Abstract
OBJECTIVE: To analyze the impact of moderate physical exercise on the total and differential leukocyte counts and red blood cell count of 36 sixty-day-old adult male Wistar rats subjected to early malnourishment. METHODS: The rats were divided in nourished (N - casein 17%) and malnourished groups (M - casein 8%) and thesegroups were then subdivided in trained (T) untrained (U) creating four groups NT, NU, MT and MU. The NT and MTgroups were submitted to moderate physical exercise using a treadmill (60 min/day, 5 days/week for 8 weeks). Onthe 1st day, before the training started T0 and 24 hours after the last training day of the week (T1 until T8), a 1 mLaliquot of blood was collected from the animals' tails for analysis. The total leukocyte count was evaluated in a cellcounter with an electronic microscope. The cyanmethemoglobin technique was used to measure the hemoglobin level. The hematocrit values were determined as a percentage using the micro-hematocrit technique with a microcapillaryreader and a cell counter was used to determine the red blood cell count. The t-test was used for statistical analysis and a p-value < 0.05 was considered significant. Data are expressed as means ± standard deviation. RESULTS: There was a significant difference in the total leukocyte count between the NT (9.1 ± 0.1) and MT groups (8.0 ± 0.1) from T1 and in neutrophils between the NT (22.1 ± 0.6) and MT groups (24.6 ± 1.8) from T7 (p < 0.05). There was no statistical significance in the hemoglobin, hematocrit and red blood cell count from T1. CONCLUSIONS: According to the results of this study, moderate physical exercise seems to have induced physiologic adaptation in adult rats from T1.
Exercise; Leukocytes; Malnutrition; Physical exertion; Animals; Rats
ORIGINAL ARTICLE
Leukocyte, red blood cell and morphological adaptation to moderate physical training in rats undernourished in the neonatal period
Marcelo Tavares VianaI; Manuella Cavalcanti PerezII; Valdenilson Ribeiro RibasI; Gilberto de Freire MartinsIII; Célia Maria Machado Barbosa de CastroI
IUniversidade Federal de Pernambuco - UFPE, Recife, PE, Brazil
IIInstituto Materno Infantil de Pernambuco - IMIP, Recife, PE, Brazil
IIIUniversidade de Pernambuco - UPE, Recife, PE, Brazil
Corresponding author Corresponding author: Marcelo Tavares Viana Universidade Federal de Pernambuco, Laboratório de Fisiologia da Nutrição/Departamento de Nutrição Rua Prof. Morais Rego, Cidade Universitária 50670-901 Recife, PE, Brazil mtviana0@yahoo.com.br
ABSTRACT
OBJECTIVE: To analyze the impact of moderate physical exercise on the total and differential leukocyte counts and red blood cell count of 36 sixty-day-old adult male Wistar rats subjected to early malnourishment.
METHODS: The rats were divided in nourished (N - casein 17%) and malnourished groups (M - casein 8%) and thesegroups were then subdivided in trained (T) untrained (U) creating four groups NT, NU, MT and MU. The NT and MTgroups were submitted to moderate physical exercise using a treadmill (60 min/day, 5 days/week for 8 weeks). Onthe 1st day, before the training started T0 and 24 hours after the last training day of the week (T1 until T8), a 1 mLaliquot of blood was collected from the animals' tails for analysis. The total leukocyte count was evaluated in a cellcounter with an electronic microscope. The cyanmethemoglobin technique was used to measure the hemoglobin level. The hematocrit values were determined as a percentage using the micro-hematocrit technique with a microcapillaryreader and a cell counter was used to determine the red blood cell count. The t-test was used for statistical analysis and a p-value < 0.05 was considered significant. Data are expressed as means ± standard deviation.
RESULTS: There was a significant difference in the total leukocyte count between the NT (9.1 ± 0.1) and MT groups (8.0 ± 0.1) from T1 and in neutrophils between the NT (22.1 ± 0.6) and MT groups (24.6 ± 1.8) from T7 (p < 0.05). There was no statistical significance in the hemoglobin, hematocrit and red blood cell count from T1.
