ABSTRACT
The effect of nutrients on the total lipid content and fatty acid profile of Scenedesmus obliquus isolated from the south coast of the Caspian Sea was evaluated. The nutritional compositions of the media impacted the growth rate and biomass of S. obliquus that ranged from 0.175 day-1 to 0.209 day-1and 0.92 gr·l-1 to 1.79 gr·l-1, respectively. The alga grew better in the medium which was characterized by higher levels of sodium and trace elements such as Fe, Mn, Mo, and Co and poor in N and P as compared with the other media. The highest level of the total lipid (32%) and the highest values of saturated fatty acids, in particular palmitic acid also were positively correlated with these nutrients. Peaks in polyunsaturated fatty acids (43.7 %), especially α-linolenic acid (28.4%) were related to N and P, but its correlation with K and Mg was more evident. The most important factors correlated with high amount of monounsaturated fatty acids were also N and P, followed by K and Mg to a lesser extent. This study demonstrated that the same algal strain may be a source of different amount of fatty acids, depending on the composition of the culture medium.
Key words:
monounsaturated fatty acids; polyunsaturated fatty acids; Canonical correspondence analysis; green algae; Caspian Sea
INTRODUCTION
Many microalgae contain considerable amounts of polar and nonpolar or neutral lipids that play important roles in different consumers 11. Renaud SM, Parry DL, Luong-Van T, Kuo C, Padovan A, Sammy N. Effect of light intensity on the proximate biochemical and fatty acid composition of Isochrysis sp. and Nannochloropsis oculata for use in tropical aquaculture. J Appl Phycol. 1991; 3: 43-53.
2. Richmond A, editor. Handbook of microalgal culture: Biotechnology and Applied Phycology. Wiley-Blackwell; 2003.
3. Gopinathan CP. Differential growth rates of microalgae in various culture media. Indian J Fish. 1986; 33: 450-456.-44. Wen ZY, Chen F. Heterotrophic production of eicosapentaenoic acid by microalgae. Biotechnol Adv. 2003; 21 (4): 273-294.. Among them, polyunsaturated fatty acids (PUFAs) especially eicosapentaenoic (EPA), docosahexaenoic (DHA), and arachidonic acids (AA) are the most valuable to create significant physiological and biochemical changes in a body, including reducing risk of cardiovascular diseases 55. Napolitano GE. Fatty acids as trophic and chemical markers in freshwater ecosystems. In: Arts MT, Wainman BC, editors. Lipids in Freshwater Ecosystems. Springer-Verlag, New York; 1999, p. 21-44.
6. Bigogno Ch, Khozin-Goldberga I, Boussibaa S, Vonshaka A, Cohen Z. Lipid and fatty acid composition of the green oleaginous alga Parietochloris incisa, the richest plant source of arachidonic acid. Phytochemistry. 2002; 60: 497-503.
7. Patil V, Källqvist T, Olsen E, Vogt G, Gislerød HR. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquacult Int. 2007; 15:1-9. doi:10.1007/s10499-006-9060-3.
https://doi.org/10.1007/s10499-006-9060-...
8. Nomura M, Kamogawa H, Susanto E, Kawagoe C, Yasui H, Saga N, Hosokawa M, Miyashita K. Seasonal variations of total lipids, fatty acid composition, and fucoxanthin contents of Sargassum horneri (Turner) and Cystoseira hakodatensis (Yendo) from the northern seashore of Japan. J Appl Phycol . 2013; 25(4): 1159-1169.-99. Le HD, Meisel JA, De Meijer VE, Gura KM, Puder M. The essentiality of arachidonic acid and docosahexaenoic acid. Prostag Leukotr Ess. 2009; 81(2): 165-170..
Definitely, the total lipid content may vary noticeably among individual species or strains within and between taxonomic groups 77. Patil V, Källqvist T, Olsen E, Vogt G, Gislerød HR. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquacult Int. 2007; 15:1-9. doi:10.1007/s10499-006-9060-3.
https://doi.org/10.1007/s10499-006-9060-...
. In recent years, species of green algae are often considered as a source of essential fatty acids that is necessary not only for improving the organism nutrition, but also for production of biodiesel 1010. Deng X, Li Y and Fei X. Microalgae: A promising feedstock for biodiesel. Afr J Microbiol Res. 2009; 3 (13): 1008-1014.
