Abstracts
Continuous culture study of the ruminal bacterium Megasphaera elsdenii using lactate as carbon source is reported. Butyrate, the minor product in batch culture, was the main product in continuous culture. Under steady state conditions the biomass concentration was increased from 0.46 to 0.72 g l-1. Addition of Yea Sacc did not result an increased availability of carbon. The produced amounts of valerate and CO2 were calculated by using a combined carbon and degree of reduction balances. According to these results, the lactate catabolism of M. elsdenii was changed by the presence of yeast additive, yielding more butyrate and diminishing the production of valerate, while acetate and propionate production was not affected. Results reported in this work suggested that soluble compounds present in the yeast additive filtrate enhanced the anabolism of M. elsdenii and modified the carbon fluxes through its pathways by increasing butyrate and decreasing valerate production in continuous culture.
Megasphaera elsdenii; butyrate; yeast additive; continuous culture
Estudo em cultura contínua da bactéria ruminal Megasphaera elsdenii que utiliza lactato como fonte de carbono foi realizada. Butirato, foi o produto produzido em menor concentração na cultura em batelada, porém em cultura contínua foi o produto de maior concentração. Sob condições definidas a concentração de biomassa aumentou de 0.46 a 0.72 g l-1. A adição do aditivo Yea Sacc não resultou num aumento da disponibilidade do carbono. As quantidades produzidas de valerato e CO2 foram calculadas pelo uso combinado do balanço e grau de redução do carbono. De acordo com esses resultados, o catabolismo de lactato de M. elsdenii foi mudado pela adição de aditivo a base de levedura. Maior rendimento em butirato e redução na produção de valerato foi observada, por outro lado a produção deacetato e propionato não foi afetada. Os resultados desse trabalho sugerem que os componentes solúveis presentes no aditivo a base de levedura aumentam o anabolismo de M. elsdenii modificando o fluxo de carbono através de suas vias de metabólicas, pelo aumento do butirato e redução do valerato. Em cultura contínua.
Stimulation of the Megasphaera elsdeniis Butyrate Production in Continuous Culture by a Yeast Additive
Oscar Soto-Cruz1, Rafael Chávez-Rivera2 and Gerardo Saucedo-Castañeda1* * Author for correscpondence
1Department of Biotechnology and 2Department of Process Engineering and Hydraulics, Autonomous Metropolitan University, Iztapalapa Campus. Av. Michoacán y Purísima s/n, A. P. 55-535, C. P. 09340, México D. F., MEXICO.
ABSTRACT
Continuous culture study of the ruminal bacterium Megasphaera elsdenii using lactate as carbon source is reported. Butyrate, the minor product in batch culture, was the main product in continuous culture. Under steady state conditions the biomass concentration was increased from 0.46 to 0.72 g l-1. Addition of Yea Sacc did not result an increased availability of carbon. The produced amounts of valerate and CO2 were calculated by using a combined carbon and degree of reduction balances. According to these results, the lactate catabolism of M. elsdenii was changed by the presence of yeast additive, yielding more butyrate and diminishing the production of valerate, while acetate and propionate production was not affected. Results reported in this work suggested that soluble compounds present in the yeast additive filtrate enhanced the anabolism of M. elsdenii and modified the carbon fluxes through its pathways by increasing butyrate and decreasing valerate production in continuous culture.
Key words: Megasphaera elsdenii, butyrate, yeast additive, continuous culture
INTRODUCTION
Megasphaera elsdenii, the main lactate utilizer rumen bacterium, is capable of fermenting up to 97% of the rumen lactate (Counotte et al., 1981). The volatile fatty acids (VFA, acetate, propionate and butyrate) are the main products of lactate metabolism (Marounek & Bartos, 1987; Marounek et al., 1989; Hino et al., 1994). On the other hand, the effect of additives on microbial activity has been reported (Wallace & Newbold, 1992), both growth and lactate uptake are stimulated by the commercial additives: Amaferm (Waldrip & Martin, 1993), Yea Sacc (Rossi et al., 1995) and Levucell SC (Chaucheyras et al., 1996). Nevertheless, their effect on microbial fermentation patterns are not well known.