CONCLUSIONS: According to the results of this study, moderate physical exercise seems to have induced physiologic adaptation in adult rats from T1.
Keywords: Exercise; Leukocytes; Malnutrition; Physical exertion; Animals; Rats
Introduction
Lack of protein results in several physiological and metabolic alterations, which have been linked to depression of the immune system(1). Thus, an adequate supply of nutrients is essential for the growth of all body systems. Studies on protein malnutrition in the intrauterine, neonatal and development stages have attempted, using animal models, to investigate the effect of exercise on nutritional recovery (NR)(2). As restoration of metabolic variables is slow, exercise may improve NR(1).
Moderate physical exercise (MPE) can trigger various biochemical, metabolic, hormonal andimmune responses(3). For example, performing MPE can induce physiological changes such as therelease of inflammatory mediators, cytokines and the activation of leukocytes(4). It may also favoraerobiosis, improving the kinetics of oxygen uptake by promoting better pulmonary absorption,oxygen transportation and its transference to cells(5). Aerobic exercise training also promotes anincrease in the basal metabolic rate or energy expenditure thus inducing weight loss(6).
Few studies using physical training protocols have analyzed weekly responses to physical exercise on NR in patients who were submitted to malnutrition in the neonatal period.
Thus, the aim of this study was to analyze changes in leukocyte and red blood cell (RBC) parameters and on the body weight related to MPE in rats submitted initially to malnourishment during the neonatal period and subsequently NR.
Methods
Thirty-six male Wistar rats, bred in the Department of Nutrition of the Universidade Federalde Pernambuco, were used in this study. The Central Animal House is maintained at a temperatureof 21 ± 2ºC with a reversed 12-hour light cycle (light - 9:01 p.m. to 9:00 a.m. and dark - 9:01 a.m. To 9:00 p.m.). In this vivarium, animals are bred in polypropylene cages with food and water ad libitum.
For this study, adult animals were first kept in the animal house for a period of 15 days to adapt to the reversed light cycle environment before being bred. A ratio of one male to two females was used during breeding. On pregnancy, defined by an increase in size, female rats wereplaced in individual cages until partum. The first day postpartum was defined as the beginning of lactation at which time six male pups were placed with each mother. These groups were allocatedto malnourishment (a low-protein diet - 8% casein) and normal nourishment (normal protein diet 17% casein) regimens. The pups were fed the diet via the mother'smilk.
From the twenty-second day of life (weaning) groups of three pups were placed in cages, respecting the diet of the neonatal period. The animals were then fed commercial Labina rat feed which is used as a standard diet in the animal house because it contains 23% mixed protein. This diet was administered until the end of the experiment (Day 120).
The total body weight (TBW) of the animals was measured from the first day of life until the end of the experiment using digital weighing scales with an accuracy of 0.1 g and a maximum capacity of 4 kg (Marte, model S-2000). The experimental period was divided into lactation (the first 21 days), weaning (22nd to 59th day) and training (60th to 120th day).
At sixty days, the initial groups (nourished and malnourished) were subdivided for physical exercise creating four groups: trained and nourished (TN; n = 6), trained and malnourished (TM; n = 6), untrained and nourished (UN; n = 6) and untrained and malnourished (UM; n = 6).
The training groups (TN and TM) were submitted to a MPE protocol on a treadmill (60 mins daily, 5 days/week for 8 weeks(7)),while the other two groups remained in their cages, which were placed near to the treadmill at the time of the exercise period.
Analysis of red blood cell and leukocyte parameters
Before the start of training (T0) and 24 hours after the lastsession of each training week (T1 to T8), a small blood sample(1 mL) was collected from the tail of animals under anesthesia. A volume of 0.5 mL was used to measure RBC indices, such as the RBC count, hemoglobin and hematocrit and 0.5 mL was used for the total and differential leukocyte counts. The drawn blood was placed in a 5 mL tube containing one drop (20 µL) of 3% ethylenediaminetetraacetic Acid (EDTA) as anticoagulant.
