Abstract
Mycoplasma gallisepticum (Mg) infection of wild native Brazilian psittacines (Psittaciformes) which died of any cause during sorting, rehabilitation, or conservation, was investigated by PCR. Two previously described PCR methodologies using Mg specific primers were employed for the analyses of 140 swab samples (cloaca, trachea, or palatine cleft). Average positive Mg detection in cloacal swabs was 51.9%, with 80.0% (n=5) of Blue-and-yellow Macaws (Ara ararauna), 60.0% (n=3) Dusky Parrots (Pionus fuscus), 52.5% (n=59) Amazon Parrots (Amazona aestiva), 50.0% (n=2) Orange-winged Parrots (Amazona amazonica), 50.0% (n=2) Jandaya Parakeetsor Jandaya Conures (Aratinga jandaya), 0% (n=2) Golden Conures or Golden Parakeets (Guarouba guarouba), and 0% (n=2) Hyacinth Macaws (Anodorhynchus hyacinthinus). Palatine cleft swab sampling was more sensitive to detect Mg, with 85.4% (n=17) detection rate, as compared to 67.4% (n=46) obtained with tracheal samples, and 53.5% (n=77) with cloacal swabs. The surprisingly high Mg incidence in psittacines kept in conservation or triage environments is possibly due to the proximity or cohabitation with several bird species during confinement and housing psittacines of different origins together. The implementation of biosecurity measures and species-specific facilities is recommended.
Amazona aestiva; Amazona amazonica; Anodorhynchus hyacinthinus; Aratinga jandaya; Brazilian psittacine fauna; Guarouba guarouba; Mycoplasma gallisepticum; PCR; Pionus fuscus; Psittaciformes; Psittacidae
Detection of Mycoplasma gallisepticum in dead captive psittacines in Belo Horizonte, Brazil
Gomes AMI; Costa LLI; Vilela DARII; Marques MVRI; Carvalhaes AGI; Marin SYI; Costa MPI; Horta RSI; Resende JSI; Martins NRSI
ISetor de Doenças das Aves. Departamento de Medicina Veterinária Preventiva. Escola de Veterinária da UFMG
IIInstituto Brasileiro de Meio Ambiente e Recursos Naturais Renováveis, IBAMA
Mail Address Mail Address: Nelson RS Martins Avenida do Contorno, 8.121 Cidade Jardim 30.110-120. Belo Horizonte, MG, Brazil Phone: 55 31 3409-2093 E-mail: rodrigo@vet.ufmg.br
ABSTRACT
Mycoplasma gallisepticum (Mg) infection of wild native Brazilian psittacines (Psittaciformes) which died of any cause during sorting, rehabilitation, or conservation, was investigated by PCR. Two previously described PCR methodologies using Mg specific primers were employed for the analyses of 140 swab samples (cloaca, trachea, or palatine cleft). Average positive Mg detection in cloacal swabs was 51.9%, with 80.0% (n=5) of Blue-and-yellow Macaws (Ara ararauna), 60.0% (n=3) Dusky Parrots (Pionus fuscus), 52.5% (n=59) Amazon Parrots (Amazona aestiva), 50.0% (n=2) Orange-winged Parrots (Amazona amazonica), 50.0% (n=2) Jandaya Parakeetsor Jandaya Conures (Aratinga jandaya), 0% (n=2) Golden Conures or Golden Parakeets (Guarouba guarouba), and 0% (n=2) Hyacinth Macaws (Anodorhynchus hyacinthinus). Palatine cleft swab sampling was more sensitive to detect Mg, with 85.4% (n=17) detection rate, as compared to 67.4% (n=46) obtained with tracheal samples, and 53.5% (n=77) with cloacal swabs. The surprisingly high Mg incidence in psittacines kept in conservation or triage environments is possibly due to the proximity or cohabitation with several bird species during confinement and housing psittacines of different origins together. The implementation of biosecurity measures and species-specific facilities is recommended.
Keywords:Amazona aestiva, Amazona amazonica, Anodorhynchus hyacinthinus, Aratinga jandaya, Brazilian psittacine fauna, Guarouba guarouba, Mycoplasma gallisepticum, PCR, Pionus fuscus, Psittaciformes, Psittacidae.
