Abstract
This trial was conducted to evaluate some beef quality attributes of Nellore, F1 Simmental × Nellore and F1 Angus × Nellore steers finished on feedlot. The effects of feeding regime and genetic group on shear force, thawing losses, cooking (leak + evaporation) losses, total losses and muscle fiber type, as well as carcass pH and temperature during 24 h of chilling were evaluated. There was a genetic group effect on shear force, where the beef from F1 Simmental × Nellore and F1 Angus × Nellore animals had lower values than Nellore animals. Beef of the animals fed the diets with 1% and 2% of body weight on concentrated lost more liquid than the meat of the animals fed at maintenance during thawing and when considering total losses. During cooking there was a difference among the feeding regimes for drip losses which were greater on the animals fed the diet of 1% of body weight on concentrate, followed by the 2% diet and, finally, by the animals fed at maintenance. The muscle of the Nellore steers had larger proportion of intermediate fibers and lower proportion of oxidative fibers than the crossbred animals. The proportion of glycolytic fibers was not influenced by genetic group. The Nellore animals had larger proportion of fibers of fast contraction and smaller proportion of fibers of slow contraction when compared with the crossbred animals. Feeding regime did not influence the proportion of muscular fibers or shear force. Nellore cattle produce tougher beef than crossbred Simmental × Nellore or Angus × Nellore, although all of them have the potential to produce an acceptable beef when slaughtered at young age. Feed restriction up to 90 days is not enough to cause modification on muscle fiber frequencies, then not affecting beef quality.
beef quality; muscular fibers; shear force
RUMINANTS
Beef quality traits of Nellore, F1 Simmental × Nellore and F1 Angus × Nellore steers fed at the maintenance level or ad libitum with two concentrate levels in the diet1 1 Research financed by FAPEMIG.
Ivanna Moraes de Oliveira; Pedro Veiga Rodrigues Paulino; Marcos Inácio Marcondes; Sebastião de Campos Valadares Filho; Jucilene Cavali; Laura Franco Prados; Marcio de Souza Duarte; Edenio Detmann
Universidade Federal de Viçosa
ABSTRACT
This trial was conducted to evaluate some beef quality attributes of Nellore, F1 Simmental × Nellore and F1 Angus × Nellore steers finished on feedlot. The effects of feeding regime and genetic group on shear force, thawing losses, cooking (leak + evaporation) losses, total losses and muscle fiber type, as well as carcass pH and temperature during 24 h of chilling were evaluated. There was a genetic group effect on shear force, where the beef from F1 Simmental × Nellore and F1 Angus × Nellore animals had lower values than Nellore animals. Beef of the animals fed the diets with 1% and 2% of body weight on concentrated lost more liquid than the meat of the animals fed at maintenance during thawing and when considering total losses. During cooking there was a difference among the feeding regimes for drip losses which were greater on the animals fed the diet of 1% of body weight on concentrate, followed by the 2% diet and, finally, by the animals fed at maintenance. The muscle of the Nellore steers had larger proportion of intermediate fibers and lower proportion of oxidative fibers than the crossbred animals. The proportion of glycolytic fibers was not influenced by genetic group. The Nellore animals had larger proportion of fibers of fast contraction and smaller proportion of fibers of slow contraction when compared with the crossbred animals. Feeding regime did not influence the proportion of muscular fibers or shear force. Nellore cattle produce tougher beef than crossbred Simmental × Nellore or Angus × Nellore, although all of them have the potential to produce an acceptable beef when slaughtered at young age. Feed restriction up to 90 days is not enough to cause modification on muscle fiber frequencies, then not affecting beef quality.
Key Words: beef quality, muscular fibers, shear force
Introduction
The preference of beef consumers and the requirements of the international market of beef have been changing the concepts of producers about the production system and type of animals used for meat production.
A major problem faced by the beef production chain in Brazil is the lack of uniformity of factors that affect beef tenderness and palatability such as age of animals, carcass fatness and marbling.
The age of the animal at the slaughter has been suggested as one of the main factors that affect beef tenderness. Therefore, the use of high concentrate diets may be an alternative to improve growth rate in order to reduce the slaughter age of cattle and consequently improve meat quality traits.
The Brazilian cattle herd is composed mainly of pure bred or crossbred Bos indicus animals (Alves et al., 2004). The beef from Bos indicus cattle has been reported as a tough meat, due to reduced activity of micro-calpain on myofibril degradation and high calpatastin activity (Rubensam et al., 1998), which is in turn a specific calpain inhibitor. The high inheritance of Bos indicus genotype associated to advanced aged that usually cattle are slaughtered in Brazil are the main reasons why Brazilian beef has been reported as less tender. However, this fact would be altered by using strategies such as the use of Taurine crossbred cattle, exploring the heterosis and complementarities of those genotypes.
