, Blackwood DJ. Effects of anodization parameters on the formation of titania nanotubes in ethylene glycol. Electrochimica Acta. 2010;56(2):905-912. DOI: 10.1016/j.electacta.2010.10.004
https://doi.org/10.1016/j.electacta.2010...
, mixture of electrolytes1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
,2121 Macak JM, Schmuki P. Anodic growth of self-organized anodic TiO2 nanotubes in viscous electrolytes. Electrochimica Acta. 2006;52(3):1258-1264. DOI: 10.1016/j.electacta.2006.07.021
https://doi.org/10.1016/j.electacta.2006...
and anodization time1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
,2525 Taveira LV, Macák JM, Tsuchiya H, Dick LFP, Schmuki P. Initiation and Growth of Self-Organized TiO2 Nanotubes Anodically Formed in NH4F/(NH4)2SO4 Electrolytes. Journal of the Electrochemical Society. 2005;152(10):B405-B410. DOI: 10.1149/1.2008980
https://doi.org/10.1149/1.2008980...
. A few examples below can elaborate their effects; electrolytes consisting of ethylene glycol2626 Albu SP, Ghicov A, Schmuki P. High aspect ratio, self-ordered iron oxide nanopores formed by anodization of Fe in ethylene glycol/NH4F electrolytes. Physica Status Solidi - Rapid Research Letters. 2009;3(2-3):64-66. DOI: 10.1002/pssr.200802285
https://doi.org/10.1002/pssr.200802285...
enable thicker oxide layers as compared to the electrolyte composed of only glycerol2121 Macak JM, Schmuki P. Anodic growth of self-organized anodic TiO2 nanotubes in viscous electrolytes. Electrochimica Acta. 2006;52(3):1258-1264. DOI: 10.1016/j.electacta.2006.07.021
https://doi.org/10.1016/j.electacta.2006...
,2727 Lee BG, Choi JW, Lee SE, Jeong YS, Oh HJ, Chi CS. Formation behavior of anodic TiO2 nanotubes in fluoride containing electrolytes. Transactions of Nonferrous Metals Society of China. 2009;19(4):842-845. DOI: 10.1016/S1003-6326(08)60361-1
https://doi.org/10.1016/S1003-6326(08)60...
.
Recently, we have synthesized TiO2 nanotubes in the mixture of glycerol and ethylene glycol and found that the double wall features of the nanotubes can be controlled by the anodization time1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
. Bervian et al.2828 Alagumurthi N, Palaniradja K, Soundararajan V. Optimization of Grinding Process Through Design of Experiment (DOE)-A Comparative Study. Materials and Manufacturing Processes. 2006;21(1):19-21. DOI: 10.1081/AMP-200060605
https://doi.org/10.1081/AMP-200060605...
have shown that the length of the TiO2 nanotubes increases with increasing anodization time; however, the diameter of the nanotubes was not changed. In addition, there is a linear relationship between the diameter of the nanotubes and the anodization potential.
Xie et al.2424 Xie ZB, Blackwood DJ. Effects of anodization parameters on the formation of titania nanotubes in ethylene glycol. Electrochimica Acta. 2010;56(2):905-912. DOI: 10.1016/j.electacta.2010.10.004
https://doi.org/10.1016/j.electacta.2010...
observed that at low NH4F concentrations a continuous layer, containing some pits, forms on the top of nanotube arrays of TiO2. However, for the higher NH4F concentration the continuous layer on the nanotubes was not observed. Xue et al.1515 Xue C, Yonezawa T, Nguyen MT, Lu X. Cladding Layer on Well-Defined Double-Wall TiO2 Nanotubes. Langmuir. 2015;31(4):1575-1580. DOI: 10.1021/la504670p
https://doi.org/10.1021/la504670p...
reported that the average internal nanotubes diameter increased with the increase of NH4F concentration. To optimize the growth of nanotubes it is necessary to evaluate the influence of these factors. Changing all these multiple parameters simultaneously is technically expensive, very laborious and time-consuming. Therefore, it is promising to use a statistical model that can help to find the best operating parameters to obtain highly ordered TiO2 nanotubular arrays with the desired geometrical features including length, diameter and wall thickness.
Statistical design of experiments refers to the process of planning the experiment so that appropriate data can be collected and analyzed by statistical methods, resulting in valid and objective conclusions. Design of Experiment (DOE) focuses on choosing the levels of controllable factors (or parameters) to ensure that the mean of the output response is at a desired level or target and to ensure that the variability around this target value is as small as possible. The number of experiments in a DOE depends on the number of levels and controlling factors2828 Alagumurthi N, Palaniradja K, Soundararajan V. Optimization of Grinding Process Through Design of Experiment (DOE)-A Comparative Study. Materials and Manufacturing Processes. 2006;21(1):19-21. DOI: 10.1081/AMP-200060605
https://doi.org/10.1081/AMP-200060605...
,2929 Pignatiello JJ Jr. An Overview of the Strategy and Tactics of Taguchi. IIE Transactions. 1988;20(3):247-254. DOI: 10.1080/07408178808966177
https://doi.org/10.1080/0740817880896617...
.
The Taguchi experimental model is a robust statistical method that minimizes the number of variations in the experiments and has been widely used in industry to improve the quality of products3030 Yiamsawas D, Boonpavanitchakul K, Kangwansupamonkon W. Optimization of experimental parameters based on the Taguchi robust design for the formation of zinc oxide nanocrystals by solvothermal method. Materials Research Bulletin. 2011;46(5):639-642. DOI: 10.1016/j.materresbull.2011.02.004
https://doi.org/10.1016/j.materresbull.2...
,3131 Mohamed AMA, Jafari R, Farzaneh M. An optimization of superhydrophobic polyvinylidene fluoride/zinc oxide materials using Taguchi method. Applied Surface Science. 2014;288:229-237. DOI: 10.1016/j.apsusc.2013.10.013
https://doi.org/10.1016/j.apsusc.2013.10...
. The Taguchi approach to DOE considers an orthogonal array of factors to reduce the number of experiments involved in the process of optimizing the activities. It is also a simple and efficient technique in research to investigate the effects of multiple factors, employing the best parameters3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
,3333 Yousefieh M, Tamizifar M, Boutorabi SMA, Borhani E. Taguchi Optimization on the Initial Thickness and Pre-aging of Nano-/Ultrafine-Grained Al-0.2 wt.%Sc Alloy Produced by ARB. Journal of Materials Engineering and Performance. 2016;25(10):4239-4248. DOI: 10.1007/s11665-016-2273-3
https://doi.org/10.1007/s11665-016-2273-...
. The Taguchi method is a combination of mathematical and statistical techniques used in an empirical study3434 Ghani JA, Choudhury IA, Hassan HH. Application of Taguchi method in the optimization of end milling parameters. Journal of Materials Processing Technology. 2004;145(1):84-92. DOI: 10.1016/S0924-0136(03)008653
https://doi.org/10.1016/S0924-0136(03)00...
.
Instead of testing all possible combinations as a planning factorial experiment, this method allows the collection of only the required data with the minimum possible experimental activities, thereby saving time and research resources. Furthermore, the Taguchi approach identifies some types of factors that cause variability in the important system response variables. These noise factors are often functions of environmental conditions such as temperature or relative humidity. Zhu et al.3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
applied Taguchi design to optimize the degradation phase parameters of TiO2 composite photocatalysis. Ghani et al.3434 Ghani JA, Choudhury IA, Hassan HH. Application of Taguchi method in the optimization of end milling parameters. Journal of Materials Processing Technology. 2004;145(1):84-92. DOI: 10.1016/S0924-0136(03)008653
https://doi.org/10.1016/S0924-0136(03)00...
have shown that the Taguchi method is suitable to solve the stated problem with the minimum number of trials as compared with a full factorial design.
