Abstract
This study was conducted with the objective of evaluating the use of concrete or hard-packed dirt floor in broiler houses. This experiment was carried out in two different phases. The following performance parameters were studied: live weight, feed intake, feed conversion ratio, and mortality. Litter moisture, pH and temperature were measured. Litter residual contamination after cleaning and disinfection was also evaluated. A dry bulb thermometer, a wet bulb thermometer, and a black bulb thermometer were placed inside each broiler house at bird height and outside the broiler house for data collection. Environmental data were collected at 3h intervals from 00:00 to 24:00 hours during weeks 4, 5, and 6 of the grow-out. Based on the collected data, air relative humidity (RH) was determined, after which wet bulb globe temperature (WBGT) and Radiant Heat Load (RHL) were calculated. There were no differences in live performance parameters. However, total mortality and sudden death were higher in birds raised on dirt floor. On days 0, 14 and 35, litter pH was higher in the dirt floor as compared to the concrete floor, but at the end of the grow-out, this difference disappeared. There was a cubic effect of bird age on litter moisture, which increased up to day 28, and then stabilized or decreased. Litter coliform contamination was higher at the end of the grow-out as compared to that found at housing, but it was not influenced by floor type. The general thermal comfort of broiler raised on dirt floor was similar to that of broilers raised on concrete floor.
Coliforms; litter pH; litter temperature; live performance; thermal comfort
Effect of floor type (dirt or concrete) on litter quality, house environmental conditions, and performance of broilers
Abreu VMN; Abreu PG de; Jaenisch FRF; Coldebella A; Paiva DP de
Embrapa Suínos and Aves
Mail Adress
ABSTRACT
This study was conducted with the objective of evaluating the use of concrete or hard-packed dirt floor in broiler houses. This experiment was carried out in two different phases. The following performance parameters were studied: live weight, feed intake, feed conversion ratio, and mortality. Litter moisture, pH and temperature were measured. Litter residual contamination after cleaning and disinfection was also evaluated. A dry bulb thermometer, a wet bulb thermometer, and a black bulb thermometer were placed inside each broiler house at bird height and outside the broiler house for data collection. Environmental data were collected at 3h intervals from 00:00 to 24:00 hours during weeks 4, 5, and 6 of the grow-out. Based on the collected data, air relative humidity (RH) was determined, after which wet bulb globe temperature (WBGT) and Radiant Heat Load (RHL) were calculated. There were no differences in live performance parameters. However, total mortality and sudden death were higher in birds raised on dirt floor. On days 0, 14 and 35, litter pH was higher in the dirt floor as compared to the concrete floor, but at the end of the grow-out, this difference disappeared. There was a cubic effect of bird age on litter moisture, which increased up to day 28, and then stabilized or decreased. Litter coliform contamination was higher at the end of the grow-out as compared to that found at housing, but it was not influenced by floor type. The general thermal comfort of broiler raised on dirt floor was similar to that of broilers raised on concrete floor.
Keywords: Coliforms, litter pH, litter temperature, live performance, thermal comfort.
INTRODUCTION
Considering broiler health, management and comfort, concrete floors are recommended for broiler houses. However, most farmers do not use this floor type due to its high building cost, and prefer to use hard-packed dirt floors. This recommendation is, in fact, empirical, as there few research studies specifically on this issue. The arguments against the use of dirt floors are that they cause thermal discomfort and impair broiler performance and house disinfection. However, a study published in Selecciones Avícolas (1996) showed that the litter on dirt floor presented 8-10% less humidity and lower fermentation and produced less ammonia, as compared to the concrete floor. According to Fiorentin (2006), most of the poultry houses in the south of Brazil have dirt floors, and farmers using this floor type have reported better litter quality relative to concrete floor in similar poultry houses. The arguments of the former EurepGAP (currently GLOBALGAP), however, is that dirt floors are more difficult to disinfect.
One of the main critical points of poultry house disinfection is the disinfection of hard-packed floors, as it is difficult to remove all the organic matter from the surface, which reduced the antimicrobial action of most disinfectants (Salle & Silva, 2000). However, lime application was shown to effectively disinfect dirt floors, according to Brito et al. (2006) and Daí Prá et al. (2009).
In fact, the biggest problem is the lack of information on this subject in international literature. Fiorentin (2006) asserts that the floor and the litter also directly affect animal welfare, as shown by the international requirements of dry litter and substrate allowing poultry to perform their natural dust-bathing behavior. The use of concrete floor on one hand, may allow better disinfection, but on the other hand, as it generates wetter litter, may not allow dust-bathing, and therefore may be the reason of non-compliance to animal welfare requirements.