Many factors may determine differences in size and frequency of muscle fibers. The period that cattle are fed may change muscles fiber frequency as well as the energy level of diets (Lehnert et al., 2006). The frequency of muscle fiber differs among muscles as well as among animals, depending on factors such as age, body weight, breed and diet (Klont, 1998). Consequently, the quality of meat may vary according to the frequency of muscle fiber.
Water holding capacity is important for maintenance of meat juiciness and to avoid losses of important nutritional elements (Cheftel et al., 1986). Carcass ultimate pH, temperature, and their decreasing rate determine most of challenges during the post mortem period that is responsible for juiciness and tenderness of meat. In addition, pH values between 5.9 and 6.0 have been considered the limit to classify the meat as DFD (dry, firm and dark) (Wirth, 1987), not being able to be exported (Condão Certificadora, 2007).
In this context, the objective of this study is to evaluate beef quality traits of Nellore, F1 Simmental × Nellore and F1 Angus × Nellore steers finished in feedlot.
Material and Methods
This experiment was conducted in the Laboratório Animal of the Departmento de Zootecnia of Universidade Federal de Viçosa (UFV), in Viçosa-MG, from May to September of 2007. Forty-eight steers with average age of 18 months (16 Nellore (N), 16 F1 Simmental × Nellore (SN), 16 F1 Angus × Nellore (AN)) were used.
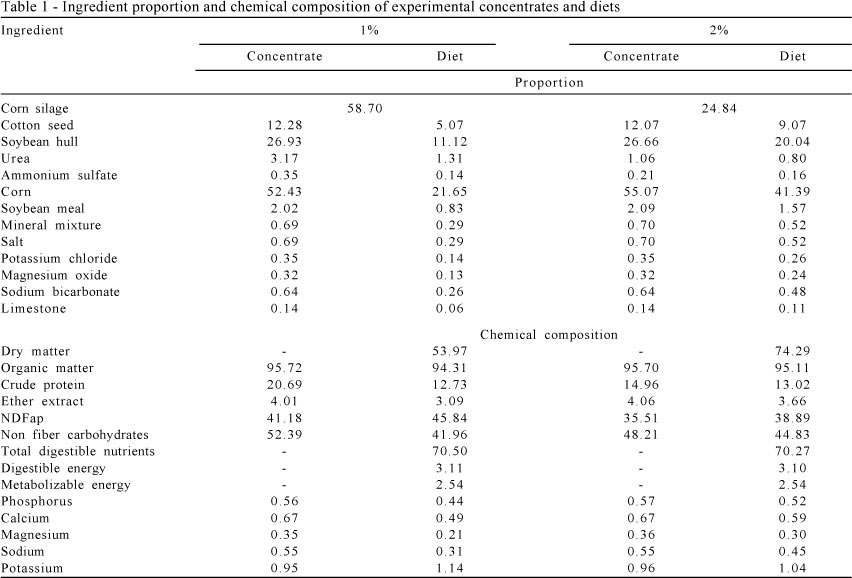
Animals were submitted to a period of 30 days of adaptation to the experiment conditions and 14 days of adaptations to experimental diets prior the beginning of the of 84 days of experiment. During the 30 days of adaptation to experimental conditions, cattle were fed the same diet with the level of concentrate at 1% of BW (Table 1). After the 30 days of adaptation, six animals from each genetic group that were assigned to receive concentrate at levels of 2% of BW were fed concentrate at 1.5% of BW for 7 days and fed concentrate at 2% of BW for 7 days for adaptation to experimental diets. Six animals from each genetic group assigned to receive concentrate at 1% of BW and four animals assigned to be fed at maintenance were fed their treatment diets after the 30 days of adaptation to the experimental conditions.
The experimental period was divided into 3 periods of 28 days. At the beginning of the experiment, cattle were weighed in order to obtain the average initial body weight (BW), which were 265.6±6.4 kg for N, 325.3±4.7 kg for SN and 324.6±6 .0 kg for AN. Cattle were also weighed on days 28 and 56 of the experimental period.
Animals were confined to individual stalls with feeders and drinkers and a total area of 30 m2 of which 8 m2 were covered with asbestos tiles. After analyzing the available ingredients, diets were formulated to be isonitrogenous with 12.5% CP. The animal feed intake was estimated as suggested by Valadares Filho et al. (2006) and the macro and micro minerals requirements were adjusted according to NRC (1996). Concentrates were formulated by using corn meal, soybean meal, cotton seed, soybean hull, urea:ammonium sulphate (9:1) sodium bicarbonate, magnesium oxide, salt and mineral mixture. Corn silage was used as source of roughage. All diets were formulated to contain 20% as the minimum amount of NDF. Through the experiment, the concentrate:roughage ratio was 58.7:41.3 and 24.84:75.16 for diets with concentrate levels at 1% and 2% of BW, respectively.
