Abstract
Production of manganese-dependent peroxidase (MnP) by the white-rot fungus Phanerochaete chrysosporium BKM-F-1767 (ATCC 24725) was monitored during growth in different media and growth conditions. The effect of some activators of MnP production, Mn2+, Tween 80, phenylmethylsulphonylfloride (PMSF), oxygen, temperature, pH, glycerol and nitrogen was studied. Supplementing the cultures with Tween 80 (0.05 %, v/v) and Mn2+ (174 µM) resulted a maximum MnP activity of 356 U/L which was approximately two times higher than that obtained in the control culture (without Tween 80). Decolourisation of Direct Blue 15 and Direct Green 6 (50 mg/L) was also achieved with MnP.
Phanerochaete chrysosporium; manganese dependent peroxidase; Tween 80; Mn2+; phenylmethylsulphonylfloride
AGRICULTURE, AGRIBUSINESS AND BIOTECHNOLOGY
Enhanced production of manganese peroxidase by Phanerochaete chrysosporium
Raziye Ozturk UrekI, * * Author for correspondence ; Nurdan Kasikara PazarliogluII
IDokuz Eylul University, Faculty of Education, Department of Chemistry, 35150 Buca-Izmir - Turkey
IIEge University, Faculty of Science, Department of Biochemistry, 35100 Bornova - Izmir - Turkey
ABSTRACT
Production of manganese-dependent peroxidase (MnP) by the white-rot fungus Phanerochaete chrysosporium BKM-F-1767 (ATCC 24725) was monitored during growth in different media and growth conditions. The effect of some activators of MnP production, Mn2+, Tween 80, phenylmethylsulphonylfloride (PMSF), oxygen, temperature, pH, glycerol and nitrogen was studied. Supplementing the cultures with Tween 80 (0.05 %, v/v) and Mn2+ (174 µM) resulted a maximum MnP activity of 356 U/L which was approximately two times higher than that obtained in the control culture (without Tween 80). Decolourisation of Direct Blue 15 and Direct Green 6 (50 mg/L) was also achieved with MnP.
Keywords: Phanerochaete chrysosporium; manganese dependent peroxidase; Tween 80; Mn2+; phenylmethylsulphonylfloride
INTRODUCTION
White-rot fungi are primarily responsible for the initial decomposition of lignin in wood, which occurs via an oxidative and relatively nonspecific process (Hatakka, 1994; Hammel et al., 2002). Phanerochaete chrysosporium, a white-rot-wood decaying basidiomycete, produces a potent lignin degrading system that oxidizes lignin completely to CO2 (Breen and Singleton, 1999). The major components of the lignin degrading system include lignin peroxidase (LiP), manganese-dependent peroxidase (MnP), laccase and hydrogen peroxide producing enzymes (Silva et al.,, 2005). Based on enzyme production pattern, white-rot fungi have been classified in different categories (Tuor et al., 1995).
Manganese and nitrogen have strong regulating effects on the ligninolytic machinery (Hamman et al., 1999). In white-rot fungus, P. chrysosporium, ligninolytic enzymes are synthesized in response to nitrogen, carbon or sulfur limitation (Hamman et al., 1997). The mode of cultivation also influences production of the enzymes by basidiomycetes (Niku-Paavola et al.,1990).
MnP (EC. 1.11.1.13) is a ligninolytic enzyme produced by most white-rot fungi (Hatakka, 1994). The catalytic cycle is similar to that of other peroxidases. The principal role of Mn2+-dependent peroxidase is related to oxidation of Mn2+ to Mn3+, which then binds to an appropriate ligand, diffuses from the enzyme, and, in turn oxidizes phenolic substrates (Warrishi et al., 1989; Glenn et al., 1986). Among ligninolytic enzymes secreted by white rot fungi, MnP has been reported as the main enzyme involved in lignin depolymerization, effluent decolourization and biobleaching of pulp (Sahoo and Gupta, 2005; Hofrichter, 2002).
