Abstract
The aim of this study is to determine the amounts of radionuclides' activity present in samples of feed for young and adult chicken and feed for dairy and beef cattle, as well as in dicalcium phosphate (DCP) (CaHPO4), used as a nutritional supplement for animal feed. Radioactivity in these samples is due to the presence of the 238U and 232Th radioactive series, as well as their daughter. In addition, as it is for environmental samples, the activity of 40K should be an important source of radioactivity in all analyzed samples. Gamma rays were measured using a standard spectroscopy system, with a high-resolution HPGe detector. Measured activities in feed samples ranged from 0.23 to 1.51 Bq.kg-1, 0.67 to 4.21 Bq.kg-1, 0.29 to 1.63 Bq.kg-1 and 236 to 402 Bq.kg-1 for 226Ra, 228 Ra, 228 Th and 40K, respectively. Measured activities in DCP samples were 46.6 Bq.kg-1, 83 Bq.kg-1, 4.20 Bq.kg-1 and 16.61 Bq.kg-1 for 40 K, 226 Ra, 228 Ra and 228 Th, respectively. Although 40K activity has reached hundreds of Becquerel in feed samples, it should not represent a risk to human health, not even to animals, since potassium is an essential mineral to living organisms.
Gamma Ray Spectrometry; Natural radioactivity; Animal Feed; dicalcium phosphate (DCP); Phosphate fertilizers
INTRODUCTION
Since radioactivity was first discovered, there has been concern regarding the damage that it might pose to humans, living creatures and the environment. Many studies have been dedicated to monitoring amounts of radionuclides' activity in food and nutritional supplements, particularly related to the human diet, seeking to determine the risks those amounts may bring. (Arruda-Neto (1997)Arruda-Neto, J.D.T, Tavares, M. V., Filadelfo, M. (1997). Concentrations of uranium in animal feed supplements: Measurements and dose estimates. Journal of Radiounalytical and Nuclear Chemistry, Vol. 221, Nº 1-2, 97-104; Carvalho (2006)Carvalho, C., Anjos, R. M., Mosquera, B., Macário, K., Veiga, R. (2006). Radiocesium contamination behavior and its effect on potassium absorption in tropical or subtropical plants. Journal of Environmental Radioactivity 86, 241-250.; Casacuberta (2009)Casacuberta, N., Masque, P., Garcia-Orellana, J., Bruach-Menché, J.M., Sanchez-Cabeza, J.A., Villa, M., Hurtado, S., Manjón, G., Garcia-Tenorio, R. (2007). Dicalcium Phosphate: Contents of Uranium Decay Series. NORM V, March 19-22, Seville, Spain.; Ramli (2009); Zagato (2007)Zagato, V. A. B. Medina, N. H.; Okuno, E.; Umisedo, N.K. "Natural Radiactivity in Bananas". 8th International Symposium (NRE VIII) AIP Conf. Proc. 1034, pp. 264-268 (2007); Tchokossa (2013)Zagato, V. A. B. Medina, N. H.; Okuno, E.; Umisedo, N.K. "Natural Radiactivity in Bananas". 8th International Symposium (NRE VIII) AIP Conf. Proc. 1034, pp. 264-268 (2007); Scheibel (2006)Shanthi, G. Kumaran, J. T. K.; Raj, G. A. G.; Maniyan, C. G. Natural radionuclides in the South Indian foods and their annual dose". Nuclear Instruments and Methods in Physics Research A. (2009), doi:10.1016/j.nima.2009.10.068
https://doi.org/10.1016/j.nima.2009.10.0...
and Shanthi (2009)Silva, N.C., Fernandes, E.A.N., Cipriani, M., Taddei, M.H.T. (2001). The natural radioactivity of Brazilian phosphogypsum. Journal of Radioanalytical and Nuclear Chemistry 249, 251-255. .)