11. Mata TM, Martinsc AA, Caetano NS. Microalgae for biodiesel production and other applications: A review. Renew Sust Energ Rev. 2010; 14: 217-232.-1212. Mandal S, Mallick N. Biodiesel production by the green microalga Scenedesmus obliquus in a Recirculatory Aquaculture System. Appl. Environ. Microbiol. 2012; 78(16): 5929-5934. doi:10.1128/AEM.00610-12.
https://doi.org/10.1128/AEM.00610-12...
. However, this may not be because green algae naturally contain considerably more lipids than other algal taxa, but rather because many green algae are ubiquitous in diverse natural habitats, can easily be isolated, and generally grow faster than species from other taxonomic groups under laboratory conditions 1313. Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M and Darzins A, Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J. 2008; 54: 621-639. doi: 10.1111/j.1365-313X.2008.03492.x.
https://doi.org/10.1111/j.1365-313X.2008...
. Among green algae, Scenedesmaceae family is one of the most popular food sources in experiments with herbivorous zooplankton 1414. Lürling M, De Lange HJ, Van Donk E. Changes in food quality of the green alga Scenedesmus induced by Daphnia infochemicals: biochemical composition and morphology. Freshwater Biol. 1997; 38: 619-628. doi: 10.1046/j.1365-2427.1997.00225.x.
https://doi.org/10.1046/j.1365-2427.1997...
, and also a potential source of lipids that constitute up to 47% DW 1515. Ferrigo D, Galla G, Sforza E, Morosinotto T, Barcaccia G, Berrini CC. Biochemical characterization and genetic identity of an oil-rich Acutodesmus obliquus isolate. J Appl Phycol . 2015; 27(1): 149-161. doi: 10.1007/s10811-014-0315-5.
https://doi.org/10.1007/s10811-014-031...
, particularly PUFAs group, including oleic, linoleic, palmitic and alpha-linolenic acids 1616. Goswami R, Kalita MK. Scenedesmus dimorphus and Scenedesmus quadricauda: Two potent indigenous microalgae strains for biomass production and CO mitigation a study on their growth behavior and lipid productivity under different concentration of urea as nitrogen source. J Algal Biomass Utilization. 2011; 2(4): 42-49.. Scenedesmus Meyen is a freshwater and marine genus with 433 known species worldwide 1717. Guiry MD, Guiry GM. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway; (2015). Available from: http://www.algaebase.org.
http://www.algaebase.org...
, of which 27 species has been reported in aquatic ecosystems of Iran 1818. Zarei Darki B. Features of algae distribution of Order Sphaeropleales (Chlorophyceae) in different water bodies of Iran. Int J Algae. 2012; 14(4): 323-330..
In nature, the cytoplasmic oil droplets are accumulated under stressful environmental conditions, such as high temperature, light intensity and rise in salinity 1919. Gushina IA, Harwood JL. Algal lipids and effect on the environment on their biochemistry. In: Arts MT, Brett MT, Kainz M, editors. Lipids in Aquatic Ecosystems. Springer-Verlag, New York; 2009, p. 1-24. doi:10.1007/978-0-387-89366-2_1.
https://doi.org/10.1007/978-0-387-89366-...
20. Lynn SG, Kilham SS, Kreeger DA, Interlandi SJ. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (Bacillariophyceae). J Phycol. 2000; 36 (3): 510-522.
21. Solovchenko AE. Physiological Role of Neutral Lipid Accumulation in Eukaryotic Microalgae under Stresses. Russ J Plant Physiol. 2012; 59 (2): 192-202.-2222. Xia L, Yang H, He Q, Hu Ch. Physiological responses of freshwater oleaginous microalgae Desmodesmus sp. NMX451 under nitrogen deficiency and alkaline pH-induced lipid accumulation. J Appl Phycol . 2014; 27(2): 649-659.. In culture, nutritional composition of the medium has more effects on the total lipid content and fatty acid profile, among which nitrogen, silicon and phosphorus have greater roles 2020. Lynn SG, Kilham SS, Kreeger DA, Interlandi SJ. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (Bacillariophyceae). J Phycol. 2000; 36 (3): 510-522.
21. Solovchenko AE. Physiological Role of Neutral Lipid Accumulation in Eukaryotic Microalgae under Stresses. Russ J Plant Physiol. 2012; 59 (2): 192-202.
22. Xia L, Yang H, He Q, Hu Ch. Physiological responses of freshwater oleaginous microalgae Desmodesmus sp. NMX451 under nitrogen deficiency and alkaline pH-induced lipid accumulation. J Appl Phycol . 2014; 27(2): 649-659.