The cyanmethemoglobin technique was used to measurethe hemoglobin level with 10 mL of blood being diluted in 5 mLof hypotonic solution. Extrusion of hemoglobin is obtained byerythrocyte lysis with the formation of cyanmethemoglobin, theproduct resulting from a reaction between hemoglobin and cyanide. The concentration was inferred by spectrophotometry (BeckmanDU-62) using a 540 nm filter and calculated in mg/dL using theformula: Hb = OD of the sample x CF, where: Hb = hemoglobinconcentration, OD = optical density and CF = conversion factor.
The hematocrit values were determined as a percentage using the micro-hematocrit technique with a microcapillary reader. A 0.1 M sodium phosphate buffer solution (PBS) at a ratio of 1:200 and pH = 7.4 was used for the RBC count in a hemocytometer. For the total leukocyte count, blood samples were diluted in Turk's solution (3% acetic acid) at a ratio of 1:200(8) and counted using a hemocytometer. The differential leukocyte count employed the blood smear technique stained with Panótico Fast LB kit (Laborclin Ltd, Brazil).
This study was approved by the Research Ethics Committee on Animal Experimentation of the Center for Biological Sciences, Universidade Federal de Pernambuco (No. 76/07 Case No.008226/2007-55) and followed the guidelines recommended by the Brazilian Committee of Animal Experimentation (COBEA).
Statistical analysis
The Normal distribution of the data was checked using theShapiro-Wilk and Bartlet tests. The Student's t-test was used to analyzevariables within and between groups with the data being expressed asmeans ± standard deviation. Significance was set for a p-value < 0.05.
Results
Animals within each group were compared and no significant differences in the variables were identified.
Body weight
Malnourished animals gained less weight than the nourishedanimals from the 7th to the 120th day of life (p-value < 0.05) (Figure 1).
No significant differences were identified on comparing the body weights between the animals of the TN and UN Groups and between those of the TM and UM Groups. Thus, for this variable the TN and UN Groups and the TM and UM Groups were combined to form just two groups: Nourished and Malnourished. On comparing these groups, the Malnourished Group gainedsignificantly less TBW (211.03 ± 10.71 g) compared to the Nourished Group (272.16 ± 31.70 g) (p-value < 0.05) from the first week of training (T1 - Figure 2).
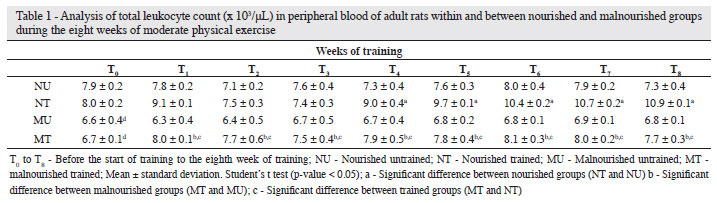
Total leukocyte count
Table 1 shows the total leukocyte counts (TLC) of thefour groups of rats over the 8 weeks of MPE. There was asignificant difference between the malnourished and nourishedanimals at T0 (effect of malnutrition) and between the TMand TN groups at T1, T4 - T8 (effect of training -p-value <0.05). Moreover, the TLC was significantly higher in the TMcompared to the UM Group from the first week of training T1(p-value < 0.05). However, a significant increase in the TLC ofthe TN Group compared to the UN Group was only observedfrom T4 (p-value < 0.05).