There are also lacking in literature studies showing the effect of poultry house floor type on bird performance and house thermal environment. In broiler production, three environmental factors are extremely important: temperature, relative humidity, and ventilation. It must be taken into account that birds change their environmental requirements with age. The thermal environment can be classified according to comfort indexes, allowing determining if the environment is adequate to desired animal production activity (Conceição et al., 2008). According to Bueno & Rossi (2006), the success or failure of broiler production is directly linked to the environmental conditions to which they are submitted.
Randón et al. (2004) assert that the essential parameters of the facilities microclimate are air temperature, air relative humidity, and litter moisture. The optimal air temperature in the broiler house must be 30 to 33 ºC during the brooding period and be gradually reduced to 20-16 ºC at the end of the grow-out. Air relative humidity must be maintained between 60-70%, with an optimal value 65% during the entire rearing period. In order to maintain thermal balance between the broilers and the environment, special care must be taken with the litter due to the direct contact of birds with the litter. Therefore, litter temperature must be similar to the air temperature required to provide comfort to the birds. According to those authors, there are no information in literature as to maximum litter humidity, but it is assumed it should be as low as possible, because high litter moisture deteriorates its mechanical and thermal properties, negatively affecting the perception of thermal environmental conditions. However, Daí Pra et al. (2009) argue that litter should not generate excessive dust, nor retain excessive humidity, preventing the proliferation of microorganisms.
Another important litter parameter is pH, which has been used in several studies on the importance of ammonia volatization and on the diversity of litter microbial population (Terzich et al., 2000; Pope & Cherry, 2000; Line, 2002; Oliveira et al., 2003; Fries et al., 2005; Iwanczuk-Czernik et al., 2007; Lovanh et al., 2007 and Daí Prá et al., 2009).
As discussed above and due to the little information in literature on broiler house floor types, this study aimed at evaluating the effects of the use of concrete floor or hard-packed dirt floor in broiler houses on litter quality, live performance, and thermal comfort of broilers.
MATERIALS AND METHODS
The experiment was carried out during two different seasons (on 22/Aug/2002 to 03/Oct/2002 winter and on 24/Oct/2002 to 05/Dec/2002- spring) in four 12m x 10m broiler houses, each divided in four pens of 20m2 each, housing 250 birds, totaling 4,000 per studied season. Broiler houses were new, and had never housed any birds. Two houses had concrete floors, and two had hard-packed dirt floors.
The following treatments were applied: concrete floor or dirt floor. A 10cm deep wood-shavings litter was placed on all houses.
Straight-run (50% males and 50% females) ROSS broilers were used. Birds and feed were weekly weighed and the following performance parameters were measured: live weight, weight gain, feed intake, and feed conversion ratio. General mortality was daily recorded and classified as sudden death, ascitis, or other causes.
Litter moisture and pH were determined by collecting five litter samples per pen. Samples were homogenized. An 80g subsample was removed from each sample and dried in an oven at 105ºC for 24h to determine litter moisture. A subsample of 10g of that sample was collected, and 100mL of distilled water were added. The sample was agitated six times for 30min, and pH was read using a pHmeter (Brasil, 2007).
Litter residual contamination after cleaning and disinfection was determined as follows. After cleaning, houses were disinfected with a quaternary ammonium product, and the houses remained closed for 10 days. Two days before chicks were housed, new wood-shavings litter was spread on the floor and equipment (drinkers, feeders, brooders) were fitted. The entire internal house environment was again disinfected and fumigated with formalin and potassium permanganate (Barro, 1994). Litter samples were collected in five sites in each pen, placed in sterile flasks, and homogenized to obtained a 25g pooled sample per pen. Samples were later processed in the laboratory of the Animal Health Diagnosis Center (CEDISA). Coliform (CFU/mL) were counted in Petrifilm plates, which are commercial kits for the recovery of the studied microorganisms. Coliforms were used as bacteriological indicators, and include non-sporulated facultative aerobic Gram-negative and Gram-negative bacilli that ferment lactose with gas production in 48 hours at 35ºC. Hygiene indicators included Escherichia coli and Enterobacter aerogens. Samples were collected at two different times: two hours before chicks were housed (1st period) and after broilers were removed from the house, when the litter had been used for 42 days (2nd period) (Siqueira, 1995).
In order to evaluate house thermal conditions, a dry-bulb thermometer, a wet-bulb thermometer, and a black-globe thermometer were place at the center of each pen at the birds' height, and in the external environment. Data were collected every three hours from 0-24h when birds were four, five, and six weeks of age. Based on the data collected at each time, air relative humidity (RU) was determined, and wet bulb globe temperature (WBGT) and radiant thermal load (RHL) were calculated.
