ABSTRACT
In broiler production, the litter is reused for consecutives flocks, and it is treated during down time between flocks to reduce its microbial load. Although covering the litter with a plastic canvas is a common litter treatment in the field, there is little scientific information available on its efficacy. The aim of this study was to evaluate the effects of covering broiler litter with a plastic canvas for eight days on litter microbiological, physical, and chemical parameters, and on the intestinal microbiota and immunity of broilers. In the first trial, reused litter from a previous flock was distributed into three treatments, with six replicates each: L1 (negative control, litter free from Salmonella Enteritidis (SE) and Eimeria maxima (EM) and not covered), L2 (positive control, litter with SE and EM, and not covered), and L3 (litter with SE and EM, and covered with plastic canvas for eight days). Litter total bacteria, Enterobacteria, Lactobacillus, SE, and EM counts, and litter pH, temperature, moisture, and ammonia emission were determined on days 1 and 8. In the second trial, broilers were housed on those litters according to the treatments described above, and their intestinal microbiota, gut CD4+ and CD8+ lymphocytes and macrophages, and liver and intestinal pro-inflammatory interleukin (IFN-γ, IL-1β e IL-18) levels were evaluated on days 14 and 28. A significant reduction of litter bacterial populations was observed in the litter covered with plastic canvas. A significantly higher mRNA IFN-γ gene expression (12.5-fold) was observed in the jejunum and liver of broilers reared on the litter with Enterobacteria counts. No EM reduction was observed in the covered litter. Covering reused broiler litter with plastic canvas reduces initial litter bacterial load as a result of the interaction between physical and chemical parameters.
Keywords:
Ammonia; cytokines; Lactobacillus; macrophages; Salmonella
INTRODUCTION
Litter is a mixture of excreta, litter substrate (e.g., wood shavings, rice hulls, peanut shells), as well as soil, feathers, and other components. It is used to cover broiler house floors to absorb excreta and provide a comfortable surface for the birds (Kelley et al., 1996Kelley TR, Pancorbo OC, Merka WC, Thompson SA, Cabrera ML, Barnhart HM. Elemental concentrations of stored whole and fractionated broiler litter. Journal of Applied Poultry Research 1996;5(3):276-281.).
Broiler litter from the first flock is typically reused for several consecutive flocks (up to 14 times) (Roll et al., 2011Roll VF, Pra MA, Roll AP. Research on Salmonella in broiler litter reused for up to 14 consecutive flocks. Poultry Science 2011;90(10):2257-2262.) for economic reasons, and also to reduce the organic load of broiler production. However, to allow its reutilization, litter needs to be treated during the downtime between flocks to reduce its microbial load (Thaxton et al., 2003Thaxton Y, Balzli CL, Tankson JD. Relationship of broiler flock numbers to litter microflora. Journal of Applied Poultry Research 2003;12(1):81-84.).
Lee et al. (2011Lee KW, Lillehoj HS, Lee SH, Jang SI, Ritter GD, Bautista DA, Lillehoj EP. Impact of fresh or used litter on the posthatch immune system of commercial broilers. Avian Diseases 2011;55(4):539-544.) observed that the exposure of broiler chickens to reused litter increased the intestinal intraepithelial lymphocyte and splenic lymphocyte subpopulations. Shanmugasundaram et al. (2012Shanmugasundaram R, Lilburn MS, Selvaraj RK. Effect of recycled litter on immune cells in the cecal tonsils of chickens. Poultry Science 2012;91(1):95-100.) showed that litter reutilization affected both cell-mediated immune response and cytokine production in broilers, reporting that broilers reared on reused litter expressed more IL-1 and IL-4 cytokines, whereas those reared on new litter expressed more IL-10 cytokine.
The main methods of litter treatment are composting (Macklin et al., 2006Macklin KS, Hess JB, Bilgili SF, Norton RA. Effects of in-house composting of litter on bacterial levels. Journal of Applied Poultry Research 2006;15(4):531-537.) and incorporation of quicklime (Pra et al., 2009Pra MA, Corrêa ÉK, Roll VF, Xavier EG, Lopes DC, Lourenço FF, et al. Uso de cal virgem para o controle de Salmonella spp. e Clostridium spp. em camas de aviário. Ciência Rural 2009;39:1189-1194.), aluminum sulfate (Line, 2002Line JE. Campylobacter and Salmonella populations associated with chickens raised on acidified litter. Poultry Science 2002;81(10):1473-1477.), or sodium bisulfate (Williams et al., 2012Williams ZT, Blake JP, Macklin KS. The effect of sodium bisulfate on Salmonella viability in broiler litter. Poultry Science 2012;91(9):2083-2088.). An alternative method is to wet the litter and cover it with a plastic canvas for a short period of time (seven days). This method is described by Muniz et al. (2014Muniz E, Mesa D, Cuaspa R, Souza A, Santin E. Presence of Salmonella spp. in reused broiler litter. Revista Colombiana de Ciencias Pecuarias 2014;27(1):12-17.), who observed a significant reduction in Salmonella spp. counts in the treated reused litter. However, the authors did not evaluate other bacteria or physical and chemical litter parameters.
Further knowledge on the method of treating used broiler litter with a plastic canvas is needed. The aim of this study was to evaluate the influence of covering used poultry litter with a plastic canvas on broiler body weight, bacterial colonization and intestinal leukocyte recruitment, host gene transcription of inflammatory and immune response genes in the intestine and liver of broilers, as well as on litter microbiological and physicochemical characteristics.
MATERIALS AND METHODS
The study was conducted at the Center for the Study of Immune Responses of Poultry (CERIA-LABMOR), of the Department of Agricultural Sciences, Federal University of Paraná, during the month of June 2013, when average temperature was 14.8°C.