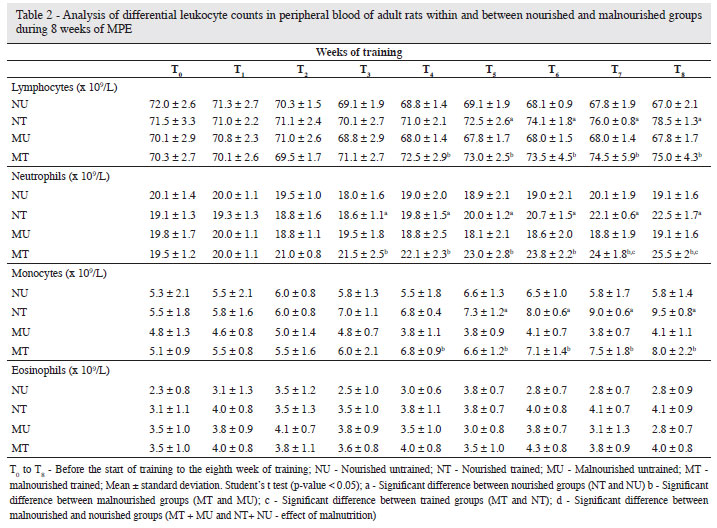
Differential leukocyte counts
Table 2 shows the differential leukocyte counts for the different groups over the 8 weeks of MPE. No significant difference was observed in the lymphocytes of malnourishment compared to nourished animals at T0 (effect of NR) and between the TM and TN groups from T1. However, a significant increase was observed on comparing the TM with the UM Group (effect of training) from T4 (p < 0.05), and between the TN and UN Groups (effect of training) from T5 (p < 0.05).
For neutrophils, there were no significant differences between themalnourished and nourished animals at T0 (effect of the NR) or between the TM and TN Groups up to T6. However there was a significantdifference between the TM and TN Groups at T7 and T8 (p < 0.05).
From T3 there were significant differences between the TM and UMGroups and between the TN and UN Groups from T3 (p < 0.05).
No significant difference was observed for monocytes betweenthe nourished and malnourished animals at T0 (effect of the NR) or between the TM and TN Groups from T1. However on comparingthe trained and untrained animals, there was a significant increase forthe TM compared to the UM Group (effect of training) from T4 and between the TN and UN Groups (effect of training) from T5 (p < 0.05).
Eosinophils did not change significantly with NR or with training; although there were slight increases in their count, no significant differences were observed between nourished and malnourished animals with training.
Red blood cell indices
Table 3 shows the changes in RBC indices over the 8 weeks of training. There were no significant differences between the hematocrit levels of the nourished and malnourished animals at the start of training (T0 - effect of the NR) or between the TM and TN Groups from T1. However a significant increase was observed for the TN Group compared to the UN Group from T2 (p <0.05); a significant increase in the TM Group compared to the UM Group occurred at T8 (p < 0.05).
There were statistical differences for hemoglobin between the nourished and malnourished animals at T0 (effect of malnutrition) and between the TM Group and TN Group at T1(p < 0.05). However, this difference was not significant from T2. A significant increase in hemoglobin was observed in the TN Group compared to the UN Group at T1 (effect of early training - break in homeostasis) and from T5 (p < 0.05); the difference was significant between the TM and UM Groups from T4 (p < 0.05).
In respect to the RBC count, there was a statistical difference between the nourished and malnourished animals at T0 (p < 0.05 - effect of malnutrition). Between the trained groups (TN and TM), there was a significant difference at T1 (p < 0.05). However, this difference was not observed from T2 to T8. On comparing TN with UN and TM with UM there were significant increases from T5 and T4, respectively (p < 0.05).
Discussion
This study analyzed physiological changes induced byMPE on the leukocytes and RBCs of the peripheral bloodand on the morphology (TBW) of adult male rats submittedto early malnourishment. The total and differential leukocytecounts (neutrophils, lymphocytes, eosinophils and monocytes)were evaluated as were the RBC count and the hematocrit and hemoglobin concentrations. To identify the impact of MPE and NRon animals that had been submitted to malnourishment and those that had not, these variables were measured at the end of each weekof training (T1 to T8) and compared both with basal measurements(T0) and between the different experimental groups.
Before the start of training (T0)
At T0, the total lymphocytes, and hemoglobin and Hematocrit concentrations of malnourished animals were lower, even following NR, compared to the nourished animals; however, there were no significant differences in the other variables. It is likely that NR was not effective for these specific variables. These results may be a consequence of the period when malnutrition was applied. At this stage, there is a greater demand for calories and proteins for cell function and to provide energy for the neonatal metabolism(9). The cellular changes caused by malnourishment seem to impact on the formation of lymphoid organs(10), as wellas on leukocyte expansion during adulthood(11).