Diets were fed as total mixed ration (roughage + concentrate) and cattle were fed twice daily (at 6:30 a.m. and 3: 30 p.m.) allowing for up to 5-10% of orts. Animals assigned to be fed at maintenance were fed once daily with the same diet of those fed concentrate at 1% of BW. At the first experimental period cattle fed at maintenance were fed concentrate levels at 1.2% of BW. However, due to a gain of weight observed in those animals, they were fed concentrate levels at 1% of BW after the second experimental period.
At the end of the trial the animals were weighed after a 16-h solid fast and harvested at the slaughter facility of Universidade Federal de Viçosa. Cattle were slaughtered by cerebral concussion followed by jugular and carotid venesection following the Normative Instruction no. 3 of 01/13/2000 (Technical Regulation of Methods for Humane Slaughtering of Livestock).
After the slaughter, each carcass was split into two identical longitudinal halves and kept in cooling chambers at 0 °C for 24 h. After the post mortem chill period, a boneless longissimus dorsi (LD) sample was taken from left half of each carcass in adequate size for each meat quality analyses, packaged and frozen at -20°C for further analysis of Warner-Bratzler shear force (WBSF), thawing loss, cooking loss (evaporative loss + drip loss), and total loss. For muscle fiber analysis, LD samples were packaged and frozen at -80 °C. All samples were previously trimmed in order to remove connective subcutaneous fat tissues.
Muscle fiber analysis was performed at the laboratory of embryology and histology of the Department of Biology of Universidade federal de Viçosa. Frozen samples were processed at -20°C in cryomicrotome Leica® CM 1850TM (Leica Microsystems, Wttzlar, Germany). Once the cut temperature was reached, samples were fixed on sectioning platforms of resin Optimal Critical Temperature Compound - OTC Tissue-Tek® (Sakura, Finetek, Zoeterwoude, The Netherlands).
Serial cuts of 12 µm section were made perpendicularly to LD the muscle fibers. After cutting, sections were placed on glass slides that were kept at room temperature. Glass slides were previously washed and immersed in chromium sulphate gelatin solution, and kept overnight at 37 °C. After cutting section on glass slides, those were kept at -20 °C. Glass slides containing the cut sections were adjusted to a room temperature prior to inking based on sensibility for activity of myofibrillar adenosine-tri-phosphatase (m-ATPase) in exposition to different pH values (adapted from Stevens & Palmer, 1996). Thus, differentiation of muscle fiber types was performed by associating two procedures of histochemical reactions of m-ATPase. After the adjustment to a room temperature, sections were submitted to a pre-incubation at pH = 9.4 for activation of ATPase. After that, half of the samples were incubated at pH = 4.2 in order to separate the low contraction fibers from high contraction fibers (I ¹ IIA+IIB) and pH = 4.7 for glycolytic and intermediate muscle fibers (IIA ¹ IIB+I).
By comparing two glass slides, muscle fibers could be classified as type I (oxidative), IIA (intermediate) or IIB (glycolytic), according to Brooke & Kaiser (1970). After inking, 20 images were captured from each glass slide by using digital camera Olympus® U-CMAD-2 (Olympus Corporation Tokyo Japan) integrated with optic microscope Olympus® BX-60TM (Olympus Corporation, Tokyo, Japan) with 10 X objective and Image-Pro® Plus v.4.5.0.29 software (Media Cybernetics, Maryland, USA). The frequency ratio (%) of fibers was calculated by counting the number of each type of fiber in empty spots of the mask grid, which, in turn, had a total of 65 spots. For each animal 15 images were selected.
Carcass temperature and pH values were recorded every 2 h through the 24 h chill in order to evaluate the decreasing rate of those variables. Both temperature and pH measurements were taken at the LD muscle (12th rib).
The Warner-Bratzler shear force (WBSF) steaks were thawed at 5 ºC for a period of 24 h and oven broiled in an electric oven pre-heated to 150 ºC. Internal steak temperature was monitored with 20-gauge copper-constantan thermocouples placed in the approximate geometric center of each steak and attached to a digital monitor. Steaks were flipped every 15 min and allowed to reach an internal temperature of 71ºC before removal from the oven. Cooked WBSF steaks were cooled for 24 h at 4 ºC. Five round cores (1.27-cm diameter) were removed from each steak parallel to the long axis of the muscle fibers. Each core was sheared once through the center, perpendicularly to fiber direction by a Warner-Bratzler® (G-R Electrical Manufacturing Company, Manhattan - KS, USA).
Steak thawing and cook losses were evaluated on steaks also used for WBSF measurement. For thawing loss evaluation, each steak was weighed when frozen and after a 24 h period of 4 ºC thawing. The cooking loss of each steak was recorded after steaks were oven-broiled during WBSF processing. Total cook loss was calculated as the difference between the weight of the steaks before and after oven-broiling. The total cook loss minus drip loss represented the evaporative loss (Arrigoni et al., 2004). The total liquid loss was calculated by the difference between the weight of frozen and cooked steak.