23. Lopez Alonso D, Belarbi EH, Fernandez Sevilla JM, Ruiz RJ, Grima EM. Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry . 2000; 54(5): 461-471.-2424. Chen M, Tang H, Ma H, Holland TC, Simon NKY, Salley SO. Effect of nutrients on growth and lipid accumulation in the green algae Dunaliella tertiolecta. Bioresource Technol. 2011; 102: 1649-1655., followed by iron 2525. Liu ZY, Wang C, Zhou BC. Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresource Technol . 2008; 99 (11): 4717-4722.
26. Gonzalez PM, Piloni NE, Puntarulo S. Iron overload and lipid peroxidation in biological systems. School of Pharmacy and Biochemistry, University of Buenos Aires; 2012, p. 89-108.
27. Dou X, Lu XH, Lu MZ, Yu LS, Xue R, Ji JB. The effects of trace elements on the lipid productivity and fatty acid composition of Nannochloropis oculata. J Renew Energy. 2013; 1-6. Available from: http://dx.doi.org/10.1155/2013 /671545.
http://dx.doi.org/10.1155/2013 /671545...
-2828. Mata TM, Almeida R, Caetano NS. Effect of the culture nutrients on the biomass and lipid productivities of microalgae Dunaliella tertiolecta. Chem Eng Tranceact. 2013; 32: 973-978.. In 1987, the lipid content and fatty acid composition of Scenedesmus obliquus was determined 2929. Choi KJ, Nakhost Z, Barzana E, Karel M. Lipid content and fatty acid composition of green algae Scenedesmus obliquus grown in a constant cell density apparatus. Food Biotechnol. 1987; 1(1): 117-28., but at a later time, the values differed within the species 3030. Makulla A. Fatty acid composition of Scenedesmus obliquus: Correlation to dilution rates. Limnologica. 2000; 30: 162-168.
31. Radmann EM, Costa JAV. Lipid content and fatty acids composition variation of microalgae exposed to CO2, SO2 and NO. Quím. Nova. 2008; 31(7). http://dx.doi.org/10.1590/S0100-40422008000700002.
http://dx.doi.org/10.1590/S0100-40422008...
-3232. Salama ES, Kim HC, Abou-Shanab RAI, Ji MK, Oh YK, Kim SH, Jeon BH. Biomass, lipid content, and fatty acid composition of freshwater Chlamydomonas mexicana and Scenedesmus obliquus grown under salt stress. Bioprocess Biosystems Eng. 2013; 36 (6): 827-833.. Therefore, the present study focused on whether the nutritional composition could affect the total lipid content and fatty acid profile of S. obliquus that was isolated from the Caspian Sea.
MATERIALS AND METHODS
The alga strain and growth conditions
Sampling was carried out in the Nour shore of the Caspian Sea shore near the Department of Marine Biology of Tarbiat Modares University (36° 35' 22"N, 52° 02' 05"E) by a plankton net with a mesh size of 55 and the Ruthner's bathometer in 2012. S. obliquus was isolated and purified using the agar plates and repeated liquid culture. To obtain a liter of suspension, S. obliquus strain from the stock culture was inoculated into the sterile medium close by the flame, until the transmission coefficient (T) got to 92-93% 3333. Trenkenshu RP, Borovkov AB, Lelekov AS. Standardized laboratory setting to study the lower phototrophs. Sevastopol; 2009. Russian.. The alga was grown in the growth chamber of Binder model maintaining the light intensity of 60 µE. m-2 s-1 for 0:24 h a dark/light cycle and temperature of 25 ±0.5 ºC for 31 days. Three media, viz. Trenkenshu, Tamiya and Guillard were used to grow the alga (Table 1).
Assessment of the microalgae growth
Cell number was daily counted with a Neubauer hemocytometer using a light microscope in triplicate for each sample and cell density was measured according to the following formula 3434. Guillard RRL. Division rates. In: Stein JR, editors. Handbook of phycological methods, Cambridge University Press, Cambridge; 1973, p. 289 - 312.:
Cell density per milliliter =Total cell counts × 104× Dilution factor
The growth of alga also was monitored by determining the absorbance using a UV-Vis spectrophotometer at 750 nm 33. The biomass values were obtained by the calibration curve of absorbance versus dry weight biomass concentration.
The specific growth rate (GR) of cultured microalgae was calculated by the following equation:
GR = ln (X1- X2)/t2 - t1
Where,
X1 = Biomass concentration at the end of selected time interval,
X2 = Biomass concentration at the beginning of selected time interval,
t2 -t1 = Elapsed time between selected time in the day.