Bird performance, litter pH and moisture data were analyzed using the theory of mixed models for repeated measures, considering the effects of season, treatment, bird age, and the interaction between these two parameters, as well as 16 variance and covariance matrix structures using PROC MIXED of SAS statistical package (2003), according to Xavier (2000). The structure used in the analysis was chosen based on the lowest value of the Akaike Information Criterion (AIC). The maximum restricted verisimilitude was used as estimation method.
The details of the analyses of pH and moisture for the effect of age were calculated by the analysis of regression of orthogonal polynomials.
Coliform CFU data were log transformed (y + 1) and analyzed as mentioned above, replacing the factor bird age by the factor related to evaluation period (before and after each grow-out).
Mortality data, as they present binomial distribution, were analyzed by logistic regression, using the LOGISTIC procedure of the SAS statistical package (2003), considering the effects of season, treatment, and the interaction between season and treatment. Internal environment data were submitted to harmonic analysis, according to Morettin & Toloi (2004), applying model (1):
where: yt is the observed value of the parameters in time t; µ, mean; R, range; ϕ, phase angle; ω, frequency, fixed in π/12; and t, random component. In order to make the estimation of the parameters of model (1) easier, model (2) was adjusted as:
where: R=  and ϕ = arctg (-B/A) where, R=
and ϕ = arctg (-B/A) where, R=  and ϕ = arctg (-B/A). In order to verify the effect of treatments on parameters µ, R and ϕ, their estimates were calculated for each combination of treatment, season, and week. Based on these estimates, the effects of season, treatment, week, and the interaction between the last two factors parameters µ, R and ϕ were evaluated using the theory of mixed models for repeated measures and 16 variance and covariance matrix structures using PROC MIXED of SAS statistical package (2003), according to Xavier (2000). External environmental parameters were submitted to harmonic analysis, estimating the parameters µ, R and ϕ per week, and finally, the general mean of the parameters was calculated to compare the internal and external environmental parameter curves.
and ϕ = arctg (-B/A). In order to verify the effect of treatments on parameters µ, R and ϕ, their estimates were calculated for each combination of treatment, season, and week. Based on these estimates, the effects of season, treatment, week, and the interaction between the last two factors parameters µ, R and ϕ were evaluated using the theory of mixed models for repeated measures and 16 variance and covariance matrix structures using PROC MIXED of SAS statistical package (2003), according to Xavier (2000). External environmental parameters were submitted to harmonic analysis, estimating the parameters µ, R and ϕ per week, and finally, the general mean of the parameters was calculated to compare the internal and external environmental parameter curves.
RESULTS AND DISCUSSION
The results of feed intake, feed conversion ratio, weight gain, and body weight were not significantly influenced (p>0.05) by treatment (type floor) or by the interaction between age and treatment, except for feed conversion ratio, which was significantly affected by this interaction (p<0.05). The main effects of age and of season significantly (p<0.05) affected all parameters. Despite the significant effect of the interaction between treatment and age on feed conversion ratio, there was no significant effect of treatment in all evaluated ages (Table 1).
Figure 1 illustrates broiler performance as a function of treatment and age, showing that floor type does not influence bird performance.
In the absence of literature data to compared these results, we decided to use the genetic line standards to determine if broiler performance was influenced or not by floor type. Birds raised on both floor types exceeded in 115g the standard live weight for straight run flocks (2.225g). Also, the obtained feed conversion ratios were better than the genetic line standard (1.74). Therefore, it is possible to conclude that there was no influence of floor type on the performance of the evaluated broilers.
The analyses showed that the main effects of age and season were significant (p<0.05) for litter pH and moisture. There was no influence (p>0.05) of treatments (floor type) on these parameters, but there was a significant effect of the interaction between treatment and age on litter pH (Table 2). The details of this interaction showed a quadratic effect of bird age on litter pH in both treatments, with pH reduction with bird age (Figure 2).
When treatments were compared within age, it was observed that only on days 0, 14, and 35, the litter on dirt floor presented significantly higher pH (p<0.05) as compared to the litter on the concrete floor (Figure 2), but this difference disappeared at the end of the grow-out, when the pH of the litters on both floors were statistically similar. Litter moisture presented a cubic effect of age, showing that litter moisture increases up 28 days of age, and stabilizes or is reduced after this age (Figure 2).
Average litter moisture values were similar between concrete floor (30.37%) and dirt floor (30.50%), with the lowest values (15.62% for concrete floor and 16.82% for dirt floor) obtained in the beginning of the grow-out, and the highest obtained on day 28 (41.37% for concrete floor and 42.11% for dirt floor). This result is opposed to the findings of Kunkle et al (1981), who studied the influence of floor type on litter moisture, and concluded that it was higher in the concrete floor (24.6%) than in the dirt floor (22.3%).
