ABSTRACT
Salmonella species have been isolated from various kinds of food and are accountable for outbreaks of foodborne diseases in humans. This study aimed at identifying the similarities between the DNA profiles of Salmonella isolated from chicken feces, chicken products, and human feces in southern Brazil. Six hundred samples were collected (200 from chicken products, 200 from broiler chicken feces, and 200 from human feces) and tested for the presence of Salmonella. Isolates proven to be Salmonella compatible by biochemical and serological tests were tested by the Polymerase Chain Reaction. Their DNA profiles were then analyzed by PFGE and rep-PCR. Salmonella was isolated from 16 out of 600 analyzed samples, with Schwarzengrund serotype presenting the highest incidence, followed by Mbandaka in chicken meat and fecal samples, and Panama in human fecal samples. Some strains isolated from chicken fecal and product samples were indistinguishable by the molecular methods used in the study, suggesting that that the contamination of the broilers on the farm can be transmitted the processed products.
Keywords:
Mbandaka; Schwarzengrund; Panama; PFGE; rep-PCR
INTRODUCTION
Food safety professionals try hard to prevent the growth of deteriorating and pathogenic microorganisms in food. Salmonella species have been isolated from various kinds of food and are accountable for outbreaks of foodborne diseases (FBD) in humans. The Centers for Disease Control and Prevention (CDC) estimate that FBD cause about 1.2 million cases of salmonellosis and about 450 deaths each year in the United States (CDC, 2015). In Brazil, between 2007 and 2014, 450 FBD outbreaks caused by Salmonella were reported, affecting 13,165 people (Portal Brasil, 2015).
Among the different sources of contamination and transmission of Salmonella to humans, food products of poultry origin are the most frequently reported, and are mainly associated with consumption of undercooked poultry meat and/or products that were contaminated with raw poultry meat (Grant et al., 2016Grant A, Hashem F, Parveen S. Salmonella and Campylobacter: Antimicrobial resistance and bacteriophage control in poultry. Food Microbiology 2016;53(Part B):104-109.). The main source of contamination of chicken products is often the farm, where the occurrence of Salmonella is also high (Kanashiro et al., 2005).
The identification of Salmonella clones from animal, food, and human samples is important to understand the epidemiological dynamics of salmonellosis in the food chain, and it is essential for the development of outline food safety programs ineffective disease control plans.
Pulsed-field gel electrophoresis (PFGE), which is currently the gold standard technique by molecular epidemiologists (CDC, 2003b), has been an important tool for the investigation of FBD outbreaks. It has been used to identify correlations between reported cases and implicated food items (Yin et al., 2016Yin M, Yang B, Wu Y, Wang L, Wu H, Zhang T, et al. Prevalence and characterization of Salmonella enterica serovar in retail meats in market place in Uighur, Xinjiang, China. Food Control 2016; 64:165-172.) by estimating the genetic distances between strains. In addition, the discrimination ability of this technique makes it a relevant tool to trace the contamination source within a food chain, thereby enabling the isolation of strains of the same species from different sites involved in food processing (Ribot et al., 2006Ribot EM, Fair MA, Gautom R, Cameron DN, Hunter SB, Swaminathan B, et al. Standardization of pulsed-field gel electrophoresis protocols for the subtyping of Escherichia coli O157:H7, Salmonella and Shigella for PulseNet. Foodborne Pathog Diseases 2006;3(1):59-67.).
Another molecular method used in the genetic distance characterization of bacterial species is the amplification of repetitive extragenic regions (repetitive extragenic palindromic sequence-based polymerase chain reaction [rep-PCR]) dispersed in their genomes, which provides distinct patterns of amplified bands. Rep-PCR is a simple and fast method that, despite its good reproducibility, has moderate discriminatory power as compared to PFGE (Tyler et al., 1997Tyler FD, Wang G,Tyler SD, JOHNSON WM. Factors affecting reliability and reproducibility of amplification-based DNA fingerprinting of representative bacterial pathogens. Journal of Clinical Microbiology 1997;35(2):339-346.).
This study aimed at identifying the similarities between the DNA profiles of Salmonella strains isolated from chicken stool samples, chicken products, and human stool samples in southern Brazil.
MATERIAL AND METHODS
Sample
Six hundred samples were collected: 200 chicken product samples, 200 broiler chicken fecal samples, and 200 human fecal samples.
The chicken product samples (40 whole legs, 40 whole wings, 40 backs, 40 ground meat, and 40 liver samples) were obtained at retail stores in southern Brazil. The originally chilled samples were kept in their packages, stored in cool boxes, and immediately submitted to the Laboratory of Animal Product inspection of the Federal University of Pelotas for further processing. Products of 16 different brands were collected. Brand A products (80 samples) were produced in a processing plant located in the studied region. The other 120 samples belonged to15 different brands (B to P) were marketed in the same region.