Continuous culture of M. elsdenii has been utilized to evaluate the effect of pH on the efficiency of growth (Russell & Dombrowsky, 1980), to study the catabolism of amino acids (Wallace, 1986) and to evaluate the role of extracellular acetate in the fermentation of glucose (Hino et al., 1991), but previous studies have not considered production of VFA by M. elsdenii in continuous culture with lactate as carbon source. In this work, M. elsdenii was cultivated in continuous culture and the effect of a yeast additive (Yea Sacc1026, AllTechTM) was assessed upon fermentation pattern.
MATERIALS AND METHODS
Micro-organism and culture medium:
Megasphaera elsdenii, Spanish Type Culture Collection (CECT) 390, was conserved in glycerol (30%) at -20°C. The culture medium for batch and continuous culture contained (g l-1): lactate (as sodium DL-lactate syrup 60% w/v), 10; casein peptone, 8; yeast extract, 2.8; beef extract, 2.8; cysteine hydrochloride, 0.5; TweenTM 80, 1 ml l-1 and mineral solution, 40 ml l-1. The mineral solution contained (g l-1): CaCl2, 0.2; MgSO47H2O 0.2; K2HPO4, 1; KH2PO4, 1; NaHCO3, 10 and NaCl, 2. pH was fixed to 6.7. Yeast additive was prepared under anaerobic conditions as follows: 4 g of Yea Sacc1026 (AllTechTM) plus 50 ml of deionized water were stirred during 1 hour. The suspension was passed through a filter paper Whatman #41 and sterilized by filtration through a 0.45 µm membrane. The filtered liquid was bubbled with O2-free CO2 during 10 minutes. To study the effect of the yeast additive, the resulting solution was added to culture medium in order to obtain a concentration of 0.675 g l-1 (2% v/v).
Reactor operation: Continuous culture was performed in a B-Braun fermenter model Biostat-B. Culture vessel with 1800 ml of medium was sterilized at 121°C during 1 hour. Oxygen was eliminated with a continuous flow of nitrogen (10 ml min-1), which was maintained during all the process in order to preserve anaerobic conditions. Medium was inoculated with 200 ml of an overnight culture of M. elsdenii CECT 390 and the fermentation was carried out at 37°C and 100 min-1. The system was first operated in batch culture during 18 h. After this time, continuous culture was started at a dilution rate of 0.1 h-1. Two stages in continuous culture were studied. In the first stage, medium without additive was used. Later, medium plus yeast additive was supplied during second stage.
Sample analysis and expression of results: Turbidimetric (Goodhue et al., 1986), enzymatic (Boehringer Mannheim, 1999) and gas chromatography (Jouany, 1982) techniques were used for biomass, DL-lactate and VFA measurements, respectively. Measured acetate, propionate and butyrate were expressed individually and as total VFA. Biomass (YXS), acetate (YAS), propionate (YPS) and butyrate (YBS) yields were calculated as indicated by Stephanopoulos and coworkers (1998). Units of these yield coefficients are given in Table 1, and expressed in terms of one C-mole.
RESULTS AND DISCUSSION
Growth and lactate consumption
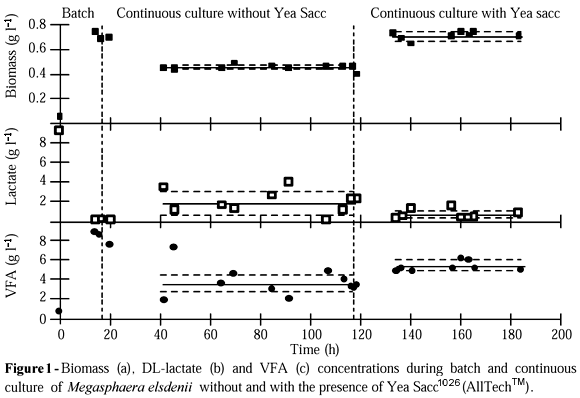
Biomass, DL-lactate and VFA evolution during the process are shown in Figure 1, where the batch and two stages of continuous culture can be observed. During batch culture, a maximum biomass concentration of 0.75 g l-1 was obtained. This result is in agreement to that obtained by Rossi et al. (1995) working with a medium of similar composition. Fig. 1a shows that biomass concentration was kept constant (0.46±0.01 g l-1) during 80 hours of continuous culture and therefore a steady state was considered. In order to simulate a step type perturbation, enough solution of Yea Sacc was added to the culture to obtain the concentration of Yea Sacc indicated previously (0.675 g l-1). Then, the culture was fed with a broth containing the yeast additive (0.675 g l-1). Concentration of biomass increased until a new steady state (0.72±0.03 g l-1, during 50 hours), as indicated in Figure 1a. Biomass concentration increased 54% in the presence of the yeast additive. A Student t test indicated significant difference (p<0.05) between steady state concentrations of biomass.