Literature presents very different results. Randón et al. (2004) comment that there are no literature information on maximum recommended litter moisture and that it should be as low as possible. However, Daí Pra et al. (2009) argue that litter moisture should not be too low, in order not to generate dust, and at the same time, it must prevent the proliferation of microorganisms. Those authors find Salmonella spp, Escherichia coli, Listeria, Campylobacter or toxigenic Staphylococcus spp in litter with 22% moisture. On the other hand, wet litter poses a high risk for the presence of spores and conidia of pathogenic fungi and of mycotoxins.
Benito (1991) observed that litter moisture increases with bird age, and recommends a maximum level of 40%. Starting with initial moisture levels of 10-15%, final moisture may reach 25-50%. According to Almeida (1986), litter moisture content should not exceed 35%, because at higher levels, litter is rapidly broken down, cakes, and contribute for high environmental ammonia levels, leading to respiratory diseases, lower weight gain, and carcass downgrading, whereas litter moisture levels lower than 20% cause dust problems.
Terzich et al. (2000) made a survey on broiler litter pathogens in 12 regions in the US and found average litter moisture values of 25.1%, with minimum and maximum values of 20.0% and 30.8%, respectively, and average pH of 8.0, ranging between 6.7 and 9.0.
In the present study, average litter pH values were 6.71 for the concrete floor and 6.82 for the dirt floor. The lowest pH values were determined in the beginning of the grow-out, with 4.77 for concrete floor and 5.05 for the dirt floor. The highest pH values were found at the end of the grow-out (42 days), with 8.08 for the litter on the concrete floor and 8.06 for the litter on the dirt floor.
According to Carr et al. (1985), litter moisture lower than 30% and pH lower than 8 reduced broiler house air ammonia levels. Benito (1991) mentioned that ammonia production may be minimal when litter moisture and pH are maintained below 30% and 7.5, respectively, as well as air relative humidity around 50%. According to Terzich (1997), litter pH has a decisive role in NH3 volatilization, and the main ureolytic bacterium (Bacillus pasteurii) cannot grow in neutral pH, but thrives in pH higher than 8.5. Low pH is beneficial for several reasons. Pope & Cherry (2000) comment that ammonia concentration increases with increasing pH. Ammonia release is insignificant when litter pH is below 7.0, it starts to be released when pH is close to 7.0 and reaches high level in litter pH of 8.0 and higher. Moreover, when litter pH is reduced from 8.0-9.0 to values below 3.0, bacterial load is also reduced.
In a deeper analysis on the effects of litter pH on bacterial levels, Terzich et al. (2000) did not find statistical differences, but rather trend lines. These trend lines were placed in scatter plots for each bacterial class, except for coliforms, and tended to increase as pH increased. Although the lowest pH level used in that study was 6.0, those authors mention that other studies found reduction of the bacterial load when litter pH was below 4.0. The correlation analysis showed that total bacterial load was highly correlated with pH. A similar result was not obtained when litter moisture was analyzed, when correlations were not statistically different.
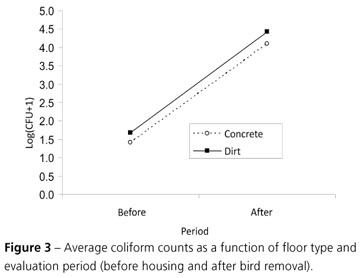
Coliform count analysis of log (y + 1)-transformed CFU showed a significant effect (p<0.05) of season and collection period. At the end of the grow-out, higher coliform CFU values were obtained, independently of treatment (Table 3). Figure 3 illustrates this behavior.
According to Pilotto et al. (2006), total and fecal coliform counts using the technique of the most probable number (MPN), performed after farm disinfection, indicate if the cleaning procedure and products were indeed effective. Total coliform indexes are used to evaluate hygiene conditions, and high counts indicate that cleaning and sanitation were not effective. On the other hand, fecal coliform counts are used as indicators of fecal contamination or health conditions, as this group largely consists of Escherichia coli.
It was demonstrated that even before chicks were housed, the broiler houses were already contaminated by coliforms. This stresses the potential risk of contamination of recently housed chicks, which immune system is not yet fully developed.
All procedures aiming at reducing the conditions that may allow the survival of microorganisms in the environment must be implemented during the grow-out, particularly during the first days after housing, as broilers' immune system is still immature during the first week of life. Logan & Bartlet (2001) evaluated coliform contamination underneath the dirt floor and found 1900 CFU/g (coliform count) 1/4 inch deep and 460 CFU/g (coliform count) 1/2 inch deep. According to Paganini (2002), this evaluation is particularly important considering the scratching behavior of chickens, which may bring these bacteria to the surface. However, in broiler houses with 10 cm deep wood-shavings litter, even though broilers perform their natural scratching behavior, it would be difficult to bring these bacteria to the surface of the litter.
