Abstract
Polyunsaturated omega-3 fatty acids (n-3 PUFA) are a family of essential fatty acids with many biological activities. These fatty acids are incorporated into cell membranes, changing their structural and functional characteristics. N-3 PUFA can act by modulating inflammatory responses at different levels. Omega-3 PUFA can be converted in the body to longer-chain n-3 PUFA at a limited rate and are differently converted in body systems. It appears that when specific longer-chain n-3 PUFA are desired these need to be supplemented directly in the diet. In different species some evidence indicates a potential effect on improving insulin sensitivity. Recently, a novel class of n-3 PUFA-derived anti-inflammatory mediators have been recognized, termed E-series and D-series resolvins, formed from EPA and DHA, respectively. N-3 PUFA derived resolvins and protectins are heavily involved in the resolution of inflammation. Supplementation with n-3 fatty acids in horses may help manage chronic inflammatory conditions such as osteoarthritis, equine metabolic syndrome, laminitis, and thereby help to improve longevity of sport horse.
alpha linolenic acid; arachidonic acid inflammation; docosahexaenoic acid; eicosapentaenoic acid; linoleic acid
Dietary n-3 and n-6 long-chain polyunsaturated fatty acids (PUFA)
Dietary fats are required to support absorption of fat-soluble vitamins and provide the essential fatty acids (NRC, 2007National Research Council - NRC. 2007. Nutrient requirements of horses. 6th rev ed. National Academic Press, Washington, DC.), linoleic acid (LA) and alpha-linolenic acid (ALA). The long-chain fatty acid family of omega-6 (n-6) and omega-3 polyunsaturated fatty acids (PUFA) are essential components of the diet and are necessary in daily physiological functions as well as for fetal development and neonatal growth. The 'parent' fatty acids are linoleic acid (LA) for the n-6 family and alpha-linolenic acid (ALA) for the n-3 family. These 'parent' fatty acids are considered essential in mammalian diets; the lack of proper enzymes prevents their endogenous synthesis. The derivatives of these 'parents' are likely of even greater importance; LA can be elongated and converted to arachidonic acid (ARA), whose primary role is to produce 20 carbon-signaling molecules known as eicosanoids. Eicosanoids have short half-lives and are localized close to their production site, influencing events within and around the cells that produce them. Arachidonic acid and other eicosanoid- producing fatty acids must be present in tissue in order for these signaling molecules to be effective. Eicosanoids are important in that they regulate a variety of cellular functions, during both physiological (normal) and inflammatory events. The most well-known classes of eicosanoids are prostaglandins (PG), thromboxanes (TX), leukotrienes (LT) and lipoxins (LX); with PG and TX being synthesized via the cyclooxygenase (COX) pathway and LT and LX being converted from ARA by lipoxgenases (Figure 1). While all classes are vital physiological components, prostaglandins are important as they are utilized by all major organ systems including reproductive, gastrointestinal and neurological. Of these active eicosanoids, prostaglandin E2 is the primary PG, synthesized exclusively from ARA, playing an important role in the inflammatory response. In terms of joint health and the development of osteoarthritis, PGE2 has been implicated as therapeutic target as it is elevated in early stages of the disease and contributes to down-stream production of degradative cartilage enzymes (McIlwraith, 2005McIlwraith, C. W. 2005. Frank Milne Lecture: From arthroscopy to gene therapy-30 years of looking in joints. Proceedings of the American Association of Equine Practitioners 51:65-113.).
Long-chain derivatives of ALA, specifically eicosapentaenoic acid (EPA), n-3 docosapentanoic acid (DPA) and docosahexaenoic acid (DHA) (Figure 2), have as equally important roles as ARA in cellular function and physiologic homeostasis. While all ALA derivatives can be converted to produce eicosanoids, EPA is the most widely recognized n-3 PUFA that is a source for anti-inflammatory PG, TX, LT and LX (Figure 1). However, research results indicate that n-3 DPA and DHA also play vital roles in mediating the inflammatory response in conjunction with EPA (Kaur et al., 2011Kaur, G.; Cameron-Smith, D.; Garg, M. and Sinlcair, A. J. 2011. Docosapentaenoic acid (22:5n-3): a review of it biological effects. Progress in Lipid Research 50:28-34.). Increased intake of EPA and DHA in a dose-dependent manner has been shown to decrease ARA amounts in cell membrane phospholipids involved in inflammation (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). Furthermore, increased EPA intake has been shown to inhibit ARA metabolism (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
) and decrease the expression of the pro-inflammatory gene COX-2 (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). One of the premier sources of EPA and DHA is marine fish oil, also known as menhaden oil. Eicosapentaenoic acid, though found in the largest quantities in marine fish oil, originates from ALA (Figure 2), which is encountered in high concentrations in plant fats. Plant oils such as linseed, soybean and flaxseed oils are all sources of ALA, therefore, when consumed, the animal may be able to synthesize EPA, DPA and DHA from these plant oils (Figure 2). Alpha-linolenic acid is converted to EPA, DPA and DHA via a desaturation and elongation pathway (Figure 2). The initial step, the addition of a double bond to ALA by the ∆6-desaturase enzyme, is the rate-limiting step in the pathway and contributes to the reported low conversion efficiency of ALA to the longer-chain PUFA (Calder, 2013Calder, P. C. 2013. Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology? British Journal of Clinical Pharmacology 75:645-662. doi: 10.1111/j.1365-2125.2012.04374.x
https://doi.org/10.1111/j.1365-2125.2012...
). Both LA and ALA share a need for ∆6-desaturase (Figure 2), and while the enzyme has a higher affinity for ALA, in humans dietary fats contain a higher percentage of LA, therefore competing for the same enzyme ALA needs for conversion to its longer derivate (Tu et al., 2010Tu, W. C.; Cook-Johnson, R. J.; James, M. J.; Muhlausler, B. S. and Gibson, R. A. 2010. Omega-3 long chain fatty acid synthesis is regulated more by substrate levels than gene expression. Prostaglandins, Leukotrienes and Essential Fatty Acids 83:61-68.). In addition, ALA has been shown to have the highest oxidation rate among all unsaturated fatty acids in human tracer studies (Nettleton, 1991Nettleton, J. A. 1991. Omega-3 fatty acids: comparison of plant and seafood sources in human nutrition. Journal of the American Dietetic Association 91:331-337.), contributing to the low conversion of ALA to its longer derivate. A review of studies investigating the efficiency of dietary ALA conversion in humans reported the conversion of ALA to EPA ranged from 8-10% with conversion efficiency being as low at 4% for DHA (Williams and Burdge, 2006Williams, C. M. and Burdge, G. 2006. Long chain n-3 PUFA: plant v. marine sources. Proceedings of the Nutrition Society 65:42-50.). Due to evidence of a low conversion rate, it is recommended to supply EPA and DHA directly in the diet (Arterburn et al., 2006Arterburn, L. M.; Hall, E. B. and Oken, H. 2006. Distrubution, interconversion and dose response of n-3 fatty acids in humans. American Journal of Clinical Nutrition 83:S1467-1476.).