The stationary phase was reached when the absorbance values stabilized, proceeding then to the biomass harvest.
Total lipid and fatty acid analysis
The total lipid was estimated from dry cells of S. obliquus collected in the stationary phase (it was for each medium on the different days) according to the protocol of Bligh and Dyer 36. Before the lipid extraction, the samples of 0.05±0.001 gr DW were fairly soaked in 4 ml of distilled water and were homogenized with a manual homogenizer for one minute. The algal samples were extracted with chloroform: methanol mixture (2:1 v/v) and kept for 5 minutes at room temperature. After a five-minute rest, they were dissolved in a methanol-chloroform-water mixture (MCWM) (3/3/1, v/v/v) and were homogenized; the piston was washed every time with 3.5 ml of MCWM. The resulting mixture was incubated for 15 minutes at room temperature, then 5 ml of chloroform was added and the mixture was given 15 minutes rest for lipid extraction. For appearing two phases in the samples, 5 milliliter of distilled water was added and the samples were heavily shaken. The lipid fractions were separated in a clean pre-weighed vial (first wt) and the solvent was evaporated using a rotary evaporator. The weight of the vial was again recorded (second wt). Total lipid was calculated by subtracting first wt from second wt. The final organic phase was dried under nitrogen.
Preparation of fatty acid methyl ester from the total lipid was performed according to Radwan 37. All analyses for identification of fatty acid contents were performed using gas chromatography (Varian CP-3800 model).
Statistical analysis
All experiments were repeated three times independently, and the data were recorded as the mean. The statistical analyses were performed using the software Statistical Program for Social Sciences 17.0. A one-way ANOVA and Duncan test were used to evaluate the differences among the treatments. Canonical-correlation analysis (CCA) and Pearson's coefficient were used to determine relationships among the nutritive composition of a culture medium and amount of total lipid content and fatty acid profile.
RESULTS
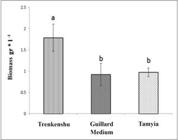
Nutrient availability had a significant impact on growth of the microalga and broad effects on its lipid and fatty acid composition. The effect of the nutritional compounds as three investigated media on the growth of was recorded by way of cell count (cell number l−1) and optical density (OD) every day, both of which showed quite similar curves (Figures 1 and 2). The algal cell growth was greatly affected in Trenkenshu and Tamyia media. In Trenkenshu medium, the maximum cell count (17×106 cell·ml-1) and OD (1.26 at A750) were recorded on the 25th day. The lowest cell number was recorded in Guillard medium (max. 14×106 cell·ml-1 on the 30th day). In Trenkenshu medium, the lag phase lasted less than in the other media that points to more quickly culture adaption. The exponential phase started at 0.2 OD750 and continued to 1.2 OD750. The longest onset of the stationary phase was observed in Guillard medium on 31th day. The nutritional compositions of the media had markedly impacted the growth rate and biomass of S. obliquus (Figures 3 and 4). The slowest growth rate (0.175 day-1) and biomass value (0.92 gr·l-1) were recorded in Guillard medium, while the maximum growth rate(0.209 day-1) and biomass(1.79 gr·l-1) were observed in Trenkenshu medium.
The highest total lipid content (32% DW) was observed inTrenkenshu medium (Table 2), which was significantly (p>0.05) higher than Tamiya (24% DW) and Guillard (21%) media, but no significant difference p<0.05) was observed between the latter two media. The highest level of SFAs (45.7%) was observed in Trenkenshu medium which was significantly different from the other two media (p>0.05), while the highest level of MUFAs (47.6%) was recorded in Guillard medium which was significantly different from the other two media (p>0.05). The highest percentage of PUFAs (43.7%) was recorded in Tamyia medium, which was significantly different from the other two media (p>0.05).The fatty acids profile of S. obliquus also varied according to medium (Table 2). Being the most common saturated fatty acid in living organisms, palmitic acid (C16:0) was also the main SFA in all the three media. Elaidic acid, the trans isomer of oleic acid (C18:1 n-9 trans), was the most important in MUFAs ranging 11.8-39.2. Moreover, cetoleic acid (C22:1n11) showed quite high value (11.8%) in Trenkenshu medium. Among PUFAs, ALA was the dominant fatty acid, for which Tamyia medium was a better source. It is interesting to note that lauric and arachidic acids with a 12- and 20-carbon atom chain, respectively, were only quantified in Trenkenshu medium.