The disinfection procedures used in poultry production do not act deep in the soil, as not enough water is used to penetrate it and the commonly used active compounds have little or no action in the presence of organic matter. This partially explains why it is difficult to eliminate some diseases from the poultry house even after cleaning, disinfection and adequate downtime. Moreover, considering the high environmental resistance of Clostridium spp, which sporulates, deep floor contamination in poultry houses should be taken into account. Pilotto et al. (2006) detected the presence of total and fecal coliform only down to 0,5cm deep in the soil, showing that coliform contamination can be restricted to the top layer of the dirt floor, regardless soil type. Logan & Bartlet (2001) found that after cleaning and disinfection of dirt soils, only a few superficial mm of the floor were relatively clean. Their evaluation of deeper soils samples revealed very high bacterial counts.
The discussion on environmental comfort conditions in the present study is based on optimal conditions during rearing, as we did not find in literature specific studies on the effect of floor type on the rearing environment.
The results of the statistical analyses of air temperature (TBS), wet bulb globe temperature (WBGT), radiant heat load (RHL), air relative humidity (RH) and litter temperature showed that main effects of season and week were significant (p<0.0001) on all parameters, except for WBGT, which was not affected by season (Table 4). Floor type had no influence on any of the environmental parameters evaluated.
As to the intercept (mean parameter values), the main effects of season and week were significant (p<0.05) for all parameters, whereas the effect of the interaction between floor type and week was significant only for air temperature (TBS) and the effect of floor type on litter temperature.
As to phi (phase angle, which determines curve behavior), there was effect of the interaction of floor type and week on TBS, WBGT and RHL, whereas RH was influenced only by weeks (p<0.0002) and litter temperature was affected by season, floor type and week (Table 4).
The intercept represents mean parameter values, which are shown in Table 5. Under optimal rearing conditions, according to Abreu et al. (2007), optimal temperatures are 23ºC 26ºC during week 4, 20ºC 23ºC during week 5, and 20ºC on weeks 6 and 7. Therefore, according to mean temperatures, floor types presented thermal comfort conditions during weeks 4 and 5, and were slightly above the optimal temperature during week 6. Mean air temperature in the houses with concrete floor was 21.16ºC, whereas with the dirt floor, it was 20.86ºC, and was not affected by floor type.
In order to ensure bird welfare, it is essential to have efficient and adequate control of the environmental conditions (Iwanczuk-Czernik et al., 2007).
According to Medeiros et al. (2005), since the beginning of the 20th century, attempts have been made to quantify animal thermal environment using correlations among the parameters temperature, humidity, air velocity and radiation. However, there are other factors that may also interact, and may cause stress, such as radiations, electro-magnetic fields, environmental noise, air pollution compounds. The most frequently used until the 1980s to evaluate animal thermal environment was the temperature humidity index (THI), and in the 1990s, wet bulb globe temperature (WBGT), which has the advantage of incorporating the effects of air temperature, humidity, ventilation, and radiation (Medeiros et al., 2005).
WBGT value obtained with the concrete floor was 70.72, and 70.40 with the dirt floor. According to Teixeira (1983), WBGT values ranging between 65 and 77 are compatible with broiler production. Moraes (2002) found that broilers after 21 days were under heat stress when WBGT was higher than 76 in the period of 12:00 to 16:00h. Therefore, in the present study, both floor types presented similar conditions, which where within the broiler comfort zone. During week 5, WBGT was lower, showing better conditions for the birds. This was also found in the level of radiant heat load (RHL) received by the birds. RHL affected air temperature and WBGT values, and had a favorable influence on relative humidity during week 5. Moraes (2002) concluded that RHL of 450 W m-2 was the upper limit of thermal comfort of adult chickens. Comparing this value with those obtained in the present study, it was observed that both floor types provided RHL values within the comfort range. Air relative humidity (RH) was higher in the houses with concrete floor (84.86%) and also when birds were 4 weeks old (87.81%). In addition, air relative humidity values during weeks 4, 5, and 6 were higher than those considered optimal for broiler rearing, which are about 60-70% (Abreu et al., 2007).