Taguchi statistical experiments were carried out to determine the influence of the morphology variables of the TiO2 nanotubes. Since in the anodization process there are multiple process parameters that influence the morphology of the anodized film. It is therefore promising to study the influence of these parameters on the morphological evolution of TiO2 nanotubes applying the Taguchi method. This study aims apply the L9 orthogonal array because reduces considerably the experiments3535 Naghibi S, Sani MAF, Hosseini HRM. Application of the statistical Taguchi method to optimize TiO2 nanoparticles synthesis by the hydrothermal assisted sol-gel technique. Ceramics International. 2014;40(3):4193-4201. DOI: 10.1016/j.ceramint.2013.08.077
https://doi.org/10.1016/j.ceramint.2013....
,3636 Kulkarni S, Duttagupta S, Phatak G. Taguchi design of experiments for optimization of ionic conductivity in nanocrystalline Gadolinium doped Ceria. Ceramics International. 2015;41(7):8973-8980. DOI: 10.1016/j.ceramint.2015.03.171
https://doi.org/10.1016/j.ceramint.2015....
.
As reported in the literature1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
,2121 Macak JM, Schmuki P. Anodic growth of self-organized anodic TiO2 nanotubes in viscous electrolytes. Electrochimica Acta. 2006;52(3):1258-1264. DOI: 10.1016/j.electacta.2006.07.021
https://doi.org/10.1016/j.electacta.2006...
,3737 Liu Z, Zhang X, Nishimoto S, Jin M, Tryk D, Murakami T, et al. Highly Ordered TiO2 Nanotube Arrays with Controllable Length for Photoelectrocatalytic Degradation of Phenol. Journal of Physical Chemistry C. 2008;112(1):253-259. DOI: 10.1021/jp0772732
https://doi.org/10.1021/jp0772732...
,3838 Wang C, Chen D, Ping G, Liu S, Huang XN, Huang YX, et al. Controllable synthesis of well-ordered TiO2 nanotubes in a mixed organic electrolyte for high-efficiency photocatalysis. Science China Chemistry. 2012;55(11):2373-2380. DOI: 10.1007/s11426-012-4680-0
https://doi.org/10.1007/s11426-012-4680-...
several parameters influence the formation of TiO2 nanotubes. Thus, it was studied parameters that have a great influence on the development of morphology of the TiO2 nanotubes which requires greater attention, as: chemical pre-treatment, ammonium fluoride concentrations, water content and applied potential.
The objective of this study via anodization process in a mixture of ethylene glycol and glycerol is to evaluate their influence on the tubular length, internal and external diameters and formation of nanograss on the film surface following the Taguchi method and aiming to obtain highly ordered TiO2 nanotubes.
2. Experimental
2.1 Materials and sample preparation
Nanotubes were grown on Ti foil grade 2 - ASTM-F67 (99.8 wt.% purity, 0.7 mm thickness, Realum), with dimensions of 1 cm × 5 cm. These foils were cleaned by ultrasonication for 15 minutes in degreasing agent and deionized water (DI) in sequence and dried under cold air. Anodization was carried out in an electrochemical cell with a two-electrode configuration and with a Minipa MPC 303 DI power supply controlled by TCXX. In the anodizing system, the Ti foil was used as a working electrode and a platinum foil was used as a counter electrode. The distance between the electrodes was fixed at 2 cm3737 Liu Z, Zhang X, Nishimoto S, Jin M, Tryk D, Murakami T, et al. Highly Ordered TiO2 Nanotube Arrays with Controllable Length for Photoelectrocatalytic Degradation of Phenol. Journal of Physical Chemistry C. 2008;112(1):253-259. DOI: 10.1021/jp0772732
https://doi.org/10.1021/jp0772732...
. The anodization was performed without stirring and applying 20, 40 and 60 V at a potential ramp of 1 Vs-1. The organic electrolyte was a mixture of glycerol (Synth 99.5%) and ethylene glycol (Synth - purity: 99.0%) in 3:1 (v/v) ratio1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
. The mixture composed of ethylene glycol and glycerol was adopted due to the influence of the geometry of the formed nanotubes. The concentration of ammonium fluoride (NH4F) was 0.25, 0.50 and 0.75 wt.% (Synth - purity: 98.0%) and the concentration of the deionized water was 0, 2 and 10 wt.%.
The pH was measured prior to NH4F addition at the organic electrolyte with a pH meter organic (Mettler Toledo HA405-60-88G-S7/120). The pH obtained was 7.8. Also was measured with the pH test strips (Acilit® pH 0-6; Merck) obtaining in the range pH 5-5.5. After addition of NH4F at the organic electrolyte the pH was measured only with pH test strips, and found to in the range pH 4.5-5. The time of anodization employed was 180 minutes at room temperature (23 ºC ± 2 ºC) and the viscosity of the mixture was 104.6 Pa s. The current transient (I-t) curves were obtained by using a system acquisition of data (Multimeter Minipa ET-2076A) recorded from the source meter connected with software TCXX. After anodization, the samples were annealed in a muffle furnace at 450 °C for 3 h with a heating ramp of 10 ºC min-1 to crystallize the TiO2 nanotubes3939 Kandiel TA, Dillert R, Feldhoff A, Bahnemann DW. Direct synthesis of photocatalytically active rutile TiO2 nanorods partly decorated with anatase nanoparticles. Journal of Physical Chemistry C. 2010,114(11):4909-4915. DOI: 10.1021/jp912008k
https://doi.org/10.1021/jp912008k...
,4040 Moro CC, Lansarin MA, Bagnara M. Nanotubos de TiO2 dopados com nitrogênio: Comparação das atividades fotocatalíticas de materiais obtidos através de diferentes técnicas. Química Nova. 2012;35(8):1560-1565. DOI: 10.1590/S0100-40422012000800013
https://doi.org/10.1590/S0100-4042201200...
.
2.2 Taguchi method design of experiment
In this work, a standard Taguchi experimental plan L9 was chosen to optimize the experimental conditions to evaluate the morphology of the TiO2 films prepared by the anodization process in a mixture of ethylene glycol and glycerol. Four essential process parameters were chosen for this study: Ti chemical pretreatment, concentration of water, NH4F concentration and the applied anodizing potential. The variation of these factors in turn was based on three different levels: 1) low, 2) medium and 3) high, as shown in Table 1.
With the aim of taking into account the highest degree of interaction, a fully balanced factorial plan was implemented. Thus, a simple factorial design was applied to assign three levels for each parameter and the numbers of permutations would be 8, i.e., degrees of freedom = 9-1 = 8. However, the fractional factorial design reduced the number of experiments to 9. This is shown in Table 2. The choice of parameters was based on the fact that these are the parameters that show the most influence in the anodization process, according to the results mentioned in the literature3737 Liu Z, Zhang X, Nishimoto S, Jin M, Tryk D, Murakami T, et al. Highly Ordered TiO2 Nanotube Arrays with Controllable Length for Photoelectrocatalytic Degradation of Phenol. Journal of Physical Chemistry C. 2008;112(1):253-259. DOI: 10.1021/jp0772732
https://doi.org/10.1021/jp0772732...
,3838 Wang C, Chen D, Ping G, Liu S, Huang XN, Huang YX, et al. Controllable synthesis of well-ordered TiO2 nanotubes in a mixed organic electrolyte for high-efficiency photocatalysis. Science China Chemistry. 2012;55(11):2373-2380. DOI: 10.1007/s11426-012-4680-0
https://doi.org/10.1007/s11426-012-4680-...
. Table 2 represents the Taguchi orthogonal array (L9)4141 Kumar RS, Sureshkumar K, Velraj R. Optimization of biodiesel production from Manilkara zapota(L.) seed oil using Taguchi method. Fuel. 2015;140:90-96. DOI: 10.1016/j.fuel.2014.09.103
https://doi.org/10.1016/j.fuel.2014.09.1...
. And the ranking was assigned on the basis of the parameters shown in Table 1.