The chicken fecal samples were collected in a processing plant with official inspection services from broilers reared on 40 different farms. At the time of slaughter, the large intestine was longitudinally sectioned immediately after the cecal region using sterile surgical scissors, and the contents were collected using swabs. Five random batch samples were simultaneously collected from each flock. Swabs were placedin tubes containing 10 mL buffered peptone water (BPW, Acumedia, Lansing, MI, USA) and submitted to laboratory.
Human fecal samples were obtained from human clinical analysis laboratories located in the studied region, which kindly provided the material for analysis. Swab samples were collected from collection containers submitted to the human laboratories, placedin tubes containing 10 mL BPW, and submitted to the laboratory. All patients whose fecal samples were included in this study reported abdominal discomfort and had been instructed by their personal physician to collect fecal samples for analysis. The human fecal samples were obtained with written consent from the subjects whose stool samples were forwarded for laboratory analysis.
Isolation and identification
The chicken product samples, depending on each case, either 25 g or the whole sample, were placed in sterile plastic bags containing 100 mL BPW and massaged for 5 min. The resulting suspension was drained and used as pre-enrichment step for the detection of Salmonella species, in compliance with the recommendations of the US Food and Drug Administration (FDA) (Andrews et al., 2014Andrews WH, Jacobson A, Hammack T. Salmonella. In: BAM - Bacteriological Analytical Manual. Silver Spring: U.S. Food And Drug Administration, Bacteriological Analytical Manual. 2014. Available from: http://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm070149.htm.
http://www.fda.gov/Food/FoodScienceResea...
). The chicken and human fecal samples were incubated in test tubes to which 10 mL BPW was added as pre-enrichment, as well as for other Salmonella-related research procedures, as mentioned in Andrews et al. (2014).
Isolates proven to be positive for Salmonella according to standard biochemical and serological tests results, as described by Andrews et al. (2014Andrews WH, Jacobson A, Hammack T. Salmonella. In: BAM - Bacteriological Analytical Manual. Silver Spring: U.S. Food And Drug Administration, Bacteriological Analytical Manual. 2014. Available from: http://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm070149.htm.
http://www.fda.gov/Food/FoodScienceResea...
), were tested by PCR as suggested by Malorny et al. (2003Malorny B, Hoorfar J, Bunge C, Helmuth R. Multicenter validation of the analytical accuracy of Salmonella PCR: towards an international standard. Applied Environmental Microbiology 2003;69(1):290-296.) to confirm their identity. The DNA was extracted according to Sambrook & Russel (2001Sambrook J, Russel DW. Molecular cloning: a laboratory manual. New York: Cold Spring Harbor Laboratory Press; 2001.). The primers used were
5'-GTGAAATTATCGCCACGTTCGGGCAA-3'
and
5'-TCATCGCACCGTCAAAGGAACC-3',
targeting invA, using the following PCR cycling parameters: initial denaturation at 95°C for 1 min; 38 cycles of denaturation at 95°C for 30 s, annealing at 64°C for 30 s, and extension at 72°C for 30 s; final extension at 72°C for 4 min. The reactions were performed in a TC-3000 thermal cycler (Techne, Staffordshire, UK).
After PCR confirmation, the strains were referred to the Department of Bacteriology of the Enterobacteria Laboratory of the Oswaldo Cruz Foundation (FIOCRUZ, Manguinhos, Rio de Janeiro, Brazil) for serotype iden-tification.
Bacteria strains were stored in Brain-Heart Infusion broth (BHI, Acumedia) with the addition of 20% glycerolat 37°C for24h and then frozen at -70°C. The strains were incubated in BHI at 37°C for recovery.
Molecular profiles
Isolates were analyzed using PFGE to compare DNA profiles following the suggested protocol from Centers for Disease Control and Preventions (CDC, 2013c). Briefly, the DNA isolated from each sample was digested with XbaI restriction endonuclease (New England Biolabs™ Inc., Beverly, MA, USA), and analyzed on a 1% agarose gel using Pulsed Field Gel Electrophoresis (Bio-Rad Laboratories, Hercules, CA, USA) apparatus. The gel was then stained with ethidium bromide, and visualized with ultraviolet light.
Rep-PCR was performed according to Versalovic et al. (1994Versalovic J, Schneider M, de Bruijn FJ, Lupski JR. Genomic fingerprinting of bacteria with repetitive sequence based polymerase chain reaction. Methds in Molecular and Cellular Biology 1994;5:25-40.), using the 5'-GTGGTGGTGGTGGTG-3' primer and the following PCR cycling parameters: initial denaturation at 94°C for 1 min; 30 cycles of denaturation at 95°C for 30 s, annealing at 45°C for 60 s, and extension at 60°C for 5 min; final extension at 60°C for 16 min. The reactions were carried out in a TC-3000 thermal cycler.