There is a relationship between the low biological value of dietary protein and iron intake and alterations in the RBC parameters(12). Malnourishment may change the structure of hematocrit and hemoglobin reducing concentrations and function(12). The lack of iron can alter oxygen transport, thereby reducing the maximum oxygen consumption (VO2 max), one of the parameters used to analyze performance in physical exercise(5). Malnourishment may have altered the protein content of the milk provided to the offspring thereby impacting the TBW(13) keepingthe weight down from the 7th day of the neonatal period until the end of the experiment (120th day).
This work corroborated, in some respects, to another studythat did not observe changes in the monocyte and eosinophil counts of malnourished compared to nourished animals, however, there was a significant reduction in total leukocyte count during this period and in the TBW of the animals from the 5th day oflife(13). The effects of restricted protein consumption during the neonatal period were also seen in rats from day 5(14). In the studyby Barone et al.(12) it is possible that the differences in the other variables are due to specific physiological responses to the type of training (swimming), as the experiment designs were similar.
In another study, malnourishment caused significant changes in rats from the 21st to the 60th day of life(1). This result is different from the current study, as although the objectives were similar, the malnourishment continued beyond the neonatal period. This does not change the effectiveness of the model utilized, but may hinder the identification of some cellular responses that are affected specifically during the neonatal period(10,11). In another model, the red and white blood cell variables of undernourished rats administered with saline did not differ from those administered lipopolysaccharides however, the body weight during adulthood was different to rats that were breastfed(15).
The results in the current study differ from a study that administered an endotoxin (lipopolysaccharides) in the neonatal period(15) as we did not use exogenous pro-inflammatory stimuli during the experimental period. Malnourishment in the neonatal period reduces the immunocompetence and may cause damage to structures and functions of organ systems such as the nervous, endocrine and immune systems(16,17). NR may re-establish these losses, but the process is slow(1) and physical exercise may help to reduce the effects caused by malnourishment.
1st week of training (T1)
During this period, the purpose of the training was to break the organic homeostasis of the animals(18); some variablesresponded with values above baseline, albeit sometimes without significance. It was also observed that malnourishment significantly affected the total leukocyte count and hemoglobin and hematocrit concentrations even with the start of training. It is possible that these variables are not responsive to short sessions (5 minutes) of MPE(19), which altered the responses of other variables, creating an increase in the number of blood cells(20).
The first week of MPE triggered physiological and metabolic responses similar to those of acute training, despite the moderate intensity. These changes induced homeostatic adjustments(21),either as a response to physical stress(22) or as pre-disposition of changes to training. The physical exercise in acute conditions can change the count, distribution and functional capacity of some cells(19). These conditions were observed in the significant increase in the total leukocyte count of animals that performed moderate physical exercise for 5 minutes compared to light physical exercise for 5 minutes(19).
A short period of physical exercise can be compared to sudden physical stress as it also induces changes in RBCs.Animals, but not humans, are capable of storing large quantities of RBCs in the spleen and expel them into circulation when the oxygen transport system is under stress(12). In the current study, the first week of MPE seems to have induced a higher demand of energy from the cardiovascular system thereby improving VO2max. as shown by the increased hematocrit and hemoglobin concentrations. Thus, the lack of nutrients does not appear to have changed the chemiosmotic coupling process of aerobic energy production in the mitochondria(4,12).
The energy production in the mitochondria results in changes that appear to be related to increased metabolism during physical exercise, and thus, the TBW has been used to evaluate this(23). During the entire period of training (T1 to T8)there were significant differences in the TBW between training groups, however, there were no differences within groups. This result of the TM Group demonstrates that NR was efficient, as energy expenditure and the catabolic phase are more intense with the association of physical exercise and protein deficiency(2),inducing a greater reduction in TBW.
In a study that used swimming with this animal model, a smaller weight gain was observed in rats from the 5th week of physical exercise compared to a control group that did not swim(12). Although the periods and type of exercise were different, the results are similar, however, in the current study the rats were first submitted to malnourishment and later to NR. It is possible that the differences in methodology contributed to the differences in the outcomes.