The experiment was conducted in a completely randomized design in a 3 × 3 factorial arrangement. Treatments consisted of 3 genetic types (Nellore; F1 Simmental × Nellore; F1 Angus × Nellore) and 3 feed regimes (cattle fed at maintenance and ad libitum with concentrate levels at 1 or 2% of BW) with six replicates in each of ad libitum levels and four replicates in the group of animals fed at maintenance.
Muscle fiber frequency, WBSF, and thawing, cooking and total losses were analyzed using SAS version 9.1 (SAS Institute, Inc., 2000). Variables were compared by contrast with significance considered at P<0.05.
Decreasing rates of carcass temperature and pH were obtained by NLIN procedure of SAS and compared by confident intervals at α = 0.05. Coefficients of regression were estimated as the following model:
Y=a*EXP (b*time)
where "a" is the initial pH or temperature and "b" the decreasing rate of pH and temperature. Comparisons between initial pH and temperature and final pH and temperature of carcass were evaluated at α = 0.05, as method described by Roy & Bose (1953).
Results and Discussion
No effects were observed (P>0.05) for feed regime and interactions between genetic type × feeding regime were observed for any of the muscle fibers evaluated (Table 2).
The number of muscle fibers is determined during the intrauterine stage, which is the phase that cells hyperplasia occurs, which can be influenced by the genetic and environmental factors such as the adequate maternal nutrition. However, the phenotype of muscle fibers is defined during the growing phase of the animal since muscle fibers can change their functionality as a response to environmental challenges. The energy and calcium metabolism of muscle fibers during the life of the animal are dependent on certain factors such as physical activity, feeding system and genetic type, which possibly affects the post mortem muscle conversion to meat (Ryu & Kim, 2005). Challenges in muscle fibers as a response to environmental factors occur due to challenges in expression of certain genes such as those that regulates feed intake and energy production, calcium metabolism.
According to Ashmore et al. (1972), LD can be considered as a white muscle which presents greater number of glycolytic fibers, which was observed in the present study (Table 2).
In general, glycolytic fiber has its frequency increased when there an increased availability of energy. Moody et al. (1980) found that different levels of energy in diets (grazing system × feedlot system) has caused physiological challenges among intermediate and white muscle fibers, where animals fed lower level of energy presented greater frequency of intermediate than glycolytic muscle fibers. Nonetheless, in the current study no differences were found (P>0.05) in frequency of intermediate and glycolytic muscle fibers among the feeding regimes (Table 2). In this case, the lack of differences in the frequency of intermediate and glycolytic muscle fibers may have occurred as a result of a low intensity of physical activity as the animals stayed in confinement through the entire experiment.
Lehnert et al. (2006), evaluating the effect of feed restriction on muscle fiber frequency reported that the period of 114 days of feeding was not sufficient to cause challenges in the number of muscle fibers. Thus, it seems that there is a reduction in the size of muscle fibers prior to the challenge in their frequency. Therefore, as in the present study the animals were fed no longer than 84 days, the time of feeding to which animals were submitted was possibly not sufficient to provide challenges in muscle fibers frequency.
Differences were observed (P<0.05) in the proportion of intermediate and oxidative muscle fibers between Nellore and crossbred cattle (Table 3). Nellore animals presented a greater proportion of intermediate muscle fibers (P<0.05) and lower proportion of oxidative muscle fibers (P<0.05) compared with crossbred animals. Regarding glycolytic fibers and color classification of fibers, no differences were observed (P>0.05) between genetic groups. Differences were observed (P<0.05) in the frequency of muscle fibers classified according to their contractible characteristics, where Nellore animals presented greater proportion of fast contraction fibers and lower proportion of low contraction fiber (P<0.05) compared with crossbred animals (Table 3).
The values of oxidative fibers frequency found in this study (approximately 19.8%) were lower than those reported by Wegner et al. (2000) and Mello (2007). According to Johnson et al. (1981) differences in muscle fiber frequency can be found not only among muscles but also in the same muscle, which is in turn affected by the genetic type of the animal.
The frequency of muscle fibers in the same muscle is highly variable (Armstrong & Phelps, 1984). According to Pearson & Young (1989) oxidative muscle fibers usually have high frequency in the interior of the muscle while the glycolytic fiber is usually concentrated in the muscle surface. Thus, even though LD samples to evaluate the frequency of muscle fibers were taken at the same region of the longissimus dorsi, the sub sampling procedures was not standardized, which is a possible reason that would cause differences in the frequency and distribution of oxidative muscle fibers among the genetic groups.