Orthogonal array (L9) proposed by Taguchi method to study the variation of four parameters in 3 different levels and the obtained experimental results during 180 minutes of anodization
The most important guidelines that support this method are the orthogonal arrays (L9) and the signal-to-noise (S/N) ratios, which are derived on the basis of loss functions that penalize even small deviations from the target performance level. The first ones allow one to design an optimal plan from a minimum number of experiments, and the S/N ratio makes it possible to estimate the variability of the system response in terms of controllable parameters and random signals3333 Yousefieh M, Tamizifar M, Boutorabi SMA, Borhani E. Taguchi Optimization on the Initial Thickness and Pre-aging of Nano-/Ultrafine-Grained Al-0.2 wt.%Sc Alloy Produced by ARB. Journal of Materials Engineering and Performance. 2016;25(10):4239-4248. DOI: 10.1007/s11665-016-2273-3
https://doi.org/10.1007/s11665-016-2273-...
.
There are three types of S/N ratios used in the Taguchi method and these are nominal-is-best, smaller-is-better and larger-is-better3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
,4141 Kumar RS, Sureshkumar K, Velraj R. Optimization of biodiesel production from Manilkara zapota(L.) seed oil using Taguchi method. Fuel. 2015;140:90-96. DOI: 10.1016/j.fuel.2014.09.103
https://doi.org/10.1016/j.fuel.2014.09.1...
. In this study, it is necessary to calculate the signal-to-noise (S/N) ratio for each experiment and thus determine the effect that each variable has on the output. The S/N for the length of the nanotubes is calculated according to the equation smaller-is-better Equation 1, aiming to maximize the S/N ratio. For the S/N ratios of the internal diameter (nm) and external diameter (nm) of the TiO2 nanotubes, the larger-is-better theorem evaluated from Equation (2) is used3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
,4242 Abbas AR, Li MH, Tai KC. Optimizing SUS 304 wire drawing process by grey relational analysis utilizing Taguchi method. Journal of University of Science and Technology Beijing, Mineral, Metallurgy, Material. 2008;15(6):714-722. DOI: 10.1016/S1005-8850(08)60276-5
https://doi.org/10.1016/S1005-8850(08)60...
,4343 Chaulia KP, Das R. Process parameter optimization for fly ash brick by Taguchi method. Materials Research. 2008;11(2):159-164. DOI: 10.1590/S1516-14392008000200008
https://doi.org/10.1590/S1516-1439200800...
,4444 Acevedo-Peña P, González F, González G, González I. The effect of anatase crystal orientation on the photoelectrochemical performance of anodic TiO2 nanotubes. Physical Chemistry Chemical Physics. 2014;16(47):26213-26220. DOI: 10.1039/C4CP03930K
https://doi.org/10.1039/C4CP03930K...
:
where: n is the number of variables and y i is the value of each variable. The samples mentioned in Table 2 are labeled as Exp. 1 to Exp. 9 from top to bottom, respectively of the first column.
2.3 Characterization
The TiO2 crystalline structure was determined by X-ray diffraction (XRD) using an XRD-6000 by SHIMADZU, operated at 40 kV and 30 mA. The X-ray source consists of Cu radiation (1.54184 Å) selected with an Ni filter. The measurements were performed with a step of 0.05° and a counting time of 0.60 seconds per step and θ/2θ geometry was used. The cross-section morphology was evaluated by the images obtained by Field Emission Gun Scanning Electron Microscope (FEG-SEM) equipment MIRA3 by TESCAN operated at 10 and 15 kV. The micrographs of the surface and cross-section of the nanotubes were taken at various magnifications.
The main effect is observed for each parameter in the plot analyzed by MINITAB. The lengths, internal and external diameters of the obtained structures were determined using the software Image J.
3. Results and Discussion
The as-anodized TiO2 nanotubes are generally amorphous; therefore, thermal treatment was required to crystallize them. Fig. 1 shows the XRD patterns of the samples annealed at 450 ºC for 3 h. The characteristic peak related to the anatase phase of TiO2 appears at 2θ = 25.2º, i.e., in agreement with the standard JCPDS No. 89-4921. The diffractograms of the samples do not present any peak assigned to rutile phase that normally appears at 2θ = 27.36º4545 Regonini D, Groff A, Sorarù GD, Clemens FJ. Suppressing Deep Traps in Self-Organized TiO2 Nanotubes by Nb Doping and Optimized Water Content. Journal of the Electrochemical Society. 2016;16(3):H243-H251. DOI: 10.1149/2.0131605jes
https://doi.org/10.1149/2.0131605jes...
. Hence, the samples prepared in this work consist of anatase crystalline phase. The peaks at 38.3º, 40.4º and 53.4º correspond to the Ti substrate. For Exp. 4 and Exp. 7, it can be observed that the relative peak intensity at 37.8º corresponding to the (004) plane of anatase phase slightly increases as compared to other samples4444 Acevedo-Peña P, González F, González G, González I. The effect of anatase crystal orientation on the photoelectrochemical performance of anodic TiO2 nanotubes. Physical Chemistry Chemical Physics. 2014;16(47):26213-26220. DOI: 10.1039/C4CP03930K
https://doi.org/10.1039/C4CP03930K...
,4646 Liang H, Li X. Effects of structure of anodic TiO2 nanotube arrays on photocatalytic activity for the degradation of 2,3-dichlorophenol in aqueous solution. Journal of Hazardous Materials. 2009;162(2-3):1415-1422. DOI: 10.1016/j.jhazmat.2008.06.033
https://doi.org/10.1016/j.jhazmat.2008.0...
,4747 Li L, Ya J, Hu F, Liu Z, Lei E. Photocatalytic decompositions of gaseous HCHO and methylene blue with highly ordered TiO2 nanotube arrays. Journal of Energy Chemistry. 2016;25(4):740-746. DOI: 10.1016/j.jechem.2016.03.003
https://doi.org/10.1016/j.jechem.2016.03...
.
XRD patterns of TiO2 samples for different experiments (Table 2) annealed at 450 °C for 3 h
Fig. 2 displays the FEG-SEM cross-sectional images of the heat treated films and their thicknesses are displayed in Table 3. It can be observed that Exp. 1 and Exp. 6 present a relatively thinner oxide layer as compared to other experiments (Table 3); however, Exp. 1 does not present a tubular morphology. It should be noted that both samples were obtained at lower applied potential level (Table 2). The oxide thicknesses for Exp. 2, Exp. 3 and Exp. 4 were 2.51 µm, 2.70 µm and 2.80 µm, respectively (Table 3). The sample in Exp. 8 shows the formation of a discontinuous and completely irregular nanostructure without formation of nanotubes. Exp. 5 and Exp. 7 resulted in the smoothest and longest nanostructure (4.72 µm and 7.66 µm, respectively). Among all the samples, smooth walls were obtained for Exp. 4, Exp. 5 and Exp. 7. The S/N ratio of aspect ratio and standard deviation of TiO2 nanotubes for 9 experiments are calculated as shown in Table 3. Then, the S/N ratio response to each factor at each level were obtained from the orthogonal array experiments which calculated from the average S/N ratio for each level of the factors3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
,4343 Chaulia KP, Das R. Process parameter optimization for fly ash brick by Taguchi method. Materials Research. 2008;11(2):159-164. DOI: 10.1590/S1516-14392008000200008
https://doi.org/10.1590/S1516-1439200800...
.
Cross-sectional images obtained by FEG-SEM of TiO2 nanotube arrays fabricated for 180 minutes of anodization and annealed at 450 °C for 3 h
Mean values of internal diameter, external diameter and thickness of the heat treated layer and their respects signal-to-noise (S/N) ratio
As illustrated in Table 3, Exp. 3 (A1B3C3D3) has value internal diameter higher of S/N 41.61 with the average nanotubes internal larger and standard deviation at 115.4 ± 22 nm. However, Exp. 6 (A2B3C1D2) with the S/N value of 31.38 and the smaller internal diameter and standard deviation at 38.2 ± 3 nm. Therefore, for the external diameter it is observed that Exp. 3 shows the largest diameter external and Exp. 6 the smallest external diameter. Thus, it can be concluded that the Exp. 3 is a strong candidate for application in the photocatalytic decomposition in formaldehyde and methylene blue4747 Li L, Ya J, Hu F, Liu Z, Lei E. Photocatalytic decompositions of gaseous HCHO and methylene blue with highly ordered TiO2 nanotube arrays. Journal of Energy Chemistry. 2016;25(4):740-746. DOI: 10.1016/j.jechem.2016.03.003
https://doi.org/10.1016/j.jechem.2016.03...