Phase 1
Litter preparation: Litter (wood shavings used for a single flock) from a previous study was used. The study aimed at assessing the impact both of Salmonella Enteritidis (SE) and Eimeria maxima oocysts (EM) on broiler chickens. The protocol of this previous study was 1mL of SE inoculated on day 7 by gavage at concentration of 105 CFU/mL. The SE strain was obtained from the stock of the Laboratory of Microbiology and Avian Pathology of UFPR. Eimeria challenge amounted to 20,000 oocysts/bird on days 1 and 20; the protozoa were donated by Elanco. Those broilers (10 birds per treatment) were reared up to 42 days, ensuring litter contamination.
One day before the beginning of the present experiment, 5L of water were added to the entire litter of the previous flock (84,000 cm3) in order to maintain 30%moisture (Lavergne et al., 2006Lavergne TK, Stephens MF, Schellinger D, Carney WA. In-house pasteurization of broiler litter. Louisiana State University Agricultural Center 2006;2955:1-16.).
A randomized experimental design including three treatments with six replicates each, was applied. Treatments consisted of: L1 (negative control: litter free from SE and EM, not covered); L2 (positive control: litter contaminated with SE and EM, not covered); L3 (litter contaminated with SE and EM, and covered with a 150µm-thick black plastic canvas for eight days).
The six replicates for each treatment were placed on concrete floor blocks (70-cm long, 40-cm wide and 30-cm high each) in two separate rooms per treatment. In order to maintain the separation between the replicates, the plastic canvas was carefully stuck between the litter and the blocks. At the time of sample collection, the canvas was gently removed and immediately placed back.
Litter microbiology: On days 1 and 8 of the trial, 100 g of litter per replicate per treatment were collected. Total bacterial (TB), Lactobacillus (LAC), Enterobacteria (EN) and SE (6 samples per treatment in total) counts were determined. TB, EN and SE counts were performed according to the official method of the Brazilian Ministry of Agriculture (Brasil, 2003Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa nº62, 26 de agosto de 2003. Métodos analíticos oficiais para análises microbiológicas para controle de produtos de origem animal e água. Brasília, DF: MAPA; 2003. p.43.), and LAC counts were determined according to Souza et al. (2007Souza M, Moreira J, Barbosa F, Cerqueira M, Nunes ÁC, Nicoli JR. Influence of intensive and extensive breeding on lactic acid bacteria isolated from Gallus gallus domesticus ceca. Veterinary Microbiology 2007;120(1-2):142-150.).
Oocysts of EM were counted on days 1 and 8 in10 g the litter per replicate per treatment. Samples were diluted in 50 mL distilled water, and filtered through a 70 mesh/cm2 sieve. A drop of the filtered solution (50 µL) was suspended in a Neubauer chamber, and oocysts were counted (Fagonde & Pedroso, 2009Fagonde CA, Pedroso D. Cultivo in vivo, in vitro e diagnostico específico de Eimeria spp. de Gallus gallus. Brasília: Embrapa; 2009. p. 219.).
Litter physical and chemical parameters: Litter temperature was measured daily using a digital thermometer INCOTERM® (Porto Alegre, Brazil) The metal probe was inserted in the center of the litter at a depth of 15 cm. Litter moisture, pH, and ammonia emission were measured on days 1, 3, 5 and 8 of the experiment. The methodology of Benabdeljelil & Ayachi (1996Benabdeljelil K, Ayachi A. Evaluation of alternative litter materials for poultry. Journal of Applied Poultry Research 1996;5(3):203-209.) was adopted to measure the pH, using a digital pH meter model 330i/SET, WTW (Weilheim, Germany). Litter moisture was determined according to Carvalho et al. (2011Carvalho MR, Moura DJ, Souza ZM, Souza GS, Bueno LG. Qualidade da cama e do ar em diferentes condições de alojamento de frangos de corte. Pesquisa Agropecuária Brasileira 2011;46:351-361.), and ammonia emission according to Hernandes & Cazetta (2001Hernandes R, Cazetta J. Método simples e acessível para determinar amônia liberada pela cama aviária. Revista Brasileira de Zootecnia 2001;30:824-829.).
Phase 2
In-vivo trial
The experiment was approved by the Ethics Committee for Animal Experimentation of the Agricultural Sciences Department, Federal University of Paraná, under protocol number 041/2013.
The six replicates from each treatment applied in phase 1 were mixed and homogenized, resulting in three litter groups, which were left to rest for two days before broilers were housed.
Animals: Sixty one-day-old male Cobb®broilers were obtained from a commercial hatchery. Birds were weighed and uniformly distributed according to average body weight into the treatments described in the phase 1. During the 28 days of experiment, birds were provided with adequate environmental temperature for this rearing phase, and received water and feed ad libitum.
Sample collection for microbiology, histopa-thology, immunohistochemistry and quantitative PCR (qPCR): For the quantitative analysis of TB, LAC, EN and presence of SE at 14 and 28 days of age, five chickens per treatment were sacrificed and necropsied for the collection of cecal content. Microbiological analyses were performed using the same methods described for phase 1. Jejunum and cecum samples of five birds per treatment were collected, and fixed in 10% buffered formalin for histology. Fragments of those same segments were embedded in Tissue-Tek® gel for CD8+ and CD4+ lymphocyte and macrophage counts. For gene expression analyses, samples of scrapings of the mucosa of the jejunum and a liver fragment were collected, placed in plastic tubes containing 1 mL of RNA storage solution RNAlater® (Thermo Fisher Scientific Inc., Waltham, USA) and refrigerated until processing. Gene expression of the cytokines interferon gamma (IFN-γ), interleukin 1 beta (IL-1β) and interleukin 18 (IL 18) were determined.