Concern with radiation levels in food samples is also very important, because ingestion is one of the most common way radionuclides enter living organisms. In this study radioactivity was measured in cattle and poultry feed samples. Normally, animal feeds are developed from an organic base and are intended to provide the most complete nutrition possible. In an attempt to optimize its nutritional value, elements that may in fact elevate the amount of radionuclide activity in the feed are sometimes added, as discussed by Arruda-Neto (1997).
Measured feeds were all developed from a base of soy, wheat, corn fibers, sodium chloride and a vitamin complex called "Premix Vitamínico Mineral" ("Vitamin Mineral Premix"). Thus, in these feed samples one must account for the natural radioactivity present in corn, wheat and soy, for example. Plants are primary receivers of radioactive contamination. The absorption of radionuclides from soil depends not only on their physiology but also in the processes that occur in there (UNSCEAR, 2000). However, due to the difference in mobility of Radium and Uranium, as published by Carvalho (2006), one should not expect radioactive equilibrium in natural series in biological samples and processed geological materials. Since radionuclides that are after 226Ra and 228Ra in their respective chains were measured in the present work, it was possible to estimate the activity of 226Ra in the Uranium series, and the activity of 228Ra and 228Th in the Thorium series.
Phosphorous is one of the elements often added to feed, since it is one of the most important minerals for living organisms, including humans. It impacts several important biological processes, such as the osmotic pressure and all metabolic reactions (Casacuberta, 2007). One phosphate source commonly used for raising cattle is dicalcium phosphate (CaHPO4), also known as DCP. It is a nutritional supplement obtained from phosphate rocks that may contain a high level of 238U and its series, depending on its source. Samples of DCP measured by Arruda-Neto (1997), Casacuberta (2009) and Saueia (2005)Saueia, C. H., Mazzilli, B. P. (2006). Distribution of natural radionuclides in the production and use of phosphate fertilizers in Brazil. Journal of Environmental Radioactivity 89, 229-239., for example, presented values for 238U activity ranging from 24 up to 2444 Bq.kg-1.
For this reason, concern with radiation levels in feed and samples is also very important, once ingestion is one of the most common ways radionuclides enter living organisms. Thus, as humans ingest both eggs and poultry, milk and beef, it is relevant to monitor the radiation levels in animal food samples (DCP and feed) since a part of the radioactivity to which these animals are exposed could possibly be transferred to humans. In fact, researchers as Casacuberta (2009) and Arruda-Neto (1997) have reported a concern with possible risk to human health due to concentration of natural radionuclides in animal feed samples and DCP. Depending on the production process of DCP brands sold in Brazil, the annual dose per Brazilian consumer could, for example, reach up to about 10 mSv.y-1, representing a value significantly higher than the dose due to natural background (1.8 - 2 mSv.y-1) and can not be negligible. (Arruda-Neto (1997) and UNSCEAR (1982))
Thus, the present work aims to determine the amount of natural radionuclides in samples of poultry and cattle feed and a sample of DCP; and investigate a possible risk to human health, which feeds the meat of these animals and their derivatives
MATERIALS AND METHODS
Sampling
Samples of cattle feed were divided into two types, one for dairy cattle and another for beef cattle. Samples for poultry feed were also divided between feed for young and adult chickens. The mass of each feed sample was of approximately 0.8 kg. DCP samples masses were around 1.1 kg.
Samples were stored in a 1 L Marinelli beaker, which was completely filled for all samples, sealed and stored for 40 days, in order to reach secular equilibrium between 226Ra and 222Rn before measurements were made. Measurement time was of 86,400 seconds and samples were measured in natura.
For each kind of feed and for DCP, six samples from the same brand were measured, totaling 30 routine samples. Measurements were made at two-month intervals and the background spectrum of the acquision system was checked on a weekly basis.
All feed samples studied were produced and sold in the city of Londrina, in the state of Paraná, Brazil, in 2007. DCP brand tested in this study was also commercialized at the time in Londrina.