In order to improve both production and stability of MnP secreted by P. chrysosporium culture conditions have been modified by adding different substances to the culture media. Among these additives, Mn2+, Tween 80, PMSF, oxygen, glycerol, nitrogen and temperature, pH as physical conditions were selected, to study their influence on the MnP secreted by cultures of P. chrysosporium. The analysis of the obtained data in flask cultures will give insight into the main production variables, which would directly affect enzymatic regulation, fungal growth and properties of the growth medium.
MATERIALS AND METHODS
Microorganism and growth medium
P. chrysosporium BKM-F-1767 (ATCC 24725) was maintained at 37°C on 2% malt agar slants and plates. Spores were harvested, filtered through glass wool, and kept at -20°C before use (Jäger et al., 1985). The growth medium was prepared according to Tien and Kirk (1988) with 10 g glucose per litre as carbon source, except that dimethylsuccinate was replaced by 20 mM acetate buffer (pH 4.5) (Tien and Kirk, 1988; Dosoretz et al., 1990a).
Culture conditions of P. chrysosporium
Spores (6 days old) were harvested in sterile water, filtered through glass-wool and diluted to 0.5 at OD650. 1 ml of this spore suspension was added to growth medium (10 ml) in 100 ml flasks. Incubation was carried out statically at 37ºC. Enzyme production was carried out growth medium containing selected inducers.
The production medium composition was identical with the growth medium. Some flasks were supplemented with Mn2+ (100-354 µM), oxygen (1.2 MPa), Tween 80 (0.03-0.12 %, v/v), others with phenylmethylsulphonylfloride (PMSF, to a final concentration of 0.1 mM) and the rest with nitrogen (11-44 mM) and glycerol (0.665-10 g/L). These were added the beginning incubation whereas PMSF was introduced after 3 and 5 days of incubation in order to inhibit protease activity. Additionally, in the experiment with oxygen, the headspace was aseptically flushed with O2 gas for 3 min at the time of inoculation and afterwards once a day.
The Erlenmeyer flasks were loosely capped with cellulose stoppers, which permitted a passive aeration, and incubated statically at 37°C and 90% humidity.
Enzymatic decolourization
The reaction mixture contained enzyme and dye (50 mg/L; Direct Blue 15 and Direct Green 6) in a total volume of 1 ml. The decolourization of dyes was determined as the relative decrease of absorbance at their maximum wavelengths by using Na-acetate buffer, pH 4.5 at 30°C.
Analytical methods
Mn (II)-dependent peroxidase activity was assayed spectrophotometrically by the method of Kuwahara et al. (1984). One unit was defined as the amount of enzyme that oxidised 1 µmol of dimethoxyphenol per minute and the activities at 30 ºC were expressed in U/L.
Protease activity was measured with azocoll as substrate (Dosoretz et al.,1990a). One unit was defined as the amount of enzyme, which releases azodye causing an absorbance change of 0.001 per min, measured at 520 nm.
Mycelial dry weight was measured daily by filtering triplicate cultures and drying for 24 h at 105ºC.
All results were the mean of the least three replicates.
RESULTS AND DISCUSSION
Since, media composition and growth conditions are important factors affecting the production of extracellular ligninolytic enzymes and, consequently lignin degradation (Niku-Paavola et al., 1990), in the present work, different supplementations were added to the culture medium in order to study the influence of such substances on the Mn (II)-dependent peroxidase production of this fungus.
Effect of Tween 80 on MnP activity
Several authors have shown an improvement in enzyme excretion in the presence of certain surfactants such as Tween 80 in cultures of P. chrysosporium (Jäger et al., 1985; Asther et al., 1987; Lestan et al., 1990 and 1993). Apparently, Tween 80 transformed the cell membrane structure and promoted the permeation of MnP from the cell into the medium. The highest MnP activity was obtained with 0.05 % Tween 80 (v/v) (Fig.1).