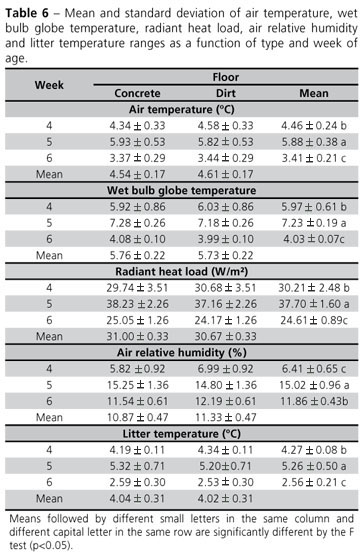
Wide environmental ranges are a challenge that both birds and farmers have to face in order to maintain adequate environmental conditions within the poultry house. Bird comfort demand the narrower range as possible. Temperatures and humidity changes may directly affect bird physiology, with negative effects on feed intake, their capacity to dissipate heat, and may ultimately place the bird's life at risk In the present study, the widest range for all evaluated parameters was observed during week 5 (Table 6).
The statistical analysis results showed lower litter temperature in the dirt floor. Analyzing litter temperature behavior during the different weeks of the grow-out, the results show that the lowest value was obtained during week 5, when, however, variation was widest. Litter temperature presented similar behavior as air temperature, and was in average 3 -4ºC higher than air temperature.
It should be noticed that both air and litter temperatures must be maintained within the birds' comfort range. Broilers try not to sit on the litter when its temperature is high during the last week of the grow-out. One way birds exchange heat with the environment is by conduction. In order to be efficient, litter temperature must be lower than body temperature. Therefore, the high litter temperature observed in the present study during the last week of grow-out, was outside the comfort range, independently of floor type (Abreu et al., 2007).
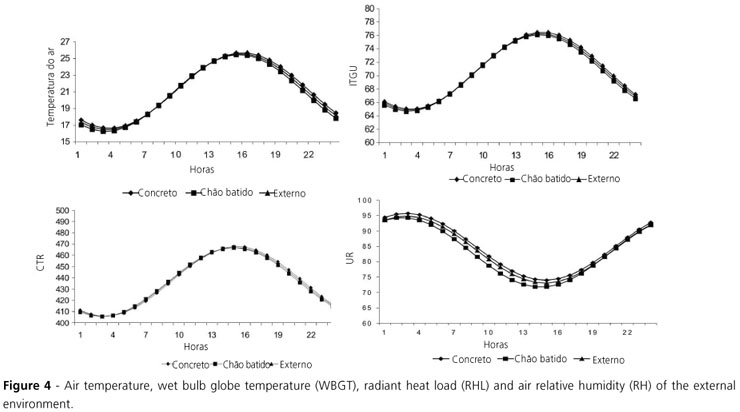
Air relative humidity presented cyclic behavior, opposite to air temperature (Figure 4). This was expected, as high temperature is usually followed by low air relative humidity. Mininal TBS, WBGT, and RHL values were observed at 03:00h and maximal values at 14:00, whereas the opposite occurred with air relative humidity (Figure 4).
Figure 1 shows compares the behavior of the variable curves between the internal and the external environment.
As expected, internal thermal conditions of poultry houses are more beneficial to broilers as compared to the external environment, as shown by the parameter means (intercept values) and the narrower range of these parameters. The best comfort conditions were always observed in the evening and in the morning, whereas in the afternoon, independently of floor type, conditions were worse. Medeiros et al. (2005) found that in environments with WBGT between 69 and 77, broilers were calm, uniformly distributed in the house, and presented high performance. In cold environments (59 67 WBGT), broilers presented 14% daily weight gain reduction and 12.1% higher mortality, in addition to significant changes in physiological parameters. On the other hand, in hot environments (78 88 WBGT), there was no mortality, but daily weight gain and feed intake were reduced in 67% and 43%, respectively.
Studying the effects of temperature and relative humidity on the performance and carcass yield of 1- to 49-day-old broilers, Oliveira et al. (2006) concluded that high environment temperatures impair performance and carcass cuts yield, and that these effects are more significant as air relative humidity increased.
Randon et al. (2004) measured air and litter temperature and humidity for two years, and concluded that it is easier to maintain optimal broiler house temperature when external air temperature was lower than the internal air temperature, because the lack of heat can be supplemented by brooding, whereas excessive heat requires its removal by air exchange. Their measurements showed that internal house temperature remained optimal during winter, spring, and fall, and during some periods in the beginning of the summer. High air temperature and low moisture emission in the beginning of the grow-out (first 10 days) resulted in very low air relative humidity. During the remaining of the grow-out, both during winter and summer, humidity presented considerable fluctuation, with periods of very high or very low levels. However, when both external air temperature and humidity are high, it is impossible to reduce internal house humidity if the air is not dried. Litter temperature showed a characteristic pattern, independently of grow-out week. It was equal to internal air temperature after day 18, and continued to rise, reaching 30-34ºC. At the end of the grow-out, litter temperature slightly decreased or was kept constant. The authors also mentioned that higher litter temperature relative to air temperature may be attributed to a heat source in the litter, which may be litter fermentation or bird presence on the litter. Finally, litter moisture increased during the grow-out. The authors concluded that changes in litter moisture significantly affect moisture emission in the poultry house. Lovanh et al. (2007), studying the changes in the space distribution of the microbial population in broiler litter associated to litter physical-chemical properties, concluded that litter moisture, temperature and pH are the main factors that affect microbial diversity.