Based on the CCA, eigenvalues of axes 1 (λ = 0.463) and 2 (λ = 0.063) explained 88.0% and 12.0% of the relation between fatty acids and nutrient data, respectively (Fig. 5, Table 3). The ranks of the nutrition variables contributing to this model were (1) sodium, cobalt and molybdenum, (2) potassium and magnesium, (3) manganese, (4) iron, (5) PUFA, C14:0, C18:1n9c, (6) C15:0, C16:0, C18:3n3, (7) C18:3n6 and C16:1n7.
Canonical correspondence analysis (CCA) biplots showing the relationships between media, nutrients growth parameters, total lipid and fatty acids.
The total lipid and SFA were correlated with sodium, potassium, magnesium, manganese, molybdenum and cobalt while MUFA and PUFA had positive correlation with nitrogen and phosphorus (Fig. 5, Table 3). Furthermore, PUFA depended strongly on potassium and magnesium. The main fatty acids such as C16:0 and C18:1n9t were correlated with potassium, magnesium, sodium and nitrogen, phosphorus, respectively. ALA (C18:3n3) was associated strongly with nitrogen, phosphorus and less with potassium and magnesium.
DISCUSSION
The quantity and quality of fatty acids content as well as the entire biochemical composition varies in response to environmental conditions 88. Nomura M, Kamogawa H, Susanto E, Kawagoe C, Yasui H, Saga N, Hosokawa M, Miyashita K. Seasonal variations of total lipids, fatty acid composition, and fucoxanthin contents of Sargassum horneri (Turner) and Cystoseira hakodatensis (Yendo) from the northern seashore of Japan. J Appl Phycol . 2013; 25(4): 1159-1169.,2020. Lynn SG, Kilham SS, Kreeger DA, Interlandi SJ. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (Bacillariophyceae). J Phycol. 2000; 36 (3): 510-522.,2424. Chen M, Tang H, Ma H, Holland TC, Simon NKY, Salley SO. Effect of nutrients on growth and lipid accumulation in the green algae Dunaliella tertiolecta. Bioresource Technol. 2011; 102: 1649-1655.,2525. Liu ZY, Wang C, Zhou BC. Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresource Technol . 2008; 99 (11): 4717-4722.,3838. Zhila NO, Kalacheva GS, Volova TG. Effect of salinity on the biochemical composition of the alga Botryococcus braunii Kütz IPPAS H-252. J Appl Phycol . 2011; 23(1): 47-52. doi: 10.1007/s10811-010-9532-8.
https://doi.org/10.1007/s10811-010-9532-...
,3939. Juneja A, Ruben Cm, Murthy Gs. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: A review. Energies. 2013; 6: 4607-4638. doi: 10.3390/en6094607.
https://doi.org/10.3390/en6094607...
. Particularly, under unfavorable environmental or stress conditions for growth, such as salinity, temperature, pH, and nutrient levels, many algae alter their lipid biosynthetic pathways towards the formation and accumulation of neutral lipids to control intracellular stress 1313. Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M and Darzins A, Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J. 2008; 54: 621-639. doi: 10.1111/j.1365-313X.2008.03492.x.
https://doi.org/10.1111/j.1365-313X.2008...
,4040. El-Kassas HY. Corrigendum to "Growth and fatty acid profile of the marine microalga Picochlorum sp. grown under nutrient stress conditions. Egyp J Aquatic Resear. 2014; 39: 233-239.. Finding the appropriate conditions to stimulate the synthesis of different fatty acids of interest is crucial for developing an efficient biological production process.
The present research showed significant differences in total lipid and fatty acid profile for the same strain of S. obliquus cultivated in three different media (Fig. 5 and Table 3).
The fatty acids content as well as the ratio between unsaturated and saturated fatty acids is an important parameter for determination of algae value. SFAs were found to increase by increasing the iron amount in Nannochloropsis oculata2020. Lynn SG, Kilham SS, Kreeger DA, Interlandi SJ. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (Bacillariophyceae). J Phycol. 2000; 36 (3): 510-522.. In Dunaliella tertiolecta and Stephanodiscus minutulus, changes of the SFA's values were directly related to changes in the amount of nitrogen 2424. Chen M, Tang H, Ma H, Holland TC, Simon NKY, Salley SO. Effect of nutrients on growth and lipid accumulation in the green algae Dunaliella tertiolecta. Bioresource Technol. 2011; 102: 1649-1655..In the present study, SFAs showed significantly positive correlationship with manganese and iron first of all, then sodium which were sufficiently present in Trenkenshu medium (Table 3 and Figure 5).In addition, trace elements such as molybdenum and cobalt are the important nutritional variables for obtaining high SFAs and total lipid. Dou et al. 2727. Dou X, Lu XH, Lu MZ, Yu LS, Xue R, Ji JB. The effects of trace elements on the lipid productivity and fatty acid composition of Nannochloropis oculata. J Renew Energy. 2013; 1-6. Available from: http://dx.doi.org/10.1155/2013 /671545.