When the culture was supplemented with 0.05 % Tween 80, MnP values were approximately 2-fold higher than those obtained in the control culture (180.2 U/L) (without Tween 80). The mechanism by which surfactants (i.e., Tween 80) enhance extracellular enzyme production in filamentous fungi has not been elucidated. The mechanism possibly consists of modification of the cytoplasmic membrane (Jäger et al., 1985) or in a change in lipid metabolism of the fungus (Asther et al.,1988a-b).
Effect of Mn2+ on enzyme activity
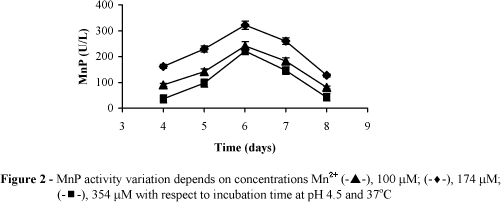
An accumulation of manganese, deposited as MnO2, can be observed in areas of intense lignin degradation by several white-rot fungi (Blanchette, 1984). The effect of adding various concentrations of Mn2+ on P. chrysosporium is shown in Fig. 2.
MnP activity increased steadily with increasing Mn2+concentration. Mn2+ is implicated in MnP production and has a triple role: (i) an essential cofactor for functioning of the MnP protein (Glenn et al., 1986); (ii) activates transcription of the mnp gene (Brown et.al., 1990) and (iii) inhibits the production of veratryl alcohol, which is highly correlated with LiP titers (Mester et al.,1995). A regulatory role in biosynthesis of MnP is ascribed to manganese ions (Perez and Jeffriez, 1992). In the present study, the highest enzyme activity of 321.1 U/L was obtained at 174 µM Mn2+. The presence of high Mn2+ concentration (174 µM Mn2+) in growth medium also elevated the production of cell mass. There might be interactions between high Mn2+ concentration and the nutrients that accounted for the differences in dry weight. Higher concentrations of Mn2+ than 174 µM did not produce a further increase in MnP level.
Effect of PMSF on MnP activity
PMSF, inhibitor of serine protease was applied to a final concentration of 0.1 mM after 3 and 5 days of incubation and the effect on the MnP activity was measured. The loss of ligninolytic activity and its decay have been investigated in P. chrysosporium, in relation to protease production (Bonnarme and Asther, 1993). The protease activity is generally induced by starvation in nitrogen limited medium and is characteristic for P. chrysosporium metabolism. Therefore, the protease takes part in the regulation of MnP activity in the culture. PMSF has been reported to be non-inhibitory to ligninase activity in crude extracellular fluid (Tien and Kirk, 1984). The incubation of PMSF with P. chrysosporium did not increase MnP activity. Therefore, protease activity in 6th day culture of P. chrysosporium was determined as low as 0.6 U/L.
Effect of temperature and pH on MnP activity
In liquid cultures, it was demonstrated that incubation temperature influenced differently the activity pattern of ligninolytic enzymes over time. The change in temperature may affect fatty acid synthesis and membrane fluidity (Vyas et al., 1994). The action of temperature on secondary metabolism occurs at some point in the transitional period between the cessation of vegetative growth and the formation of secondary end products (Asther et al.,1988a). MnP production in medium containing 0.05 % Tween 80 and 174 µM Mn2+ was investigated at different temperatures (20-45ºC) and at pH 4.5 or at different pH values (3-5.5) at 37ºC (Figs. 3, 4).
MnP production was dependent on the cultivation temperature and no activity was detected at 25ºC. Increasing the temperature to 30ºC resulted in the production of small amounts of MnP. A decrease of enzyme production was observed at 40-45ºC. The optimum temperature of 37ºC reported for enzyme production for the fungus support its ecological adaptation in nature (Heinzkill et al., 1998). The MnP production rate and the maximum MnP activity varied with temperature and pH. For MnP production by P. chrysosporium the optimal value of medium pH was 4.5. The maximum MnP activity was obtained at 37ºC and pH 4.5 after 6 days incubation.