Biochemical pathway for the incorporation of n-3 and n-6 fatty acids.
Source: Reprinted from Arterburn et al. (2006), with permission.
A source of ALA in the equine diet is forage (hay or pasture), which is the largest portion of equine diets. Many horses are fed forage alone; however, performance horses require additional energy-dense feeds such as grains or oils due to their higher energy demands. Vegetable oils are also a source of ALA, such as flaxseed and linseed oil, which can be top-dressed onto feed. Linoleic acid is very abundant in cereal grains such as corn and barley, and makes up the majority of the fat in corn oil as well. Algae produce the n-3 PUFA, EPA and DHA; therefore they are present in fish because algae is an ordinary diet of fish. For that reason, marine foods and fish oil are a good source of EPA and DHA and may be consumed by humans and other carnivores or omnivores. Additionally, they have been used as supplements in equine diets. Horse studies indicated that supplementation with ALA would not lead to increases in circulating DHA (Hansen et al., 2002Hansen, R. A.; Savage, C. J.; Reidlinger, K.; Traub-Dargatz, J. L.; Ogilvie, G. K.; Mitchell D. and Fettman, M. J. 2002. Effects of dietary flaxseed oil supplementation on equine plasma fatty acid concentrations and whole blood platelet aggregation. Journal of Veterinary Internal Medicine 16:457-463.; Vineyard et al., 2010Vineyard, K. R.; Warren, L. K. and Kivipelto, J. 2010. Effect of dietary omega-3 fatty acid source on plasma and red blood cell membrane composition and immune function in yearling horses. Journal of Animal Science 88:248-257.), only increases in circulating EPA.
Inflammation and PUFA
Cellular activities involved in inflammatory responses are designed to be harmful to pathogens; however, they can cause damage to the host tissues (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). Inflammation usually is self-limiting, and resolves rapidly due to the activation of negative feed-back mechanisms like secretion of anti-inflammatory cytokines or pro-resolving lipid mediators, shedding of receptors for inflammation, and activation of regulatory cells (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). Loss of this regulatory process can result in excessive, inappropriate or chronic inflammation that can cause damage to the host organism (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). As part of these anti-inflammatory and pro-resolving mediators, a novel class of n-3 PUFA-derived anti-inflammatory mediators have been recognized, termed E-series and D-series resolvins, formed from EPA and DHA, respectively (Calder, 2009Calder, P. C. 2009. Polyunsaturated fatty acids and inflammatory processes: New twists in an old tale. Biochimie 91:791-795.). Protectins are another class of mediators produced from DHA (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). While information is limited, it appears n-3 PUFA-derived resolvins and protectins are heavily involved in the resolution of inflammation (Kohli and Levy, 2009Kohli, P. and Levy, B. D. 2009. Resolvins and protectins: mediating solutions to inflammation. British Journal of Pharmacology 158:960-971.). Protectins D1 are produced from DHA, and known as protectins D1, owing to their protecting activity in inflammatory and neural systems. Biological effects of resolvins and protectins have been studied in cell culture and animal models of inflammation and have been shown to stimulate resolution and reduce magnitude of inflammatory response in vivo (Serhan et al., 2008Serhan, C.; Chiang, N. and Van Dyke, T. 2008. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nature Reviews Immunology 8:349-361.). Resolvin E1 reduces inflammation in vivo and blocks human transendothelial migration (Serhan et al., 2004Serhan, C.; Gotlinger, K.; Hong S. and Arita, M. 2004. Resolvins, docosatrienes, and neuroprotectins, novel omega-3-derived mediators, and their aspirin-triggered endogenous epimers: an overview of their protective roles in catabasis. Prostaglandins and other Lipid Mediators 73:155-172.). Resolvin E2 reduces zymosan initiated neutrophil infiltration (Tjonahem et al., 2006Tjonahen, E.; Oh, S. F.; Siegelman, J.; Elangovan, S.; Percapio, K. B.; Hong, S.; Arita, M. and Serhan, C. N. 2006. Resolvin E2: identification and anti-inflammatory actions: pivotal role of human 5-lipoxygenase in resolvin E series biosynthesis. Chemistry and Biology 13:1121-1122.). Resolvin D1 has been shown to be a potent regulator of mouse and human neutrophils (Serhan et al., 2004Serhan, C.; Gotlinger, K.; Hong S. and Arita, M. 2004. Resolvins, docosatrienes, and neuroprotectins, novel omega-3-derived mediators, and their aspirin-triggered endogenous epimers: an overview of their protective roles in catabasis. Prostaglandins and other Lipid Mediators 73:155-172.; Sun et al., 2007Sun, Y.; Oh, S.; Uddin, J.; Yang, R.; Gotlinger, K.; Campbell, E.; Colgan, S. P.; Petasis, N. A. and Serhan, C. N. 2007. Resolvin D1 and its aspirin-triggered 17R Epimer stereochemical assignments, anti-inflammatory properties, and enzymatic inactivation. Journal of Biological Chemistry 282:9323-9334.). Resolvin E1 also has been shown to initiate resolution of inflammation causing a decrease in the number of neutrophils in exsudates sooner than during spontaneous resolution. Resolvin E1 and resolvin D1 prevented the infiltration of neutrophils into sites of inflammation, and inhibited IL-1β production (Calder, 2014Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
https://doi.org/10.1016/j.bbalip.2014.08...
). Protectin D1 also blocked T cell migration in vivo, reducing TNF-α (tumor necrosis factor alpha), IL-1β, interferon-γ (IF-γ) secretion, and promoting T cell apoptosis (Ariel et al., 2006Ariel, A.; Li, P. L.; Wang, W.; Tang, W. X.; Fredman, G.; Hong, S.; Gotlinger, K. H. and Serhan, C. N. 2006. The docosatriene protectin D1 is produced by TH2 skewing and promotes human T cell apoptosis via lipid raft clustering. Journal of Biological Chemistry 280:43079-43086.). Protectin D1 shifted the onset of resolution to an earlier time point in addition to shortening the time to reduce the number of maximum neutrophils by half (Serhan et al., 2008Serhan, C.; Chiang, N. and Van Dyke, T. 2008. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nature Reviews Immunology 8:349-361.).