The PFGE and rep-PCR patterns were interpreted in accordance with the criteria suggested by Tenover et al. (1995Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, et al. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. Journal of Clinical Microbiology 1995;33(9):2233-2239.) using the following classifications: indistinguishable (no different bands), closely related (2 to 3 distinct bands), possibly related (4 to 6 distinct bands), and different (over 7 distinct bands).
RESULTS AND DISCUSSION
Salmonella was isolated from 16 out of 600 analyzed samples, 8 (8/200; 4%) from chicken products, 4 (4/200; 2%) from chicken fecal samples, and 4 (4/200; 2%) from human fecal samples. All strains phenotypically characterized as Salmonella contained invA, a highly preserved DNA region of this genus, which can be used to confirm its identity at the molecular level.
Among the 200 chicken product samples analyzed from 16 different brands (A to P), 8 samples from 3 brands, A (5/80); L (2/18), and N (1/3), were contaminated with Salmonella: 3 from the liver (brands A, L, and N), 3 from whole legs(brands A [2 samples] and L), 1 from whole wings (brand A),and 1 from the back (brand A).This low prevalence was also reported by other authors. Panzenhagen et al. (2016Panzenhagen PHN, Aguiar WS, Frasão BS, Pereira VLA, Abreu BLC, Rodrigues DP, et al. Prevalence and fluoroquinolones resistance of Campylobacter and Salmonella isolates from poultry carcasses in Rio de Janeiro, Brazil. Food Control 2016;61:243-247.) analyzed 60 chicken carcasses from six processing plants in Rio de Janeiro, and found 6.67% and 8.33% Salmonella contamination, using the conventional method and PCR, respectively. Duarte et al. (2009Duarte DAM, Ribeiro AR, Vasconcelos AMM, Santos SB, Silva JVD, Andrade PLA, et al. Occurrence of Salmonella spp. in broiler chicken carcasses and their susceptibility to antimicrobial agents. Brazilian Journal of Microbiology 2009;40(3):569-573.) analyzed 260 chicken carcasses bought from five different processing plants, and found 9.6% carcasses positive for Salmonella.
Among the 200 analyzed fecal samples of broilers derived from 40 farms, Salmonella was isolated from four chicken fecal samples from three different farms that supplied brand A at time of this study.
Four distinct serotypes were identified in chicken fecal samples (Table 1). One broiler harbored two distinct serotypes simultaneously. These two strains (FF02 and FF03), confirmed as Salmonella by serology and PCR, showed phenotypically distinct colonies (one of the colonies showed typical biochemical characteristics, and the other grew on completely acidified TSI agar). The serotypes of these isolates were identified as Schwarzengrund (FF02) and Mbandaka (FF03).
The serotypes Enteritidis and Typhimurium have been reported in other research studies (Abd-Elghany et al., 2015Abd-Elghany SM, Sallam KI, Abd-Elkhalek A, Tamura T. Occurrence, genetic characterization and antimicrobial resistance of Salmonella isolated from chicken meat and giblets. Epidemiology and Infection 2015;143(5):997-1003.; Suresh et al., 2011Suresh T, Hatha AAM, Harsha HT, Lakshmanaperumalsamy P. Prevalence and distribution of Salmonella serotypes in marketed broiler chickens and processing environment in Coimbatore City of southern India. Food Research International 2011;4(3):823-825.; Thakur et al., 2013Thakur S, Brake J, Keelara S, Zou M, Sisick E. Farm and environmental distribution of Campylobacter and Salmonella in broiler flocks. Researchin Veterinary Science 2013;94(1):33-42.) as the most common in chickens. This study, however, did not detect any isolates of these serotypes in chicken product or fecal samples. The most frequent isolate was serotype Schwarzengrund, followed by Mbandaka. Other studies have also reported a higher prevalence of serotypes other than Enteritidis and Typhimurium, e.g., that of Aslam et al. (2012Aslam M, Checkley S, Avery B, Chalmers G, Bohaychuk V, Gensler G, et al. Phenotypic and genetic characterization of antimicrobial resistance in Salmonella serovars isolated from retail meats in Alberta, Canada. Food Microbiology 2012;32(1):110-117.) in Canada, in ground beef samples, and Le Bouquin et al. (2010Le Bouquin S, Allain V, Rouxel S, Petetin I, Picherot M, Michel V, et al. Prevalence and risk factors for Salmonella spp. contamination in French broiler-chicken flocks at the end of the rearing period. Preventive Veterinary Medicine 2010;97(3-4):245-251.) who found a higher prevalence of serotype Hadar in France in chicken samples, differently from the present study.