Effect of oxygenation conditions
Since O2 is necessary to support the ligninolytic system, two methods of oxygen supply (headspace and 1.2 MPa) were tested. Oxygenation at 1.2 MPa did not increase enzyme activity, because the polysaccharide production might increase (Dosoretz et al., 1990a-b). An effect was found with the oxygenation supply, which presented no regulatory effect directly on enzyme synthesis. The growth-limiting factor could not be attributed to oxygen because increasing it's concentration in the media had no effect on the growth and production of enzyme. Similar result was observed in Bjerkandera sp. strain BOS55 (Mester et al.,1996). Mechanically agitated cultures are known to have an inhibitory effect on ligninolytic enzyme production due to the shear stress for mycelia (Kirk et al.,1978). Therefore, the static cell culture is desirable for the production of MnP because it increases the contacting area between cells and oxygen without shear stress.
Effect of nitrogen concentration
Ligninolytic enzymes are produced by carbon, nitrogen or sulfur limitation (Faison and Kirk, 1985). It was demonstrated that the regulatory role of nitrogen concentration affected lignin degradation and the biosynthesis of MnP. To test the effect of nitrogen source levels on P. chrysosporium (NH4)2SO4 (11-44 mM) was added to growth medium (Fig. 5). Results showed that increased nitrogen concentration decreased enzyme activity. The appearance of MnP activity with high nitrogen conditions could be due to nutrient limitation into the mycelial mat (Mester et al., 1996). The effect of nitrogen concentration on MnP activity could be related with the C/N ratio. The highest enzyme activity was obtained at 22 mM concentration of nitrogen.
Effect of glycerol concentration
It is assumed that the metabolized substrate is essential for fungi not only for the synthesis of lignin degrading enzymes, but also for the production of peroxide and synthesis of effectors of the ligninolytic system (Faison and Kirk, 1985). Therefore, experiments were conducted to determine the effect of glycerol concentration on MnP production. The addition of various glycerol concentrations (0.665-10 g/L) did not increase the MnP activity than obtained with glucose (Table 1). Thus, glycerol was a poorer carbon source compared to glucose (Kern, 1990). Growth on glycerol leads to carbon limitation which affects the onset of secondary metabolism (Buswell et al., 1984). Therefore, glucose (10 g/L) was selected as carbon source.
CONCLUSION
Highest MnP activity was achieved as 356 U/L at optimized conditions: 174 µM Mn2+, 0.05 % Tween 80, 22 mM nitrogen, 10 g/L glucose, 37ºC and pH 4.5 (Fig.6). Under these conditions, growth of the microorganism was achieved with a maximum value of 3.1 g/L biomass.
Decolourisation studies by enzyme
Synthetic dyes such as Direct Blue 15 (50 mg/L; l=610 nm) and Direct Green 6 (50 mg/L; l= 620 nm) were treated with MnP. The decolourization of these dyes was performed for Direct Blue 15 as 12 % and Direct Green 6 as 5.4 % after 5 min of treatment. This was an extremely short period to achieve azo dye degradation. According to the results, MnP produced by P.chrysosporium was able to play a role for the decolourization of these direct azo dyes (Kasikara Pazarlioglu et al., 2005; Harazono et al., 2003).
Received: June 21, 2005;
Revised: June 09, 2006;
Accepted: May 18, 2007.
- Asther, M., Corrieu, G., Drapron, R., Odier, E. (1987), Effect of Tween 80 and oleic acid on ligninase production by Phanerochaete chrysosporium INA-12. Enzyme and Microbial Technol., 9, 245249.
- Asther, M., Capdevila, C., Correu, G. (1988a), Control of lignin peroxidase production by Phanerochaete chrysosporium INA-12 by temperature shifting. Appl. Env. Microbiol., 54, 3194-3196.
- Asther, M., Lesage, L., Drapron, R., Corrieu, G., Odier, E. (1988b), Phospholipid and fatty acid enrichment of Phanerochaete chrysosporium INA-12 in relation to ligninase production. Appl. Env. Microbiol., 27, 393_398.