Other anti-inflammatory effects of n-3 PUFA from plant and animal sources include a reduced cytokine production in vitro (De Caterina et al., 1994De Caterina, R.; Cybulsky M. I.; Clinton S. K.; Gimbrone, M. A. and Libby, P. 1994. The omega-3 fatty acid docosahexaenoate reduces cytokine-induced expression of proatherogenic and proinflammatory proteins in human endothelial cells. Artheriosclerosis and Thrombosis 14:1829-1836.) and in vivo (Meydani et al., 1993Meydani, S.; Lichtenstein, A. H.; Cornwall, S.; Meydani, M.; Goldin, B. R.; Rasmussen, H.; Dinarello, C. A. and Schaefer, E. J. 1993. Immunologic effects of national cholesterol education panel step-2 diets with and without fish-derived N-3 fatty acid enrichment. Journal of Clinical Investigation 92:105-113.; Grimm et al., 1994Grimm, H.; Tibell, A.; Norrlind, B.; Blecher, C.; Wilker, S. and Schwemmle, K. 1994. Immunoregulation by parenteral lipids: impact of the n-3 to n-6 fatty acid ratio. Journal of Parenteral and Enteral Nutrition 18:417-421.; McCann et al., 2000McCann, M.; Moore, J. N.; Carrik, J. B. and Barton, M. H. 2000. Effect of intravenous infusion of omega-3 and omega-6 lipid emulsions on equine monocyte fatty acid composition and inflammatory mediator production in vitro. Shock 14:222-228.).
Sources of fatty acids in the diet
In humans, supplementation with long-chain n-3 PUFA has been shown to improve inflammatory status, prevent cardiovascular diseases (Calder, 2001Calder, P. C. 2001. Omega 3 polyunsaturated fatty acids, inflammation and immunity. World Review on Nutrition and Dietetics 88:109-116.), and reduce pain and inflammation in patients with rheumatoid arthritis (MacLean et al., 2004MacLean, C. H.; Mojica, W. A.; Morton, S. C.; Pencharz, J.; Hasenfeld, R.; Garland, T. W.; Newberry, S. J.; Jungvig, L. K.; Grossman, J.; Khanna, P.; Rhodes, S. and Shekelle, P. 2004. Effects of omega-3 fatty acids on lipids and glycemic control in type II diabetes and the metabolic syndrome and on inflammatory bowel disease, rheumatoid arthritis, renal disease, systemic lupus erythematosus, and osteoporosis. Evidence Report/Technology Assessment No. 89 (Prepared by Southern California/RAND Evidence-based Practice Center, under Contract No. 290-02-0003). AHRQ Publication No. 04-E012-2. Agency for Healthcare Research and Quality, Rockville, MD.). In arthritic horses, supplementation with EPA and DHA increased stride length (Woodward et al., 2005Woodward, A. D.; Nielsen, B. D.; O'Conner, C. I.; Webel, S. K. and Orth, M. W. 2005. Supplementation of dietary long-chain polyunsaturated fatty acids high in docosahexaenoic acid (DHA) increases plasma DHA concentrations and may increase trot stride lengths in horses. Equine and Comparative Exercise Physiology 4:71-78.) and reduced inflammatory markers (Manhart et al., 2009Manhart, D. R.; Scott, B. D.; Gibbs, P. G.; Honnas, C. M.; Hood, D. M. and Coverdale, J. A. 2009. Markers of inflammation in arthritic horses fed omega-3 fatty acids. Professional Animal Scientist 25:155-160.). In horses, feeding Menhaden fish oil modulated leucotriene synthesis influencing inflammatory conditions (Hall et al., 2004Hall, J.; Van Saun, R. and Wander, R. C. 2004. Dietary (n-3) fatty acids from Menhaden fish oil alter plasma fatty acid and leukotriene B synthesis in healthy horses. Journal of Veterinary Internal Medicine 18:871-879.).
Proposed mechanisms of EPA and DHA on inflammation
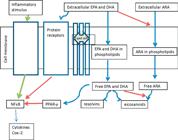
Several mechanisms have been proposed regarding the processes by which EPA and DHA act on the inflammatory response in tissues. It is well established that these lipids act on both a direct (by alteration of eicosanoid production via cyclooxygenase and lipoxygenase pathways) and indirect (modification of gene transcription) mechanism (Calder, 2006Calder, P. C. 2006. n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. American Journal of Clinical Nutrition 83(suppl):1505S-1519S.). Direct modification of prostaglandin and leukotriene synthesis was outlined previously. Supplementation of n-3 PUFA in feeds will also exert an effect on the expression of inflammatory genes (Renier et al., 1993Renier, G.; Skamene, E.; de Sanctis, J. and Radzioch, D. 1993. Dietary n-3 polyunsaturated fatty acids prevent the development of artherosclerotic lesions in mice: modulation of macrophage secretory activities. Journal of Artherosclerosis and Thrombosis 13:1515-1524.; Curtis et al., 2000Curtis, C.; Hughes C. E.; Flannery C. R.; Little C. B.; Harnson B. and Carterson, B. 2000. n-3 fatty acids specifically modulate catabolic factors involved in articular cartilage degradation. Journal of Biological Chemistry 275:721-724.; Wallace et al., 2001Wallace, F.; Miles, E. A.; Evans, C.; Stock, T. E.; Yaqoob, P. and Calder, P. C. 2001. Dietary fatty acids influence the production of Th1-but not TH-2 type cytokines. Journal of Leukocyte Biology 69:449-457.).
It is hypothesized that particular fatty acids, such as EPA and/or DHA, may modify transcription factors in the nucleus and thus influence cytokine and eicosanoid production at the level of gene expression (Figure 3). Another theory is that n-6 (i.e., ARA) and n-3 (i.e., EPA and DHA) fatty acids modify protein synthesis of inflammatory mediators via modification of cell surface receptors on lipid rafts or within the cell by suppression of nuclear receptor activation (Chapkin et al., 2009Chapkin, R. S.; Kim W.; Lupton J. R. and McMurray, D. N. 2009. Dietary docoahexaenoic and eicosapentaenoic acid: emerging mediators of inflammation. Prostaglandins, Leukotrienes and Essential Fatty Acids 81:187-191.). Additional regulation comes in the form of peroxisome proliferator-activated receptors, which are key nuclear receptors that regulate transcription of genes through ligand binding with a variety of lipophilic metabolites, having a high affinity for PUFA, in particular DHA (Stulnig, 2003Stulnig, T. M. 2003. Immunomodulation by polyunsaturated fatty acids: Mechanisms and effects. International Archives of Allergy and Immunology 132:310-321.). Peroxisome proliferator-activated receptors usually have three isoforms; α, β, γ and are believed to be potent regulators of adipocyte function as well as immune molecules such as lymphocytes and macrophages (Marx et al., 2002Marx, N.; Kehrle, B.; Kohlhammer, K.; Grub, M.; Koenig, W.; Homback, V.; Libby, P. and Plutzky, J. 2002. PPAR activators as anti-inflammatory mediators in human T lymphocytes: Implications for atherosclerosis and transplantation-associated arterosclerosis. Circulation Research 90:703-710.) influencing downstream transcription of inflammatory cytokines (Figure 3).
Summary of the anti-inflammatory actions of n-3 polyunsaturated fatty acids; modified from Calder (2013)Calder, P. C. 2013. Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology? British Journal of Clinical Pharmacology 75:645-662. doi: 10.1111/j.1365-2125.2012.04374.x
https://doi.org/10.1111/j.1365-2125.2012... .EPA - eicosapentaenoic acid; DHA - docosahexaenoic acid; ARA - arachidonic acid; COX - cyclooxigenase; NFkB - nuclear factor kappa B; PPAR - peroxisome proliferator activated receptor. Solid red lines indicate inhibition. Solid blue lines indicate sources of n-3 PUFA. Solid green lines indicate inflammatory activation.