INTRODUCTION
Blue-and-yellow Macaws (Ara ararauna), Dusky Parrots (Pionus fuscus), Amazon Parrots (Amazona aestiva), Orange-winged Parrots (Amazona amazonica), Jandaya Parakeets or Jandaya Conures (Aratinga jandaya), Golden Conures or Golden Parakeets (Guarouba guarouba) and Hyacinth Macaws (Anodorhynchus hyacinthinus) are psittacines of the Brazilian avian fauna (Brettas & Sigrist, 2007). Mycoplasma gallisepticum (Mg) is one of the most important avian pathogens, responsible for chronic respiratory disease, which may cause conjunctivitis, tracheitis, pneumonia, air sacculitis, arthritis, infertility, embryonic death, and death (Ley, 2003). The eradication of infections by Mycoplasma gallisepticum, M. synoviae, and M. meleagridis is legally mandatory in Brazil and it is also adopted by the Brazilian poultry industry to comply with international trade requirements. Mycoplasmosis has a huge economic impact, and biosecurity is essential for the maintenance of mycoplasma-free flocks. Although there is extensive literature on mycoplasmas in commercial poultry (Ley, 2003), information relative to the Brazilian avian fauna is scarce.
The routine diagnosis of diseases of wild captive birds in our laboratory has shown a high frequency of respiratory diseases. This study aimed to investigating the occurrence of Mg in psittacines that died of any cause during quarantine in the triage center CETAS (Brazilian Institute of Natural Environment, IBAMA, Instituto Brasileiro de Meio Ambiente e Recursos Naturais Renováveis, Belo Horizonte) and in captivity at the ecological park Parque Ecológico Fazenda Vale Verde, Minas Gerais, Brazil, using partial Mg DNA detection by PCR.
MATERIALS AND METHODS
One hundred and forty (140) biological samples of dead psittacines, being 77 cloacal, 46 tracheal and 17 palatine cleft swabs, of 77 individuals maintained in captivity were collected between December 2007 and August 2008. Samples were submitted to the Avian Diseases Laboratory, School of Veterinary Medicine, UFMG, Brazil for diagnosis.
The applied DNA extraction and Mg PCR protocols were previously described (OIE, 1992), except for proteinase K DNA purification. Standard Mg DNA samples were obtained from a commercial lyophilized vaccine strain MG-70 (Myco-Galli MG70, Biovet, Brazil), reconstituted to contain 107.52 CFU/100µl, and from an inactivated oil-emulsion R-980 vaccine (MG-BAC, Fort Dodge, USA), and stored at -20ºC. The oil-based vaccine emulsion was broken down by freezethawing. Swab samples were transferred to Eppendorf tubes containing 500µl of PBS 160mM (pH 7,2-7,4)-Tween 0,05% and maintained overnight at 4ºC. Microtubes containing swabs were centrifuged, the swabs were discarded, and 1µl proteinase K (20mg/mL) and 100µl of 10mM-Tris-EDTA-SDS (sodium dodecyl sulfate at 2,5% in TE) were added. Tubes were incubated for 15 min at 37 ºC with 100µl 8mM ammonium acetate, kept for 15 min at room temperature (25ºC), and centrifuged at 14,000 x g/ 5min. The supernatant was transferred to a fresh tube, 420µl isopropanol at 6ºC were added, gently mixed, and the tubes were centrifuged. The resulting sediment was washed with 800µl ethanol and eluted in 25µl 10mM Tris-EDTA. In each reaction tube, 100ng DNA in 2µl autoclaved Milli-Q water were added to the 48µl premix (10mM Tris-HCl, 50mM KCl, 1.5mM MgCl2), containing 1µl 10mM dNTP, 1 unit (0.2µl) of Taq polimerase (Phoneutria, Brazil) and, for the OIE protocol (OIE, 1992), 1µl of 10 mol forward MG 1 (5'-GAGCTAATCTGTAAAGTTGGTC-3') and 10 mol of reverse MG 2 (5'-GCTTCCTTGCGGTTAGCAAC-3'). Products were amplified at 94ºC/5min., 35 cycles of 94ºC/1min., 55ºC/1min., 72ºC/2min., and final extension was carried out at 72ºC for 5min. The PCR protocol, using the forward primer MG-f 5'-GGATCCCATCTCG ACCACGAGAAAA-3' and the reverse primer MG-r 5'-CTTTCAATCAGTGAGTAACTGATGA-3', was previously described (Silveira et al., 1996), and a 732bp product was used to confirm the reactions. PCR products were detected in 1% agarose gel electrophoresis in TBE (1x) performed at 100v/40min, stained with ethidium bromide (6,0ng/mL), and visualized under UV light. The generated product sizes were estimated according to a molecular weight marker ladder at 1µg/µl (Invitrogen, USA).