,4848 Pasikhani JV, Gilani N, Pirbazari AE. The effect of the anodization voltage on the geometrical characteristics and photocatalytic activity of TiO2 nanotube arrays. Nano-Structures & Nano-Objects. 2016;8:7-14. DOI: 10.1016/j.nanoso.2016.09.001
https://doi.org/10.1016/j.nanoso.2016.09...
. However, for the thickness of the TiO2 nanotubes it is observed (Table 3) that the higher the thickness of the nanotubes the higher the S/N.
As mentioned before, the signal-to-noise (S/N) ratio analysis is adapted to improve the statistical properties of the Taguchi design method used in this work to evaluate the conditions required to obtain ordered TiO2 nanotubular arrays by anodization (Fig. 3). In this study, for measurable quality characteristics, the equation smaller-is-better was used as in Eq. (1)
3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
.
The trend diagram of each parameter on the mean for the length of the nanotubes (signal-to-noise (S/N) ratio)
In the Fig. 3, Fig. 5 to Fig. 7 for the parameters the chemical pretreatment (A) and water concentration (B) the level 1 means that the lower level of chemical treatment was performed in time equal to zero, it means without any chemical attack, and for parameter B (water concentration) there was no addition of water in the organic electrolyte. Therefore, level 1 was chosen in this way to verify their behavior/influence of these variables in all processes of the production of TiO2 nanotubes.
Fig. 3 shows the S/N ratios calculated from Eq.(1) for different lengths of the TiO2 nanotubes (Table 3). Optimal levels obtained from S/N ratios are marked on the graphs with a circle. This parameter (smaller-is-better) was chosen because, according to the literature, it is reported that shorter TiO2 nanotubes had a better efficiency in degradation of pollutants and hydrogen production, compared to longer nanotubes1818 Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research. 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
https://doi.org/10.1590/1980-5373-MR-201...
,4949 Gui Q, Yu D, Li D, Song Y, Zhu X, Cao L, et al. Efficient suppression of nanograss during porous anodic TiO2 nanotubes growth. Applied Surface Science. 2014;314:505-509. DOI: 10.1016/j.apsusc.2014.07.046
https://doi.org/10.1016/j.apsusc.2014.07...
. The longer nanotubes have a slower internal diffusion for reactants, which is detrimental to the reaction rate1212 Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews. 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
https://doi.org/10.1021/cr500061m...
,4949 Gui Q, Yu D, Li D, Song Y, Zhu X, Cao L, et al. Efficient suppression of nanograss during porous anodic TiO2 nanotubes growth. Applied Surface Science. 2014;314:505-509. DOI: 10.1016/j.apsusc.2014.07.046
https://doi.org/10.1016/j.apsusc.2014.07...
. The Taguchi orthogonal array (L9) is an approach to reduce the number of experiments optimizing the parameters and the response variable as reported in the literature3030 Yiamsawas D, Boonpavanitchakul K, Kangwansupamonkon W. Optimization of experimental parameters based on the Taguchi robust design for the formation of zinc oxide nanocrystals by solvothermal method. Materials Research Bulletin. 2011;46(5):639-642. DOI: 10.1016/j.materresbull.2011.02.004
https://doi.org/10.1016/j.materresbull.2...
,4141 Kumar RS, Sureshkumar K, Velraj R. Optimization of biodiesel production from Manilkara zapota(L.) seed oil using Taguchi method. Fuel. 2015;140:90-96. DOI: 10.1016/j.fuel.2014.09.103
https://doi.org/10.1016/j.fuel.2014.09.1...
.
Therefore, by means of the Fig. 3, it is possible to see that the studied parameters ((A) chemical pretreatment, (B) concentration of water, (C) applied potential and (D) concentration of NH4F) influence in the following order (from larger to smaller importance) when the objective is to obtain smaller nanotubes: applied potential, concentration of water, concentration of NH4F and chemical pretreatment. So to get smaller nanotubes what should be taken into account is the ranking described above.
Fig. 4 shows the top-view FEG-SEM images of the heat treated samples and the external and internal diameters are displayed in Table 3. It can be observed that Exp. 1 (Fig. 4) presents no formation of nanotubes. However, the presence of the oxide on the surface can be observed from the cross-sectional view (Fig. 2), which clearly identifies that the formation of the TiO2 nanotubes did not occur. For Exp. 4 and Exp. 7 is observed cracks in top. In the literature2020 Macak JM, Hildebrand H, Marten-Jahns U, Schmuki P. Mechanistic aspects and growth of large diameter self-organized TiO2 nanotubes. Journal of Electroanalytical Chemistry. 2008;621(2):254-266. DOI: 10.1016/j.jelechem.2008.01.005
https://doi.org/10.1016/j.jelechem.2008....
,5050 Mor GK, Varghese OK, Paulose M, Ong KG, Grimes CA. Fabrication of hydrogen sensors with transparent titanium oxide nanotube-array thin films as sensing elements. Thin Solid Films. 2006;496(1):42-48. DOI: 10.1016/j.tsf.2005.08.190
https://doi.org/10.1016/j.tsf.2005.08.19...
,5151 Meen TH, Tsai JK, Tu YS, Wu TC, Hsu WD, Chang SJ. Optimization of the dye-sensitized solar cell performance by mechanical compression. Nanoscale Research Letters. 2014;9:523. DOI: 10.1186/1556-276X-9-523
https://doi.org/10.1186/1556-276X-9-523...
reports that these cracks can be caused by various forms, such as through the from chemical and field-assisted dissolution of the oxide at local points of high energy, or through the capillary force that appears when solvent rapidly evaporate from film surface during drying process of the TiO2 nanotubes, decreasing of bond strength among TiO2 nanotubes when the film is thick.