Insulin sensitivity and n-3 PUFA
Insulin resistance (decreased insulin sensitivity) in horses has been linked to the development of laminitis, osteochondrosis, and metabolic syndrome (Coffman and Coles, 1983Coffman, J. R. and Colles, C. M. 1983. Insulin tolerance in laminitic ponies. Canadian Journal of Comparative Medicine 47:347-351.; Ralston, 1996Ralston, S. L. 1996. Hyperglycemia/hyperinsulinemia after feeding a meal of grain to young horses with osteochondritis dissecans (OCD) lesions. Pferdeheilkunde 12:320-322.; Treiber et al., 2006Treiber, K. H.; Kronfeld, D. S.; Hess, T. M.; Byrd, B. M.; Splan, R. K. and Staniar, W. B. 2006. Evaluation of genetic and metabolic predispositions and nutritional risk factors for pasture-associated laminitis in ponies. Journal of American Veterinary Medical Association 228:1538-1545.; Frank et al., 2010Frank, N.; Geor, R. J.; Durham, A. E. and Johnson, P. J. 2010. Equine metabolic syndrome. Journal of Veterinary Internal Medicine 24:467-475.) and is therefore considered a problem in the equine industry. These diseases can result in a loss of function and performance of the horse. Several factors may predispose a horse to developing insulin resistance, such as diet, age, breed/genetics and obesity (Jeffcot et al., 1986Jeffcott, L. B.; Field, J. R.; McLean, J. G. and O'Dea, K. 1986. Glucose tolerance and insulin sensitivity in ponies and standardbred horses. Equine Veterinary Journal 18:97-101.; Treiber et al., 2006Treiber, K. H.; Kronfeld, D. S.; Hess, T. M.; Byrd, B. M.; Splan, R. K. and Staniar, W. B. 2006. Evaluation of genetic and metabolic predispositions and nutritional risk factors for pasture-associated laminitis in ponies. Journal of American Veterinary Medical Association 228:1538-1545.; Vick et al., 2007Vick, M. M.; Adams, A. A.; Murphy, B. A.; Sessions, D. R.; Horohov, D. W.; Cook, R. F.; Shelton, B. J. and Fitzgerald, B. P. 2007. Relationships among inflammatory cytokines, obesity, and insulin sensitivity in the horse. Journal of Animal Science 85:1144-1155.). Supplementation with certain dietary components could increase insulin sensitivity in animals that have insulin resistance, reducing the risk for the development of diseases such as metabolic syndrome and laminitis.
Dietary supplementation with n-3 PUFA has been shown to increase insulin sensitivity in pigs and rats (Behme, 1996Behme, M. T. 1996. Dietary fish oil enhances insulin sensitivity in miniature pigs. Journal of Nutrition 126:1549-1553.; Luo et al., 1996Luo, J.; Rizkalla, S. W.; Boillot, J.; Alamowitch, C.; Chaib, H.; Bruzzo, F.; Desplanque, N.; Dalix, A. M.; Durand, G. and Slama, G. 1996. Dietary (n-3) polyunsaturated fatty acids improve adipocyte insulin action and glucose metabolism in insulin-resistant rats: Relation to membrane fatty acids. Journal of Nutrition 126:1951-1958.). Dietary EPA and DHA incorporate into cell membranes increasing membrane fluidity due to greater unsaturation of the membrane improving glucose transport function (Lardinois et al., 1987Lardinois, C. K. 1987. The role of omega 3 fatty acids on insulin secretion and insulin sensitivity. Medical Hypotheses 24:243-248.; Zhao et al., 2008Zhao, S. M.; Jia, L.W.; Gao, P.; Li, Q. R.; Lu, X.; Li, J. S. and Xu, G. W. 2008. Study on the effect of eicosapentaenoic acid on phospholipids composition in membrane microdomains of tight junctions of epithelial cells by liquid chromatography/electrospray mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis 47:343-350.). Incorporation of EPA and DHA in muscle cell membrane has been shown to increase binding of insulin (Storlein et al., 1991Storlien, L. H.; Jenkins, A. B.; Chisholm, D. J.; Pascoe, W. S.; Khouri, S. and Kraegen, E. W. 1991. Influence of dietary fat composition on development of insulin resistance in rats. Relationship to muscle triglyceride and omega-3 fatty acids in muscle phospholipid. Diabetes 40:280-289.) and m-RNA expression of GLUT-4 transporters in rats (Figueiras et al., 2010Figueiras, M.; Olivan, M.; Busquets, S.; Lopez-Soriano, F. J. and Argiles, J. M. 2010. Effects of eicosapentaenoic acid (EPA) treatment on insulin sensitivity in an animal model of diabetes: improvement of the inflammatory status. Obesity 19:362-369.). Supplementation with EPA and DHA has shown to improve insulin sensitivity in rats (Storlein et al., 1991Storlien, L. H.; Jenkins, A. B.; Chisholm, D. J.; Pascoe, W. S.; Khouri, S. and Kraegen, E. W. 1991. Influence of dietary fat composition on development of insulin resistance in rats. Relationship to muscle triglyceride and omega-3 fatty acids in muscle phospholipid. Diabetes 40:280-289.), pigs (Behme, 1996Behme, M. T. 1996. Dietary fish oil enhances insulin sensitivity in miniature pigs. Journal of Nutrition 126:1549-1553.), and in humans (Rasic-Milutinovic et al., 2007Rasic-Milutinovic, Z.; Perunicic, G.; Pljesa, S.; Gluvic, Z.; Sobajic, S.; Djuric, I. and Ristic, D. 2007. Effects of n-3 PUFA supplementation on insulin resistance and inflammatory biomarkers in hemodialysis patients. Renal Failure 29:321-329.).