- Blanchette, R.A. Manganese accumulation in wood decayed by white-rot fungi. Phytopath., 74, 725730.
- Bonnarme, P., Asther, M. (1993), Influence of primary and secondary proteases produced by free or immobilized cells of the white-rot fungus Phanerochaete chrysosporium on lignin peroxidase activity. J. Biotechnol., 30, 271-282.
- Breen,A., Singleton, F.L. (1999), Fungi in lignocellulose breakdown and biopulping. Curr. Opin. Biotechnol., 10, 252258.
- Brown, J.A., Glenn, J.K., Gold, M.H. (1990), Manganese regulates expression of manganese peroxidase by Phanerochaete chrysosporium J. Bacteriol., 172, 3125-3130.
- Buswell, J.A., Mollet, B., Odier, E. (1984), Ligninolytic enzyme production on by Phanerochaete chrysosporium under conditions of nitrogen sufficiency. FEMS Microbiol. Lett., 25, 295-299.
- Dosoretz, C.G. Chen, H.C. Grethlein, H.E. (1990a), Effect of environmental conditions on extracellular protease activity in ligninolytic cultures of Phanerochaete chrysosporium Appl. Environ. Microbiol, 56, 395400.
- Dosoretz, C.G., Chen, H.C., Grethlein, M.E. (1990b), Effect of oxygenation conditions on submerged cultures of Phanerochaete chrysosporium Appl. Microb. Biotech., 34,131137.
- Faison, B.D., Kirk, T.K. (1985), Factors involved in the regulation of a ligninase activity in Phanerochaete chrysosporium Appl. Environ. Microbiol., 49, 299-304.
- Glenn, J.K., Akileswaran, L., Gold, M.H. (1986), Mn(II) oxidation is principal function of the extracellular Mn-peroxidase from Phanerochaete chrysosporium. Arch. Biochem. Biophys, 251, 688-396.
- Hatakka, A. (1994), Lignin modifying enzymes from selected white rot fungi- production and role in lignin degradation. FEMS Microbiol. Rev., 13, 125-135.
- Heinzkill, M., Bech, L., Halkier, T., Scheider, F., Anke, T. (1988), Characterization of laccases and peroxidases from wood rotting fungi. Appl. Environ. Microbial., 64, 1601-1606.
- Hofrichter, M. (2002), Review: Lignin conversion by manganese peroxidase. Enzyme Microb. Technol., 30, 454-466.
- Jäger, A., Croan, C., Kirk, T.K. (1985), Production of ligninases and degradation of lignin in agitated submerged cultures of Phanerochaete chrysosporium Appl. Environ. Microbiol., 50, 12741278.
- Hamman, O. B., Rubai, T., Martínez, J. (1997), Effect of carbon and nitrogen limitation on lignin peroxidase and manganese peroxidase production by Phanerochaete flavido-alba. J. App. Microbiol, 83, 751-757.
- Hamman, O. B., Rubai, T., Martínez, J. (1999),The effect of manganese on the peroxidases in nitrogen limited cultures. FEMS Microb. Lett.,177, 137-142. production of Phanerochaete flavido-alba ligninolytic
- Hammel, K.E., Kapich, A.N., Jensen, K.A., Ryan, Z.C. (2002), Reactive oxygen species as agents of wood decay by fungi, Enzyme Microb. Technol., 30, 445453.
- Harazono, K., Watanabe, Y., Nakamura, K. (2003), Decolorization of azo dye by the white-rot basidiomycete Phanerochaete sordida and by its manganese peroxidase. J. Biosci. Bioeng., 95, 455-459.
- Kasikara Pazarlioglu, N., Ozturk Urek, R., Ergun, F. (2005), Biodecolorization of Direct Blue 15 by Immobilized Phanerochaete chrysosporium Process Biochem., 40/5, 1923-1929.