Processing and reading material for histopa-thology and immunohistochemistry: Samples of the jejunum and the cecum were processed routinely for histology and stained with Alcian blue according to Smirnov et al. (2004Smirnov A, Sklan D, Uni Z. Mucin dynamics in the chick small intestine are altered by starvation. The Journal of Nutrition 2004;134(4):736-742.) for goblet cell counting. Twenty fields per treatment were scanned under a light microscope at 40x magnification. For the analysis of CD4+ and CD8+ lymphocytes and macrophages by immunohistochemistry, samples were processed according to Muniz et al., (2013Muniz E, Pickler L, Lourenço MC, Westphal P, Kuritza LN, Santin E. Probióticos na ração para o controle de Salmonella minnesota em frangos de corte Archives of Veterinary Science 2013;18(3):52-60.) and 20 fields were scanned per experimental group, using a light microscope at 100x magnification (Olympus BX41 Olympus USA).
Primers for qPCR: The sequences of primers for the genes cytokines IFN-γ, IL-1β, IL-18, and the three reference genes β-actin, glyceralde-hyde 3-phosphate dehydrogenase (GAPDH) and ubiquitin (UB) were presented in Table 1.
Primer sequences, housekeeping genes (β-actin, UB and GAPDH). Genes assessed (IFN-γ, IL-1β, IL-18). Primer direction: Forward (F) and Reverse (R)
RNA extraction: RNA was extracted using TRIzol® reagent, following the manufacturer’s protocol (Invitrogen®). The extracted RNA was quantified by spectrophotometry at 260 nm using the spectrophotometer (NanoDrop® 2000). In order to evaluate the integrity of the extracted RNA, all samples were run by electrophoresis in agarose gel1%, stained with ethidium bromide solution at 1% and visualized under UV light transilluminator.
cDNA synthesis: Samples were treated with DNase I (Ambion®), following the manufacturer’s recommendations. The kit High Capacity cDNA Reverse Transcription (Invitrogen®) was used for the synthesis of cDNA, according to the manufacturer’s recommendations.
Real-time qPCR: The kit SYBR Green PCR Master Mix (Applied Biosystems®) was used according to the protocol: 2.5 µL of Sybr, 0.5 µL of Milli-Q water, 1.0 µL of primer mix, and 1.0 µL of sample, summing up 5.0 µL of reaction volume in each well. Gene expression levels were analyzed in a Step One Plus thermocycler (Applied Biosystems®) following the protocol: 95°C for 15 minutes, 40 cycles of 15s to 95°C, 30s to 58°C, and 30s to 72°C.The results of real-time PCR were analyzed according to Schmittgen & Livak (2008Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nature Protocols 2008;3(6):1101-1108.) using the comparative method 2-CT.
Statistical analysis: Data were analyzed using the statistical program Statistix 9®. Normality of the data was assessed using the Shapiro-Wilk normality test. Parametric data were submitted to analysis of variance (ANOVA) followed by Tukey’s test, while nonparametric data were submitted to Kruskal-Wallis test. The presence of SE was analyzed by the chi-square test. In statistical tests, a significant level of 5% was used.
RESULTS
Phase 1
Litter microbiology: The counts of TB, LAC and EN in the litters are show in Table 2. In L3, there was a significant reduction in TB counts between days 1 and 8. L3 presented higher EN counts compared with L2 and L1 both on days 1 and 8. This was unexpected and we do not have a plausible for this result. However, EN counts in L3 decreased over time (from days 1 to 8). No LAC was detected on day 1in any dilutions (104 and 105) in none of the treatments, only on day 8,at dilutions of 103 and 104. However, LAC counts were not significantly different among treatments. No differences in EM oocyst counts were detected among treatments or experimental days (data not shown).
The presence of the SE over time was reduced only in L3 (Table 3).
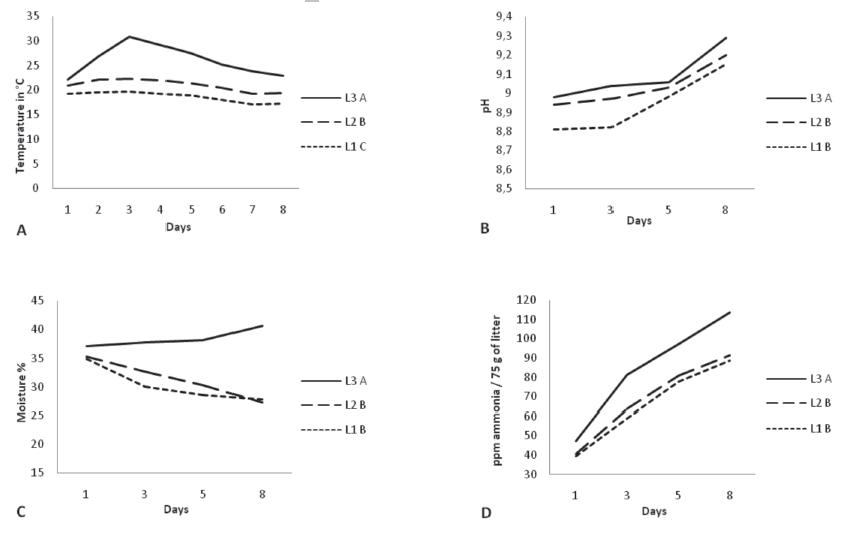
Physical and chemical parameters: Average maximum litter temperature in L3 was measured on day 3 (30.8°C) and then gradually decreased to 22.97°C on day 8. The temperatures recorded in L1 and L2 showed the same trend, with a slight increase on the first days (maximum temperatures of 19.70 and 22.26°C, respectively, on day 3) and gradually decreased till day 8 (to 17.18 and 19.34°C, respectively) (Figure 1A). Average pH value determined in L3 replicates was 8.98 at day 1 and 9.29 at day 8. For L1 and L2, average pH values of 8.81 and 8.94 were recorded on day 1, and 9.15 and 9.20 at day 8, respectively (Figure 1B). Average litter moisture of L3 replicates was stable until day 5, and then gradually increased from 37 to 40% on day 8. Conversely, average litter moisture of L1 and L2 replicates gradually decreased from 35% on day 1 to 27% on day 8 (Figure 1C). Average ammonia level of L3 replicates increased from 47 ppm/75 g of litter on day 1to 114 ppm/75 g of litter on day 8.The average ammonia concentration ofL1replicates increased from 39 ppm/75 g of litter on day 1 to 89 ppm/75 g of litter on day 8, where as L2 presented 40 ppm ammonia/75 g of litter on day 1 and 91 ppm ammonia/75 g of litter on day 8 (Figure 1D). All these physicochemical parameters were significantly different among treatments on day 8 (p≤0.05).