Gamma-ray Spectrometry
The acquisition system is the standard for the employed technique, and is composed of an HPGe detector with a relative efficiency of 66% and standard nuclear electronic chain for gamma ray spectrometry. This system is installed in the Applied Nuclear Physics Laboratory (LFNA) at State University of Londrina. Sample and detector were put inside a shielding (Ortec HPLBS1) to reduce the interference of background radiation. Detector energy resolution at 1.17 MeV and 1.33 MeV of 60Co measured 1.95 keV and 2.03 keV, respectively. Gamma spectra were acquired using Maestro 32 software and its analysis was performed with GammaVision software.
Detector efficiency was calibrated by measuring calibration samples of feed and DCP prepared at LFNA by the addition of 50.0 g of RGTh-1, RGU-1 and RGK-1(certified materials that have known activity concentrations and were provided by IAEA) to routine samples. Since these calibration samples were prepared from routine samples, one is assured that they have practically the same density and matrix composition, avoiding attenuation corrections. The process of homogenizing these samples was carried out according to the international standard protocol to ensure homogeneity.
In order to verify the accuracy of the methodology, two samples were produced in the same manner as the calibration sample, as certified samples of feed and DCP were not available in the market. To prepare these two accuracy verification samples, about 43.0 grams of each of three materials mentioned in the previous paragraph with known activity concentrations were used. The comparison between predicted and measured activity in these samples was performed using the international standard of inter-comparison procedure, as employed by IAEA (IAEA (2007)IAEA, (2003). Guidelines for radioelement mapping using gamma ray spectrometry data, Technical Report Series No. 1363. International Atomic Energy Agency, Vienna.; IAEA (2010)).
Background spectra for the feed samples were obtained by measuring empty beakers. In order to determine the background spectrum for DCP, deionized water in a beaker was measured since DCP density is about 1,100 kg/m3. Measurement time for background spectra was also 24 hours.
From 238U series, 214Pb and 214Bi were measured; and from 232Th series, the following radionuclides were quantified: 212Pb, 212Bi, 228Ac and 208Tl.
Specific activities for each gamma line were detected by applying the following equation:
where A is the specific activity; NL, the peak net count; m, the mass of the sample; , the efficiency; p, the probability of decay for this gamma ray line and t is the measurement time.
To determine the deviation in the value of the calculated activity, uncertainties in measurements of net counts and masses were propagated. (Barros-Neto et. al., 2001Barros Neto, B. B., Scarminio, I. S., Bruns, R. E. (2001). Como fazer experimentos: pesquisa e desenvolvimento na ciência e na indústria. Editora da Unicamp.)
To determine the Minimum Detectable Activity (MDA), equation 1 was applied in the same manner as for the calculation of the activity. However, net count value (NL) extracted from photopeak of a background spectrum instead of a routine sample, was employed.
RESULTS AND DISCUSSION
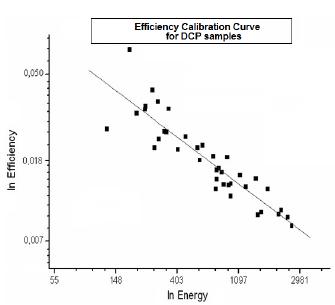
After measuring the calibration samples, the efficiency curves were obtained, which are presented at figures 1 and 2.
Table 1 shows the efficiency curves parameters and ANOVA analysis obtained by linear fit applied to the data from both curves using Microcal Origin 8 software. Quality of the linear fits was very good, as attested by ANOVA results at Table 1.
Table 2 presents the values obtained for accuracy verification.
Accuracy verification by measurement of standard samples. Uncertainties are expanded ones taking into account statistical errors and masses deviations, as usual in the literature of gamma ray spectrometry.
In the method used for verification of accuracy, values found for A1 parameters should not be greater than those for A2. A1 is the magnitude of the difference between predicted activity and measured activity, and parameter A2 is the propagation of measurement uncertainty (() of predicted and measured activities values. It can be determined by equation 2:
where 2.58 is a statistical factor related to 95% confidence rating.