n-3 PUFA supplementation and exercise
Supplementation with EPA and DHA in horses has also been shown to lower heart rate, plasma glycerol, free fatty acids and cholesterol during an exercise test compared to horses supplemented with corn oil (O'Connor et al., 2004O'Connor, C. I.; Lawrence, L. M.; St Lawrence, A. C.; Janicki, K. M.; Warren, L. K. and Hayes, S. 2004. The effect of dietary fish oil supplementation on exercising horses. Journal of Animal Science 82:2978-2984.). In human trained athletes supplemented with fish oil, heart rate and oxygen consumption was lower than in subjects supplemented with olive oil (Peoples et al., 2008Peoples, G. E.; McLennan, L. P.; Howe, P. R. C. and Groeller, H. 2008. Fish oil reduces heart rate and oxygen consumption during exercise. Journal of Cardiovascular Pharmacology 52:540-547.). Incorporation of n-3 PUFA to muscle membranes increased insulin sensitivity (Pan et al., 1995Pan, D. A.; Lillioja, S.; Milner, M. R.; Kritetos, A. D.; Baur, L. A.; Borgadus, C. and Storlein, L. H. 1995. Skeletal muscle membrane lipid composition is related to adiposity and insulin action. Journal of Clinical Investigation 96:2802-2808.) and resulted in increased ability of skeletal muscle to take up glucose. Furthermore, studies have shown that lower proportions of long-chain PUFA and higher proportions of saturated fatty acids in skeletal muscle phospholipids are associated with insulin resistance. Skeletal muscle characteristics (i.e., fatty acid profile) have some genetic influence (Baur et al., 1999Baur, L. A.; O'Connor, J.; Pan, D. A. and Storlein, L. H. 1999. Relationships between maternal risk of insulin resistance and the child's muscle membrane fatty acid composition. Diabetes 48:112-116.) but diet and physical activity also influence skeletal muscle fatty acid profile in rats, humans, and horses (Ayre et al., 1996Ayre, K. J. and Hulbert, A. J. 1996. Dietary fatty acid profile influences the composition of skeletal muscle phospholipids in rats. Journal of Nutrition 126:653-662.; Andersson et al., 2000Andersson, A.; Sjodin, A.; Hedman, A.; Olsson, R. and Vessby, B. 2000. Fatty acid profile of skeletal muscle phospholipids in trained and untrained young men. American Journal of Physiology: Endocrinology and Metabolism 279:E744-E751.; Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.).
Specific horse studies
Effects of two different dietary sources of n-3 PUFA on incorporation into the plasma, red blood cell, and skeletal muscle in horses (Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.)
In humans, the PUFA DHA and EPA need to be supplemented in order for them to be incorporated into tissues. There is limited conversion of ALA to DHA in humans (Burdge et al., 2001Burdge, G. C.; Jones, A. E. and Wootton, S. A. 2002. Eicosapentaenoic and docosapentaenoic acids are the principal products of α-linolenic acid metabolism in young men. British Journal of Nutrition 88:355-363.; Arterburn et al., 2006Arterburn, L. M.; Hall, E. B. and Oken, H. 2006. Distrubution, interconversion and dose response of n-3 fatty acids in humans. American Journal of Clinical Nutrition 83:S1467-1476.). As stated before, in horses, supplementation with ALA did not lead to increases in circulating DHA (Hansen et al., 2002Hansen, R. A.; Savage, C. J.; Reidlinger, K.; Traub-Dargatz, J. L.; Ogilvie, G. K.; Mitchell D. and Fettman, M. J. 2002. Effects of dietary flaxseed oil supplementation on equine plasma fatty acid concentrations and whole blood platelet aggregation. Journal of Veterinary Internal Medicine 16:457-463.; Vineyard et al., 2010Vineyard, K. R.; Warren, L. K. and Kivipelto, J. 2010. Effect of dietary omega-3 fatty acid source on plasma and red blood cell membrane composition and immune function in yearling horses. Journal of Animal Science 88:248-257.), only EPA. Feeding of n-3 PUFA to horses may increase circulating levels and increase incorporation into muscle tissue. This could potentially improve chronic inflammatory conditions in horses; however, the optimal type of fatty acid to be supplemented needed to be investigated.
In a related study in our laboratory (Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.) in order to compare different sources of dietary omega-3 fatty acid supplementations on plasma, red blood cell and skeletal muscle fatty acid compositions in horses, three (alfalfa/bromegrass) hay and barley diets were fed: one was supplemented with an algae and fish oil containing DHA and EPA (MARINE; Magnitude; JBS United, Sheridan, IN); another group was supplemented with the same amount of ALA in flaxseed (FLAX) as FLAX, and a third group (control; CON) was fed hay and barley to make all diets have the same calorie amount. Treatments were supplemented for 90 d. Blood samples and muscle middle gluteal biopsies were collected on d 0, 30, 60, and 90 of supplementation.
Direct supplementation of EPA and DHA through a marine source increased PUFA concentrations in the plasma, red blood cell and muscle tissue of equines (Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.). Although present in muscle tissue at baseline, EPA and DHA increased in horses supplemented with a marine source containing these specific fatty acids. In plasma and red blood cells, EPA and DHA were below detection levels in all groups and increased only in MARINE-supplemented horses. Supplementation with dietary sources containing EPA and DHA may be indicated when increased incorporation of these n-3 fatty acids to muscle and red blood cells is desired. Conversion of ALA from flaxseed and forages to EPA and DHA occurs in the skeletal muscle, as seen in this study (Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.) by the detection of these fatty acids in equine muscles at baseline. Supplementation with ALA through flaxseed lead to higher incorporation of muscle n-3 DPA, a derivate of EPA, compared to MARINE supplementation. Some positive effects of n-3 DPA on inflammation have been reported; however, the effects of such increases should be evaluated in further studies addressing inflammatory responses and compared to EPA and DHA in horses (Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.). In this study, conversion of ALA to EPA and DHA did not occur after supplementation with extra ALA (above the control dietary n-3 PUFA level) from FLAX.
Effects of n-3 PUFA supplementation on insulin sensitivity in horses (Hess et al., 2013Hess, T. M.; Rexford, J.; Hansen, D. K.; Ahrens, N. S.; Harris, M.; Engle, T.; Ross, T. and Allen, K. G. 2013. Effects of omega-3 (n-3) fatty acid supplementation on insulin sensitivity in horses. Journal of Equine Veterinary Science 33:446-453.)
Dietary supplementation with n-3 PUFA has been shown to increase insulin sensitivity as described above. In order to test the hypothesis that supplementation with n-3 PUFA would improve insulin sensitivity, the same diets described for the previous study were fed to a group of 21 adult mares to test glucose and insulin dynamics. Specific tests to determine insulin sensitivity (frequent sampling intravenous glucose tolerance tests) were performed on days 0, 30, 60, and 90 of supplementation (Hess at al., 2013Hess, T. M.; Rexford, J.; Hansen, D. K.; Ahrens, N. S.; Harris, M.; Engle, T.; Ross, T. and Allen, K. G. 2013. Effects of omega-3 (n-3) fatty acid supplementation on insulin sensitivity in horses. Journal of Equine Veterinary Science 33:446-453.).
No overall treatment effect was observed when all mares within each treatment were compared among themselves. However, when treatments were compared among mares with the lowest quintile in insulin sensitivity (Treiber et al., 2005Treiber, K. H.; Kronfeld, D. S.; Hess, T. M.; Boston, R. C. and Harris, P. A. 2005. Insulin sensitivity and pancreatic β-cell response in the horse: screening proxies and reference quintiles assessed by the minimal model. American Journal of Veterinary Research 66:2114-2121.) (11 mares) there was a trend (P = 0.08) for a treatment effect, where MARINE (n = 5 and FLAX (n = 3) horses had higher insulin sensitivity (SI = 1.18±0.16 in FLAX, and 1.05± 0.16 in MARINE compared to 0.59±0.16 in CON, n = 3). Although a small number of insulin resistant mares were compared, further studies should be performed in a larger group of insulin resistant horses. If proven effective, supplementation with omega-3 fatty acids could help to reduce problems associated with insulin resistance in horses (Hess et al., 2013Hess, T. M.; Rexford, J.; Hansen, D. K.; Ahrens, N. S.; Harris, M.; Engle, T.; Ross, T. and Allen, K. G. 2013. Effects of omega-3 (n-3) fatty acid supplementation on insulin sensitivity in horses. Journal of Equine Veterinary Science 33:446-453.).