Physical and chemical parameters measured in the litter. A,B,CDifferent letters indicate significant difference among treatments on day 8 by Tukey’s test at p≤0.05.
Phase 2
Microbiology: No significant difference in TB and EN colony-forming units (CFUs) was detected among treatments either on day14 or day 18. The presence of LAC (at103 and 104 dilutions) in L3 broilers was not detected on days 14 and 28 of the live assay. No LAC colonies or SE presence were detected in the cecal content of birds of any of the three treatments on day 28 (Table 4).
CD4+ and CD8+ lymphocytes, macrophages, and goblet cells: All cell counts evaluated both in the jejunum and the cecum were significantly higher in L3 compared with L2 and L1 broilers. In addition, more macrophages were observed in the jejunum of chicken in L3 compared withL1 on day 28 (Table 5).
Cytokine mRNA quantification by real-time PCR: The expression of mRNA for the IFN-γ gene was significantly higher (12.5-fold) in the jejunum and liver of L3 compared with L2 broilers. Higher expression of IL-1β and IL-18 was detected in liver of L3 compared with L2birds, both on days 14 and 28 (Table 6).
Broiler body weight: Broilers reared on theL1 litter presented higher average body weight compared with L2 and L3 broilers at the end of the experiment (L1 = 1.842A, L2 = 1.757B, L3 =1.753B g; p≤0.05).
DISCUSSION
The microbiological analysis of the litters detected an average total bacterial count of 8.5 log10 CFU/g (108 CFU) on day 1, in agreement with the findings of other authors (Terzich et al., 2000Terzich M, Pope M, Cherry T, Hollinger J. Survey of pathogens in poultry litter in the United States. Journal of Applied Poultry Research 2000;9(3):287-291.; Macklin et al., 2005Macklin KS, Hess JB, Bilgili SF, Norton RA. Bacterial levels of pine shavings and sand used as poultry litter. Journal of Applied Poultry Research 2005;14(2):238-245.; Rothrock et al., 2008Rothrock MJ, Cook KL, Lovanh N, Warren JG, Sistani K. Development of a quantitative real-time polymerase chain reaction assay to target a novel group of ammonia-producing bacteria found in poultry litter. Poultry Science 2008;87(6):1058-1067.). The average EN count was 2.4 log10 CFU/g of litter (103 CFU), as found in other studies (Martin et al., 1998Martin S, Mccann M, Waltman D. Microbiological survey of Georgia poultry litter. Journal of Applied Poultry Research 1998;7(1):90-98.; Fries et al., 2005Fries R, Akcan M, Bandick N, Kobe A. Microflora of two different types of poultry litter. British Poultry Science 2005;46(6):668-672.).A significant decrease in the populations of TB, EN and SE was observed when the litter was covered with the plastic canvas compared with those not covered. This may be explained by interaction of physical and chemical factors. In the present experiment, bacterial litter counts were possibly reduced by the presence of ammonia. Ammonia affects cellular metabolism, such as glycolysis and the citric acid cycle, in addition, it causes intracellular pH changes, which affect proton gradients and inhibits endocytosis and exocytosis, quickly causing cell death (Martinelle & Häggström, 1993Martinelle K, Häggström L. Mechanisms of ammonia and ammonium ion toxicity in animal cells: Transport across cell membranes. Journal of Biotechnology 1993;30(3):339-350.; Schneider et al., 1996Schneider M, Marison I, Stockar U. The importance of ammonia in mammalian cell culture. Journal of Biotechnology 1996;46(3):161-185.). The average of ammonia level measured in L3 litter replicates was significantly higher than that of the L1 and L2 litters, suggesting that covering the litter with a plastic canvas inhibits ammonia emission into the air. In this case, ammonia is retained in the litter, and may reach toxic levels for bacteria. Moreover, in the present study, the different litter abiotic parameters, including pH, moisture and temperature, were directly linked with the production of ammonia. It was shown in others studies that increases in the values of those parameters, individually or combined, triggers the production of ammonia in broiler litter (Weaver & Meijerhof, 1991Weaver W, Meijerhof R. The effect of different levels of relative humidity and air movement on litter conditions, ammonia levels, growth, and carcass quality for broiler chickens. Poultry Science 1991;70(4):746-755.; Derikx et al., 1994Derikx JL, Willers HC, Have JW. Effect of pH on the behaviour of volatile compounds in organic manures during dry-matter determination. Bioresource Technology 1994;49(1):41-45.; Nahm, 2003Nahm KH. Evaluation of the nitrogen content in poultry manure. World's Poultry Science Journal 2003;59(1):77-88.; Lovanh et al., 2007Lovanh N, Cook KL, Rothrock MJ, Miles DM, Sistani K. Spatial shifts in microbial population structure within poultry litter associated with physicochemical properties. Poultry Science 2007;86(9):1840-1849.; Miles et al., 2011Miles DM, Rowe DE, Cathcart TC. Litter ammonia generation: Moisture content and organic versus inorganic bedding materials. Poultry Science 2011;90(6):1162-1169.).