In a study conducted by the Department of Bacteriology of the Oswaldo Cruz Foundation, Hofer et al. (1997Hofer E, Silva Filho SJ, Reis EMF. Prevalência de sorovares de Salmonella isolados de aves no Brasil. Pesquisa Veterinária Brasileira 1997;17(2):55-62.) reported that the serotype Mbandaka belongs to a common Salmonella group; serotype Schwarzengrund, on the other hand, is thought to belong to a rare, or accidental, Salmonella group, according to occurrence levels recorded during 1962-1991. Kanashiro et al. (2005) also investigated the presence of Salmonella spp. in samples received from diverse chicken processing plants located in different regions in Brazil between July 1997 and December 2004, and did not report the serotype Schwarzengrund as one of the main isolates. Nevertheless, as previously mentioned, Schwarzengrund was the predominant serotype identified both in chicken fecal samples (3 isolates from 2 aviaries) and in chicken products (6 isolates) in the present study. Boni et al. (2011Boni HFK, Carrijo AS, Fascina VB. Ocorrência de Salmonella spp. em aviários e abatedouro de frangos de corte na região central de Mato Grosso do Sul. Revista Brasileira de Saúde e Produção Animal 2011;12(1):84-95.) also reported this serotype as the most frequently isolated in broiler carcasses obtained between August 2005 and December 2006 from a processing plant located in Mato Grosso do Sul State, Brazil. However, the serotype Schwarzengrund was not isolated on the farms supplying this plant, which suggests that carcasses were contaminated in the processing plant rather than on the chicken farm. Chen et al. (2010Chen MH, Wang SW, Hwang WZ, Tsai SJ, Hsih YC, Chiou CS, et al. Contamination of Salmonella Schwarzengrund cells in chicken meat from traditional marketplaces in Taiwan and comparison of their antibiograms with those of the human isolates. Poultry Science 2010;89(2):359-365.) reported a high prevalence of this serotype in raw chicken meat (30.5%) in Taiwan.
The nine Salmonella Schwarzengrund strains isolated were submitted to genotyping by PFGE with the XbaI restriction enzyme, and no differences were observed between the band patterns, except for the FF04 strain (Figure 1A). However, the rep-PCR results of the other identified showed further differences (Figure 1B). The strains CF03, CF04, CF06, CF07, and CF08 isolated from chicken products were indistinguishable from each other, and were closely related with CF05. Notwithstanding, they are possibly related with the FF01 and FF02 strains, which were isolated from chicken fecal samples, and were indistinguishable from each other, but different from FF04, which was also isolated in chicken fecal samples.
Photographs of pulsed-field gel electrophoresis (A) and repetitive extragenic palindromic sequence-based polymerase chain reaction (B) electrophoresis gels of band profiles of 9 Salmonella Schwarzengrund isolates. CF03: isolate from brand A whole leg; CF04: isolate from brand L whole leg; CF05: isolate from brand N liver; CF06: isolate from brand A liver; CF07: isolate from brand L liver; CF08: isolate from brand A back; FF01: isolate from fecal sample of chicken from farm G02; FF02 and FF04: isolates from fecal sample of chicken from farm G15; M1: DNA Size Standards - Lambda ladder; M2: GeneRuler™ 1 kb DNA ladder.
The strains CF03 and CF06 were isolated from products of a specific commercial brand (brand L), which suggests the same source of contamination, possibly at the processing plant that supplied brand L. In the case of the CF04, CF07, and CF08 strains, isolated from brand A products, the results indicate a common contamination source. Moreover, the correlation found between the strains isolated in brand A products with the strains FF01 and FF02 from chicken fecal samples obtained in the processing plant that supplied this brand suggests that the source contamination was the chicken farms.
Aarestrup et al. (2007Aarestrup FM, Hendriksen RS, Lockett J, Gay K, Teates K, McDermott PF, et al. International spread of multidrug-resistant Salmonella Schwarzengrund in food products. Emerging Infectious Diseases 2007;13(5):726-731.), using PFGE for the detection of Salmonella Schwarzengrund clones in chicken products and human isolates, demonstrated the transmission of this microorganism from food to man, as opposed to our study, where chicken isolates were not found to be related with those obtained from humans.