Evaluation of synovial fluid in horses fed two different sources n-3 PUFA: a pilot study (Ross-Jones et al., 2014Ross-Jones, T.; Hess, T. M.; Rexford, J.; Ahrens, N.; Engle, T. and Hansen, K. 2014. Effects of omega-3 long chain polyunsaturated fatty acid supplementation on equine synovial fluid fatty acid composition and prostaglandin E2. Journal of Equine Veterinary Science 34:779-783.)
Elevating n-3 PUFA in mammalian diets may downregulate inflammatory processes in the joint (Proudman et al., 2008Ralston, S. L. 1996. Hyperglycemia/hyperinsulinemia after feeding a meal of grain to young horses with osteochondritis dissecans (OCD) lesions. Pferdeheilkunde 12:320-322.) and has been shown to have symptom-modifying effects in inflammatory diseases (Lau et al., 1993Lau, C. S.; Morley, K. D. and Belch, J. J. 1993. Effects of fish oil supplementation on non-steroidal anti-inflammatory drug requirement in patients with mild rheumatoid arthritis-A double-blind placebo controlled study. British Journal of Rheumatology 32:982-989.). One hypothesized mechanism is the potential of long-chain n-3 PUFA to reduce the production of potent inflammatory eicosanoids (Chapkin et al., 2009Chapkin, R. S.; Kim W.; Lupton J. R. and McMurray, D. N. 2009. Dietary docoahexaenoic and eicosapentaenoic acid: emerging mediators of inflammation. Prostaglandins, Leukotrienes and Essential Fatty Acids 81:187-191.), primarily PGE2. In order to test the effects of different sources of n-3 PUFA on synovial fluid composition, the diets described on the first study (Hess et al., 2012Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.) were supplemented for 90 days (Ross-Jones et al., 2014Ross-Jones, T.; Hess, T. M.; Rexford, J.; Ahrens, N.; Engle, T. and Hansen, K. 2014. Effects of omega-3 long chain polyunsaturated fatty acid supplementation on equine synovial fluid fatty acid composition and prostaglandin E2. Journal of Equine Veterinary Science 34:779-783.).
On day 90 of supplementation, approximately 3 mL of synovial fluid were extracted from the right carpus of each horse. Fluid was analyzed for fatty acid concentration and PGE2 concentration. Synovial fluid samples from the MARINE group exhibited EPA and DHA, whereas the FLAX and CON groups did not express detectable concentrations. Synovial prostaglandin E2 concentration in the MARINE group tended to be lower compared to CON horses (P<0.10).
Synovial fluid fatty acid levels for EPA and DHA were significantly higher in the MARINE group compared to either CON or FLAX groups, indicating that direct supplementation of EPA and DHA is required if higher fluid concentrations of those fatty acids are desired (Ross-Jones et al., 2014Ross-Jones, T.; Hess, T. M.; Rexford, J.; Ahrens, N.; Engle, T. and Hansen, K. 2014. Effects of omega-3 long chain polyunsaturated fatty acid supplementation on equine synovial fluid fatty acid composition and prostaglandin E2. Journal of Equine Veterinary Science 34:779-783.). A small difference between treatment and control in synovial fluid PGE2 concentration in the current study may be due to all horses being healthy and free of existing joint inflammation or disease. Inducing experimental inflammation in healthy animals receiving a dietary n-3 PUFA supplement may cause measurable differences in eicosanoid levels.
Results indicated a possible inhibition of inflammatory eicosanoid prostaglandin E2 production in equine synovial fluid by oral supplementation of the polyunsaturated fatty acids EPA and DHA. Further research is needed to determine if oral n-3 PUFA supplementation can be therapeutically advantageous in horses experiencing joint inflammation.
Implications
Based on results from several studies, supplementation with n-3 polyunsaturated fatty acids has the potential to benefit humans and horses in diverse ways. Future studies should address the effects of n-3 polyunsaturated fatty acid supplementation on inflammation and specifically in horses with chronic inflammatory diseases such as laminitis, metabolic syndrome, pituitary pars intermedia disease and osteoarthritis improving the horses' health and wellbeing.
References
- Andersson, A.; Sjodin, A.; Hedman, A.; Olsson, R. and Vessby, B. 2000. Fatty acid profile of skeletal muscle phospholipids in trained and untrained young men. American Journal of Physiology: Endocrinology and Metabolism 279:E744-E751.
- Ariel, A.; Li, P. L.; Wang, W.; Tang, W. X.; Fredman, G.; Hong, S.; Gotlinger, K. H. and Serhan, C. N. 2006. The docosatriene protectin D1 is produced by TH2 skewing and promotes human T cell apoptosis via lipid raft clustering. Journal of Biological Chemistry 280:43079-43086.
- Arterburn, L. M.; Hall, E. B. and Oken, H. 2006. Distrubution, interconversion and dose response of n-3 fatty acids in humans. American Journal of Clinical Nutrition 83:S1467-1476.
- Ayre, K. J. and Hulbert, A. J. 1996. Dietary fatty acid profile influences the composition of skeletal muscle phospholipids in rats. Journal of Nutrition 126:653-662.
- Baur, L. A.; O'Connor, J.; Pan, D. A. and Storlein, L. H. 1999. Relationships between maternal risk of insulin resistance and the child's muscle membrane fatty acid composition. Diabetes 48:112-116.
- Behme, M. T. 1996. Dietary fish oil enhances insulin sensitivity in miniature pigs. Journal of Nutrition 126:1549-1553.
- Burdge, G. C.; Jones, A. E. and Wootton, S. A. 2002. Eicosapentaenoic and docosapentaenoic acids are the principal products of α-linolenic acid metabolism in young men. British Journal of Nutrition 88:355-363.
- Calder, P. C. 2001. Omega 3 polyunsaturated fatty acids, inflammation and immunity. World Review on Nutrition and Dietetics 88:109-116.
- Calder, P. C. 2006. n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. American Journal of Clinical Nutrition 83(suppl):1505S-1519S.
- Calder, P. C. 2009. Polyunsaturated fatty acids and inflammatory processes: New twists in an old tale. Biochimie 91:791-795.
- Calder, P. C. 2013. Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology? British Journal of Clinical Pharmacology 75:645-662. doi: 10.1111/j.1365-2125.2012.04374.x
» https://doi.org/10.1111/j.1365-2125.2012.04374.x - Calder, P. C. 2014. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochinica et Byophysica Acta, doi: 10.1016/j.bbalip.2014.08.010 (in press).