Mortality was classified as sudden death, ascitis, other causes and total mortality. Sudden death was significantly (p<0.05) influenced by season and floor type, whereas total mortality was affected only by floor type (Table 6).
Sudden death and total mortality were higher when broilers were kept on the dirt floor (Table 7). The odds ratio analysis showed that sudden death occurred 1.813 times and total mortality 1.729 times more than on concrete floor (Table 8).
CONCLUSIONS
Broiler live performance was not influenced by floor type, but total mortality and sudden death were higher in broilers raised on hard-packed dirt floor. Floor type did not influence litter moisture, pH, or coliform contamination at the end of the grow-out. The general thermal comfort conditions were not different as a function of floor type.
Mail Adress:
VMN Abreu
BR 153 - Km 110
Caixa Postal 21
89.700-000. Concórdia, SC, Brasil
Fone: (49) 441-0400, Fax (049) 441-0497
E-mail: valeria@cnpsa.embrapa.br
Submitted: October/2009
Approved: February/2011
- Almeida MAC. Fatores que afetam a umidade da "cama". Avicultura Industrial 1986; 76:16-18.
- Barro DR. Manejo sanitário e preparo das instalações. In: Pinheiro MR, editor. Manejo de frangos de corte. Campinas: Facta; 1994. p. 21-40.
- Benito LM. La problemática de las camas húmedas en las granjas de broilers. Selecciones Avícolas 1991; 33:437- 452.
- Bueno L, Rossi LA. Comparação entre tecnologias de climatização para criação de frangos quanto a energia, ambiência e produtividade. Revista Brasileira de Engenharia Agricola Ambiental 2006; 10(2):497-504.
- Brasil. Ministério da Agricultura, Pecuária e Abastecimento (MAPA). Manual de métodos analíticos oficiais para fertilizantes minerais, orgânicos, organominerais e corretivos [citado 2007 nov 23]. Disponível em: http://www.agricultura.gov.br
- Brito BG, Tagliari KC, Pinheiro AR, Gomes LM, Berbel MM. Efeito da utilização de cal no controle de Salmonella e Escherichia Coli em cama de criações de frango de corte. Revista Brasileira de Ciência Avícola 2006; 8(Supl):246.
- Campos EJ. Programa de alimentação e nutrição para aves de acordo com o clima-Reprodutoras. Anais do Simpósio Internacional sobre Ambiência e Instalação na Agricultura Industrial; 1995; Campinas, São Paulo. Brasil. p.251-257.
- Carr LE, Malone GW, Wabeck CJ. Broiler production environment associted with using brooding papers. ASAE 1985; p.4024. (Paper 85)
- Conceição MN da, Alves SP, Telatin Júnior A, Silva IJO da, Piedade SMS, Savastano Júnior H, Tonoli G. Desempenho de telhas de escória de alto forno e fibras vegetais em protótipos de galpões. Revista Brasileira de Engenharia Agrícola Ambiental 2008; 12(5):536-539.
- Daí Pra MA, Corrêa EK, Roll VF, Xavier EG, Lopes DCN, Lourenço FF, Zanusso JT, Roll AP. Uso de cal virgem para o controle de Salmonella spp. e Clostridium spp. em camas de aviário. Ciência Rural 2009; 39(4):1189-1194.
- De Abreu PG, Abreu VMN, Coldebella A, Jaenisch FRF, Paiva DP. Condições térmicas ambientatis e desempenho de aves criadas em aviários com e sem o uso de forro. Arquivos Brasileiro de Medicina Veterinária e Zootecnia 2007; 59(4):1014-1020.
- Fioretin LA. Reutilização da cama de aviário no contexto do benchmarking. Avicultura Industrial 2006; 97(6):12-18.
- Fioretin LA. Processos de tratamento para a reutilização de cama de aviário: aspectos bacteriológicos. Anais da Conferência APINCO de Ciência e Tecnologia Avícolas; 2006; Santos. São Paulo. Brasil. p.17-24.
- Fries R, Akcan M, bandick N, Kobe A. Microflora of two different types of poultry litter. British Poultry Science 2005; 46(6):668-672.
- Iwaczuk-Czernik K, Witkowska D, Sowinska J, Wójcik A, Mituniewicz T. The effect of a microbiological and a disinfecting preparation on the physical an chemical properties of litter and the results of broiler chicken breeding. Polish Journal of Natural Science 2007; 22(3):395-406.