Besides this requirement, the precision value (P) should not be greater than the value of Acceptance Limits for Precision (LAP). The precision value (P), previously presented in Table 2, is obtained by equation 3.
The results shown at Table 2 attest the validity of our methodology.
This method of values comparison is recognized and published by IAEA. (IAEA (2007); IAEA (2010))
Feeds
The activity of 226Ra in feed samples was estimated by measuring 214Pb and 214Bi activities. The 228Ra activity was calculated from 228Ac gamma lines. 228Th activity was estimated from the values of activities of 212Pb, 212Bi, and 208Tl. Weighted average activity was used to calculate the activity of the sub-series.
The results obtained for activity in the measured feed samples are shown in table 3. These results are presented with their deviations within 95% confidence rating, according to Student statistics.
40K, 226Ra, 228Th and 228Ra activity in Feed Samples with deviation within 95% confidence rating.
Table 3 shows the lack of radioactive equilibrium between 228Ra and 228Th in Thorium series, confirming the prior expectation.
Taking the respective deviations into consideration, the recorded values of the activity for 228Th and 226Ra are equivalent, if one compares the two types of cattle feed. The same occurs in the comparison of the activity values of 228Th in the two types of poultry feed.
One also observes that the values of activity of these two radionuclides are equivalent between themselves for the same type of cattle feed. However, in adult poultry feed the value of 226Ra and 228Th are not equivalent due fact that the value of the Thorium series is slightly higher. The values are very close, though. One can see also that in terms of absolute values the 228Ra sub-series presents a higher activity in all feed types.
Cattle feed presents higher concentrations of activity for both daughter isotopes of the Thorium and Uranium series than the poultry feed.
The amount of activity for 40K is also greater in cattle feed than in poultry feed. One should note that the greater concentration of this radionuclide is found in feed destined to dairy cattle.
DCP
As for feeds, radioactive equilibrium in decay series is also not expected for DCP, since its industrial manufacturing processes can cause separation between the Radium and Uranium (Gäfvert (2001)). Radionuclides used to determine the activity of natural series and subseries were the same as in feed samples
By the results obtained for DCP, one sees that the activity of 226Ra is much greater than the activity for Thorium series. The value for 226Ra was (83 + 26) Bq.kg-1, while the value of 228Ra reached (4.20 + 0.54) Bq.kg-1 and 228Th reached (16.61 + 0.89) Bq.kg-1. According to Silva et. al. (2001)United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and biological effects. UNSCEAR, 1982. New York, NY. 1982. 226Ra is the major source of radioactivity in Phosphogypsum and might be incorporated into living organisms, because it follows the same biological pathway of calcium
One notes also that the activities of these radionuclides in this sample are much greater than the activity of the parents of the respective sub-series, 226Ra and 228Ra, in the feed samples. However, activity for 40K was (46.6 + 2.8) Bq.kg-1, a value well below the activity of this radionuclide for the feed samples.
DCP samples measured by Casacuberta are from Spain. Samples of other references in Table 4 are commercialized in Brazil
Comparison of natural series activities in DCP samples with literature. Uncertainties are expanded ones taking into account statistical and masses deviations.
The activity for 232Th measured by Saueia (2005) had a similar value to that obtained in this study for 228Ra and 228Th, which was 10 Bq.kg-1. However, it must be noted that the concentration of Uranium presented by Saueia (2005) is much lower than the values found in the literature.
The activity of 40K measured in present work is of the same magnitude as the results of Casacuberta (2007) in the samples of DCP B, but around three times smaller than the value presented by Saueia (2005).