Top-view images obtained by FEG/SEM of TiO2 nanotube arrays fabricated for 180 minutes of anodization and annealed at 450 °C for 3 h
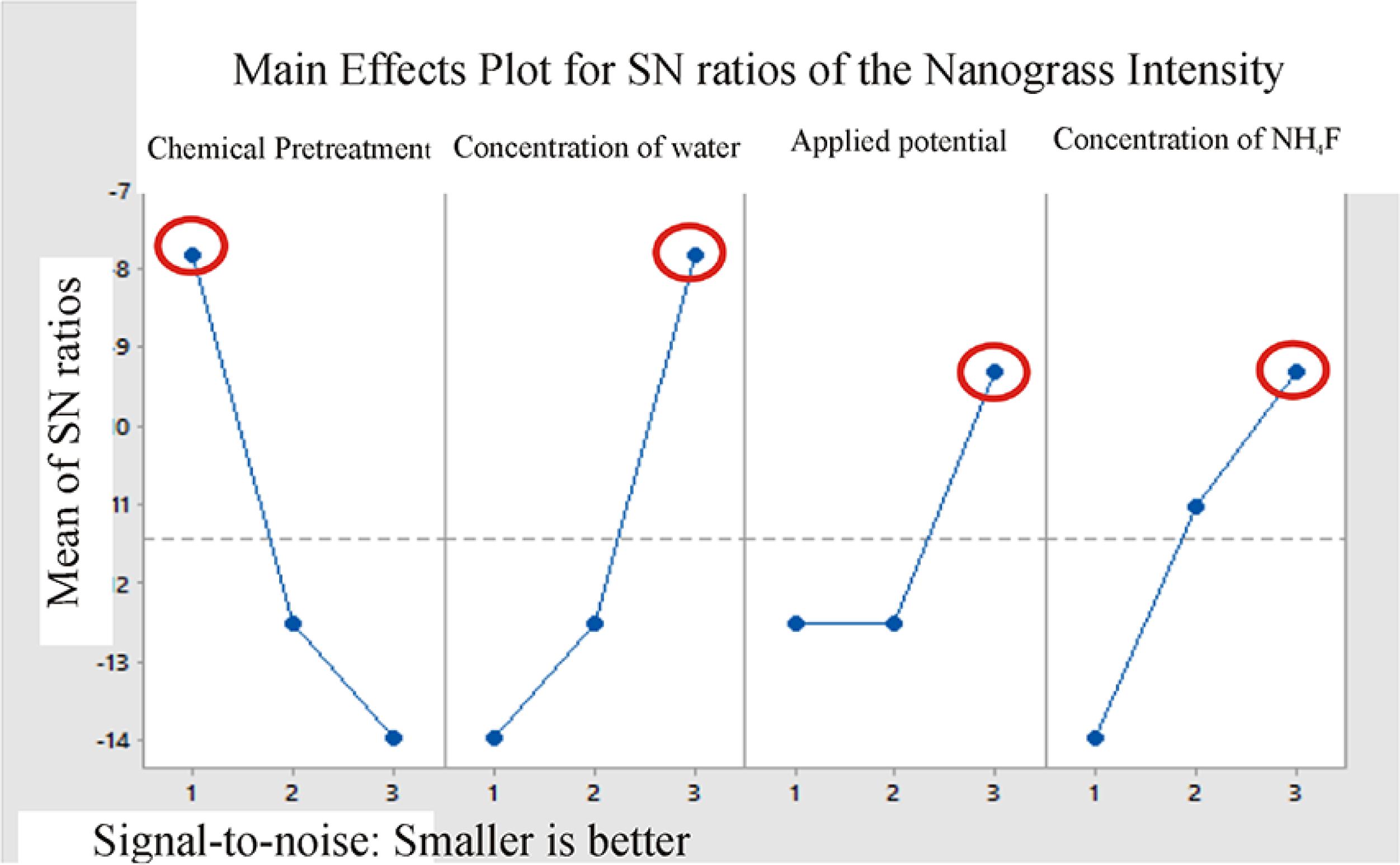
The trend diagram of each parameter on the mean for the nanograss (signal-to-noise (S/N) ratio)
The trend diagram of each parameter on the mean of the internal diameter of the nanotubes (signalto-noise (S/N) ratio)
The trend diagram of each parameters on the mean on the external diameter of the nanotubes (signal-to-noise (S/N) ratio)
It is also evident that after 180 minutes of anodization, there is a compact oxide region on the top of these samples, called nanograss5252 Paulose M, Shankar K, Yoriya S, Prakasam HE, Varghese OK, Mor GK, et al. Anodic growth of highly ordered TiO2 nanotube arrays to 134 µm in length. Journal of Physical Chemistry B. 2006;110(33):16179-16184. DOI: 10.1021/jp064020k
https://doi.org/10.1021/jp064020k...
. Thus, the tubular structure of the nanotubes could not be visualized, but only the presence of very open pores can be observed, indicating that the nanograss is covering the top of the nanotubes5353 Tsui L, Zangari G. Water content in the anodization electrolyte affects the electrochemical and electronic transport properties of TiO2 nanotubes: a study by electrochemical impedance spectroscopy. Electrochimica Acta. 2014;121:203-209. DOI: 10.1016/j.electacta.2013.12.163
https://doi.org/10.1016/j.electacta.2013...
. For Exp. 2, Exp. 3 and Exp. 6 the top surfaces clearly present tubular structures. In Exp. 5 (Fig. 4) the top is covered with nanograss and after removal of the surface oxide, the top-view image clearly shows that these nanotubes are composed of well-defined inner and outer shells.
According to the literature1212 Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews. 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
https://doi.org/10.1021/cr500061m...
,5454 Kim D, Ghicov A, Schmuki P. TiO2 Nanotube arrays: Elimination of disordered top layers ("nanograss") for improved photoconversion efficiency in dye-sensitized solar cells. Electrochemistry Communications. 2008;10(12):1835-1838. DOI: 10.1016/j.elecom.2008.09.029
https://doi.org/10.1016/j.elecom.2008.09...
,5555 Song YY, Schmidt-Stein F, Bauer S, Schmuki P. Amphiphilic TiO2 Nanotube Arrays: An Actively Controllable Drug Delivery System. Journal of the American Chemical Society. 2009;131(12):4230-4232. DOI: 10.1021/ja810130h
https://doi.org/10.1021/ja810130h...
,5656 Liao Y, Zhang D, Wang Q, Wen T, Jia L, Zhong Z, et al. Open-top TiO2 nanotube arrays with enhanced photovoltaic and photochemical performances via a micromechanical cleavage approach. Journal of Materials Chemistry A. 2015;3(27):14279-14283. DOI: 10.1039/C5TA02799C
https://doi.org/10.1039/C5TA02799C...
, nanograss on the surface of TiO2 nanotubes is undesirable, and for this reason it is important to obtain nanotubes free of nanograss. Using the smaller-is-better criterion levels for the operating parameters of the nanograss on top of TiO2 nanotubes, the S/N ratio was obtained from Eq. (1)
3232 Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
https://doi.org/10.1109/ICRSE.2015.73664...
. The criterion adopted to determine the presence of the nanograss was by percentage of covered surfaces, with values of 10, 50 and 100% being assigned according to the amount that covered the surface of the sample. The 10% value corresponds to samples with free or low amount of nanograss. The 50% value corresponds to the surface of the sample with approximately half of the surface covered with nanograss and the value 100% percent corresponds to the samples completely covered by this oxide.
It can be seen that the studied parameters influence in the following order (from more to less important) as shown in Fig. 5, when the objective is to obtain nanotubes free of nanograss: (A) chemical pretreatment, (B) concentration of water, (C) applied potential and (D) concentration of NH4F. As can be observed for the parameters; (B) concentration of water, (C) applied potential and (D) concentration of NH4F presenting a higher level, therefore, occurs a smaller formation of nanograss on top of TiO2 nanotubes, except for the parameter (A) chemical pretreatment which presents a lower level (no chemical treatment), which means that no pretreatment is required to avoid the nanograss formation.
The Taguchi design method was also used to identify the influences on the internal and external diameters (Table 3) of TiO2 nanotubes (Fig. 6 and 7). In this second stage of the work, for measurable quality characteristics, the equation larger-is-better was used as in Eq. (2). Figs 6 and 7 show the S/N ratios calculated from Eq. (2) for different internal and external diameters of the TiO2 nanotubes, as set out in Table 3.
This parameter (larger-is-better) for the internal and external diameters was chosen because, according to the literature1919 Albertin KF, Tavares A, Pereyra I. Optimized Ti polishing techniques for enhanced order in TiO2 NT arrays. Applied Surface Science. 2013;284:772-779. DOI: 10.1016/j.apsusc.2013.08.005
https://doi.org/10.1016/j.apsusc.2013.08...
,5555 Song YY, Schmidt-Stein F, Bauer S, Schmuki P. Amphiphilic TiO2 Nanotube Arrays: An Actively Controllable Drug Delivery System. Journal of the American Chemical Society. 2009;131(12):4230-4232. DOI: 10.1021/ja810130h
https://doi.org/10.1021/ja810130h...
,it is reported that TiO2 nanotubes with larger diameter presents an better efficiency in several applications as in environmental and energy55 Paramasivam I, Jha H, Liu N, Schmuki P. A Review of Photocatalysis using Self-organized TiO2 Nanotubes and Other Ordered Oxide Nanostructures. Small. 2012;8(20):3073-3103. DOI: 10.1002/smll.201200564
https://doi.org/10.1002/smll.201200564...
,1212 Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews. 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
https://doi.org/10.1021/cr500061m...
,5656 Liao Y, Zhang D, Wang Q, Wen T, Jia L, Zhong Z, et al. Open-top TiO2 nanotube arrays with enhanced photovoltaic and photochemical performances via a micromechanical cleavage approach. Journal of Materials Chemistry A. 2015;3(27):14279-14283. DOI: 10.1039/C5TA02799C
https://doi.org/10.1039/C5TA02799C...
,5757 Nakata K, Fujishima A. TiO2 photocatalysis: Design and applications. Journal of Photochemistry and Photobiology C: Photochemistry Reviews. 2012;13(3):169-189. DOI: 10.1016/j.jphotochemrev.2012.06.001
https://doi.org/10.1016/j.jphotochemrev....
.