2nd and 3rd week of training (T2 and T3)
This stage of the physical training was characterized by a transition between the breakdown of homeostasis to stability, which led to some responsive variables. Malnourishment did not impede significant increases in the total leukocyte and neutrophil counts of the TM compared to UM animals, and the total leukocyte count in relation to the TN Group. Important, albeit non-significant, increases were observed for the other variables both between the groups of malnourished animals and compared to the nourished rats. It is possible that these responses occurred due to the MPE, which, combined with NR, induced physiological adaptations in cells thereby minimizing the expected differences.
The leukocyte counts of malnourished and nourished rats progressively increased at T2 and T3 compared to T0 and T1,suggesting a physiological leukocytosis in response to this type of stress(23). This increase seems to be an adaptive response to MPE, as in the interval between T0 and T3, there was a mean total leukocyte count of 9.340/mm3, a lower value than what is suggestive of inflammatory leukocytosis(24).
An increase in the lymphocyte count was observed in agroup of rats after swimming to exhaustion(25) and in another group submitted to 8 weeks of MPE on a treadmill(18). In another experiment, regular exercise sessions at submaximal intensitiesdid not alter the lymphocyte count(26). However, leukocytosis andlymphocytosis was identified in rats subjected to 5 and 15 minutesof moderate and acute physical exercise(27). Despite the similarity, itis difficult to compare the results of this study with the literature, ashere the changes in weight each week were compared to T0.
Physical exercise seems to have influenced the increase in RBC indices in the period from T2 to T3 compared to the period of malnourishment. This increase in oxygen transportation may have improved the VO2max., thereby improving the aerobic capacity of the animals in the trained group. These results, in principle, suggest that the TM group did not suffer from a lack of nutrients such as iron, responsible for transporting oxygen and co-factordisorders such as anemia(27). There is a possibility of a reduction in the RBCs with training, which would cause pseudoanemia(27).
The hematocrit and hemoglobin concentrations and RBCcount of Wistar rats submitted to swimming for 6 weeks, didnot change statistically(6). It is possible that the type of exercise,the environment and diet given in the neonatal period may havecorroborated to the differences found compared to this work.Increases in hematocrit and hemoglobin concentrations wereobserved in another study that used psychological stress employingrestraint(28). These results corroborate the current work, even thoughanother type of stress and conditioning was used.
Animals are able to retain RBCs in the spleen and release them into circulation when the oxygen transport system is under stress(28). This mechanism, via the sympathetic nervous system, results in a contraction of the smooth muscles of the spleen reducing its volume(28). Thus, chronic physical stress, as in the current study, may have caused changes in oxygen supply, with possible increases in the concentrations of 2,3-diphosphoglycerate in RBCs(28). This compound binds to hemoglobin, reducing its affinity to oxygen thereby making it available to the tissues.
From the 4th to 8th week of training (T4 to T8)
This period was described as the beginning of the period ofphysiological adaptation to training. Most variables tended to havemoderate increases in their values. NR helped to consolidate the effectscaused by the MPE on the TM Group for almost all study variablesexcept for the eosinophil count. This, despite the changes throughoutthe period of training, was not responsive to MPE. As the eosinophilcount did not differ within the groups or between groups, the oxidativestress to overtraining was minimized(29). These responses observed inthe study variables were similar in the TN Group.
In this work, malnourishment did not change, in general,the leukocyte, hematological and morphological responses in thegroup of malnourished animals in respect to physical exerciseduring adulthood. These results corroborate other trials that did notobserve significant cellular changes induced by malnourishment inadult females, even using the same malnutrition during pregnancy and lactation, weaning at 30 days and swimming as MPE(30). It maybe noted that important variables such as gender and low-protein diet during pregnancy did not influence the results. The influence of malnourishment on linear and muscle growth was also studied in young rats and children during NR(29). It was observed that groups subjected to light physical exercise improved in terms of anthropometric indices of growth(29). It seems that the recovering organism preserves the homeostatic mechanisms that guarantee physiological adaptations to physical exercise, which stimulate linear growth(29). Our results, in line with other publications(30,29), tend to suggest a possible interactionbetween NR and physical exercise in response to malnourishment.
