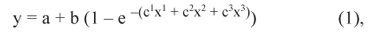Abstract
The objective of this trial was to evaluate the bioavailability of DL-2-hydroxy-4-(methyl) butanoic acid (DL-HMBA) and a polyherbal ingredient (PHI) in relation to DL-methionine (DLM) on broilers. Nine hundred male broiler chickens of the Cobb 500 strain were fed from 22 to 42 days of age either a basal diet without industrial methionine supplementation or the basal diet supplemented with DL-HMBA at one of three levels (0.143, 0.286 and 0.429%) or DLM at one of three levels (0.093, 0.186 and 0.279%, each of which is 65% of the respective DL-HMBA level by weight) or PHI at one of the same three levels used for DLM (0.093, 0.186 and 0.279%). The weight gain, feed conversion ratio and relative weights of breast and abdominal fat were improved over that of basal diet-fed broilers by the addition of DL-HMBA and DLM to the diet. A simultaneous exponential regression analysis revealed that the relative bioavailability values for DL-HMBA and PHI were 52% and 5% of that of DLM, respectively, for weight gain, and 57% and 4%, respectively, for feed conversion ratio. Concerning breast meat yield, a simultaneous linear regression analysis (slope ratio) showed that the relative bioavailability for DL-HMBA was 65% of that of DLM. Considering all studied parameters together, the relative bioavailability values for DL-HMBA and PHI are 58% and 4.5% of that of DLM on a product basis.
dl-2-hydroxy-4-methylthio butanoic acid; dl-methionine; polyherbal ingredient; sulphur amino acids
NON-RUMINANTS
Bioavailability of different methionine sources for growing broilers
Cleiton Pagliari SangaliI; Luís Daniel Giusti BrunoI; Ricardo Vianna NunesI; Adhemar Rodrigues de Oliveira NetoII; Paulo Cesar PozzaIII; Taciana Maria Moraes de OliveiraI; Rafael FrankI; Rodrigo André SchöneI
IUniversidade Estadual do Oeste do Paraná, Marechal Cândido Rondon, PR, Brasil
IIEvonik Industries, São Paulo, SP, Brasil
IIIUniversidade Estadual de Maringá, Maringá, PR, Brasil
ABSTRACT
The objective of this trial was to evaluate the bioavailability of DL-2-hydroxy-4-(methyl) butanoic acid (DL-HMBA) and a polyherbal ingredient (PHI) in relation to DL-methionine (DLM) on broilers. Nine hundred male broiler chickens of the Cobb 500 strain were fed from 22 to 42 days of age either a basal diet without industrial methionine supplementation or the basal diet supplemented with DL-HMBA at one of three levels (0.143, 0.286 and 0.429%) or DLM at one of three levels (0.093, 0.186 and 0.279%, each of which is 65% of the respective DL-HMBA level by weight) or PHI at one of the same three levels used for DLM (0.093, 0.186 and 0.279%). The weight gain, feed conversion ratio and relative weights of breast and abdominal fat were improved over that of basal diet-fed broilers by the addition of DL-HMBA and DLM to the diet. A simultaneous exponential regression analysis revealed that the relative bioavailability values for DL-HMBA and PHI were 52% and 5% of that of DLM, respectively, for weight gain, and 57% and 4%, respectively, for feed conversion ratio. Concerning breast meat yield, a simultaneous linear regression analysis (slope ratio) showed that the relative bioavailability for DL-HMBA was 65% of that of DLM. Considering all studied parameters together, the relative bioavailability values for DL-HMBA and PHI are 58% and 4.5% of that of DLM on a product basis.
Key Words: dl-2-hydroxy-4-methylthio butanoic acid, dl-methionine, polyherbal ingredient, sulphur amino acids
Introduction
Broilers diets are usually based on corn and soybean meal; in most cases, animal by-products are also used as raw material. Considering that the main source of crude protein in broiler diets is soybean meal, the first limiting amino acid for broilers is methionine, which has to be supplemented in the feed from industrial sources available on the market (Lemme et al., 2002).