- Kunkle WE, Carr LE, Carter TA, Bossard EH. Effect of flock and floor type on the levels of nutrients and heavy metals in broiler litter. Poultry Science 1991; 60:1160-1164.
- Line JE. Campylobacter and Salmonella populations associated with chickens raised on acidified litter. Poultry Science 2002; 81:14731477.
- Logan M, Bartlet S. Evaluation of coliform removal at ˝ inch depth of poultry house floor using Impact-S process. Virginia: Environmental Dynamics; 2001. 41 p.
- Lovanh N, Cook KL, Rothrock MJ, Miles DM, Sistani K. Spatial shifts in microbial population structure within poultry litter associated with physicochemical properties. Poultry Science 2007; 86:18401849.
- Medeiros CM, Baêta FC, OLiveira RFM de, Tinôco IFF, Albino LFT, Cecon PR. Efeitos da temperatura, umidade relativa e velocidade do ar em frangos de corte. Engenharia na Agricultura 2005; 13(4): 277-286.
- Moraes SRP. Caracterização de sistemas de semiclimatização de ambiente, em galpões para frangos de corte, no sudoeste de Goiás [dissertação]. Viçosa (MG): Universidade Federal de Viçosa; 2002.
- Morettin PA, Toloi CMC. Análise de séries temporais. São Paulo: Edgar Blücher; 2004.535p.
- Oliveira COM, Almeida CV, Andrade DO, Rodrigues SMMR. Teor de matéria seca, pH e amônia volatilizada da cama de frango tratada ou não com diferentes aditivos. Revista Brasileira de Zootecnia 2003; 32(4):951-954.
- Oliveira RFM, Donzele JL, Abreu MLT, Ferreira RA, Vaz RGMV, Cella PS. Efeitos da temperatura e da umidade relativa sobre o desempenho e o rendimento de cortes nobres de frangos de corte de 1 a 49 dias de idade. Revista Brasileira de Zootecnia 2006, 35(3):797-803.
- Paganini FJ. Reutilização de cama na produção de frangos de corte: porquê, quando e como fazer. Anais da Conferência APINCO de Ciência e Tecnologia Avícolas; 2002; Campinas, São Paulo. Brasil. p194-206.
- Pilotto F, Klein VA, Wald VB, Rodrigues LB, Santos, LRdos, Colussi F de M, Nascimento VP do. Determinação do volume saturante de solução desinfetante em piso de chão batido de granja avícola em função da carga microbiana (coliformes) e composição do solo. Ciência Rural 2006; 36(6):1837-1841.
- Pope MJ, Cherry TE. Na evaluation of the presence of pathogens on broilers raises on poultry litter treatment® - treated litter. Poultry Science 2000; 79:13511355.
- Randon J, Bieda W, Nawalany G. Broiler house microclimate in light of studies. EJPAU 2004; 7(2) [cited 2004]. Available from: http://www.ejpau.media.pl/volume7/issue2/engineering/art-04.html
- Salle CTP, Silva AB. Prevenção de Doenças, Manejo Profilático, Monitoração In: Berchieri Jr. A, Macari M. Doenças das aves. Campinas: Facta; 2000. p. 3-12.
- SAS Institutec. System for microsoft windows: release 91 [CD-ROM]. Cary; 2002-2003.
- Selecciones Avícolas. Un buen suelo para mucho tiempo. Selecciones Avicolas 1996; 38:97-98.
- Siqueira RS. Manual de microbiologia de alimentos. Rio de Janeiro: Embrapa, Centro Nacional de Pesquisa de Tecnologia Agroindustrial de Alimentos; 1995. p.55-72
- Teixeira VH. Estudo dos índices de conforto em duas instalações de frango de corte para as regiões de Viçosa e Visconde do Rio Branco [dissertação]. Viçosa (MG): Universidade Federal de Viçosa; 1983.
- Terzich MAC. Amônia dos galpões avícolas e o pH da cama. Anais da Conferência de Ciência e Tecnologia Avícolas; 1997; São Paulo, SP. Brasil. p.141-146.
- Terzich MAC, Pope MJ, Cherry TE, Hollinger J. Survey of pathogens in poultry litter in the United States. Applied Poultry Research 2000; 9:287-291.
- Xavier LH. Modelos univariado e multivariado para análise de medidas repetidas e verificação da acurácia do modelo univariado por meio de simulação [dissertação]. Piracicaba (SP): Escola Superior de Agricultura "Luiz de Queiroz", Universidade de São Paulo; 2000.
Publication Dates
-
Publication in this collection
18 July 2011 -
Date of issue
June 2011
History
-
Received
Oct 2009 -
Accepted
Feb 2011