Carcass initial pH values were greater (P<0.05) for the animals from the Nellore and Angus × Nellore groups than those from the Angus × Nellore group. However, no differences were observed (P>0.05) for carcass ultimate pH between the genetic groups, which might be explained by the difference observed (P<0.05) in the decreasing rate of carcass pH observed among those groups (Table 4). Angus × Nellore animals had higher decreasing rates values (P<0.05) than animals from the Simmental × Nellore group. Carcass initial temperature and the decreasing rate of temperature did not differ (P>0.05) between genetic groups. However, greater values (P<0.05) of carcass final temperature was observed in carcass of NE animals compared with Angus × Nellore animals, which presented the lowest values of carcass final temperature among the genetic groups (Table 4).
The decreasing rate of carcass pH is influenced by glycolytic rates, which is in turn influenced by the availability of glycogen in the muscle. Muscle glycogen is converted into lactic acid in the anaerobic environment resulting in the decreasing of carcass pH during the post mortem period. However, post mortem metabolism is affected by muscle fiber frequency. In general, the glycolysis is faster in white than red muscles. According to Ryu & Kim (2005) muscles that present high proportion of glycolytic fibers have high carcass pH decreasing rate, while muscles with high frequency of oxidative and intermediate fibers present a slower decreasing rate of carcass pH.
The greater temperature values observed in carcass of Nellore animals may be related to their position in the cooling chambers. Since the Nellore group was the last group of animals slaughter at the end of the trial, their carcass stayed close to the doors of cooling chamber. Thus, since the cooling chamber was frequently open due to the measurement of carcass pH and temperature, the carcass of Nellore animals were not able to reach the same final temperature as the carcass of the other genetic groups at the end of the 24-h chill.
Glycolysis is highly affected by temperature. Muscle contractions occur when the carcass is exposed to low temperatures, reducing the levels of glycogen and consequently increasing the level of lactic acid in the muscle tissue (Roça, 2009). Degradation of glycogen occurring during the post mortem period associated with high temperatures can increase muscle temperature. Consequently, metabolic reactions such as ATP hydrolysis and glycolysis are catalyzed. Thus, the lack of effects on carcass ultimate pH among animals fed different concentrate levels is possibly related to the similarly variation of decreasing rate of temperature of carcass.
Carcass initial pH observed in this study was slightly lower that commonly observed for beef cattle carcass pH after slaughter (6.9 - 7.2). This fact possibly occurred due to pre-slaughter stress, which led to a decrease in glycogen concentration and increase of acid lactic levels, and consequently reduced the final pH of the carcass.
Carcass from all animals presented ultimate pH between 5.4 - 5.6. Meat from carcass that presents ultimate pH values greater than 6.0 cannot be exported, since there is an association between carcass ultimate pH and stressful pre-slaughter handling, which was not observed in this trial. Nevertheless, a mean value of 7±1 °C was found for carcass final temperature, which is desirable after a 24-h post mortem chill. It should be noted that the pH measurements through the post mortem period affected the final temperature of carcass, as the chill chamber was opened several times through the 24 h of chill. Thus, it can be inferred that new methods of pH measurements are needed in order to minimize the chilling rate of the carcass.
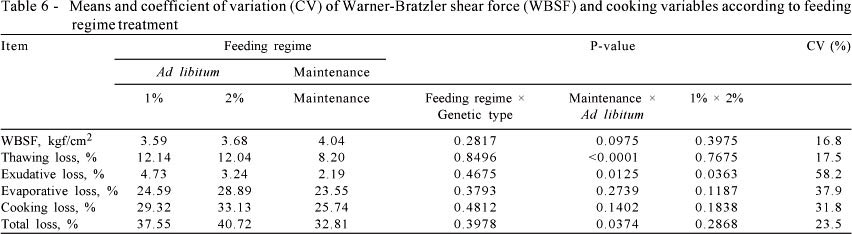
Differences were found (P<0.05) for WBSF among genetic groups where beef from Simmental × Nellore and Angus × Nellore animals presented lower values of WBSF than those observed in beef from Nellore animals (Table 5). These data suggest that beef from crossbred animals can be classified as more tender than beef from Bos indicus cattle. Beef from Nellore animals had greater values (P<0.05) of thawing, cooking, evaporative and total loss compared with crossbred animals (Table 5).
According to Shackelford et al. (1994), approximately 65% of variation on beef tenderness is related to genetic factors and only 35% due to environmental factors. Morgan et al. (1991) reported that beef tenderness is lower in areas where Bos indicus are the main type of cattle used in beef cattle systems, which is commonly seen in Brazil. Several studies have indicated that beef from Bos indicus presents less tenderness due to lower levels of intramuscular fat and greater amount of connective tissue when compared with beef from Bos taurus cattle (Bailey, 1985; Belew et al., 2003).
In addition, Bonilha et al. (2008) reported that beef from Bos indicus cattle has lower levels of micro-calpain and greater levels of calpatastin, which are the main enzymes that are related to tenderization of meat (Koohmaraie, 1994). It should be noted that the WBSF values found classifies beef as tender, even for beef from Nellore animals. According to Shackelford et al. (1997) those values should be lower than 6.0 kgf/cm2 or lower than 4.5 kgf/cm2 (Johnson et al. 1988; Knapp et al., 1989). Nellore animals used in this trial were from the same contemporary group, originally from the same farm and presented the same subcutaneous fat thickness which might explain the low values of WBSF observed.