The values of the internal and external diameters of the samples Exp. 1 and Exp. 8 are not mentioned in Table 3 due to their irregular structure, which prevented us from estimating them (Fig. 4), but in order to execute the Taguchi method program an arbitrary value was added.
The signal-to-noise (S/N) ratio analysis is adopted to improve the statistical properties (Fig. 6 and 7). Therefore, it can be observed that the medium chemical pretreatment interferes with the internal diameter, as observed in Fig. 6. Besides, a high water concentration and the applied potential are important factors that increase the internal diameter of the nanotubes, as the fluoride concentration at a medium level favors the increase in the internal diameter (Fig. 6).
Therefore, to obtain a larger internal diameter of the TiO2 nanotubes, it is important to note the parameters that influence in the following order: (C) applied potential, (B) concentration of water, (D) concentration of NH4F and (A) chemical pretreatment.
Fig. 7 shows the ranking of the factors that contribute to the increase of the external diameter in the following order (larger-is-better): (C) applied potential, (B) concentration of water, (A) chemical pretreatment and (D) concentration of NH4F. In this way, nanotubes with larger external diameters are obtained.
According to the Taguchi experimental method, we show a ranking of the importance of the factors that influence the formation of the TiO2 nanotube morphology. The parameter levels combination that simultaneously satisfy the various aspects considered (length of the nanotubes, nanograss, internal and external diameter of the nanotubes) in obtaining the TiO2 nanotube morphology described in the literature33 Li Y, Yu H, Zhang C, Song W, Li G, Shao Z, et al. Effect of water and annealing temperature of anodized TiO2 nanotubes on hydrogen production in photoelectrochemical cell. Electrochimica Acta. 2013;107:313-319. DOI: 10.1016/j.electacta.2013.05.090
https://doi.org/10.1016/j.electacta.2013...
,4747 Li L, Ya J, Hu F, Liu Z, Lei E. Photocatalytic decompositions of gaseous HCHO and methylene blue with highly ordered TiO2 nanotube arrays. Journal of Energy Chemistry. 2016;25(4):740-746. DOI: 10.1016/j.jechem.2016.03.003
https://doi.org/10.1016/j.jechem.2016.03...
,5858 Sun Y, Yan KP. Effect of anodization voltage on performance of TiO2 nanotube arrays for hydrogen generation in a two-compartment photoelectrochemical cell. International Journal of Hydrogen Energy. 2014;39(22):11368-11375., for the photocatalysis application, correspond the parameters used to obtain the sample Exp. 3.
4. Conclusions
In this paper, the synthesis of TiO2 nanotubes via the anodization method has been investigated by a Taguchi method experiment. Applied potential, chemical pretreatment, water concentration and the fluoride content were considered as the main factors that influence the anodization process. The results are summarized as follows:
-
The results obtained from Taguchi's test design (smaller-is-better) show that it is preferable to obtain nanotubes with less thickness. Thus, it is necessary to observe the following order: applied potential, concentration of water, concentration of NH4F and chemical pretreatment. Therefore, to avoid the formation of nanograss at the top of the TiO2 nanotubes, the following order must be followed (from more to less important) when the aim is obtain nanotubes free of nanograss: chemical pretreatment, concentration of water, applied potential and concentration of NH4F.
-
Also the Taguchi method was used to identify the influences on the internal and external diameters of TiO2 nanotubes with the parameter larger-is-better. Therefore, to obtain a larger internal diameter of the TiO2 nanotubes, it is important to note the parameters that influence in the following order: applied potential, concentration of water, concentration of NH4F and chemical pretreatment. To increase the external diameter, it is important to note the parameters that influence in the following order: applied potential, concentration of water, chemical pretreatment and concentration of NH4F.
-
The morphological analyses (Fig. 4) have shown that the sample Exp. 3 resulted in the smallest amount of surface nanograss, i.e., this condition makes it possible to obtain open top nanotubes, obtained from Ti foil without chemical pretreatment and high levels of water, potential and concentration of NH4F, as represented in Fig. 5.
Therefore, according to the Taguchi experimental method, exhibited a ranking of the importance of the factors that influence the morphology of TiO2 nanotubes. Thus, concludes a combination for photocatalysis applications, according to the ranking; applied potential (level 3); concentration of water (level 3); concentration of NH4F (level 2) and chemical pretreatment (level 1).
5. Acknowledgments
The present work was carried out with the support of FAPERGS, CNPq and CAPES (a Brazilian Government entity focused on human resources formation), we also thank C-LABMU, UEPG, for carrying out the FEG analysis. The authors also thank the LCMicro-UCS.
6. References
-
1Roy P, Albu SP, Schmuki P. TiO2 nanotubes in dye-sensitized solar cells: Higher efficiencies by well-defined tube tops. Electrochemistry Communications 2010;12(7):949-951. DOI: 10.1016/j.elecom.2010.04.029
» https://doi.org/10.1016/j.elecom.2010.04.029 -
2Roy P, Kim D, Lee, K, Spiecker E, Schmuki P. TiO2 nanotubes and their application in dye-sensitized solar cells. Nanoscale 2010;2(1):45-59. DOI: 10.1039/B9NR00131J
» https://doi.org/10.1039/B9NR00131J -
3Li Y, Yu H, Zhang C, Song W, Li G, Shao Z, et al. Effect of water and annealing temperature of anodized TiO2 nanotubes on hydrogen production in photoelectrochemical cell. Electrochimica Acta 2013;107:313-319. DOI: 10.1016/j.electacta.2013.05.090
» https://doi.org/10.1016/j.electacta.2013.05.090 -
4Varghese OK, Gong D, Paulose M, Ong KG, Grimes CA. Hydrogen sensing using titania nanotubes. Sensors Actuators B: Chemical 2003;93(1-3):338-344. DOI: 10.1016/S0925-4005(03)00222-3
» https://doi.org/10.1016/S0925-4005(03)00222-3 -
5Paramasivam I, Jha H, Liu N, Schmuki P. A Review of Photocatalysis using Self-organized TiO2 Nanotubes and Other Ordered Oxide Nanostructures. Small 2012;8(20):3073-3103. DOI: 10.1002/smll.201200564
» https://doi.org/10.1002/smll.201200564 -
6Zheng L, Xiao X, Li Y, Zhang W. Enhanced photocatalytic activity of TiO2 nanoparticles using WS2/g-C3N4 hybrid as co-catalyst. Transactions of Nonferrous Metals Society of China 2017;27:1117-1126. DOI: 10.1016/S1003-6326(17)60130-4
» https://doi.org/10.1016/S1003-6326(17)60130-4 -
7Khan S, Teixeira SR, Santos MJL. Controlled thermal nitridation resulting in improved structural and photoelectrochemical properties from Ta3N5 nanotubular photoanodes. RSC Advances 2015;5(125):103284-103291. DOI: 10.1039/C5RA17227F
» https://doi.org/10.1039/C5RA17227F -
8Oller I, Malato S, Sánchez-Pérez JA. Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination-A review. Science of The Total Environment 2011;409(20):4141-4166. DOI: 10.1016/j.scitotenv.2010.08.061
» https://doi.org/10.1016/j.scitotenv.2010.08.061 -
9Zhou Q, Huang Y, Xie G. Investigation of the applicability of highly ordered TiO2 nanotube array for enrichment and determination of polychlorinated biphenyls at trace level in environmental water samples. Journal of Chromatography A 2012;1237:24-29. DOI: 10.1016/j.chroma.2012.02.066
» https://doi.org/10.1016/j.chroma.2012.02.066 -
10Tsuchiya H, Macak JM, Müller L, Kunze J, Müller F, Greil P, et al. Hydroxyapatite growth on anodic TiO2 nanotubes. Journal of Biomedical Materials Research. Part A. 2006;77A(3):534-541. DOI: 10.1002/jbm.a.30677
» https://doi.org/10.1002/jbm.a.30677 -
11Byranvand MM, Kharat AN, Fatholahi L, Beiranvand ZM. A Review on Synthesis of Nano-TiO2 via Different Methods. Journal of Nanostructures 2013;3:1-9.