The most common sources of methionine in the supplements available to the poultry industry are DL-methionine (DLM) and DL-2-hydroxy-4-(methyl) butanoic acid (DL-HMBA). Recently, however, some natural sources of this amino acid have been launched, including polyherbal ingredient - PHI (Narayanswamy and Bhagwat, 2010). According to the manufacturers of these products, DLM is a pure powder product with over 99% purity, while DL-HMBA is liquid and has 88% active compounds. In addition, there is powdered PHI, which contains the dipeptides and oligopeptides of methionine, as well as the precursors and intermediates of this amino acid.
In addition to these physical differences, there are also known biochemical and metabolic differences among the methionine sources, which may contribute to the differences in intestinal absorption in birds (Maenz and Engele-Schaan, 1996ab; Drew et al., 2003). Thus, it is crucial to estimate the real methionine values given to DL-HMBA and PHI compared with DLM, since this is an important prerequisite for deciding which source of methionine should be used, considering also the costs of ingredients used in diet formulation and animal production (Brugalli, 2003).
There has already been a lot of discussion concerning the relative bioavailability of methionine from different sources, as well as a considerable number of experiments comparing various sources, mainly DL-HMBA and DLM, conducted on birds (Esteve-Garcia and Llaurado, 1997, Hoehler et al., 2005a; Elwert et al., 2008). Furthermore, the correct statistical design to evaluate the bioavailability of different sources of essential nutrients has been under debate (Littell et al., 1997; Kratzer and Littell, 2006; Piepho, 2006). Standardised method of statistical analysis would make comparisons of experiments assessing different sources of nutrients easier and more accurate (Littell et al., 1997).
Thus, the objective of this study was to evaluate the relative bioavailability of DL-HMBA and PHI compared with DLM in diets for male broilers from 22 to 42 days of age.
Material and Methods
The experiment was conducted at the Poultry Sector of the Center of Experimental Stations of Universidade Estadual do Oeste do Paraná - UNIOESTE, campus of Marechal Cândido Rondon, Paraná, Brazil.
All animal procedures were approved by the Institutional Animal Care and Use Committee.
A total of nine hundred male broilers with an average initial weight of 887 ± 2.8 g were used from 22 to 42 days of age. Birds were subdivided into 50 pens, each measuring 1.00 × 1.35 m. The experimental design was completely randomised in a 3 × 3 factorial arrangement (three sources of methionine × three supplementation levels) and an additional treatment without supplemental methionine (basal diet), with five replications and 18 birds per experimental unit, for a total of 13.33 birds/m2.
All of the diets were in mashed form and were based on corn, soybean meal, meat-and-bone meal and offal (Table 1). The basal diets contained starch, which was replaced with crystalline amino acids to formulate the different treatment diets. An analysis of the amino acid and protein contents of the experimental diets (Llames and Fontaine, 1994; Fontaine et al., 1998) confirmed the calculated values. Thus, these values were used in further statistical analysis.
Three increasing DLM (0.093, 0.186 and 0.279%), DL-HMBA (0.143, 0.286 and 0.429%) and PHI (0.093, 0.186 and 0.279%) levels were added to the basal diet in order to achieve 33, 66 and 100% of utilized methionine levels by the poultry integrator company. Three DLM levels were added, always considering 65% of the DL-HMBA amount, thus, a 65:100 ratio (product basis) was kept. For PHI, three increasing levels were used in order to replace DLM, always one-to-one (product basis), so a 100:100 ratio was kept.