Average EM counts were not different among litter treatments, differently from the lethal effect of ammonia on these oocysts found by Horton et al. (1940Horton SC, Taylor EL, Turtle EE. Ammonia fumigation for coccidial disinfection. Veterinary Record 1940;52:829-832.), who observed 100% death Eimeria spp. oocysts placed in 1% ammonia hydroxide solution for 24 hours. Those authors also observed that ammonia gas had lethal effects on oocysts, as a concentration of 25 mg/L killed 100% of the oocysts in 1 hour. Hamed et al. (2013Hamed TS, Yasser NH, Mohammed SN, Heba MH. Assessment of the efficiency of some chemical disinfectants used in poultry farms against coccidiosis. Alexandria Journal of Veterinary Sciences 2013;39(1):82-90.) observed the death of 99% of the of Eimeriatenella oocysts placed in an 5% ammonium hydroxide solution for 24 hours. These results indicate that ammonia concentrations higher than those obtained in the present study are required to reduce oocyst counts. And possibly perhaps the conditions created in the litter covered with the plastic canvas for 8 days were not sufficient to kill Eimeria maxima, suggesting further studies are needed to evaluate the survival of oocysts using this method of litter treatment. The number of oocysts recovered in the phase 1 was low. In addition, during the necropsy of the birds in phase 2, no macroscopic lesions caused by EM were detected in the gut.
The experiment carried out in phase 2 aimed at assessing the effects of the microbial populations remaining in the litter after the treatments applied in phase 1 on the immune response, intestinal microbiota, and broiler body weight. It was observed that the broilers housed on the contaminated litter covered with the plastic canvas (L3) presented higher number of immune cells in the intestinal mucosa compared with L1 and L2, as well as higher levels of pro-inflammatory cytokines in the liver relative to those on L2, which was contaminated and not covered. This may be explained by the fact that EN counts in L3 litter were initially (d 1 of phase 1) higher in L2.Some genera of the Enterobacteriaceae family, such as Proteus, Pseudomonas, and Escherichia coli are related to the ammonia production in broiler litter (Alexander et al., 1968Alexander DC, Carrière JA, Mckay KA. Bacteriological studies of poultry litter fed to livestock. The Canadian Veterinary Journal 1968;9(6):127-131.; Ivanov, 2001Ivanov IE. Treatment of broiler litter with organic acids. Research in Veterinary Science 2001;70(2):169-173.) and may also become pathogenic (Rocha et al., 2002Rocha GP, Silva AB, Brito BG, Souza HL, Pontes AP, Cé MC, et al. Virulence factors of avian pathogenic Escherichia coli isolated from broilers from the south of Brazil. Avian Diseases 2002;46(3):749-753.; Cortes et al., 2004Cortes C, Téllez I, López C, Villaseca F, Anderson R, Eslava C. Aislamiento bacteriano a partir de huevo fértil, huevo incubable y aves con infección del saco vitelino. Revista Latinoamericana de Microbiología 2004;46:12-16.; Nasrin et al., 2013Nasrin S, Islam M, Khatun M, Akhter L, Sultana S. Characterization of bacteria associated with omphalitis in chicks. Bangladesh Veterinarian 2013;29(2):63-68.). These EN present flagella made of flagellin, which are proteins identified as pathogen-associated molecular patterns (PAMPs), and recognized by receptor Toll-like 5 (TLR5) (Hayashi et al., 2001Hayashi F, Smith KD, Ozinsky A, Hawn TR, Yi EC, Goodlett DR, et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 2001;410(6832):1099-1103.; Yoon et al., 2012Yoon S, Kurnasov O, Natarajan V, Hong M, Gudkov AV, Osterman AL, et al. Structural basis of TLR5-flagellin recognition and signaling. Science 2012;335(6070):859-864.). TLR5 are expressed on intestinal epithelial cells and tissue macrophages, leading to the activation and secretion of pro-inflammatory cytokines, such as IL-1β (Haiko & Westerlund, 2013Haiko J, Westerlund WB. The role of the bacterial flagellum in adhesion and virulence. Biology 2013;2(4):1242-1267.; Keestra et al., 2013Keestra AM, Zoete MR, Bouwman L, Vaezirad MM, Putten PM. Unique features of chicken Toll-like receptors. Developmental & Comparative Immunology 2013;41(3):316-323.). The observed increase in IL-1β may have also been the result of stimulation by the lipopolysaccharide (LPS) present in those EN genera. Indeed, LPS recognized by TLR4 (Madsen et al., 2014Madsen HR, Viertlböeck B, Härtle S, Smith AL, Göbel TW. Innate immune responses. Avian Immunology 2014;2:121-147.) increase cytokine expression in both the liver and the intestine of chickens (Iqbal et al., 2005Iqbal M, Philbin VJ, Smith AL. Expression patterns of chicken Toll-like receptor mRNA in tissues, immune cell subsets and cell lines. Veterinary Immunology Immunopathology 2005;104(1-2):117-127.).
It has been shown that IL-18 is released by activated macrophages and that it is a potent inducer of IFN-γ (Li et al., 2013Li K, Gao H, Gao L, Qi X, Gao Y, Qin L, et al. Adjuvant effects of interleukin-18 in DNA vaccination against infectious bursal disease virus in chickens. Vaccine 2013;31(14):1799-1805.) and CD4+ lymphocytes (Hung et al., 2010Hung LH, Li HP, Lien YY, Wu ML, Chaung HC. Adjuvant effects of chicken interleukin-18 in avian Newcastle disease vaccine. Vaccine 2010;28(5):1148-1155.). In the liver of L3 birds, a significant increase in the expression of both IL-18 and IFN-γ was detected on day 28. This emphasizes a potential link between these two cytokines.