If complete utilization of yeast additive (0.675 g l-1) as carbon source was assumed and considering the highest reported yield of biomass (0.25 g g-1, for glucose at D 0.1 h-1) for M. elsdenii (Russell and Baldwin, 1979), it explained up to 68% of the observed biomass increase. Then, biomass concentration increase could not be explained by a yield related to the used amount of additive and Yea Sacc filtrate did not mean an increased availability of carbon.
Lactate concentration in the feed was 9.32±0.21 g l-1 along of continuous culture. Residual concentration of lactate during steady state without additive was 1.84±1.03 g l-1 (Figure 1b), then the consumed amount of lactate was 7.48±0.82 g l-1 in this stage. In the second steady state (in the presence of the yeast additive), the consumed amount of lactate was 8.63±0.26 g l-1 (residual concentration, 0.69±0.47 g l-1). According to this when the yeast additive was present, a greater consumption of lactate was observed. Steady state DL-lactate consumption was increased 18% and significant differences were found by using a Student t test (p<0.05).
Volatile fatty acids production
Total VFA concentration increased 37% (p<0.05) when the additive was present in continuous culture (Figure 1c). Butyrate was the minor product in batch culture, but it was
the main product in continuous culture while acetate and propionate proportions were decreased. Hino and coworkers (1991) found that the molar fraction of butyrate was decreased from 0.66 to 0.50 when batch culture was changed to continuous culture, using glucose as carbon source. Nevertheless, in this work it increased from 0.11 to 0.53 while using lactate as carbon source. It would worth to indicate that
M. elsdenii did not produce propionate during fermentation of glucose (Hino & Kuroda, 1993).
Figure 2 shows the individual evolution of VFA. Concentrations of acetate and propionate decreased during the early stage of continuous culture and remained nearly constant from 40 to 120 h. A Student t test did not show significant differences (p<0.05) between steady state concentrations. In fact, the variation of the total concentration of VFA could be attributed mainly to the variation of butyrate concentration. On the other hand, under steady state conditions yeast additive did not affect the concentrations of acetate and propionate, but increased the level of butyrate. A Student t test indicated significant difference (p<0.05) between steady state concentrations of butyrate when yeast additive was added. This result was different from that reported by Callaway & Martin (1997), who found that a yeast additive increased the production of acetate and propionate and did not change the butyrate production, working in batch culture and using a different yeast additive (Diamond V XPTM).
Table 1 presents the yields of biomass and formed products, where all species present in the fermentation are expressed on the basis of one C-mole. Values in this table showed that yields of acetate and propionate were not changed by the presence of the yeast additive, while biomass and butyrate yields were increased. In each stage of continuous culture, all yields were added and addition should be equal to one, in order to obtain a carbon balanced fermentation. The addittion of yields for steady states were not equal to one, then other products must be formed. On the other hand, according to actual knowledge of the metabolism of M. elsdenii (Gottschalk, 1986; Marounek et al., 1989) valerate and CO2 must have been also produced under experimental conditions used. Metabolic engineering fundamentals indicate that in a black-box model carbon balance:
and degree of reduction balance:
must be satisfied (Stephanopoulos et al., 1998). Variables in equations (1) and (2) are:
YVS = Yield of valerate
YCS = Yield of CO2
gS = Degree of reduction of lactate = 4
gX = Degree of reduction of biomass = 4.14
gA = Degree of reduction of acetate = 4
gP = Degree of reduction of propionate = 4.67
gB = Degree of reduction of butyrate = 5
gV = Degree of reduction of valerate = 5.2
Biomass degree of reduction was calculated for an average biomass composition (Stepahnopoulos, et al., 1998). It must be noted that CO2 did not participate in the degree of reduction balance (equation 2), because it is considered redox neutral. Solving to unknown values of YVS and YCS for the steady state without additive:
YVS = 0.148 (mol C-valerate) (mol C-lactate)-1
YCS = 0.176 (mol C-CO2) (mol C-lactate)-1
In similar way for the steady state with Yea Sacc:
YVS = 0.001 (mol C-valerate) (mol C-lactate)-1
YCS = 0.165 (mol C-CO2) (mol C-lactate)-1
According to these results, the lactate catabolism of M. elsdenii was changed yielding more butyrate and diminishing the production of valerate, while acetate and propionate production was not affected. The effect of some additives on the growth and lactate uptake by M. elsdenii has been mainly studied in batch culture. In this work data of continuous culture by M. elsdenii indicated that biomass and butyrate production were stimulated due to the presence of Yea Sacc filtrate.