The effects of NR and MPE attenuated possibleconsequences of malnourishment in trained rats, establishing for some variables, significantly higher increases compared to well-nourished rats. The MPE seems to have induced adaptations of leukocyte and RBC variables of adult male Wistar rats that were submitted to early malnourishment as well as attenuated the reduction in their TBW.
Submitted: 1/23/2012
Accepted: 5/21/2012
Conflict-of-interest disclosure: The authors declare no competing financial interest
- 1. Santhiago V, Silva AS, Gobatto CA, Mello MA. Treinamento físico durante a recuperação nutricional não afeta o metabolismo muscular da glicose de ratos. Rev Bras Med Esporte. 2006;12(2):76-80.
- 2. Papoti M, Almeida PB, Prada FJ, Eleno TG, Hermini HA, Gobatto CA, Mello MA. Máxima fase estável de lactato durante a natação em ratos recuperados de desnutrição protéica. Motriz, Rio Claro [Internet]. 2003 [cited 2011 Nov 21] ;9(2):103-10. Available from: http://www.rc.unesp.br/ib/efisica/motriz/09n2/14Papoti.pdf
- 3. Lapin LP, Prestes J, Pereira GB, Palanch AC, Cavaglieri CR, Verlengia R. Respostas metabólicas e hormonais ao treinamento físico. Rev Bras Educ Física Esporte Lazer Dança. 2007;2(4):115-24.
- 4. Moraska A, Deak T, Spencer RL, Roth D, Fleshner M. Treadmill running produces both positive and negative physiological adaptations in Sprague- Dawley rats. Am J Physiol Regul Integr Comp Physiol. 2000;279(4):R1321-9.
- 5. Cruz AM da. Resistência aeróbia e eritropoetina. Estudos (Goiânia). 2006;33(7/8):553-72.
- 6. Nascimento E, Cavalcante T, Pereira S, Palmeira A, Rocha MC, Viana MT, et al. O exercício físico crônico altera o perfil leucocitário e a taxa de fagocitose de ratos estressados. Rev Port Ciênc Desp. 2004;4(3):26-33.
- 7. Leandro CG. Influência do treino de resistência moderado na resposta imunológica de ratos submetidos a contenção aguda [thesis]. Portugal: Faculdade de Ciências do Desporto e de Educação Física - Universidade do Porto; 2005.
- 8. Stibbe W, Weise M, Seidel D. [Automated platelet count in thrombocytopenic patients-a comparison of methods]. J Clin Chem Clin Biochem. 1985;23(7):399-404. German.
- 9. Pithon-Curi TC, Schumacher RI, Freitas JJ, Lagranha C, Newsholme P, Palanch AC, et al. Glutamine delays spontaneous apoptosis in neutrophils. Am J Physiol Cell Physiol. 2003;284(6):C1355-61.
- 10. Chandra RK. Nutrition and the immune system from birth to old age. Eur J Clin Nutr. 2002;56(Suppl 3):S73-6
- 11. Cunningham-Rundles S, McNeeley DF, Moon A. Mechanisms of nutrient modulation of the immune response. J Allergy Clin Immunol. 2005;115(6):1119-28, quiz 1129. Comment in: J Allergy Clin Immunol. 2005;115(6):1235-7.
- 12. Barone A, Harper RG, Wapnir RA. Placental copper transport in the rat. III. Interaction between copper and iron in maternal protein deficiency. Placenta. 1988;19(1):113-8.
- 13. Porto SM, Araújo FR, Melo JF, Silva RR, Silva KM, De Castro CM. Cinética do perfil leucocitário do sangue, antes e após treinamento físico moderado, em ratos adultos desnutridos no período neonatal. An Fac Med Univ Fed Pernamb. 2006;51(1):59-65.