-
12Lee K, Mazare A, Schmuki P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chemical Reviews 2014;114(19):9385-9454. DOI: 10.1021/cr500061m
» https://doi.org/10.1021/cr500061m -
13Coser E, Bervian A, Khan S, De Léon A, Bussi J, Pianaro SA, et al. Photodegradation of glycerol using nanostructured TiO2 catalyst. Renewable Energies and Power Quality Journal 2017;15(1):468-472. DOI: 10.24084/repqj15.35
» https://doi.org/10.24084/repqj15.35 -
14Nischk M, Mazierski P, Gazda M, Zaleska A. Ordered TiO2 nanotubes: The effect of preparation parameters on the photocatalytic activity in air purification process. Applied Catalysis B: Environmental 2014;144:674-685. DOI: 10.1016/j.apcatb.2013.07.041
» https://doi.org/10.1016/j.apcatb.2013.07.041 -
15Xue C, Yonezawa T, Nguyen MT, Lu X. Cladding Layer on Well-Defined Double-Wall TiO2 Nanotubes. Langmuir 2015;31(4):1575-1580. DOI: 10.1021/la504670p
» https://doi.org/10.1021/la504670p -
16Raja KS, Misra M, Paramguru K. Formation of self-ordered nano-tubular structure of anodic oxide layer on titanium. Electrochimica Acta 2005;51(1):154-165. DOI: 10.1016/j.electacta.2005.04.011
» https://doi.org/10.1016/j.electacta.2005.04.011 -
17Khan S, Zapata MJM, Baptista DL, Gonçalves RV, Fernandes JA, Dupont J, et al. Effect of Oxygen Content on the Photoelectrochemical Activity of Crystallographically Preferred Oriented Porous Ta3N5 Nanotubes. Journal of Physical Chemistry C 2015;119(34):19906-19914. DOI: 10.1021/acs.jpcc.5b05475
» https://doi.org/10.1021/acs.jpcc.5b05475 -
18Bervian A, Coser E, Khan S, Pianaro SA, Aguzzoli C, Marcuzzo JS, et al. Evolution of TiO2 Nanotubular Morphology Obtained in Ethylene Glycol Mixed with Glycerol Mixture and its Photoelectrochemical Performance. Materials Research 2017;20(4):962-972. DOI: 10.1590/1980-5373-MR-2016-0878
» https://doi.org/10.1590/1980-5373-MR-2016-0878 -
19Albertin KF, Tavares A, Pereyra I. Optimized Ti polishing techniques for enhanced order in TiO2 NT arrays. Applied Surface Science 2013;284:772-779. DOI: 10.1016/j.apsusc.2013.08.005
» https://doi.org/10.1016/j.apsusc.2013.08.005 -
20Macak JM, Hildebrand H, Marten-Jahns U, Schmuki P. Mechanistic aspects and growth of large diameter self-organized TiO2 nanotubes. Journal of Electroanalytical Chemistry 2008;621(2):254-266. DOI: 10.1016/j.jelechem.2008.01.005
» https://doi.org/10.1016/j.jelechem.2008.01.005 -
21Macak JM, Schmuki P. Anodic growth of self-organized anodic TiO2 nanotubes in viscous electrolytes. Electrochimica Acta 2006;52(3):1258-1264. DOI: 10.1016/j.electacta.2006.07.021
» https://doi.org/10.1016/j.electacta.2006.07.021 -
22Wong YH, Affendy MG, Lau SK, Teh PC, Lee HJ, Tan CY, et al. Effects of anodisation parameters on thin film properties: a review. Materials Science and Technology 2017;33(6):699-711. DOI: 10.1080/02670836.2016.1193654
» https://doi.org/10.1080/02670836.2016.1193654 -
23Ghicov A, Schmuki P. Self-ordering electrochemistry: a review on growth and functionality of TiO2 nanotubes and other self-aligned MOx structures. Chemical Communications 2009;20:2791-2808. DOI: 10.1039/b822726h
» https://doi.org/10.1039/b822726h -
24Xie ZB, Blackwood DJ. Effects of anodization parameters on the formation of titania nanotubes in ethylene glycol. Electrochimica Acta 2010;56(2):905-912. DOI: 10.1016/j.electacta.2010.10.004
» https://doi.org/10.1016/j.electacta.2010.10.004 -
25Taveira LV, Macák JM, Tsuchiya H, Dick LFP, Schmuki P. Initiation and Growth of Self-Organized TiO2 Nanotubes Anodically Formed in NH4F/(NH4)2SO4 Electrolytes. Journal of the Electrochemical Society 2005;152(10):B405-B410. DOI: 10.1149/1.2008980
» https://doi.org/10.1149/1.2008980 -
26Albu SP, Ghicov A, Schmuki P. High aspect ratio, self-ordered iron oxide nanopores formed by anodization of Fe in ethylene glycol/NH4F electrolytes. Physica Status Solidi - Rapid Research Letters 2009;3(2-3):64-66. DOI: 10.1002/pssr.200802285
» https://doi.org/10.1002/pssr.200802285 -
27Lee BG, Choi JW, Lee SE, Jeong YS, Oh HJ, Chi CS. Formation behavior of anodic TiO2 nanotubes in fluoride containing electrolytes. Transactions of Nonferrous Metals Society of China 2009;19(4):842-845. DOI: 10.1016/S1003-6326(08)60361-1
» https://doi.org/10.1016/S1003-6326(08)60361-1 -
28Alagumurthi N, Palaniradja K, Soundararajan V. Optimization of Grinding Process Through Design of Experiment (DOE)-A Comparative Study. Materials and Manufacturing Processes 2006;21(1):19-21. DOI: 10.1081/AMP-200060605
» https://doi.org/10.1081/AMP-200060605 -
29Pignatiello JJ Jr. An Overview of the Strategy and Tactics of Taguchi. IIE Transactions 1988;20(3):247-254. DOI: 10.1080/07408178808966177
» https://doi.org/10.1080/07408178808966177 -
30Yiamsawas D, Boonpavanitchakul K, Kangwansupamonkon W. Optimization of experimental parameters based on the Taguchi robust design for the formation of zinc oxide nanocrystals by solvothermal method. Materials Research Bulletin 2011;46(5):639-642. DOI: 10.1016/j.materresbull.2011.02.004
» https://doi.org/10.1016/j.materresbull.2011.02.004 -
31Mohamed AMA, Jafari R, Farzaneh M. An optimization of superhydrophobic polyvinylidene fluoride/zinc oxide materials using Taguchi method. Applied Surface Science 2014;288:229-237. DOI: 10.1016/j.apsusc.2013.10.013
» https://doi.org/10.1016/j.apsusc.2013.10.013 -
32Zhu C, He Y, Li T. Application of a Taguchi method for optimizing the preparation and degradation parameters of composite photocatalyst TiO2-MWCNTs. In: Proceedings of 2015 First International Conference on Reliability Systems Engineering (ICRSE); 2015 Oct 21-23;Beinjing, China. DOI: 10.1109/ICRSE.2015.7366496
» https://doi.org/10.1109/ICRSE.2015.7366496 -
33Yousefieh M, Tamizifar M, Boutorabi SMA, Borhani E. Taguchi Optimization on the Initial Thickness and Pre-aging of Nano-/Ultrafine-Grained Al-0.2 wt.%Sc Alloy Produced by ARB. Journal of Materials Engineering and Performance 2016;25(10):4239-4248. DOI: 10.1007/s11665-016-2273-3
» https://doi.org/10.1007/s11665-016-2273-3 -
34Ghani JA, Choudhury IA, Hassan HH. Application of Taguchi method in the optimization of end milling parameters. Journal of Materials Processing Technology 2004;145(1):84-92. DOI: 10.1016/S0924-0136(03)008653
» https://doi.org/10.1016/S0924-0136(03)00865-3 -
35Naghibi S, Sani MAF, Hosseini HRM. Application of the statistical Taguchi method to optimize TiO2 nanoparticles synthesis by the hydrothermal assisted sol-gel technique. Ceramics International 2014;40(3):4193-4201. DOI: 10.1016/j.ceramint.2013.08.077