One sees that the activity of 226Ra in the DCP measured is much lower than the majority of activity for 238U in DCP samples in quoted references, with the exception of DCP F, measured by Arruda-Neto (1997) and of DCP measured by Saueia (2005). In the latter reference, the activity of 238U, did not exceed the MDA. However, radioactive equilibrium between 226Ra and 238U was not expected for DCP samples, as mentioned earlier. For example, Casacuberta (2009) measured 12 samples of DCP for animal feed. Results obtained in cited reference shows, as expected, the lack of equilibrium for DCP samples in both natural decay series. It is important to note that for the DCP sample measured in present work, concentration of 228Th is higher than for 228Ra. The same pattern is observed at Casacuberta (2009). In his paper, this author separates these samples in one group by clustering analysis. According to this reference, one characteristic of this group is a low Radium concentration. Studies from literature indicate that when the phosphate rock is digested with sulphuric acid, it leads to little amounts of 226Ra in DCP (<2%) (Roessler (1990)Roessler, C.E. Control of radium in phosphatemining, beneficiation and chemical processing, The Environmental Behaviour ofRadium, vol. 2, TechnicalReport Series No. 310, IAEA Vienna, 1990, pp. 269-279. and Rutherford (1996)Saueia, C. H., Mazzilli, B. P., Fávaro, D. I. T. (2005). Natural radioactivity in phosphate rock, phosphogypsum and phosphate fertilizers in Brazil. Journal of Radioanalytical and Nuclear Chemistry 264, 445.448.)
Figure 3 presents a comparison among 226Ra activity values of the literature and the present work.
- Comparison of 226Ra activity (Bq.kg-1) in the twelve DCP samples from Casacuberta (2009) and value interval of 226Ra activity (Bq.kg-1) obtained in the present work
Values displayed in Figure 3 shows that six of the samples measured by Casacuberta have lower activity for 226Ra than the samples measured in present work. In the other hand, five DCP samples from Casacuberta are statistically equivalent to values shown in the present work regarding 226Ra activity.
CONCLUSIONS
The activity of 40K, is the main activity observed in the feed samples and an important fraction of the activity in the DCP samples. By the way, the 40K activity in DCP is rather smaller than the activity of this radionuclide in feed samples. Furthermore, this activity represents only around half of 226Ra activity in DCP. Similar behavior is observed in Casacuberta (2007), where the activity of 40K is much lower than of 238U. Furthermore, Martinez-Aguirre (1997)Martinez-Aguirre, A., García-León, M., 1997. Radioactive impact of phosphate ore precessing in a wet marshland in southwestern Spain. Journal of Environmental Radioactivity 34, 45-57. points out that, typically, 40K does not have a high activity in Phosphate ores. Anyway, K is an essential element for living organisms, therefore the 40K radioactivity cannot be avoided. Thus, the activity results for this radionuclide in the present work for both feed and DCP samples are consistent with the expected and do not generate any additional concern.
Results shows that DCP measured has a considerable activity, especially for the 226Ra. Considering the concentration of radionuclide in the DCP brand, one can infer that a large percentage of the radioactivity measured in cattle feed is a result of the feed containing DCP as part of its composition. However, one cannot ascertain how much of the radioactivity is directly attributable to DCP since the manufacturer does not specify the amount of DCP in the feed. This could also be the explanation for the higher values of activity measured in cattle feed when compared to poultry feed. The determination of the amount of DCP added in bovine feeds through the measurement of phosphorus concentration in these samples could be a possible point of future study.
Taking into account the activity of 226Ra in the DCP that was measured in the present work, it was observed that this value was greater than a half of twelve samples measured by Cascuberta (2009) and greater than the sample measured by Saueia (2005). However, 228Th has a higher activity than 228Ra. Taking into account the results of Casacoberta (2009), it might mean that the DCP sample measured in present work was produced by digestion with sulfuric acid. If it is the case, only a small fraction of radium is incorporated by the resulting DCP. (Roessler (1990) and Rutherford (1996))
If this is the case for analyzed DCP sample, although 226Ra activity should not generate any concern by itself since its value is similar to those found in literature, it is very important to note that due to lake of equilibrium in the radioactive series of Uranium, such value should not be equivalent to the 238U activity. In fact, according to what literature indicates, the DCP samples which are produced by phosphate rock digestion with sulfuric acid, 238U activity is usually ten of times greater than 226Ra, and may be up to 150 times higher. Thus, 226Ra activity value cannot rule out the possibility of a higher concentration of 238U in the DCP measured.