Salmonella Mbandaka was found in 2 chicken fecal samples from 2 different origins and in 1 chicken product sample (whole wing). This serotype accounted for 17% (3/17) of the isolated samples. Suresh et al. (2011Suresh T, Hatha AAM, Harsha HT, Lakshmanaperumalsamy P. Prevalence and distribution of Salmonella serotypes in marketed broiler chickens and processing environment in Coimbatore City of southern India. Food Research International 2011;4(3):823-825.) and Hue et al. (2011Hue O, le Bouquin S, Lalande F, Allain V, Rouxel S, Petetin I, et al. Prevalence of Salmonella spp. on broiler chicken carcasses and risk factors at the slaughterhouse in France in 2008. Food Control 2011;22(8):1158-1164.) reported low occurrence of this serotype in chicken carcasses in India and France, respectively. Oliveira (2012Oliveira APD. Salmonella sp. em frango e ambiente de abate [dissertation]. Goiânia (GO): Universidade Federal de Goiás; 2012.) analyzed samples from four broiler processing plants in Goiás State, Brazil, and found a 3.45% prevalence of the Mbandaka serotype. Upon analyzing different chicken carcass parts, Suresh et al. (2011) observed that this was one of the least common serotypes in southern India; yet, it was present in different chicken parts, in addition to having been found in environmental sample collected from a cage. In France, Hue et al. (2011) also recorded low occurrence of this serotype, having identified only 1 isolate from 425 chicken carcasses in a processing plant. On the other hand, Murgia et al. (2015Murgia M, Bouchrif B, Timinouni M, Al-Qahtani A, Al-Ahdal MN, Cappuccinelli P, et al. Antibiotic resistance determinants and genetic analysis of Salmonella enterica isolated from food in Morocco. International Journal of Food Microbiology 2015;215(23):31-39.) reported that, out of 94 Salmonella strains isolated from food in Morocco, 36% were Salmonella Mbandaka. Despite its usually low occurrence, the serotype Mbandaka has been implicated in FBD (CDC, 2013a), and its presence in foods represents a hazard to consumers.
PFGE (Figure 2A) and rep-PCR (Figure 2B) results show that the strains CF02 and FF03 are indistinguishable from each other, suggesting that the sources of contamination to these samples are related. The FF05 strain, which is not related to the strains CF02 and CF03, probably had a different origin. Hoszowski & Wasyl (2001Hoszowski A, Wasyl D. Typing of Salmonella enterica subsp. enterica serovar Mbandaka isolates. Veterinary Microbiology 2001;80(2):139-148.) reported that biotyping and antimicrobial susceptibility and plasmid profiles were not sufficient to differentiate the analyzed Salmonella Mbandaka strains, and only genomic macrorestriction proved to be an efficient method for epidemiological studies of this serotype. However, in the present study, the discriminatory power of rep-PCR was similar to that of PFGE for this serotype, with the advantage that the former is less expensive and faster than PFGE.
Photographs of pulsed-field gel electrophoresis (A) and repetitive extragenic palindromic sequence-based polymerase chain reaction (B) electrophoresis gels of band profiles of 3 Salmonella Mbandaka isolates. M1(A): DNA Size Standards - Lambda ladder; M1(B): GeneRuler™ 1 kb DNA ladder; CF02: isolate from brand A whole wing; FF03 and FF05: isolates from G15 farm and G28 chicken fecal samples, respectively.
Auto-agglutinable Salmonella enterica was isolated from 1 chicken product sample. Alcocer et al. (2006Alcocer I, Oliveira KMP, Vidotto MC, Oliveira TCRM. Discriminação de sorovares de Salmonella spp. isolados de carcaças de frango por REP e ERIC-PCR e fagotipagem do sorovar Enteriditis. Ciência e Tecnologia de Alimentos 2006;26(2):414-420.), evaluating 25 Salmonella strains obtained from chicken carcasses from four processing plants located in Paraná State, Brazil, found only one auto-agglutinable strain. Other authors have reported the isolation of auto-agglutinable strains in chicken fecal samples in Brazil (Salles et al., 2008Salles RPR, Cardoso WM, Teixeira RSC, Siqueira AA, Silva EE, Castro SB. Monitoramento bacteriológico para Salmonella spp. em poedeira comercial na recria e produção de empresas avícolas da região metropolitana de Fortaleza, CE, Brasil. Ciência Animal Brasileira 2008;9(2):427-432.; Andreatti Filho et al., 2009Andreatti Filho RL, de Lima ET, Menconi A, et al. Pesquisa de Salmonella spp. em suabes de arrasto provenientes de granjas avícolas. Veterinária e Zootecnia 2009;16(1):190-194.). Nevertheless, in the present study, auto-agglutinable strains were isolated from chicken products rather than chicken fecal samples. Although not investigated, the possibility that the chicken product samples were previously contaminated with chicken feces cannot be ruled out.
Children up to five years of age are more affected by salmonellosis, as well as the elderly and people with weakened immune systems (CDC, 2015). However, in a study reporting an outbreak in São Paulo, Brazil, Matsuoka et al. (2004Matsuoka DM, Costa SF, Mangini C, Almeida GMD, Bento CN, Van der Heijden IM, et al. A nosocomial outbreak of Salmonella enteritidis associated with lyophilized enteral nutrition. Journal of Hospital Infection 2004;58(2):122-127.) observed that the average age of the affected people was 36.5 years. In the present study, Salmonella was isolated from fecal samples of patients whose age ranged from nine months to 40 years, with no gender predominance.