» https://doi.org/10.1016/j.ceramint.2013.08.077 -
36Kulkarni S, Duttagupta S, Phatak G. Taguchi design of experiments for optimization of ionic conductivity in nanocrystalline Gadolinium doped Ceria. Ceramics International 2015;41(7):8973-8980. DOI: 10.1016/j.ceramint.2015.03.171
» https://doi.org/10.1016/j.ceramint.2015.03.171 -
37Liu Z, Zhang X, Nishimoto S, Jin M, Tryk D, Murakami T, et al. Highly Ordered TiO2 Nanotube Arrays with Controllable Length for Photoelectrocatalytic Degradation of Phenol. Journal of Physical Chemistry C 2008;112(1):253-259. DOI: 10.1021/jp0772732
» https://doi.org/10.1021/jp0772732 -
38Wang C, Chen D, Ping G, Liu S, Huang XN, Huang YX, et al. Controllable synthesis of well-ordered TiO2 nanotubes in a mixed organic electrolyte for high-efficiency photocatalysis. Science China Chemistry 2012;55(11):2373-2380. DOI: 10.1007/s11426-012-4680-0
» https://doi.org/10.1007/s11426-012-4680-0 -
39Kandiel TA, Dillert R, Feldhoff A, Bahnemann DW. Direct synthesis of photocatalytically active rutile TiO2 nanorods partly decorated with anatase nanoparticles. Journal of Physical Chemistry C 2010,114(11):4909-4915. DOI: 10.1021/jp912008k
» https://doi.org/10.1021/jp912008k -
40Moro CC, Lansarin MA, Bagnara M. Nanotubos de TiO2 dopados com nitrogênio: Comparação das atividades fotocatalíticas de materiais obtidos através de diferentes técnicas. Química Nova 2012;35(8):1560-1565. DOI: 10.1590/S0100-40422012000800013
» https://doi.org/10.1590/S0100-40422012000800013 -
41Kumar RS, Sureshkumar K, Velraj R. Optimization of biodiesel production from Manilkara zapota(L.) seed oil using Taguchi method. Fuel 2015;140:90-96. DOI: 10.1016/j.fuel.2014.09.103
» https://doi.org/10.1016/j.fuel.2014.09.103 -
42Abbas AR, Li MH, Tai KC. Optimizing SUS 304 wire drawing process by grey relational analysis utilizing Taguchi method. Journal of University of Science and Technology Beijing, Mineral, Metallurgy, Material 2008;15(6):714-722. DOI: 10.1016/S1005-8850(08)60276-5
» https://doi.org/10.1016/S1005-8850(08)60276-5 -
43Chaulia KP, Das R. Process parameter optimization for fly ash brick by Taguchi method. Materials Research 2008;11(2):159-164. DOI: 10.1590/S1516-14392008000200008
» https://doi.org/10.1590/S1516-14392008000200008 -
44Acevedo-Peña P, González F, González G, González I. The effect of anatase crystal orientation on the photoelectrochemical performance of anodic TiO2 nanotubes. Physical Chemistry Chemical Physics 2014;16(47):26213-26220. DOI: 10.1039/C4CP03930K
» https://doi.org/10.1039/C4CP03930K -
45Regonini D, Groff A, Sorarù GD, Clemens FJ. Suppressing Deep Traps in Self-Organized TiO2 Nanotubes by Nb Doping and Optimized Water Content. Journal of the Electrochemical Society 2016;16(3):H243-H251. DOI: 10.1149/2.0131605jes
» https://doi.org/10.1149/2.0131605jes -
46Liang H, Li X. Effects of structure of anodic TiO2 nanotube arrays on photocatalytic activity for the degradation of 2,3-dichlorophenol in aqueous solution. Journal of Hazardous Materials 2009;162(2-3):1415-1422. DOI: 10.1016/j.jhazmat.2008.06.033
» https://doi.org/10.1016/j.jhazmat.2008.06.033 -
47Li L, Ya J, Hu F, Liu Z, Lei E. Photocatalytic decompositions of gaseous HCHO and methylene blue with highly ordered TiO2 nanotube arrays. Journal of Energy Chemistry 2016;25(4):740-746. DOI: 10.1016/j.jechem.2016.03.003
» https://doi.org/10.1016/j.jechem.2016.03.003 -
48Pasikhani JV, Gilani N, Pirbazari AE. The effect of the anodization voltage on the geometrical characteristics and photocatalytic activity of TiO2 nanotube arrays. Nano-Structures & Nano-Objects 2016;8:7-14. DOI: 10.1016/j.nanoso.2016.09.001
» https://doi.org/10.1016/j.nanoso.2016.09.001 -
49Gui Q, Yu D, Li D, Song Y, Zhu X, Cao L, et al. Efficient suppression of nanograss during porous anodic TiO2 nanotubes growth. Applied Surface Science 2014;314:505-509. DOI: 10.1016/j.apsusc.2014.07.046
» https://doi.org/10.1016/j.apsusc.2014.07.046 -
50Mor GK, Varghese OK, Paulose M, Ong KG, Grimes CA. Fabrication of hydrogen sensors with transparent titanium oxide nanotube-array thin films as sensing elements. Thin Solid Films 2006;496(1):42-48. DOI: 10.1016/j.tsf.2005.08.190
» https://doi.org/10.1016/j.tsf.2005.08.190 -
51Meen TH, Tsai JK, Tu YS, Wu TC, Hsu WD, Chang SJ. Optimization of the dye-sensitized solar cell performance by mechanical compression. Nanoscale Research Letters 2014;9:523. DOI: 10.1186/1556-276X-9-523
» https://doi.org/10.1186/1556-276X-9-523 -
52Paulose M, Shankar K, Yoriya S, Prakasam HE, Varghese OK, Mor GK, et al. Anodic growth of highly ordered TiO2 nanotube arrays to 134 µm in length. Journal of Physical Chemistry B 2006;110(33):16179-16184. DOI: 10.1021/jp064020k
» https://doi.org/10.1021/jp064020k -
53Tsui L, Zangari G. Water content in the anodization electrolyte affects the electrochemical and electronic transport properties of TiO2 nanotubes: a study by electrochemical impedance spectroscopy. Electrochimica Acta 2014;121:203-209. DOI: 10.1016/j.electacta.2013.12.163
» https://doi.org/10.1016/j.electacta.2013.12.163 -
54Kim D, Ghicov A, Schmuki P. TiO2 Nanotube arrays: Elimination of disordered top layers ("nanograss") for improved photoconversion efficiency in dye-sensitized solar cells. Electrochemistry Communications 2008;10(12):1835-1838. DOI: 10.1016/j.elecom.2008.09.029
» https://doi.org/10.1016/j.elecom.2008.09.029 -
55Song YY, Schmidt-Stein F, Bauer S, Schmuki P. Amphiphilic TiO2 Nanotube Arrays: An Actively Controllable Drug Delivery System. Journal of the American Chemical Society 2009;131(12):4230-4232. DOI: 10.1021/ja810130h
» https://doi.org/10.1021/ja810130h -
56Liao Y, Zhang D, Wang Q, Wen T, Jia L, Zhong Z, et al. Open-top TiO2 nanotube arrays with enhanced photovoltaic and photochemical performances via a micromechanical cleavage approach. Journal of Materials Chemistry A 2015;3(27):14279-14283. DOI: 10.1039/C5TA02799C
» https://doi.org/10.1039/C5TA02799C -
57Nakata K, Fujishima A. TiO2 photocatalysis: Design and applications. Journal of Photochemistry and Photobiology C: Photochemistry Reviews 2012;13(3):169-189. DOI: 10.1016/j.jphotochemrev.2012.06.001
» https://doi.org/10.1016/j.jphotochemrev.2012.06.001 -
58Sun Y, Yan KP. Effect of anodization voltage on performance of TiO2 nanotube arrays for hydrogen generation in a two-compartment photoelectrochemical cell. International Journal of Hydrogen Energy 2014;39(22):11368-11375.
Publication Dates
-
Publication in this collection
2018
History
-
Received
30 Aug 2017 -
Reviewed
29 Nov 2017 -
Accepted
18 Jan 2018