» https://doi.org/10.1016/j.bbalip.2014.08.010 - Chapkin, R. S.; Kim W.; Lupton J. R. and McMurray, D. N. 2009. Dietary docoahexaenoic and eicosapentaenoic acid: emerging mediators of inflammation. Prostaglandins, Leukotrienes and Essential Fatty Acids 81:187-191.
- Coffman, J. R. and Colles, C. M. 1983. Insulin tolerance in laminitic ponies. Canadian Journal of Comparative Medicine 47:347-351.
- Curtis, C.; Hughes C. E.; Flannery C. R.; Little C. B.; Harnson B. and Carterson, B. 2000. n-3 fatty acids specifically modulate catabolic factors involved in articular cartilage degradation. Journal of Biological Chemistry 275:721-724.
- De Caterina, R.; Cybulsky M. I.; Clinton S. K.; Gimbrone, M. A. and Libby, P. 1994. The omega-3 fatty acid docosahexaenoate reduces cytokine-induced expression of proatherogenic and proinflammatory proteins in human endothelial cells. Artheriosclerosis and Thrombosis 14:1829-1836.
- Figueiras, M.; Olivan, M.; Busquets, S.; Lopez-Soriano, F. J. and Argiles, J. M. 2010. Effects of eicosapentaenoic acid (EPA) treatment on insulin sensitivity in an animal model of diabetes: improvement of the inflammatory status. Obesity 19:362-369.
- Frank, N.; Geor, R. J.; Durham, A. E. and Johnson, P. J. 2010. Equine metabolic syndrome. Journal of Veterinary Internal Medicine 24:467-475.
- Grimm, H.; Tibell, A.; Norrlind, B.; Blecher, C.; Wilker, S. and Schwemmle, K. 1994. Immunoregulation by parenteral lipids: impact of the n-3 to n-6 fatty acid ratio. Journal of Parenteral and Enteral Nutrition 18:417-421.
- Hall, J.; Van Saun, R. and Wander, R. C. 2004. Dietary (n-3) fatty acids from Menhaden fish oil alter plasma fatty acid and leukotriene B synthesis in healthy horses. Journal of Veterinary Internal Medicine 18:871-879.
- Hansen, R. A.; Savage, C. J.; Reidlinger, K.; Traub-Dargatz, J. L.; Ogilvie, G. K.; Mitchell D. and Fettman, M. J. 2002. Effects of dietary flaxseed oil supplementation on equine plasma fatty acid concentrations and whole blood platelet aggregation. Journal of Veterinary Internal Medicine 16:457-463.
- Hess, T. M.; Rexford, J.; Hansen, D. K.; Ahrens, N. S.; Harris, M.; Engle, T.; Ross, T. and Allen, K. G. 2013. Effects of omega-3 (n-3) fatty acid supplementation on insulin sensitivity in horses. Journal of Equine Veterinary Science 33:446-453.
- Hess, T. M.; Rexford, J. K.; Hansen, D. K.; Harris, M.; Schauermann, N.; Ross, T.; Engle, T. E.; Allen, K. G. D. and Mulligan, C. M. 2012. Effects of two different dietary sources of long chain omega-3, highly unsaturated fatty acids on incorporation into the plasma, red blood cell, and skeletal muscle in horses. Journal of Animal Science 90:3023-3031.
- Jeffcott, L. B.; Field, J. R.; McLean, J. G. and O'Dea, K. 1986. Glucose tolerance and insulin sensitivity in ponies and standardbred horses. Equine Veterinary Journal 18:97-101.
- Kaur, G.; Cameron-Smith, D.; Garg, M. and Sinlcair, A. J. 2011. Docosapentaenoic acid (22:5n-3): a review of it biological effects. Progress in Lipid Research 50:28-34.
- Kohli, P. and Levy, B. D. 2009. Resolvins and protectins: mediating solutions to inflammation. British Journal of Pharmacology 158:960-971.
- Lardinois, C. K. 1987. The role of omega 3 fatty acids on insulin secretion and insulin sensitivity. Medical Hypotheses 24:243-248.
- Lau, C. S.; Morley, K. D. and Belch, J. J. 1993. Effects of fish oil supplementation on non-steroidal anti-inflammatory drug requirement in patients with mild rheumatoid arthritis-A double-blind placebo controlled study. British Journal of Rheumatology 32:982-989.
- Luo, J.; Rizkalla, S. W.; Boillot, J.; Alamowitch, C.; Chaib, H.; Bruzzo, F.; Desplanque, N.; Dalix, A. M.; Durand, G. and Slama, G. 1996. Dietary (n-3) polyunsaturated fatty acids improve adipocyte insulin action and glucose metabolism in insulin-resistant rats: Relation to membrane fatty acids. Journal of Nutrition 126:1951-1958.
- MacLean, C. H.; Mojica, W. A.; Morton, S. C.; Pencharz, J.; Hasenfeld, R.; Garland, T. W.; Newberry, S. J.; Jungvig, L. K.; Grossman, J.; Khanna, P.; Rhodes, S. and Shekelle, P. 2004. Effects of omega-3 fatty acids on lipids and glycemic control in type II diabetes and the metabolic syndrome and on inflammatory bowel disease, rheumatoid arthritis, renal disease, systemic lupus erythematosus, and osteoporosis. Evidence Report/Technology Assessment No. 89 (Prepared by Southern California/RAND Evidence-based Practice Center, under Contract No. 290-02-0003). AHRQ Publication No. 04-E012-2. Agency for Healthcare Research and Quality, Rockville, MD.
- Manhart, D. R.; Scott, B. D.; Gibbs, P. G.; Honnas, C. M.; Hood, D. M. and Coverdale, J. A. 2009. Markers of inflammation in arthritic horses fed omega-3 fatty acids. Professional Animal Scientist 25:155-160.
- Marx, N.; Kehrle, B.; Kohlhammer, K.; Grub, M.; Koenig, W.; Homback, V.; Libby, P. and Plutzky, J. 2002. PPAR activators as anti-inflammatory mediators in human T lymphocytes: Implications for atherosclerosis and transplantation-associated arterosclerosis. Circulation Research 90:703-710.
- McIlwraith, C. W. 2005. Frank Milne Lecture: From arthroscopy to gene therapy-30 years of looking in joints. Proceedings of the American Association of Equine Practitioners 51:65-113.
- McCann, M.; Moore, J. N.; Carrik, J. B. and Barton, M. H. 2000. Effect of intravenous infusion of omega-3 and omega-6 lipid emulsions on equine monocyte fatty acid composition and inflammatory mediator production in vitro. Shock 14:222-228.
- Meydani, S.; Lichtenstein, A. H.; Cornwall, S.; Meydani, M.; Goldin, B. R.; Rasmussen, H.; Dinarello, C. A. and Schaefer, E. J. 1993. Immunologic effects of national cholesterol education panel step-2 diets with and without fish-derived N-3 fatty acid enrichment. Journal of Clinical Investigation 92:105-113.