One point that contributes to this hypothesis that the sample analyzed was produced by digestion of phosphate rock with sulfuric acid is that the activity of 40K in DCP measured in the present work has the same magnitude only in five out of 12 samples measured by Casacuberta (2009). These five samples are exactly the ones that the author identifies as produced by digestion with sulfuric acid. Therefore, in addition to agreeing with the hypothesis raised, this result suggests that Potassium might be slightly more soluble in sulfuric acid than in hydrochloric acid, which would explain the difference in the values of this radionuclides activity in the two groups of samples.
Thus, a very interesting continuation of present work would be to measure daughters of 238U which comes before 226Ra in the Uranium series in DCP samples. Thus, it would be possible to study the radioactivity equilibrium in the series, and so improve the analysis of processes used in the production DCP.
ACKNOWLEDGMENTS
The authors would like to thank the National Council of Scientific and Technological Development (CNPq) for the financial support.
REFERENCES
- Arruda-Neto, J.D.T, Tavares, M. V., Filadelfo, M. (1997). Concentrations of uranium in animal feed supplements: Measurements and dose estimates. Journal of Radiounalytical and Nuclear Chemistry, Vol. 221, Nº 1-2, 97-104
- Azouazi, M., Ouahidi, Y., Fakhi, S., Andres, Y., Abbe, J.Ch., Benmansour, M. (2001). Natural radioactivity in phosphates, phosphogypsum and natural waters in Morocco. Journal of Environmental Radioactivity 54, 231-242.
- Barros Neto, B. B., Scarminio, I. S., Bruns, R. E. (2001). Como fazer experimentos: pesquisa e desenvolvimento na ciência e na indústria. Editora da Unicamp.
- Carvalho, C., Anjos, R. M., Mosquera, B., Macário, K., Veiga, R. (2006). Radiocesium contamination behavior and its effect on potassium absorption in tropical or subtropical plants. Journal of Environmental Radioactivity 86, 241-250.
- Casacuberta, N., Masque, P., Garcia-Orellana, J., Bruach-Menché, J.M., Sanchez-Cabeza, J.A., Villa, M., Hurtado, S., Manjón, G., Garcia-Tenorio, R. (2007). Dicalcium Phosphate: Contents of Uranium Decay Series. NORM V, March 19-22, Seville, Spain.
- Casacuberta, N., Masque, P., Garcia-Orellana, J., Bruach, J.M., Anguita, M., Gasa, J., Villa, M., Hurtado, S., Garcia-Tenorio, R. (2009). Radioactivity contents in dicalcium phosphate and the potential radiological risk to human populations. Journal of Hazardous Materials 170, 814-823
- Gäfvert, T., Holm, E., Roos, P. (2001). Radionuclide fluxes at a plant manufacturing dicalcium phosphate for domestic animals. Journal of Environmental Radioactivity, 61-73.
- IAEA, (2003). Guidelines for radioelement mapping using gamma ray spectrometry data, Technical Report Series No. 1363. International Atomic Energy Agency, Vienna.
- IAEA, (1994). Handbook of Parameters Values for the Prediction of Radionuclide Transfer in Temperate Environments. Technical Report Series No. 364. International Atomic Energy Agency, Vienna.
- IAEA, (2010). Individual Evaluation Report for Laboratory. Technical Report No. 175. (The IAEA-CU-2009-03 World-wide open proficiency test). International Atomic Energy Agency, Vienna.
- IAEA, 1989. Measurements of radionuclides in food and the environment.Technical Reports Series, 295. International Atomic Energy Agency, Vienna.
- IAEA, (2007). Worldwide Open Proficiency Test on the Determination of Radionuclides in Spinach, Soil and Water. IAEA Analytical Quality in Nuclear Applications Series No. 8. (IAEA-CU-2007-03). International Atomic Energy Agency, Vienna.