Although no direct relationship between IL-18 mRNA expression in the liver and CD4 + lymphocyte counts in the intestinal mucosa was observed, the values of both parameters increased on days 14 and 28. Interestingly, both the increase of CD4+ and IFN-γ are related to intracellular pathogens (Guo et al., 2013Guo P, Thomas JD, Bruce MP, Hinton TM, Bean AG, Lowenthal JW. The chicken TH1 response: Potential therapeutic applications of ChIFN-?. Developmental & Comparative Immunology 2013;41(3):389-396.).
The broilers reared L1 litter presented higher body weight than L2 and L3 broilers. The L1 litter was not contaminated, which may have favored the better performance of L1 broilers when compared of the others treatments. On the other hand, the higher moisture content of the L3 litter before birds were housed L3 (40%) compared with L1 and L2 (27%) may be responsible for the higher survival of EN populations during the in-vivo assay in phase (Miles et al., 2011Miles DM, Rowe DE, Cathcart TC. Litter ammonia generation: Moisture content and organic versus inorganic bedding materials. Poultry Science 2011;90(6):1162-1169.), and therefore, to higher ammonia production, affecting broiler performance (Carlile, 1984Carlile FS. Ammonia in poultry houses: A literature review. World's Poultry Science Journal 1984;40(2):99-113.; Beker et al., 2004Beker A, Vanhooser SL, Swartzlander JH, Teeter RG. Atmospheric ammonia concentration effects on broiler growth and performance. Journal of Applied Poultry Research 2004;13(1):5-9.). In addition, lactobacilli were detected in the cecal cecum of L1 birds, while they were absent inL3, contributing for the higher body weight of L1 relative to L3 birds. The beneficial effect of this bacterial genus on the performance of broiler has been previously documented (Huang et al., 2004Huang M, Choi Y, Houde R, Lee J, Lee B, Zhao X. Effects of Lactobacilli and an acidophilic fungus on the production performance and immune responses in broiler chickens. Poultry Science 2004;83(5):788-795.; Salim et al., 2013Salim HM, Kang HK, Akter N, Kim DW, Kim JH, Kim MJ, et al. Supplementation of direct-fed microbials as an alternative to antibiotic on growth performance, immune response, cecal microbial population, and ileal morphology of broiler chickens. Poultry Science 2013;92(8):2084-2090.).
Our study confirms the previously reported relationship between the intestinal microbiota and the host immune response (Honjo et al., 1993Honjo K, Hagiwara T, Itoh K, Takahashi E, Hirota Y. Immunohistochemical analysis of tissue distribution of B and T cells in germfree and conventional chickens. Journal of Veterinary Medical Science 1993;55(6):1031-1034.; Mwangi et al., 2010Mwangi WN, Beal RK, Powers C, Wu X, Humphrey T, Watson M, et al. Regional and global changes in TCR?? T cell repertoires in the gut are dependent upon the complexity of the enteric microflora. Developmental & Comparative Immunology 2010;34(4):406-417.). Additionally, the bacteria present in the litter may influence the intestinal microbiota and performance of broilers (Cressman et al., 2010Cressman MD, Yu Z, Nelson MC, Moeller SJ, Lilburn MS, Zerby HN. Interrelations between the microbiotas in the litter and in the intestines of commercial broiler chickens. Applied Environmental Microbiology 2010;76(19):6572-6582.; Wei et al., 2013Wei S, Gutek A, Lilburn M, Yu Z. Abundance of pathogens in the gut and litter of broiler chickens as affected by bacitracin and litter management. Veterinary Microbiology 2013;166(3-4):595-601.).
High Enterobacteria counts in the litter increase the number of CD4+ and CD8+ lymphocytes and macrophages in the gut, as well as the mRNA expression of pro-inflammatory cytokines, such as IL-1β, IL-18, and IFN-γ in the liver and intestine of broilers. For the sake of clarity, it is perhaps worthwhile noting that these immune responses are associated with significantly reduced chicken growth and may be implicated in it.
Overall, our results show that the method of treating broiler litter by covering it with a plastic canvas for eight days allows reducing the initial bacterial load due to the interaction of physical and chemical parameters, and seems to be a promising treatment to be applied for litter reutilization.
Further studies are recommended using longer periods of coverage and litters infested with Eimeria oocysts. Moreover, such method may be useful not only for the control of microscopic pathogens, but also of the darkling beetle (Alphitobius diaperinus), which has shown resistance to the many commercial chemicals. Finally, the authors recommend the use for this method for the reutilization of litter on broiler farms.
ACKNOWLEDGMENTS
The authors are grateful to Agrogen for supplying the birds, to Eliane Leite for motivating the discussion, and to the research agencies CNPq and REUNI for the scholarships granted.
REFERENCES
- Alexander DC, Carrière JA, Mckay KA. Bacteriological studies of poultry litter fed to livestock. The Canadian Veterinary Journal 1968;9(6):127-131.
- Beker A, Vanhooser SL, Swartzlander JH, Teeter RG. Atmospheric ammonia concentration effects on broiler growth and performance. Journal of Applied Poultry Research 2004;13(1):5-9.
- Benabdeljelil K, Ayachi A. Evaluation of alternative litter materials for poultry. Journal of Applied Poultry Research 1996;5(3):203-209.
- Boever S, Vangestel C, Backer P, Croubels S, Sys SU. Identification and validation of housekeeping genes as internal control for gene expression in an intravenous LPS inflammation model in chickens. Veterinary Immunology and Immunopathology 2008;122(3-4):312-317.
- Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa nº62, 26 de agosto de 2003. Métodos analíticos oficiais para análises microbiológicas para controle de produtos de origem animal e água. Brasília, DF: MAPA; 2003. p.43.
- Carlile FS. Ammonia in poultry houses: A literature review. World's Poultry Science Journal 1984;40(2):99-113.