M. elsdenii is the main lactate utilizer in the rumen (Counotte et al., 1981), and has a very important role to prevent lactic acidosis (Kung & Hession, 1995).
Results presented here suggested that Yea Sacc filtrate could contain growth factors that improved the growth of M. elsdenii, increasing the YXS from 0.071 to 0.095 in the presence of addtive, and enhancing its capacity to diminish the lactate concentration in the medium. On the other
hand, Dawson and coworkers (1990) pointed out that live yeast cells were necessary in the rumen bacteria growth stimulation. Our results suggested that soluble compounds in the filtrate without living cells have an important effect. Callaway & Martin (1997) reported similar results for M. elsdenii growing in batch culture. Their results suggested that soluble factors in the filtrate were involved in stimulating growth.
Results reported in this work suggested that soluble compounds present in the yeast additive filtrate enhanced the anabolism of M. elsdenii and modified the carbon fluxes through its pathways, by increasing butyrate and decreasing valerate production in continuous culture.
ACKNOWLEDGEMENTS
We thank Spanish Type Culture Collection (CECT) for strain donation, UAM and EC (Contract IC18*CT970185) for financial support and CONACyT for OSC scholarship.
We also thank Dr. Ernesto Favela-Torres for helpful suggestions.
RESUMO
Estudo em cultura contínua da bactéria ruminal Megasphaera elsdenii que utiliza lactato como fonte de carbono foi realizada. Butirato, foi o produto produzido em menor concentração na cultura em batelada, porém em cultura contínua foi o produto de maior concentração.
Sob condições definidas a concentração de biomassa aumentou de 0.46 a 0.72 g l-1. A adição do aditivo Yea Sacc não resultou num aumento da disponibilidade do carbono. As quantidades produzidas de valerato e CO2 foram calculadas pelo uso combinado do balanço e grau de redução do carbono. De acordo com esses resultados, o catabolismo de lactato de M. elsdenii foi mudado pela adição de aditivo a base de levedura. Maior rendimento em butirato e redução na produção de valerato foi observada, por outro lado a produção deacetato e propionato não foi afetada. Os resultados desse trabalho sugerem que os componentes solúveis presentes no aditivo a base de levedura aumentam o anabolismo de M. elsdenii modificando o fluxo de carbono através de suas vias de metabólicas, pelo aumento do butirato e redução do valerato. Em cultura contínua.
Received: August 15, 2000;
Revised: September 22, 2000;
Accepted: February 16, 2001.
- Boehringer Mannheim. (1999), Biochemicals Catalog. Boehringer Mannheim, Mexico.
- Callaway, E. S. and Martin, S. A. (1997), Effects of a Saccharomyces cerevisiae culture on ruminal bacteria that utilize lactate and digest cellulose. J Dairy Sci, 80, 2035-2044
- Chaucheyras, F.; Fonty, G.; Bertin, G.; Salmon, J. M. and Gouet, P. (1996), Effects of a strain of Saccharomyces cerevisiae (Levucell SC), a microbial additive for ruminants, on lactate metabolism in vitro. Can J Microbiol, 42, 927-933
- Counotte, G. H.; Prins, R. A.; Janssen, R. H. A. M. and deBie, M. J. A. (1981), Role of Megasphaera elsdenii in the fermentation of DL[2-13C] lactate in the rumen of dairy cattle. Appl Environ Microbiol, 42, 649-655
- Dawson, K. A.; Newman, K. E. and Boling, J. A. (1990), Effects of microbial supplements containing yeast and lactobacilli on roughage-fed ruminal microbial activities. J Anim Sci, 68, 3392
- Goodhue, C. T.; Rosazza, J. P. and Peruzzotti, G. P. (1986), Methods for transformation of organic compounds. In- Manual of Industrial Microbiology and Biotechnology, eds. Demain, A. L. and N. A. Solomon, American Society for Microbiology, Washington D. C., 97-121
- Gottschalk, G. (1986), Bacterial Metabolism. Second edition. Springer-Verlag. N. Y.