The elaboration processes applied to produce all of the diets used in this study was to produce firstly the basal diets (without any methionine sources) and also the highest level for the methionine source DLM, DL-HMBA and PHI (Table 1). All the intermediate levels for every source were produced by mixing the basal diet with the highest level of every methionine source, which was done in different percentages in order to achieve the methionine level desired. The treatment 2 of DLM was produced by mixing 66% of treatment 1 (basal diet) with 33% of treatment 4 (highest level of DLM), and the treatment 3 of DLM was achieved by adding 33% of basal diet to 66% of treatment 4. The same procedure was done for every source studied.
Diet and water were provided ad libitum during the entire experimental period; to achieve this, tubular feeders and water nipple drinkers were used. Maximum and minimum temperatures were measured with thermometers and recorded daily.
The evaluated parameters were feed intake, weight gain, feed conversion ratio, and carcass parameters such as breast, thighs, drumsticks and wing yield. Liver and abdominal fat relative weights were also measured. For performance, final weight was based on the average weight of a bird pen (18 birds), while to assess the carcass, various cuts, the liver and abdominal fat, two birds per replicate were used, totalling 10 broilers per treatment.
The relative bioavailability values of DL-HMBA and PHI in relation to DLM, for variables of performance and carcass characteristics, were determined by simultaneous exponential regression model (for non-linear responses) according to Equation 2 or by simultaneous linear regression design or slope-ratio, according to Equation 3, as suggested by Littell et al. (1997), using the general linear design procedure (GLM) of SAS computer program (Statistical Analysis System, version 6.11).
in which y = performance criterion; a = performance obtained with the basal diet (y-intercept); b = asymptotic response (difference between 'a' and asymptote); a + b = common asymptote (maximum performance level); c1, c2 and c3 = slope coefficients to DLM, DL-HMBA and PHI, respectively; and x1, x2 and x3 = dietary levels of DLM, DL-HMBA and PHI, respectively.
in which y = performance criterion; a = performance obtained with the basal diet (y-intercept); b1, b2 and b3 = linear regression coefficients for DLM, DL-HMBA and PHI, respectively; and x1, x2 and x3 = dietary levels of DLM, DL-HMBA and PHI, respectively. According to Littell et al. (1997), the bioavailability values of DL-HMBA and PHI in relation to DLM are provided by the regression coefficient ratios: c2/c1 and c3/c1 with multi-exponential regression, and b2/b1 and b3/b1 using multi-linear regression.
Results and Discussion
Along the experimental period, the average minimum and maximum temperatures were 26.4 and 19.6 ºC, respectively. Oliveira et al. (2006) indicate 25 ºC as animal comfort temperature between 1 and 49 days, and the air temperature of 35 ºC as stressful for broilers at this age.
Broiler performance was improved over that of basal diet-fed broilers by the addition of DL-HMBA and DLM to the diet (Table 2). This suggests that the aim of the basal diet, as identified by Lemme et al. (2002), namely, the detection of any differences in bioavailability of dietary methionine source or deficiency in methionine + cystine, was achieved. Birds fed the basal diet achieved only 1,657 g of weight gain and 2.03 g/g feed conversion ratio. The best broiler performance was associated with the largest amount of either DLM or DL-HMBA supplementation, in which case the broilers achieved 1,998 g and 1.67 g/g: a 20.6% improvement in weight gain and an 18.0% improvement in feed conversion ratio. These results are in accordance with Payne et al. (2006), who have also noted improvements in the performance of broilers supplemented with increasing levels of either DL-HMBA or DLM, in which the supplemented quantity of DLM was always 65% of the DL-HMBA (product basis).
Birds supplemented with PHI, which was provided in the same quantities as DLM, showed the worst performance among the supplemented groups. Among these birds, the best weight gain was 1,746 g and the best feed conversion ratio was 1.98 g/g (Table 2), not differing from the basal diet.
Regarding carcass parameters (Table 3), the birds fed the basal diet showed relative weights of 35.18 and 2.67% for breast and abdominal fat, respectively. The best values for each of these variables were associated with the larger amount of either DLM or DL-HMBA supplementation, which yielded improvements of 9.7, 3.9 and 32.1% for breast and abdominal fat, respectively. Birds supplemented with PHI, on the other hand, showed no improvement in the carcass parameters evaluated. No improvement was found in the relative weights of carcass, thigh and drumstick, wing and liver of the birds supplemented with DLM or DL-HMBA either.