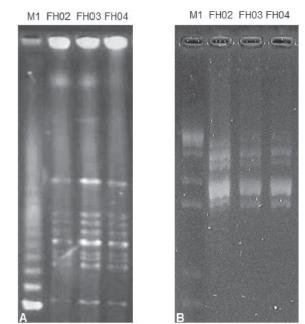
In human fecal samples, four strains (three Panama and one Typhimurium serotypes) were isolated. These serotypes have been shown to cause gastroenteritis in humans, both in Brazil (Fernandes et al., 2006Fernandes SA, Tavechio AT, Ghilardi ACR, Dias AMG, Almeida IAZC, Melo LCV. Salmonella serovars isolated from humans in São Paulo state, Brazil, 1996-2003. Revista do Instituto de Medicina Tropical de São Paulo 2006;48(4):179-184.; Antunes et al., 2016Antunes P, Mourão J, Campos J, Peixe L. Salmonellosis: the role of poultry meat. Clinical Microbiology and Infection 2016;22(2):110-121.) and in other countries (Soto et al., 2001Soto SM, Guerra B, del Cerro A ,González-Hevia MA, Mendoza MC. Outbreaks and sporadic cases of Salmonella serovar Panama studied by DNA fingerprinting and antimicrobial resistance. International Journal of Food Microbiology 2001;71(1):35-43.; Tsai et al., 2007Tsai KS, Yang YJ, WangSM, Chiou CS, Liu CC. Change of serotype pattern of group D non-Typhoidal Salmonella isolated from pediatric patients in southern Taiwan. Journal of Microbiology, Immunology, and Infection 2007;40(3):234-239.). However, during the period 2006-2015, there was no report by the CDC (2015) of Panama Salmonella outbreaks in humans related with the consumption of animal products. In our study, Panama serotype band profiles were indistinguishable (Figure 3) from each other, which is suggestive of an outbreak, insofar as the three samples were collected from patients at the same location on the same day.
Photographs of pulsed-field gel electrophoresis (A) and repetitive extragenic palindromic sequence-based polymerase chain reaction (B) electrophoresis gels of band profiles of three Salmonella Panama isolates. M1(A): DNA Size Standards - Lambda ladder; M1(B): GeneRuler™ 1 kb DNA ladder; FH02, FH03, and FH04: isolates from human fecal samples.
CONCLUSIONS
Salmonella is present in broiler chickens in southern Brazil, as well as in chicken products available for consumption, which represents a health risk for consumers. Salmonella Schwarzengrund is a common serotype in southern Brazil, followed by Mbandaka, both on farms and in chicken products. In this region, despite the few reports, Panama serotype occurs in humans. The strains whose genotypes were indistinguishable by the molecular methods used in the study were detected in chicken fecal and product samples. This finding suggests that the contamination of the broilers on the farm can reach the processed product. This emphasizes the need for better hygienic and health practices in the processing plants for the control of undesirable microorganisms and disease eradication in animals. In addition, stricter farm biosecurity measures are required in order to minimize the risk of contamination of the final product.
The serotypes of the strains isolated in humans were different from those found in chicken products. The fact that the human strains were indistinguishable among each other by the applied techniques used suggests the occurrence of an outbreak. In addition, other salmonellosis cases and outbreaks may not be reported to the authorities, contributing for the underestimation of the incidence of this disease in humans in Brazil.
ACKNOWLEDGEMENTS
We would like to thank the Fundação de Amparo à Pesquisa do Estado Rio Grande do Sul (FAPERGS)for funding this research study, the clinical analysis laboratories for providing the human fecal samples, and the Laboratório deCultura de Tecidos of the Instituto de Biologia, UniversidadeFederal de Pelotas, where PFGE gels were stained.
REFERENCES
- Aarestrup FM, Hendriksen RS, Lockett J, Gay K, Teates K, McDermott PF, et al. International spread of multidrug-resistant Salmonella Schwarzengrund in food products. Emerging Infectious Diseases 2007;13(5):726-731.
- Abd-Elghany SM, Sallam KI, Abd-Elkhalek A, Tamura T. Occurrence, genetic characterization and antimicrobial resistance of Salmonella isolated from chicken meat and giblets. Epidemiology and Infection 2015;143(5):997-1003.
- Alcocer I, Oliveira KMP, Vidotto MC, Oliveira TCRM. Discriminação de sorovares de Salmonella spp. isolados de carcaças de frango por REP e ERIC-PCR e fagotipagem do sorovar Enteriditis. Ciência e Tecnologia de Alimentos 2006;26(2):414-420.