Water hold capacity can be increased by different factors such as low rate of post mortem glycolysis, high carcass ultimate pH and inadequate decreasing rate of carcass temperature. In addition, muscles that present high levels of intramuscular fat may present greater water hold capacity, as the intramuscular fat may distend the micro-structure of the muscles allowing a better water hold capacity.
In the present study, the values of carcass ultimate pH were above 5.2 - 5.3, the limit value where water hold capacity is reduced (Lawrie, 1998), which corresponds to the isoelectric point of most of myofibrillar proteins. Therefore, carcass ultimate pH is not a reason that would explain the greater liquid loss of beef from Nellore animals.
Muscular tissue has higher capacity to retain water than fat tissue; the protein molecules have high attractiveness to water molecules. Thus, since Nellore cattle had lower amount of fat in carcass compared with crossbred animals (Table 6), beef from Nellore cattle were more vulnerable to liquid loss than beef from crossbred animals.
Most of the water found in the muscle is retained by myofibrillar protein and only 3% of the total of water in muscles is retained by the sarcoplasmic proteins. The amount of water retained depends on the space available between the muscle fibers. During the rigor mortis establishment there is a sarcomere shortening that reduces the space available within the muscle cell. According to Honikel et al. (1986), the thawing loss can be linearly increased with a reduction of the sarcomere length. In this case, the water is mobilized from the intra-myofibrillar to extra-myofibrillar space, and the liquid loss increases. Thus, the greater values of thawing loss from beef of Nellore animals possibly occurred due to a sarcomere shortening, as these animals had lower back fat thickness (3.81 mm) compared with crossbred animals (5.5 mm).
Feed regime did not affect (P>0.05) the WBSF, cooking or evaporative loss of LD steaks (Table 6). Beef from cattle fed 1 and 2% concentrate had greater values (P<0.05) of thawing and total loss than beef from cattle fed at maintenance (Table 6). At cooking, differences were observed (P<0.05) for exudative loss among feeding regime treatments, where animals fed concentrate at 1% of BW presented a greater values of exudative loss than cattle fed concentrate at 2% of BW and at maintenance, respectively (Table 6).
According to Koohmaraie (1992), 85% of differences in meat tenderness occurs due to post mortem challenges, and 15% due to ante mortem differences. The main ante mortem factors are genetic type, age of the animals, carcass fat thickness, growth rate, collagen composition, among others. On the order hand, the main post mortem factors that affect meat tenderness are carcass chilling rate, pH decreasing rate, ultimate pH, proteolytic activity and aging time.
Aberle et al. (1981) suggested that the increased growth rates of animals associated to an increased protein turn over would allow obtaining a tender meat. In addition, Crouse et al. (1986) reported that greater weight gain rate allows the animal to reach the maximum muscular growth in a short period of time presenting greater solubility of collagen, as there is a lower number of cross-links in collagen molecules of animals that have high weight gain rate (Harper, 1999). The intermolecular cross-links are associated with substantial increases in stiffness and insolubility of collagen and, subsequently, a reduction in tenderness of meat. The lack of differences between animals fed concentrate at 1% and 2% of BW can be explained by the similar growth rate among those treatments (1.24 kg/d and 1.32 kg/d for animals fed concentrate at 1% and 2%. respectively).
At normal conditions, the muscle fiber diameter increases in animals fed diets with levels of energy that allows an adequate growth (Pardi et al. 1995). Based on muscle tissue growth, it was expected that cattle fed at maintenance would present a tender beef, as the lack of growth due to the restriction of energy intake would provide a high frequency of muscle fibers with low diameter and consequently reduce the shear-force of beef.
However, animals fed diets with lower energy level during the finishing period prior to harvest present lower collagen solubility as there is a lower turnover rate. Fishell et al. (1985) reported that steers with low growth rate had lower collagen solubility of Semimembranosus muscle and that the concentration of intramuscular collagen was greater when animals had feed restriction. Based on those statements, it can be inferred that the diameter of the muscle fiber and the collagen solubility would equate the beef tenderness of animals fed at maintenance, resulting in similar values of WBSF between cattle fed at maintenance and ad libitum.
Animals fed at maintenance presented low carcass fat thickness, which may have caused a sudden drop of carcass temperature before the establishment of the rigor mortis, and consequently caused the cold shortening. In addition, the feed restriction of animals fed at maintenance would lead to a low level of muscular glycogen causing an inadequate drop of carcass pH. This would explain the lower liquid loss observed in beef from animals fed at maintenance compared with those fed ad libitum (Table 6).