The low performance for birds fed the basal diet or supplemented with PHI is probably related to the deficiency of amino acids in their diets (Albino et al., 1999), specifically methionine deficiency; this indicates that PHI as a methionine source did not meet the sulphur amino acid requirements. It is noteworthy that broilers fed diets deficient in methionine + cystine exhibit lower levels of protein synthesis. The result is an inefficient use of the other amino acids, which can be observed in this study in birds fed the basal diet or supplemented with PHI. The amino acid imbalance caused by methionine + cystine deficiency can also be validated by the fact that the highest mean values of abdominal fat (Table 3) were observed in birds either fed the basal diet (2.67%) or supplemented with PHI (2.98%). According to Silva et al. (2005), this imbalance limits the accumulation of lean tissue and drives calories to adipocytes. This probably occurred in the present study.
The response of bird performance to increasing levels of the effective methionine sources starting from the methionine + cystine-deficient basal diet followed a nonlinear trend corresponding to the law of decreasing yields (Figures 1, 2 and 3). In this pattern, animal performance increases as the quantity of nutrients increases until no additional unit of nutrients yields an increase in animal performance (Timmler and Rodehutscord, 2003). According to Littell et al. (1997), the most accurate way to describe the performance response presented by the broilers in the present study to the increasing levels of methionine sources would be a simultaneous regression design. This design provides a way to determine unbiased estimates of bioavailability among the tested substances (DL-HMBA and PHI) and the reference substance (DLM).
The use of simultaneous regression to determine the bioavailability of dietary methionine sources has been further highlighted by recent studies (Hoehler et al., 2005b; Piepho, 2006; Hoehler et al., 2006; Elwert et al., 2008).
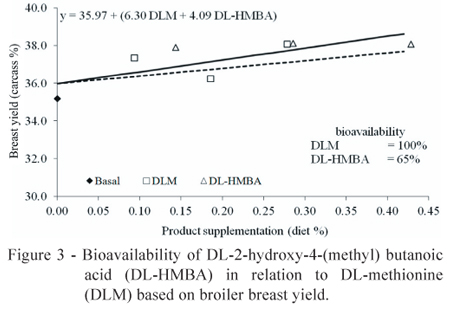
Our simultaneous regression analysis of the current trial data conclusively showed that the bioavailability of DL-HMBA is only 52% of that of DLM for weight gain (Figure 1), 57% of that of DLM for feed conversion ratio (Figure 2) and 65% of that of DLM for breast yield (Figure 3) on a product basis. A similar relation between the bioavailability of DL-HMBA and that of DLM based on breast yield results (64%) was found by Lemme et al. (2002) using a simultaneous exponential regression with mean values, although that study found that the relative bioavailability of DL-HMBA was actually higher than that of DLM for weight gain (68%) and feed conversion ratio (67%). A study by Payne et al. (2006) calculated the bioavailability of DL-HMBA as 64% of that of DLM for weight gain and 59% for feed conversion ratio.
The simultaneous regression design was also used to determine the relative bioavailability of PHI compared with that of DLM, which we estimated at 5% for weight gain and 4% for feed conversion ratio (Figures 1 and 2), which represents an average PHI bioavailability of 4.5% compared with that of DLM.
The relative weights of the carcass, legs, liver and abdominal fat of birds supplemented with DL-HMBA and PHI as well as the breast yield data of birds supplemented with PHI were also analyzed by simultaneous regression. Nevertheless, there was no significant data adjustment of those parameters in the GLM procedure on SAS software. Thus it was not possible to determine DL-HMBA and PHI bioavailability in relation to DLM for those variables.