- Martinez-Aguirre, A., García-León, M., 1997. Radioactive impact of phosphate ore precessing in a wet marshland in southwestern Spain. Journal of Environmental Radioactivity 34, 45-57.
- Mazzilli, B., Palmiro, V., Saueia C., Nisti, M. B., 2000. Radiochemical Characterization of Brazilian Phosphogypsum. Journal of Environmental Radioactivity, 49, 113-122. doi:10.1016/S0265-931X(99)00097-1
- Ramli, A.I., Ampriantoro, N. H., Wagiran, H., Wood, A. K., Kuan, S. L. 2009. Health Risk Implications of High Background Radiation Dose Rate in Kampung Sungai Durian, Kinta District, Perak, Malaysia. Global Journal of Health Science 1, 140-149.
- Roessler, C.E. Control of radium in phosphatemining, beneficiation and chemical processing, The Environmental Behaviour ofRadium, vol. 2, TechnicalReport Series No. 310, IAEA Vienna, 1990, pp. 269-279.
- Roessler, C.E., Smith, Z.A., Bolch, W.E., Prince, R.J. (1979). Uranium and Radium-226 in Florida phosphate materials, Health Physics, 37, 269-277.
- Rutherford, P.M., Dudas, M.J., Arocena, J.M. (1996). Heterogeneous distribution of radionuclides, barium and strontium in phosphogypsum by-product, The Science of the Total Environment. 180, 201-209.
- Saueia, C. H., Mazzilli, B. P., Fávaro, D. I. T. (2005). Natural radioactivity in phosphate rock, phosphogypsum and phosphate fertilizers in Brazil. Journal of Radioanalytical and Nuclear Chemistry 264, 445.448.
- Saueia, C. H., Mazzilli, B. P. (2006). Distribution of natural radionuclides in the production and use of phosphate fertilizers in Brazil. Journal of Environmental Radioactivity 89, 229-239.
- Saueia, C. H., Mazzilli, B. P., Le Bourlegat, F.M., Costa, G. J. L. (2013) Distribution of potentially toxic elements in the Brazilian phosphogypsum and phosphate fertilizers. Proceedings of the 16th International Conference on Heavy Metals in the Environment, 1.
- Scheibel, V. ; Appoloni, C.R. ; Schechter, H. . Natural radioactivity traces in South-Brazilian cereal flours by gamma-ray spectrometry. Journal of Radioanalytical and Nuclear Chemistry, v. 270, n.1, p. 163-165, 2006
- Shanthi, G. Kumaran, J. T. K.; Raj, G. A. G.; Maniyan, C. G. Natural radionuclides in the South Indian foods and their annual dose". Nuclear Instruments and Methods in Physics Research A. (2009), doi:10.1016/j.nima.2009.10.068
» https://doi.org/10.1016/j.nima.2009.10.068 - Silva, N.C., Fernandes, E.A.N., Cipriani, M., Taddei, M.H.T. (2001). The natural radioactivity of Brazilian phosphogypsum. Journal of Radioanalytical and Nuclear Chemistry 249, 251-255. .
- United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and biological effects. UNSCEAR, 1982. New York, NY. 1982.
- United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and effects of ionizing radiation. UNSCEAR 2000. Report to General Assembly. United Nations.
- Tchokossa P., Olomo J.B., balogun F.A. and C.A. adesanmi C.A. "Assessment of Radioactivity Contents of Food in the Oil and Gas Producing Areas in Delta State, Nigeria" International Journal of Science and Technology Vol 3, No. 4, pp 245 - 250. 2013
- Zagato, V. A. B. Medina, N. H.; Okuno, E.; Umisedo, N.K. "Natural Radiactivity in Bananas". 8th International Symposium (NRE VIII) AIP Conf. Proc. 1034, pp. 264-268 (2007)
Publication Dates
-
Publication in this collection
2016
History
-
Received
30 Oct 2015 -
Accepted
07 Apr 2016