- Henderson, C. (1980), The influence of extracellular hydrogen on the metabolism of Bacteroides rumicola, Anaerovibrio lipolytica and Selenomonas ruminantium J Gen Microbiol, 119, 485-491
- Hino, T.; Miyazaki, K. and Kuroda, S. (1991), Role of extracellular acetate in the fermentation of glucose by a ruminal bacterium, Megasphaera elsdenii J Gen Appl Microbiol 37, 121-129
- Hino, T. and Kuroda, S. (1993), Presence of lactate dehydrogenase and lactate racemase in Megasphaera elsdenii grown on glucose or lactate. Appl Environ Microbiol, 59, 255-259
- Hino, T., Shimada, K. and Maruyama, T. (1994), Substrate preference in a strain of Megasphaera elsdenii, a ruminal bacterium, and its implications in propionate production and growth competition. Appl Environ Microbiol, 60, 1827-1831
- Jouany, J. P. (1982), Volatile fatty acid and alcohol determination in digestive contents, silage juice, bacterial cultures and anaerobic fermentor contents. Sci Aliments, 2, 131-144
- Kung, L. and Hession, A. (1995), Preventing in vitro lactate acumulation in ruminal fermentations by inoculation with Megasphaera elsdenii J Anim Sci, 73, 250-256.
- Marounek, M. and Bartos, S. (1987), Interactions between rumen amylolytic and lactate-utilizing bacteria in growth on starch. J Appl Bacteriol, 63, 233-238
- Marounek, M.; Fliegrova, K. and Bartos, S. (1989), Metabolism and some characteristics of ruminal strains of Megasphaera elsdenii Appl Environ Microbiol, 55, 1570-1573
- Papoutsakis, E. T. (1984), Equations and calculations for fermentation of butyric acid bacteria. Biotechnol Bioeng, 26, 174-187
- Rossi, F.; Cocconcelli, P. S. and Masoero, F. (1995), Effect of a Saccharomyces cerevisiae culture on growth and lactate utilization by the ruminal bacterium Megasphaera elsdenii Ann Zootech, 44, 403-409
- Russell, J. B. and Baldwin, R. L. (1979), Comparison of maintenance energy expeditures and growth yields among several rumen bacteria grown in continuous culture. Appl Environ Microbiol, 39, 604-610
- Russell, J. B. and Dombrowsky, D. B. (1980), Effect of pH on the efficiency of growth by pure cultures of rumen bacteria in continuous culture. Appl Environ Microbiol, 37, 537-543
- Russell, J. B.; Cotta, M. A. and Dombrowski, D. B. (1981), Rumen bacterial competition in continuous culture: Streptococcus bovis versus Megasphaera elsdenii Appl Environ Microbiol, 41, 1394-1399
- Stephanopoulos, G.; Nielsen, J and Aristodou, A. (1998), Metabolic engineering. Principles and methodologies. Academic Press, N. Y.
- Waldrip, H. M. and Martin, S. A. (1993), Effects of an Aspergillus oryzae fermentation extract and other factors on lactate utilization by the ruminal bacterium Megasphaera elsdenii J Anim Sci, 71, 2770-2776
- Wallace, R. J. (1986), Catabolism of amino acids by Megasphaera elsdenii LC1. Appl Environ Microbiol, 51, 1141-1143
- Wallace, R. J. and Newbold, C. J. (1992), Probiotics for ruminants. In- Probiotics: the scientific basis, ed. R. Fuller, Chapman and Hall, London, 317-353
Publication Dates
-
Publication in this collection
07 Nov 2001 -
Date of issue
June 2001
History
-
Reviewed
22 Sept 2000 -
Received
15 Aug 2000 -
Accepted
16 Feb 2001