- Andreatti Filho RL, de Lima ET, Menconi A, et al. Pesquisa de Salmonella spp. em suabes de arrasto provenientes de granjas avícolas. Veterinária e Zootecnia 2009;16(1):190-194.
- Andrews WH, Jacobson A, Hammack T. Salmonella. In: BAM - Bacteriological Analytical Manual. Silver Spring: U.S. Food And Drug Administration, Bacteriological Analytical Manual. 2014. Available from: http://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm070149.htm
» http://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm070149.htm - Antunes P, Mourão J, Campos J, Peixe L. Salmonellosis: the role of poultry meat. Clinical Microbiology and Infection 2016;22(2):110-121.
- Aslam M, Checkley S, Avery B, Chalmers G, Bohaychuk V, Gensler G, et al. Phenotypic and genetic characterization of antimicrobial resistance in Salmonella serovars isolated from retail meats in Alberta, Canada. Food Microbiology 2012;32(1):110-117.
- Boni HFK, Carrijo AS, Fascina VB. Ocorrência de Salmonella spp. em aviários e abatedouro de frangos de corte na região central de Mato Grosso do Sul. Revista Brasileira de Saúde e Produção Animal 2011;12(1):84-95.
- Brasil. Ministério da Saúde. Saúde. Vigilância. Higiene no preparo de alimentos evita contaminação por Salmonella; 2015. Available from: http://www.brasil.gov.br/saude/2015/01/higiene-no-preparo-de-alimentos-evita-contaminacao-por-salmonella
» http://www.brasil.gov.br/saude/2015/01/higiene-no-preparo-de-alimentos-evita-contaminacao-por-salmonella - CDC - Centers for Disease Control and Prevention. Multistate outbreak of human Salmonella infections linked to live poultry. 2013a. Available from: http://www.cdc.gov/salmonella/live-poultry-04-13
» http://www.cdc.gov/salmonella/live-poultry-04-13 - CDC - Centers for Disease Control and Prevention. National center for emerging and zoonotic infectious diseases. division of foodborne, waterborne, and environmental diseases. Salmonella. 2015. Available from: http://www.cdc.gov/salmonella/general/technical.html
» http://www.cdc.gov/salmonella/general/technical.html - CDC - Centers for Disease Control and Prevention. National center for emerging and zoonotic infectious diseases. division of foodborne, waterborne, and environmental diseases. What is pulsed-field gel electrophoresis (PFGE)? 2013b. Available from: http://www.cdc.gov/pulsenet/index.html
» http://www.cdc.gov/pulsenet/index.html - CDC - Centers for Disease Control and Prevention. Standard operating procedure for pulsenet PFGE of Escherichia coli O157:H7, Escherichia coli non-O157 (STEC), Salmonella serotypes, Shigella sonnei and Shigella flexneri. 2013c. Available from: http://www.cdc.gov/pulsenet/pdf/ecoli-shigella-salmonella-pfge-protocol-508c.pdf.
- Chen MH, Wang SW, Hwang WZ, Tsai SJ, Hsih YC, Chiou CS, et al. Contamination of Salmonella Schwarzengrund cells in chicken meat from traditional marketplaces in Taiwan and comparison of their antibiograms with those of the human isolates. Poultry Science 2010;89(2):359-365.
- Duarte DAM, Ribeiro AR, Vasconcelos AMM, Santos SB, Silva JVD, Andrade PLA, et al. Occurrence of Salmonella spp. in broiler chicken carcasses and their susceptibility to antimicrobial agents. Brazilian Journal of Microbiology 2009;40(3):569-573.
- Fernandes SA, Tavechio AT, Ghilardi ACR, Dias AMG, Almeida IAZC, Melo LCV. Salmonella serovars isolated from humans in São Paulo state, Brazil, 1996-2003. Revista do Instituto de Medicina Tropical de São Paulo 2006;48(4):179-184.
- Grant A, Hashem F, Parveen S. Salmonella and Campylobacter: Antimicrobial resistance and bacteriophage control in poultry. Food Microbiology 2016;53(Part B):104-109.
- Hofer E, Silva Filho SJ, Reis EMF. Prevalência de sorovares de Salmonella isolados de aves no Brasil. Pesquisa Veterinária Brasileira 1997;17(2):55-62.
- Hoszowski A, Wasyl D. Typing of Salmonella enterica subsp. enterica serovar Mbandaka isolates. Veterinary Microbiology 2001;80(2):139-148.
- Hue O, le Bouquin S, Lalande F, Allain V, Rouxel S, Petetin I, et al. Prevalence of Salmonella spp. on broiler chicken carcasses and risk factors at the slaughterhouse in France in 2008. Food Control 2011;22(8):1158-1164.