- 14. Queiros-Santos A. Desnutrição precoce e estresse agudo: aspectos da resposta imunitária em ratos adultos [dissertation]. Recife: Universidade Federal de Pernambuco; 2000.
- 15. Ferreira e Silva WT. Aspectos da resposta inflamatória em ratos adultos endotoxêmicos submetidos à desnutrição no período de aleitamento. [dissertation]. Recife. Universidade Federal de Pernambuco; 2002.
- 16. Rocha-de-Melo AP, Picanço-Diniz CW, Borba JM, Santos-Monteiro JG, Guedes RC. NADPH-diaphorase histochemical labeling patterns in the hippocampal neuropil and visual cortical neurons in weaned rats reared during lactation on different litter sizer. Nutrit Neurosci. 2004;7(4):207-16.
- 17. Costa Rosa LF, Vaisberg MW. Influência do exercício na resposta imune. Rev Bras Med Esporte [Internet].2002 [cited 2011 Nov 21];8(4):167:72. Available from: http://www.scielo.br/pdf/rbme/v8n4/v8n4a06.pdf
- 18. Dias R, Frollini AB, Prestes J, Ferreira CK, Donatto FF, Verlengia R, et al. Efeito do exercício agudo de curta duração em leucócitos circulantes e linfócitos teciduais de ratos. Rev Bras Educ Fis Esp. 2007;21(3):229-49.
- 19. Green KJ, Croaker SJ, Rowbottom DG. Carbohydrate supplementation and exercise-induced changes in T-lymphocyte function. J Appl Physiol. 2003;95(3):1216-23.
- 20. Pereira B, Souza Jr TP. Adaptação e rendimento físico - considerações biológicas e antropológicas. Rev Bras Ci Mov [Internet]. 2005 [cited 2011 Nov 21];13(2):145-52. Available from: http://portalrevistas.ucb.br/index.php/RBCM/article/view/636/647
- 21. Afonso M, Souza CN, Zagatto AM, Luciano E. Respostas metabólicas agudas ao exercício físico moderado em ratos wistar. Motriz, Rio Claro [Internet]. 2003 [cited 2011 Nov 21] ;9(2):87-92. Available from: http://www.rc.unesp.br/ib/efisica/motriz/09n2/Afonso.pdf
- 22. Green KJ, Rowbottom DG, Mackinnon LT. Exercise and T-lymphocyte function: a comparison of proliferation in PBMC and NK cell-depleted PBMC culture. J Appl Physiol. 2002;92(6):2390-5.
- 23. Prestes J, Ferreira CK, Frollini AB, Dias R, Donatto FF, Guereschi MG, et al. Influência do exercício físico agudo realizado até a exaustão sobre o número de leucócitos, linfócitos e citocinas circulantes. Fit Perf J. 2007;6(1):32-7.
- 24. Kim H, Shin MS, Kim SS, Lim BV, Kim HB, Kim YP, et al. Modulation of immune responses by treadmill exercises in Sprague-Dawley rats. J Sports Med Phys Fitness. 2003;43(1):99-104.
- 25. Pedersen BK, Hoffman-Goetz L. Exercise and the immune system regulation, integration and adaptation. Physiol Rev. 2000;80(3):1055-81.
- 26. Bricks LF. Ferro e infecções. Atualização Pediat. (São Paulo). 1994;16(1):34-43.
- 27. Araújo CF, Moraes MS, Diniz AP, Cosendey AE. Pseudoanemia dilucional e os atletas olímpicos. Rev Bras Anal Clin. 2004;36(4):197-200.
- 28. Stewart IB, McKenzie DC. The human spleen during physiological stress. Sports Med. 32(6):361-9.
- 29. Torun B, Viteri FE. Influence of exercise on linear growth. Eur J Clin Nutr. 1994;48(Suppl 1):S186-9.
- 30. The early history of the biochemistry of muscle contraction. J Gen Physiol. 2004;123(6):631-41.
Corresponding author:
Publication Dates
-
Publication in this collection
06 Sept 2012 -
Date of issue
2012
History
-
Received
23 Jan 2012 -
Accepted
21 May 2012