Combining the bioavailability values for weight gain, feed conversion ratio and breast yield, the average bioavailability of DL-HMBA was 58% of that of DLM. Although this value is lower than that found in some other studies (Lemme et al., 2002; Jansman et al., 2003; Hoehler et al., 2005b; Sauer et al. 2008), it agrees with the results obtained by Payne et al. (2006), in which the average bioavailability of DL-HMBA in relation to that of DLM was 57%.
There are several possible reasons for the lower bioavailability of DL-HMBA in relation to DLM. Physical and chemical differences between DLM and DL-HMBA can be directly related to the efficiency with which each substance is used by birds as a source of methionine. DL-methionine is a pure product with 99% activity of methionine, while DL-HMBA is composed of 12% water and only 88% active compounds (Boebel and Baker, 1982). Of these 88%, 65% are in monomeric form and the remaining 23% consist of dimers and oligomers (Boebel and Baker, 1982). Some authors (Van Weerden et al., 1992) suggest that DL-HMBA polymers have a lower bioavailability than pure DL-HMBA does. Pure DL-HMBA has a 66% relative bioavailability, while polymers have a 49% bioavailability when compared with DLM on a product basis (Van Weerden et al., 1992). Hasseberg (2002) and Mitchell and Lemme (2008) have also suggested that the use of DL-HMBA in less efficient polymeric forms is a major reason for its lower bioavailability in relation to that of DLM.
Another potential reason for the lower DL-HMBA bioavailability is that both D and L-isomers should receive NH3+ from an amino acid and be transformed into L-methionine (Hasseberg, 2002; Rombola et al., 2008). In practice, however, only the D-isomers of DLM must be transformed to be used in the active substance (L-methionine), mainly to build muscles, but also to build other protein components. It is important to note that, before any conversion of L-isomers, methionine sources have to be absorbed as well. The body has specific mechanisms responsible for nutrient absorption, and there seems to be some variation in the effectiveness of these mechanisms at absorbing methionine from different sources. Maenz and Engele-Schaan (1996a) have already reported that different mechanisms of active transport are involved in the uptake of methionine (Na+ dependent) and methionine analogues (H+ dependent). These authors reported that affinity with the carrier and maximum speed of transport were higher for methionine than for methionine analogues (DL-HMBA). Therefore, differences between methionine sources in the transport mechanism used can lead to differences in the quantity that is transported.
There are also several studies suggesting a possible interaction between DL-HMBA molecules and the gastrointestinal tract. Maenz and Engele-Schaan (1996a) reported that a significant quantity of DL-HMBA was converted into non-absorbable by-products during its passage through the gastrointestinal tract. Furthermore, Lingens and Molnar (1996) have indicated that methionine absorption rates into broilers bodies were significantly higher when it was given as DLM than when it was given as DL-HMBA. This means that there is a definite relationship between the availability of dietary methionine sources and methionine absorption into muscle tissue. Even though these scientific studies have suggested that DL-HMBA absorption is reduced, there has been no clear explanation for this phenomenon. However, the interaction between DL-HMBA and the gastrointestinal tract was very clear in the study by Drew et al. (2003). The authors reported that over 10% of 3H-DL-HMBA activity remained in the distal ileum of conventional chickens, while only 4.7% remained in the distal ileum of germ-free chickens. Moreover, there was no difference in the H3+-DLM residual amounts in the distal ilea of conventional and germ-free chickens (3.0% vs 3.7%, respectively). Thus, it seems that DL-HMBA is absorbed and metabolised by certain intestinal microorganisms during its passage through the gastrointestinal tract, which means that gut flora have a significant impact on the availability of DL-HMBA to a broiler.
As described above, there have been a considerable number of studies comparing DL-HMBA with DLM. Few studies, however, have compared natural sources of methionine with DLM or investigated the possible metabolic loss of methionine activity from natural sources in poultry metabolism. These are promising directions for future research.