- Carvalho MR, Moura DJ, Souza ZM, Souza GS, Bueno LG. Qualidade da cama e do ar em diferentes condições de alojamento de frangos de corte. Pesquisa Agropecuária Brasileira 2011;46:351-361.
- Cortes C, Téllez I, López C, Villaseca F, Anderson R, Eslava C. Aislamiento bacteriano a partir de huevo fértil, huevo incubable y aves con infección del saco vitelino. Revista Latinoamericana de Microbiología 2004;46:12-16.
- Cressman MD, Yu Z, Nelson MC, Moeller SJ, Lilburn MS, Zerby HN. Interrelations between the microbiotas in the litter and in the intestines of commercial broiler chickens. Applied Environmental Microbiology 2010;76(19):6572-6582.
- Derikx JL, Willers HC, Have JW. Effect of pH on the behaviour of volatile compounds in organic manures during dry-matter determination. Bioresource Technology 1994;49(1):41-45.
- Eldaghayes I, Rothwell L, Williams A, Withers D, Balu S, Davison F, Kaiser P. Infectious bursal disease virus: Strains that differ in virulence differentially modulate the innate immune response to infection in the chicken bursa. Viral Immunology 2006;19(1):83-91.
- Fagonde CA, Pedroso D. Cultivo in vivo, in vitro e diagnostico específico de Eimeria spp. de Gallus gallus. Brasília: Embrapa; 2009. p. 219.
- Fan WQ, Wang HN, Zhang Y, Guan ZB, Wang T, Xu CW, et al. Comparative dynamic distribution of avian infectious bronchitis virus M41, H120, and SAIBK strains by quantitative real-time RT-PCR in SPF chickens. Bioscience, Biotechnology, and Biochemistry 2012;76(12):2255-2260.
- Fries R, Akcan M, Bandick N, Kobe A. Microflora of two different types of poultry litter. British Poultry Science 2005;46(6):668-672.
- Guo P, Thomas JD, Bruce MP, Hinton TM, Bean AG, Lowenthal JW. The chicken TH1 response: Potential therapeutic applications of ChIFN-?. Developmental & Comparative Immunology 2013;41(3):389-396.
- Haiko J, Westerlund WB. The role of the bacterial flagellum in adhesion and virulence. Biology 2013;2(4):1242-1267.
- Hamed TS, Yasser NH, Mohammed SN, Heba MH. Assessment of the efficiency of some chemical disinfectants used in poultry farms against coccidiosis. Alexandria Journal of Veterinary Sciences 2013;39(1):82-90.
- Hayashi F, Smith KD, Ozinsky A, Hawn TR, Yi EC, Goodlett DR, et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 2001;410(6832):1099-1103.
- Hernandes R, Cazetta J. Método simples e acessível para determinar amônia liberada pela cama aviária. Revista Brasileira de Zootecnia 2001;30:824-829.
- Hong YH, Lillehoj HS, Lee SH, Dalloul RA, Lillehoj EP. Analysis of chicken cytokine and chemokine gene expression following Eimeria acervulina and Eimeria tenella infections. Veterinary Immunology and Immunopathology 2006;114(3/4):209-223.
- Honjo K, Hagiwara T, Itoh K, Takahashi E, Hirota Y. Immunohistochemical analysis of tissue distribution of B and T cells in germfree and conventional chickens. Journal of Veterinary Medical Science 1993;55(6):1031-1034.
- Horton SC, Taylor EL, Turtle EE. Ammonia fumigation for coccidial disinfection. Veterinary Record 1940;52:829-832.
- Huang M, Choi Y, Houde R, Lee J, Lee B, Zhao X. Effects of Lactobacilli and an acidophilic fungus on the production performance and immune responses in broiler chickens. Poultry Science 2004;83(5):788-795.
- Hung LH, Li HP, Lien YY, Wu ML, Chaung HC. Adjuvant effects of chicken interleukin-18 in avian Newcastle disease vaccine. Vaccine 2010;28(5):1148-1155.
- Iqbal M, Philbin VJ, Smith AL. Expression patterns of chicken Toll-like receptor mRNA in tissues, immune cell subsets and cell lines. Veterinary Immunology Immunopathology 2005;104(1-2):117-127.
- Ivanov IE. Treatment of broiler litter with organic acids. Research in Veterinary Science 2001;70(2):169-173.
- Kaiser P, Rothwell L, Galyov EE, Barrow PA, Burnside J, Wigley P. Differential cytokine expression in avian cells in response to invasion by Salmonella Typhimurium, Salmonella Enteritidis and Salmonella Gallinarum. Microbiology 2000;146(12):3217-3226.
- Keestra AM, Zoete MR, Bouwman L, Vaezirad MM, Putten PM. Unique features of chicken Toll-like receptors. Developmental & Comparative Immunology 2013;41(3):316-323.
- Kelley TR, Pancorbo OC, Merka WC, Thompson SA, Cabrera ML, Barnhart HM. Elemental concentrations of stored whole and fractionated broiler litter. Journal of Applied Poultry Research 1996;5(3):276-281.
- Lavergne TK, Stephens MF, Schellinger D, Carney WA. In-house pasteurization of broiler litter. Louisiana State University Agricultural Center 2006;2955:1-16.
- Lee KW, Lillehoj HS, Lee SH, Jang SI, Ritter GD, Bautista DA, Lillehoj EP. Impact of fresh or used litter on the posthatch immune system of commercial broilers. Avian Diseases 2011;55(4):539-544.
- Li K, Gao H, Gao L, Qi X, Gao Y, Qin L, et al. Adjuvant effects of interleukin-18 in DNA vaccination against infectious bursal disease virus in chickens. Vaccine 2013;31(14):1799-1805.