RESULTS AND DISCUSSION
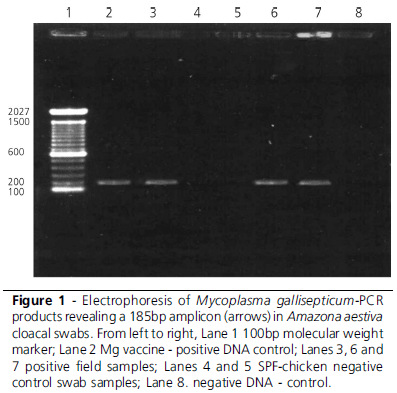
The expected 185bp product was obtained using the OIE protocol, as described in literature (OIE, 1992), as well as the 732bp product of the confirmatory PCR protocol (Silveira et al., 1996). Both PCR protocols presented similar efficiency in the detection of Mg DNA. However, Mg DNA detection in palatine cleft swabs was more successful, obtaining 85.4% (14/17) positive results, as compared to 67.4% in tracheal and 53.5% (41/77) in cloacal swabs. The sensitivity of the swab sampling sites is presented in Table 1. The highest sensitivity was obtained with palatine cleft swabs (82,4%; n=17), followed by tracheal (67,4%; n=46) and cloacal (53,5%; n=77) swabs. Mg infection is primarily a respiratory infection, although it may commonly develop to a systemic disease. The higher detection rate obtained in the palatine cleft may indicate the early infection of the upper respiratory tract, or expectoration of tracheal mucus.
The results of the two previously described PCR methodologies (OIE, 1992 & Silveira, 1996), each protocol using different Mg specific primers, were similar. More samples (59/77) were obtained from the Amazon Parrot, also the most abundant species in captivity and most frequently illegally captured, which might favor Mg transmission. Insufficient samples were obtained for an epidemiological study with other species, such as Blue-and-yellow Macaw (n=5), Dusky Parrot (n=3), Orange-winged Parrot (n=2), Jandaya Parakeet (n=2), which presented high infection rates of80%, 60.0%, 50.0%, 50.0%, respectively. Mg was not detected (0%) in the Golden Parakeet (n=2) and Hyacinth Macaw (n=2). The highest detection rate in Blue-and-yellow Macaw may be due to the fact that this macaw species is the most successfully bred, abundant, and frequently exchanged amongst breeders. On the other hand, Mg was not detected in the Golden Parakeet and Hyacinth Macaw possibly because the low number of tested individuals and the much less frequent exchange among breeders. Capture and captivity conditions may play a role in the respiratory transmission of the disease amongst birds of different origins and health status by inhalation of infected aerosols, and may account for the high degree of infection in Amazona aestiva and Ara ararauna. Due to the high detection rate of Mg in psittacines and its importance of in birds, this pathogen may be partially responsible for the observed mortality. The incidence of Mg in psittacines in captivity may also be relevant for the poultry industry and worth considering in terms of biosecurity.
Click to enlarge

Although respiratory signs, apathy, wasting, and anorexia may indicate Mg infection, other conditions, such as stress and immunodepression, trauma, and malnutrition, commonly affect captured birds. Therefore, it was not possible to directly correlated the observed clinical signs with Mg, despite its detection. A low correlation between Mg infection and clinical signs was also previously described in Melopsittacus undulatus (Budgerigar), Amazona auropalliata (Yellow-naped Parrot), and Carpodacus mexicanus (House Finch) experimentally inoculated with Mg chicken isolates (Farmer et al., 2005). Mg can be found in the respiratory system of normal birds (Yoder, 1998), which may be reservoirs. Fecal-oral habits may also play a role in the transmission and dissemination of Mg (Lemos et al., 2007), and the risk of infection can be associated with the proximity with free-living urban or peri-urban birds and free-range backyard birds (O'Connor et al., 1999; Nolan et al., 2000). Conjunctivitis by Mg in Carpodacus mexicanus in Maryland and Georgia, as diagnosed by PCR, indicated the disseminative potential of this pathogen (Ley et al., 1996).
Respiratory infections are frequent in psittacines in captivity (Bozeman et al., 1984). All materials investigated in the present study were obtained from dead birds, and the detection of Mg suggest that it may have been partially involved in the infection of these birds. Other infectious agents may have been present, including Chlamydophila psittaci and Aspergillus spp., which were previously detected in Amazona aestiva and other psittacines in our laboratory (results not shown). The high occurrence of Mg in psittacines may be a consequence of the proximity or cohabitation with several other bird species during confinement, including passerine species, as previously described (Luttrell et al., 2001; Farmer et al., 2005). Free-living pigeons are regularly seen in the vicinity of captivity facilities, and Mg was previously detected in Columbiformes (Bradbury, 1998) by PCR, but not by serology (Keymer et al., 1983). The transmission to and from a few common species of passerines and free-range chickens in the region may also have been possible. Considering the higher susceptibility of psittacines to Mg as compared to chickens (Bozeman et al., 1984), especially in coinfections, it is essential to implement biosecurity in captive bird environments, including separation by species, dedicated workers, the use of protective garments, and regular monitoring. In industrial poultry production, broilers, turkeys or other industrial birds are reared in bird-proof facilities, preventing the entrance of free-living passerines and maintaining adequate distance from other birds.