Conclusions
The data indicates that Nellore purebred cattle has less tender beef than Nellore × Simmental and Nellore × Angus crossbred cattle. However, both Nellore purebred and crossbred cattle are capable of producing beef with acceptable tenderness when slaughtered at a young age. With regard to feed regime, 90 days of feed restriction is not enough to cause challenges in muscle fibers frequency not affecting beef quality.
Received June 15, 2009 and accepted July 28, 2011.
Corresponding author: imoraesdeoliveira@yahoo.com.br
- ABERLE, E.D.; REEVES, M.D. Palatability and muscle characteristics of cattle with controlled weight gain: Time on a high-energy diet. Journal of Animal Science, v.52, n.4, p.757-764, 1981.
- ALVES, D.D.; PAULINO, M.F.; BACKES, A.A. et al. Características de carcaça de bovinos Zebu e cruzados Holandês-Zebu (F1) nas fases de recria e terminação. Revista Brasileira de Zootecnia, v.33, n.5, p.1274-1284, 2004.
- ARMSTRONG, R.B.; PHELPS, R.D. Muscle fiber type composition of rat hindlimb. American Journal of Anatomy, v.171, n.2, p.259-272, 1984.
- ARRIGONI, M.B.; ALVES JÚNIOR, A.; DIAS, P.M.A. et al. Desempenho. fibras musculares e carne de bovinos jovens de três grupos genéticos. Pesquisa Agropecuária Brasileira, v.39, n.10, p.1033-1039, 2004.
- ASHMORE, C. R.; THOMPKINS, G.; DOERR, L. Postnatal development of muscle fiber types in domestic animals. Journal of Animal Science, v.34, n. 1, p.37-41, 1972.
- BAILEY,A.J. The role of collagen in the development of muscle and relationship to eating quality. Journal of Animal Science v.60. n.6, p.1580-1587, 1985.
- BELEW, J.B.; BROOKS, J.C.; MCKENNA, D.R. et al. Warner-Bratzler shear evaluations of 40 bovine muscles. Meat Science, v.64, n.4, p.507-512, 2003.
- BONILHA, S.F.M.; TEDESCHI, L.O.. PACKER, I.U. et al. Evaluation of carcass characteristics of Bos indicus and tropically adapted Bos taurus breeds selected for postweaning weight. Journal of Animal Science, v.86, n.8, p.1770-1780, 2008.
- BROOKE, M.H.; KAISER, K.K. Three 'myosin adenosine triphosphatase' systems: the nature of their pH lability and sulphydryl dependence. Journal of Histochemistry and Cytochemistry, v.18, p.670-672, 1970.
- CHEFTEL, J.C.; CUQ, J.L.; LORIENT, D. Proteínas alimentarias Zaragoza: Acribia, 1986. 346p.
- CONDÃO CERTIFICADORA [2007]. Influência do transporte sobre a qualidade da carne produzida Available at: <http://www.condaocertificadora.com.br/noticias.aspx?not=226>. Accessed on: Jan. 10, 2009.
- CROUSE, J.D.; CALKINS, C.R.; SEIDEMAN, S.C. The effects of rate of change in body weight I tissue development and meat quality of youthfull bulls. Journal of Animal Science, v.63, n.6, p.1824-1829, 1986.
- FISHELL, V.K.; ABERLE, E.D.; JUDGE, M.D. et al. Palatability and muscle properties of beef as influenced by preslaughter growth rate. Journal of Animal Science, v.61, n.1, p.151-157, 1985.
- HARPER, G.S. Trends in skeletal muscle biology and the understanding of toughness in beef. Australian Journal of Agricultural Research, v.50, n.7, p.1105-1129, 1999.
- HONIKEL, K.O.; KIM, C.J.; HAMM, R. et al. Sarcomere shortening of prerigor muscles and its influence on drip loss. Meat Science, v.16, n.4, p.267-282, 1986.
- JOHNSON, D.M.; MOODY, W.G.; BOLING, J.A. et al. Influence of breed type. sex. feeding systems. and muscle bundle size on bovine fiber type characteristics. Journal of Food Science, v.46, n.6, p.1760-1765, 1981.
- JOHNSON, D.D.; LUNT, D.K.; SAVELL, J.W. et al. Factors affecting carcass characteristics and palatability of young bulls. Journal of Animal Science, v.66, n.10, p.2568-77, 1988.
- KLONT, R.E.; BROCKS, L.; EIKELENBOOM, G. Muscle fibre type and meat quality. Meat Science, v.49, n.1, p.219-229, 1998.
- KNAPP, R.H.; TERRY, C.A.; SAVELL, J.W. et al. Characterization of cattle types to meet specific beef targets. Journal of Animal Science, v.67, n.9, p.2294-308, 1989.
- KOOHMARAIE, M. Role of the neutral proteinases in postmortem muscle protein degradation and meat tenderness. In: RECIPROCAL MEAT CONFERENCE, 45., 1992, Knoxville. Proceedings... Knoxville: American Meat Science Association, 1992. p.63-71.