- Kanashiro AMI, Stoppa GFZ, Cardoso ALSP, Tessari ENC, Castro AGM. Serovars of Salmonella spp isolated from broiler chickens and commercial breeders in diverse regions in Brazil from July 1997 to December 2004. Brazilian Journal of Poultry Science;7(3):195-198.
- Le Bouquin S, Allain V, Rouxel S, Petetin I, Picherot M, Michel V, et al. Prevalence and risk factors for Salmonella spp. contamination in French broiler-chicken flocks at the end of the rearing period. Preventive Veterinary Medicine 2010;97(3-4):245-251.
- Malorny B, Hoorfar J, Bunge C, Helmuth R. Multicenter validation of the analytical accuracy of Salmonella PCR: towards an international standard. Applied Environmental Microbiology 2003;69(1):290-296.
- Matsuoka DM, Costa SF, Mangini C, Almeida GMD, Bento CN, Van der Heijden IM, et al. A nosocomial outbreak of Salmonella enteritidis associated with lyophilized enteral nutrition. Journal of Hospital Infection 2004;58(2):122-127.
- Murgia M, Bouchrif B, Timinouni M, Al-Qahtani A, Al-Ahdal MN, Cappuccinelli P, et al. Antibiotic resistance determinants and genetic analysis of Salmonella enterica isolated from food in Morocco. International Journal of Food Microbiology 2015;215(23):31-39.
- Oliveira APD. Salmonella sp. em frango e ambiente de abate [dissertation]. Goiânia (GO): Universidade Federal de Goiás; 2012.
- Panzenhagen PHN, Aguiar WS, Frasão BS, Pereira VLA, Abreu BLC, Rodrigues DP, et al. Prevalence and fluoroquinolones resistance of Campylobacter and Salmonella isolates from poultry carcasses in Rio de Janeiro, Brazil. Food Control 2016;61:243-247.
- Ribot EM, Fair MA, Gautom R, Cameron DN, Hunter SB, Swaminathan B, et al. Standardization of pulsed-field gel electrophoresis protocols for the subtyping of Escherichia coli O157:H7, Salmonella and Shigella for PulseNet. Foodborne Pathog Diseases 2006;3(1):59-67.
- Salles RPR, Cardoso WM, Teixeira RSC, Siqueira AA, Silva EE, Castro SB. Monitoramento bacteriológico para Salmonella spp. em poedeira comercial na recria e produção de empresas avícolas da região metropolitana de Fortaleza, CE, Brasil. Ciência Animal Brasileira 2008;9(2):427-432.
- Sambrook J, Russel DW. Molecular cloning: a laboratory manual. New York: Cold Spring Harbor Laboratory Press; 2001.
- Soto SM, Guerra B, del Cerro A ,González-Hevia MA, Mendoza MC. Outbreaks and sporadic cases of Salmonella serovar Panama studied by DNA fingerprinting and antimicrobial resistance. International Journal of Food Microbiology 2001;71(1):35-43.
- Suresh T, Hatha AAM, Harsha HT, Lakshmanaperumalsamy P. Prevalence and distribution of Salmonella serotypes in marketed broiler chickens and processing environment in Coimbatore City of southern India. Food Research International 2011;4(3):823-825.
- Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, et al. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. Journal of Clinical Microbiology 1995;33(9):2233-2239.
- Thakur S, Brake J, Keelara S, Zou M, Sisick E. Farm and environmental distribution of Campylobacter and Salmonella in broiler flocks. Researchin Veterinary Science 2013;94(1):33-42.
- Tsai KS, Yang YJ, WangSM, Chiou CS, Liu CC. Change of serotype pattern of group D non-Typhoidal Salmonella isolated from pediatric patients in southern Taiwan. Journal of Microbiology, Immunology, and Infection 2007;40(3):234-239.
- Tyler FD, Wang G,Tyler SD, JOHNSON WM. Factors affecting reliability and reproducibility of amplification-based DNA fingerprinting of representative bacterial pathogens. Journal of Clinical Microbiology 1997;35(2):339-346.
- Versalovic J, Schneider M, de Bruijn FJ, Lupski JR. Genomic fingerprinting of bacteria with repetitive sequence based polymerase chain reaction. Methds in Molecular and Cellular Biology 1994;5:25-40.
- Yin M, Yang B, Wu Y, Wang L, Wu H, Zhang T, et al. Prevalence and characterization of Salmonella enterica serovar in retail meats in market place in Uighur, Xinjiang, China. Food Control 2016; 64:165-172.
Publication Dates
-
Publication in this collection
Oct-Dec 2016
History
-
Received
May 2016 -
Accepted
Aug 2016