Mg may pose an important disease challenge for the conservation and breeding of wild and captive birds, including companion birds. It impairs rehabilitation, it must be controlled to reduce the mortality of embryos as well as of young and mature birds. Mg infection also directly affects fertility and reproduction, reducing hatchability and livability of young birds (Ley, 2003), thereby compromising the development of healthy breeders.
ACKNOWLEDGEMENTS
The authors are indebted to IBAMA and Vale Verde Parque Ecológico Fazenda for biological materials and CNPq, FAPEMIG and Fundação de Ensino e Pesquisa em Medicina Veterinária e Zootecnia for financial support.
Arrived: September/2009
Approved: April/2010
- Bozeman LH, Kleven SH, Davis RB. Mycoplasma challenge studies in budgerigars (Melopsittacus undulatus) and chickens. Avian Diseases 1984; 28:426-34.
- Bradbury JM. Recovery of mycoplasmas from birds. In: Miles R, Nicholas R. Methods in molecular biology: mycoplasma protocols. New York: Humana Press; 1993; 104:45-51.
- Brettas EP, Sigrist T. Aves do Brasil oriental: birds of eastern Brazil. São Paulo: Ed. Avisbrasilis; 2007. 448p.
- Farmer KL, Hill GE, Roberts SR. Susceptibility of wild songbirds to the house finch strain of Mycoplasma gallisepticum Journal of Wildlife Diseases 2005; 41:317-325.
- Keymer IF, Leach RH, Clarke RA, Bardsley ME, Mcintyre RR. Isolation of Mycoplasma spp. from racing pigeons (Columba livia). Avian Pathology 1984; 13:65-74.
- Lemos M, Fuki LT, Barreto ML, Nascimento ER. Prevalência de micoplasmas em aves de rapina no estado do Rio de Janeiro. Anais do 1ş Encontro Internacional de Medicina de Conservação; 2007; Vitória, Espírito Santo. Brasil. p.25.
- Ley DHJ, Berkhoff JE, Mclaren JM. Mycoplasma gallisepticum isolated from house finches (Carpodacus mexicanus) with confunctivitis. Avian Diseases 1996; 40:480-483.
- Ley DH. Mycoplasma gallisepticum infection. In: Saif YM, Barnes HJ, Glisson JR, Fadly AM, McDougald LR, Swayne DE, editors. Diseases of poultry. Ames: Iowa State Press; 1996; p.722-744.
- Luttrell MP, Stallknecht DE, Kleve SH, Kavanaugh DM, Corn JL, Fischer JR. Mycoplasma gallisepticum in house finches (Carpodacus mexicanus) and other wild birds associated with poultry production facilities. Avian Diseases 2003; 45:321-329.
- Nascimento ER. Micoplasmoses. In: Berchieri Jr A. Macari M, editors. Doenças das aves. Campinas: FACTA; 2000. p. 217-224.
- Nolan PM, Duckworth RA, Hill GE, Roberts SR. Maintenance of a captive flock of house finches free of infection by Mycoplasma gallisepticum Avian Diseases 2000; 44:948-952.
- O'connor RJ, Turner KS, Sander JE, Kleven SH, Brown TP, Gómez Jr L, Cline JL. Pathogenic effects on domestic poultry of a Mycoplasma gallisepticum strain isolated from a wild house finch 1999. Avian Diseases 2000; 43:640-648.
- OIE Office International des Epizooties. Manual of standards for diagnostic tests and vaccines for list A and B diseases of mammals, birds and bees. 2Ş ed. Paris; 1992.
- Silveira RM, Fiorentin L, Marques EK. Polymerase chain reaction optimization for Mycoplasma gallisepticum and M. synoviae diagnosis. Avian Disease 1996; 40:218-222.
- Yoder Jr HW. 1998. Avian mycoplasmosis. In: Hofstadt MS, editor. Diseases of poultry. 8th ed. Ames: Iowa State University Press; 1998. p.187-200.
Mail Address:
Publication Dates
-
Publication in this collection
11 Aug 2010 -
Date of issue
June 2010
History
-
Accepted
Apr 2010 -
Received
Sept 2009