Conclusions
The relative bioavailability values of butanoic acid and polyherbal ingredient compared with that of DL-methionine are 52 and 5% for weight gain and 57 and 4% for feed conversion ratio, respectively. For breast meat yield, the relative bioavailability for butanoic acid compared with that of DL-methionine is 65%. When all parameters analysed in the present study are considered together, the average bioavailability values of butanoic acid and polyherbal ingredient in relation to that of DL-methionine are 58% and 4.5%, respectively, on a product basis.
Received February 5, 2013 and accepted November 8, 2013.
Corresponding author: luis.bruno@unioeste.br
- Albino, L. F. T.; Silva, S. H. M.; Vargas Jr., J. G.; Rostagno, H. S. and Silva, M. A. 1999. Níveis de metionina + cistina para frangos de corte de 1 a 21 e 22 a 42 dias de idade. Revista Brasileira de Zootecnia 28:519-525.
- Boebel, K. P. and Baker, D. H. 1982. Efficacy of the calcium salt and free forms of methionine hydroxy-analog for chicks. Poultry Science 61:1167-1175.
- Brugalli, I. 2003. Eficácia relativa das fontes de metionina Por que a indústria está repensando esta questão? Revista AveWorld 4:31-35.
- Drew, M. D.; Van Kessel, A. G. and Maenz, D. D. 2003. Absorption of methionine and 2-hydroxy-4-methylthiobutanoic acid in conventional and germ-free chickens. Poultry Science 82:1149-1153.
- Elwert, C.; Fernandes, E. A. and Lemme, A. 2008. Biological effectiveness of methionine hydroxy-analogue calcium salt in relation to DL-methionine in broiler chickens. Asian-Australasian Journal of Animal Sciences 21:1506-1515.
- Esteve-Garcia, E. and Llaurado, L. 1997. Performance, breast meat yield and abdominal fat deposition of male broiler chickens fed diets supplemented with DL-methionine or DL-methionine hydroxy analogue free acid. British Poultry Science 38:397-404.
- Fontaine, J.; Bech-Anderson, S.; Bütikofer, U. and De Froidmont-Görtz, I. 1998. Determination of tryptophan in feed by HPLC - Development of an optimal hydrolysis and extraction procedure by the EU Commission DG XII in three international collaborative studies. Agribiolycal Research 51:97-108.
- Hasseberg, H. A. 2002. No hay una monomerización importante en metionina hidroxianáloga. Amino NewsTM 3:37-42.
- Hoehler, D.; Lemme, A.; Roberson, K. and Turner, K. 2005a. Impact of methionine sources on performance in turkeys. Journal of Applied Poultry Research 14:296-305.
- Hoehler, D.; Lemme, A.; Jensen, S. K. and Vieira, S. L. 2005b. Relative effectiveness of methionine sources in diets for broiler chickens. Journal of Applied Poultry Research 14:679-693.
- Hoehler, D. 2006. Letter to the editor. No evidence for different dose responses of commercial methionine sources in broilers. Poultry Science 85:2047.
- Jansman, A. J. M.; Kan, C. A. and Wiebenga, J. 2003. Comparison of the biological efficacy of DL-methionine and hydroxy-4-methyl-thiobutanoic acid (HMB) in pigs and poultry. Documentation Report No. 29. Centraal Veevoederbureau (CVB, Central Bureau for Livestock Feeding), The Netherlands.
- Kratzer, D. D. and Littell, R. C. 2006. Appropriate statistical methods to compare dose responses of methionine sources. Poultry Science 85:947-954.
- Lemme, A.; Hoehler, D.; Brennan, J. J. and Mannion, P. F. 2002. Relative effectiveness of methionine hydroxy analogue compared to DL-methionine in broiler chick. Poultry Science 81:838-845.
- Lingens, G. and Molnar, S. 1996. Studies on metabolism of broilers by using 14C-labelled DL-methionine and DL-methionine hydroxy analogue Ca-salt. Archives of Animal Nutrition 49:113-124.