- Line JE. Campylobacter and Salmonella populations associated with chickens raised on acidified litter. Poultry Science 2002;81(10):1473-1477.
- Lovanh N, Cook KL, Rothrock MJ, Miles DM, Sistani K. Spatial shifts in microbial population structure within poultry litter associated with physicochemical properties. Poultry Science 2007;86(9):1840-1849.
- Macklin KS, Hess JB, Bilgili SF, Norton RA. Bacterial levels of pine shavings and sand used as poultry litter. Journal of Applied Poultry Research 2005;14(2):238-245.
- Macklin KS, Hess JB, Bilgili SF, Norton RA. Effects of in-house composting of litter on bacterial levels. Journal of Applied Poultry Research 2006;15(4):531-537.
- Madsen HR, Viertlböeck B, Härtle S, Smith AL, Göbel TW. Innate immune responses. Avian Immunology 2014;2:121-147.
- Martin S, Mccann M, Waltman D. Microbiological survey of Georgia poultry litter. Journal of Applied Poultry Research 1998;7(1):90-98.
- Martinelle K, Häggström L. Mechanisms of ammonia and ammonium ion toxicity in animal cells: Transport across cell membranes. Journal of Biotechnology 1993;30(3):339-350.
- Miles DM, Rowe DE, Cathcart TC. Litter ammonia generation: Moisture content and organic versus inorganic bedding materials. Poultry Science 2011;90(6):1162-1169.
- Muniz E, Mesa D, Cuaspa R, Souza A, Santin E. Presence of Salmonella spp. in reused broiler litter. Revista Colombiana de Ciencias Pecuarias 2014;27(1):12-17.
- Muniz E, Pickler L, Lourenço MC, Westphal P, Kuritza LN, Santin E. Probióticos na ração para o controle de Salmonella minnesota em frangos de corte Archives of Veterinary Science 2013;18(3):52-60.
- Mwangi WN, Beal RK, Powers C, Wu X, Humphrey T, Watson M, et al. Regional and global changes in TCR?? T cell repertoires in the gut are dependent upon the complexity of the enteric microflora. Developmental & Comparative Immunology 2010;34(4):406-417.
- Nahm KH. Evaluation of the nitrogen content in poultry manure. World's Poultry Science Journal 2003;59(1):77-88.
- Nasrin S, Islam M, Khatun M, Akhter L, Sultana S. Characterization of bacteria associated with omphalitis in chicks. Bangladesh Veterinarian 2013;29(2):63-68.
- Pra MA, Corrêa ÉK, Roll VF, Xavier EG, Lopes DC, Lourenço FF, et al. Uso de cal virgem para o controle de Salmonella spp. e Clostridium spp. em camas de aviário. Ciência Rural 2009;39:1189-1194.
- Rocha GP, Silva AB, Brito BG, Souza HL, Pontes AP, Cé MC, et al. Virulence factors of avian pathogenic Escherichia coli isolated from broilers from the south of Brazil. Avian Diseases 2002;46(3):749-753.
- Roll VF, Pra MA, Roll AP. Research on Salmonella in broiler litter reused for up to 14 consecutive flocks. Poultry Science 2011;90(10):2257-2262.
- Rothrock MJ, Cook KL, Lovanh N, Warren JG, Sistani K. Development of a quantitative real-time polymerase chain reaction assay to target a novel group of ammonia-producing bacteria found in poultry litter. Poultry Science 2008;87(6):1058-1067.
- Salim HM, Kang HK, Akter N, Kim DW, Kim JH, Kim MJ, et al. Supplementation of direct-fed microbials as an alternative to antibiotic on growth performance, immune response, cecal microbial population, and ileal morphology of broiler chickens. Poultry Science 2013;92(8):2084-2090.
- Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nature Protocols 2008;3(6):1101-1108.
- Schneider M, Marison I, Stockar U. The importance of ammonia in mammalian cell culture. Journal of Biotechnology 1996;46(3):161-185.
- Shanmugasundaram R, Lilburn MS, Selvaraj RK. Effect of recycled litter on immune cells in the cecal tonsils of chickens. Poultry Science 2012;91(1):95-100.
- Smirnov A, Sklan D, Uni Z. Mucin dynamics in the chick small intestine are altered by starvation. The Journal of Nutrition 2004;134(4):736-742.
- Souza M, Moreira J, Barbosa F, Cerqueira M, Nunes ÁC, Nicoli JR. Influence of intensive and extensive breeding on lactic acid bacteria isolated from Gallus gallus domesticus ceca. Veterinary Microbiology 2007;120(1-2):142-150.
- Terzich M, Pope M, Cherry T, Hollinger J. Survey of pathogens in poultry litter in the United States. Journal of Applied Poultry Research 2000;9(3):287-291.
- Thaxton Y, Balzli CL, Tankson JD. Relationship of broiler flock numbers to litter microflora. Journal of Applied Poultry Research 2003;12(1):81-84.
- Weaver W, Meijerhof R. The effect of different levels of relative humidity and air movement on litter conditions, ammonia levels, growth, and carcass quality for broiler chickens. Poultry Science 1991;70(4):746-755.
- Wei S, Gutek A, Lilburn M, Yu Z. Abundance of pathogens in the gut and litter of broiler chickens as affected by bacitracin and litter management. Veterinary Microbiology 2013;166(3-4):595-601.
- Williams ZT, Blake JP, Macklin KS. The effect of sodium bisulfate on Salmonella viability in broiler litter. Poultry Science 2012;91(9):2083-2088.
- Yoon S, Kurnasov O, Natarajan V, Hong M, Gudkov AV, Osterman AL, et al. Structural basis of TLR5-flagellin recognition and signaling. Science 2012;335(6070):859-864.
Publication Dates
-
Publication in this collection
Oct-Dec 2016
History
-
Received
Apr 2015 -
Accepted
Feb 2016