- Kern, H.W. (1990), Production and stability of lignin peroxidase of Phanerochaete chrysosporium cultivated on glycerol in the presence of salid manganese (IV) oxide. Appl. Microbiol. Biotech., 33, 582-588.
- Kirk, T.K., Schultz, E., Connors, W.J., Lorenz, L.F., Zeikus, F.G. (1978), Influence of culture parameters on lignin metabolism by Phanerochate chrysosporium Arch. Microbial., 117, 277-285.
- Kuwahara, M., Glenn, J.K.. Morgan, M.A., Gold, M.H. (1984), Separation and characterization of two extracellular H2O2-dependent oxidases from ligninolytic cultures of Phanerochaete chrysosporium FEBS Lett., 169, 247250.
- Lestan, D., Strancar, A., Perdih, A. (1990), Influence of some oils and surfactants on ligninolytic activity, growth and lipid fatty acids of Phanerochaete chrysosporium Appl. Microbiol. Biotechnol., 34, 426428.
- Lestan, D., Cernilec, M., Strancar, A., Perdih, A. (1993), Influence of some surfactants and related compounds on ligninolytic activity of Phanerochaete chrysosporium FEMS Microbiol. Lett., 106, 1722.
- Mester, T., de Long, E., Field, J.A. (1995), Manganese regulation of veratryl alcohol in white rot fungi and its indirect effect on lignin peroxidase. Appl. Environ. Microbiol., 61, 1881-1887.
- Mester, T., Pena, M., Field, J.A. (1996), Nutrient regulation of extracellular peroxidases in the white-rot fungus Bjerkandera sp. strain BOS55. Appl. Microb.Biotechnol., 44, 778-784.
- Niku-Paavola, M.L., Karhunen, E., Kantelinene, A., Viikari, L., Lundell, T., Hatakka A. (1990), The effect of culture conditions on the production of lignin modifying enzymes by the white rot fungus Phlebia radiata J. Biotechnol., 13, 211-221.
- Perez, J., Jeffriez, T.W. (1992), Roles of manganese and organic acid chelators in regulating lignin degradation and biosynthesis of peroxidases by Phanerochaete chrysosporium Appl. Environ. Microbiol., 58, 2402-2408.
- Sahoo, D. K., Gupta, R. (2005), Evaluation of ligninolytic microorganisms for efficient decolorization of a small pulp and paper mill effluent. Process Biochem., 40, 1573-1578.
- Silva, C.M.M.S., Melo, I.S., Oliveira, P.R. (2005), Ligninolytic enzyme production by Ganoderma spp. Enzyme Microb. Technol., 37, 324-329.
- Tien, M., Kirk, T.K. (1984), Lignin-degrading enzyme from Phanerochaete chrysosporium Purification, characterization and catalytic properties of an unique H2O2-requiring oxygenase. Proc. Natl. Acad. Sci. USA., 81, 22802284.
- Tien, M., Kirk, T.K. (1988), Lignin peroxidase of Phanerochate chrysosporium Meth. Enzymol., 161, 238249.
- Tuor, U., Winterhalter, K., Fiechter, A. (1995), Enzymes of white rot fungi involved in lignin degradation and ecological determinants for wood decay. J. Biotechnol, 41, 1-17.
- Vyas, B.R.M., Volc, J., Sasek, V. (1994), Effects of temperature on the production of manganese peroxidase and lignin peroxidase by Phanerochaete chrysosporium Folia Microbiol., 39, 19-22.
- Warrishi, H., Dunford, H.B., Mcdonald, I.D., Gold M.H. (1989), Manganese peroxidase from the lignin-degrading basidiomycete Phanerochaete chrysosporium Transient state kinetics and reaction mechanism. J. Biol. Chem., 264, 3335-3340.
Publication Dates
-
Publication in this collection
03 Mar 2008 -
Date of issue
Nov 2007
History
-
Received
21 June 2005 -
Reviewed
09 June 2006 -
Accepted
18 May 2007