- KOOHMARAIE, M. Muscle proteinases and meat aging. Meat Science, v.36, n.1-2, p.93-104, 1994.
- LAWRIE, R.A. Constituição química e bioquímica do músculo. In: LAWRIE, R.A. (Ed.) Ciência da carne São Paulo: Artmed, 1998. p.79-119.
- LEHNERT, S.A.; BYRNE, K.A.; REVERTER, A. et al. Gene expression profiling of bovine skeletal muscle in response to and during recovery from chronic and severe undernutrition. Journal of Animal Science, v.84, n.12, p.3239-3250, 2006.
- MELLO, R.O. Eficiência produtiva e econômica, características da carcaça e qualidade da carne de bovinos mestiços confinados e abatidos com diferentes pesos corporais 2007. 160f. Dissertação (Doutorado em Zootecnia) Universidade Federal de Viçosa, Viçosa, MG.
- MOODY, W.G.; KEMP, J.D.; MAHYUDDIN, M. et al. Effect of feeding systems. slaughter weight and sex on histological properties of lamb carcasses. Journal of Animal Science, v.50, n.2, p.249-256, 1980.
- MORGAN, J.B.; SAVELL, J.W.; HALE, D.S. et al. National beef tenderness survey. Journal of Animal Science, v.69, n.8, p.3274-3283, 1991.
- NATIONAL RESEARCH COUNCIL - NRC. Nutrient requirements of beef cattle 7.ed. Washington, D.C.: National Academic Press, 1996. 242p.
- PARDI, M.C.; SANTOS, I.F.; SOUZA, E.R. et al. Ciência, higiene e tecnologia da carne Goiânia: UFG, 1995. 584p.
- PEARSON, A.M.; YOUNG, R.B. Skeletal muscle fibers. In: PEARSON, A.H.; YOUNG, R.B. (Eds.) Muscle and meat biochemistry San Diego: Academic Press, 1989. p.235-265.
- ROÇA, A.O. [2009]. Modificações post mortem. Available at: <http://dgta.fca.unesp.br/docentes/roca/carnes/Roca105.pdf> Accessed on: Jan. 10, 2009.
- ROY, S.N.; BOSE, R.C. Simultaneous confidence interval estimation. Annals of Mathematical Statistics, v.24. p.513-36, 1953.
- RUBENSAM, J.M.; FELÍCIO, P.E.; TERMIGNONI, C. Influência do genótipo Bos indicus na atividade de calpastatina e na textura da carne de novilhos abatidos no sul do Brasil. Ciência e Tecnologia de Alimentos, v.18, n.4, p.405-409, 1998.
- RYU, Y.C.; KIM, B.C. The relationship between muscle fiber characteristics. postmortem metabolic rate. and meat quality of pig longissimus dorsi muscle. Meat Science, v.71, n.2, p.351-357, 2005.
- SHACKELFORD, S.D.; WHEELER, T.L.; KOOHMARAIE, M. Tenderness classification of beef: I. Evaluation of beef longissimus shear force at 1 or 2 days postmortem as a predictor of aged beef tenderness. Journal of Animal Science, v.75, n.9, p.2417-2422, 1997.
- SHACKELFORD, S.D.; KOOHMARAIE, M.; CUNDIFF, L.V. et al. Heritabilities and phenotypic and genetic correlations for bovine post rigor calapstatin activity. intramuscular fat content. Warner-Bratzler shear force. retail product yield and growth rate. Journal of Animal Science, v.72, n.4, p.857-863, 1994.
- STATISTICAL ANALYSIS SYSTEM - SAS. SAS/STAT User's guide v.8.0, v. I. Cary: SAS Institute, 2000. (CD-ROM).
- STEVENS, A.; PALMER, J. Enzyme histochemistry: diagnostic applications. In: BANCROFT, J.D.; STEVENS, A. (Eds.) Theory and practice of histological techniques 4.ed. New York: Churchill Livingstone, 1996. 766p.
- VALADARES FILHO, S.C.; PAULINO, P.V.R.; MAGALHÃES, K.A. Exigências nutricionais de zebuínos e tabelas de composição de alimentos - BR CORTE Viçosa, MG: UFV, Suprema Gráfica Ltda, 2006. 142p.
- WEGNER, J.; ALBRECHT, E.; FIEDLER, I. et al. Growth- and breed-related changes of muscle fiber characteristics in cattle. Journal of Animal Science, v.78, n.6, p.1485-1496, 2000.
- WIRTH, F. El pH y la elaboración de productos cárnicos. Fleischwirtschaft, v.2, p.24-33, 1987.
Publication Dates
-
Publication in this collection
19 Jan 2012 -
Date of issue
Dec 2011
History
-
Received
15 June 2009 -
Accepted
28 July 2011