- National Research Council - NRC. 2007. Nutrient requirements of horses. 6th rev ed. National Academic Press, Washington, DC.
- Nettleton, J. A. 1991. Omega-3 fatty acids: comparison of plant and seafood sources in human nutrition. Journal of the American Dietetic Association 91:331-337.
- O'Connor, C. I.; Lawrence, L. M.; St Lawrence, A. C.; Janicki, K. M.; Warren, L. K. and Hayes, S. 2004. The effect of dietary fish oil supplementation on exercising horses. Journal of Animal Science 82:2978-2984.
- Pan, D. A.; Lillioja, S.; Milner, M. R.; Kritetos, A. D.; Baur, L. A.; Borgadus, C. and Storlein, L. H. 1995. Skeletal muscle membrane lipid composition is related to adiposity and insulin action. Journal of Clinical Investigation 96:2802-2808.
- Peoples, G. E.; McLennan, L. P.; Howe, P. R. C. and Groeller, H. 2008. Fish oil reduces heart rate and oxygen consumption during exercise. Journal of Cardiovascular Pharmacology 52:540-547.
- Proudman, S. M.; Cleland, L. G. and James, M. J. 2008. Dietary omega-3 fats for treatment of inflammatory joint disease: efficacy and utility. Rheumatic Disease Clinic of North America 34:469-479.
- Ralston, S. L. 1996. Hyperglycemia/hyperinsulinemia after feeding a meal of grain to young horses with osteochondritis dissecans (OCD) lesions. Pferdeheilkunde 12:320-322.
- Rasic-Milutinovic, Z.; Perunicic, G.; Pljesa, S.; Gluvic, Z.; Sobajic, S.; Djuric, I. and Ristic, D. 2007. Effects of n-3 PUFA supplementation on insulin resistance and inflammatory biomarkers in hemodialysis patients. Renal Failure 29:321-329.
- Renier, G.; Skamene, E.; de Sanctis, J. and Radzioch, D. 1993. Dietary n-3 polyunsaturated fatty acids prevent the development of artherosclerotic lesions in mice: modulation of macrophage secretory activities. Journal of Artherosclerosis and Thrombosis 13:1515-1524.
- Ross-Jones, T.; Hess, T. M.; Rexford, J.; Ahrens, N.; Engle, T. and Hansen, K. 2014. Effects of omega-3 long chain polyunsaturated fatty acid supplementation on equine synovial fluid fatty acid composition and prostaglandin E2. Journal of Equine Veterinary Science 34:779-783.
- Serhan, C.; Gotlinger, K.; Hong S. and Arita, M. 2004. Resolvins, docosatrienes, and neuroprotectins, novel omega-3-derived mediators, and their aspirin-triggered endogenous epimers: an overview of their protective roles in catabasis. Prostaglandins and other Lipid Mediators 73:155-172.
- Serhan, C.; Chiang, N. and Van Dyke, T. 2008. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nature Reviews Immunology 8:349-361.
- Storlien, L. H.; Jenkins, A. B.; Chisholm, D. J.; Pascoe, W. S.; Khouri, S. and Kraegen, E. W. 1991. Influence of dietary fat composition on development of insulin resistance in rats. Relationship to muscle triglyceride and omega-3 fatty acids in muscle phospholipid. Diabetes 40:280-289.
- Stulnig, T. M. 2003. Immunomodulation by polyunsaturated fatty acids: Mechanisms and effects. International Archives of Allergy and Immunology 132:310-321.
- Sun, Y.; Oh, S.; Uddin, J.; Yang, R.; Gotlinger, K.; Campbell, E.; Colgan, S. P.; Petasis, N. A. and Serhan, C. N. 2007. Resolvin D1 and its aspirin-triggered 17R Epimer stereochemical assignments, anti-inflammatory properties, and enzymatic inactivation. Journal of Biological Chemistry 282:9323-9334.
- Tjonahen, E.; Oh, S. F.; Siegelman, J.; Elangovan, S.; Percapio, K. B.; Hong, S.; Arita, M. and Serhan, C. N. 2006. Resolvin E2: identification and anti-inflammatory actions: pivotal role of human 5-lipoxygenase in resolvin E series biosynthesis. Chemistry and Biology 13:1121-1122.
- Treiber, K. H.; Kronfeld, D. S.; Hess, T. M.; Boston, R. C. and Harris, P. A. 2005. Insulin sensitivity and pancreatic β-cell response in the horse: screening proxies and reference quintiles assessed by the minimal model. American Journal of Veterinary Research 66:2114-2121.
- Treiber, K. H.; Kronfeld, D. S.; Hess, T. M.; Byrd, B. M.; Splan, R. K. and Staniar, W. B. 2006. Evaluation of genetic and metabolic predispositions and nutritional risk factors for pasture-associated laminitis in ponies. Journal of American Veterinary Medical Association 228:1538-1545.
- Tu, W. C.; Cook-Johnson, R. J.; James, M. J.; Muhlausler, B. S. and Gibson, R. A. 2010. Omega-3 long chain fatty acid synthesis is regulated more by substrate levels than gene expression. Prostaglandins, Leukotrienes and Essential Fatty Acids 83:61-68.
- Vick, M. M.; Adams, A. A.; Murphy, B. A.; Sessions, D. R.; Horohov, D. W.; Cook, R. F.; Shelton, B. J. and Fitzgerald, B. P. 2007. Relationships among inflammatory cytokines, obesity, and insulin sensitivity in the horse. Journal of Animal Science 85:1144-1155.
- Vineyard, K. R.; Warren, L. K. and Kivipelto, J. 2010. Effect of dietary omega-3 fatty acid source on plasma and red blood cell membrane composition and immune function in yearling horses. Journal of Animal Science 88:248-257.
- Wallace, F.; Miles, E. A.; Evans, C.; Stock, T. E.; Yaqoob, P. and Calder, P. C. 2001. Dietary fatty acids influence the production of Th1-but not TH-2 type cytokines. Journal of Leukocyte Biology 69:449-457.
- Williams, C. M. and Burdge, G. 2006. Long chain n-3 PUFA: plant v. marine sources. Proceedings of the Nutrition Society 65:42-50.
- Woodward, A. D.; Nielsen, B. D.; O'Conner, C. I.; Webel, S. K. and Orth, M. W. 2005. Supplementation of dietary long-chain polyunsaturated fatty acids high in docosahexaenoic acid (DHA) increases plasma DHA concentrations and may increase trot stride lengths in horses. Equine and Comparative Exercise Physiology 4:71-78.
- Zhao, S. M.; Jia, L.W.; Gao, P.; Li, Q. R.; Lu, X.; Li, J. S. and Xu, G. W. 2008. Study on the effect of eicosapentaenoic acid on phospholipids composition in membrane microdomains of tight junctions of epithelial cells by liquid chromatography/electrospray mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis 47:343-350.
Publication Dates
-
Publication in this collection
Dec 2014
History
-
Received
22 May 2014 -
Accepted
01 Oct 2014