- Littell, R. C.; Henry, P. R.; Lewis, A. J. and Ammerman, C. B. 1997. Estimation of relative bioavailability of nutrients using SAS procedures. Journal of Animal Science 75:2672-2683.
- Llames, C. R. and Fontaine, J. 1994. Determination of amino acids in feeds: Collaborative study. Journal of AOAC International 77:1362-1402.
- Maenz, D. D. and Engele-Schaan, C. M. 1996a. Methionine and 2-hydroxy-4-methylthiobutanoic acid are transported by distinct Na+-dependent and H+-dependent systems in the brush border membrane of the chick intestinal epithelium. Journal of Nutrition 126:529-536.
- Maenz, D. D. and Engele-Schaan, C. M. 1996b. Methionine and 2-hydroxy-4-methylthiobutanoic acid are partially converted to nonabsorbed compounds during passage through the small intestine and heat exposure does not affect small intestinal absorption of methionine sources in broiler chicks. Journal of Nutrition 126:1438-1444.
- Mitchell, M. A. and Lemme, A. 2008. Examination of the composition of the luminal fluid in the small intestine of broilers and absorption of amino acids under various ambient temperatures measured in vivo. International Journal of Poultry Science 7:223-233.
- Narayanswamy, H. D. and Bhagwat, V. G. 2010. Evaluating the efficacy of methionine supplementation options in commercial broiler chickens. Poultry Line 7:1-4.
- Oliveira, R. F. M.; Donzele, J. L.; Abreu, M. L. T.; Ferreira, R. A.; Vaz, R. G. M. V. and Cella, P. S. 2006. Efeitos da temperatura e da umidade relativa sobre o desempenho e o rendimento de cortes nobres de frangos de corte de 1 a 49 dias de idade. Revista Brasileira de Zootecnia. 35:797-803.
- Payne, R. L.; Lemme, A.; Seko, H.; Hashimoto, Y.; Fujisaki, H.; Koreleski, J.; Swiatkiewicz, S.; Szczurek, W. and Rostagno, H. 2006. Bioavailability of methionine hydroxy analog-free acid relative to DL-methionine in broilers. Animal Science Journal 77:427-439.
- Piepho, H. P. 2006. Letter to the editor: A cautionary note on appropriate statistical methods to compare dose responses of methionine sources. Poultry Science 85:1511-1512.
- Rombola, L. G.; Faria, D. E.; Deponti, B. J.; Silva, F. H. A.; Faria Filho, D. E. and Junqueira, O. M. 2008. Fontes de metionina em rações formuladas com base em aminoácidos totais ou digestíveis para frangas de reposição leves e semipesadas. Revista Brasileira de Zootecnia 37:1990-1995.
- Sauer, N.; Emrich, K. and Piepho, H.P. 2008. Meta-analysis of the relative efficiency of methionine-hydroxy-analogue-free-acid compared with DL- methionine in broilers using nonlinear mixed models. Poultry Science 87:2023-2031.
- Silva Jr., R. G. C.; Lana, G. R. Q.; Rabello, C. B.; Barboza, W. A. and Lana, S. R. V. 2005. Exigências de metionina + cistina para frangos de corte machos de 1 a 21 e de 22 a 42 dias de idade, em clima tropical. Revista Brasileira de Zootecnia 34:2399-2407.
- Timmler, R. and Rodehutscord, M. 2003. Dose-response relationships for valine in the growing White Pekin duck. Poultry Science 82:1755-1762.
- Van Weerden, E. J.; Schutte, J. B. and Bertram, H. L. 1992. Utilization of the polymers of methionine hydroxy analogue free acid (MHA-FA) in broiler chicks. Archives Geflügelk 56:63-68.
Publication Dates
-
Publication in this collection
11 Mar 2014 -
Date of issue
Mar 2014
History
-
Received
05 Feb 2013 -
Accepted
08 Nov 2013