http://dx.doi.org/10.1155/2013 /671545...
also found that the addition of Fe3+, Zn2+, Mn2+, Mo6+, and EDTA can increase the lipid productivity. S. obliquus cultivated in Trenkenshu medium can be used for biofuel because quite high content of palmitic acid obtained in this medium is desirable for good quality biodiesel 1212. Mandal S, Mallick N. Biodiesel production by the green microalga Scenedesmus obliquus in a Recirculatory Aquaculture System. Appl. Environ. Microbiol. 2012; 78(16): 5929-5934. doi:10.1128/AEM.00610-12.
https://doi.org/10.1128/AEM.00610-12...
. Furthermore, palmitic, lauric and myristic acids are not good substances in nutrition of living organisms because they are responsible for raising bad cholesterol levels in blood serum 4141. Tholstrup T, Marckmann P, Jespersen J, Vessby B, Jart A and Sandström B. Effect on Blood Lipids, Coagulation and Fibrinolysis of a Fat High in Myristic Acid and a Fat High in Palmitic Acid. Am. J. Clin. Nutr. 1994; 59: 371-377.. According to Napolitano et al. 4242. Napolitano GE, Ratnayake WMN, Ackman RG. Fatty acid components of larval Ostrea edulis (L.) importance of triacylglycerols as fatty acid reserve. Comp Biochem Physiol. 1988; 90B: 875-883., SFAs may play a double role: as a store of saturated fatty acids to be used as a source of energy and as a store of PUFAs required for phospholipid synthesis to various membrane structures or to be integrated in several metabolic processes.
PUFAs are one of the most nutritionally important and essential fatty acids because they are key nutrients in animal nutrition, and the most algae are rich in these acids 43. Nitrogen and phosphate are two important macronutrients for phospholipids because nitrogen and phosphorus starvation shifts the lipid metabolism from membrane lipid synthesis to neutral lipid storage 3939. Juneja A, Ruben Cm, Murthy Gs. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: A review. Energies. 2013; 6: 4607-4638. doi: 10.3390/en6094607.
https://doi.org/10.3390/en6094607...
,4444. Solovchenko AE, Khozin-Goldberg I, Didi-Cohen S, Cohen Z, Merzlyak MN. Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa. J Appl Phycol . 2008; 20: 245-251. doi: 10.1007/s10811-007-9233-0.
https://doi.org/10.1007/s10811-007-9233-...
45. Naderi Farsani M, Meshkiniy S, Manaffar R, Asal Pishe Z. Response of growth, protein and fatty acid content of Desmodesmus cuneatus to the repletion and depletion of nitrogen. Biolog J Microorg. 2015; 3(12): 59-68.-4646. Ruiz-Domínguez MC, Vaquero I, Obregón V, De La Morena B, Vílchez C, Vega JM. Lipid accumulation and antioxidant activity in the eukaryotic acidophilic microalga Coccomyxa sp. (strain onubensis) under nutrient starvation. J Appl Phycol . 2015; 27(3): 1099-1108.. Results of the present study also showed clearly that peaks in PUFAs are related to nitrogen and phosphorus, but its correlation with potassium and magnesium was more evident. In Tamiya medium, the most dominant fatty acid ALA also was related to nitrogen, phosphorus, potassium and magnesium. The ratio between PUFA and SFA content observed in Tamiya medium can be considered as nutritional value of S. obliquus cultivated in this medium because the higher value of P/S index means a smaller deposition of lipids in the body 4747. Lawton CL, Delargry HJ, Brockman J, Simith RC and Blundell JE. The degree of saturation of fatty acids influences in post ingestive satiety. Brit J Nutr. 2000; 83 (5): 473 - 482..
There are reports that amount of MUFAs goes up due to the reduction of nitrogen in the culture medium 2020. Lynn SG, Kilham SS, Kreeger DA, Interlandi SJ. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (Bacillariophyceae). J Phycol. 2000; 36 (3): 510-522.,2323. Lopez Alonso D, Belarbi EH, Fernandez Sevilla JM, Ruiz RJ, Grima EM. Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry . 2000; 54(5): 461-471.. The potential relationship between phosphorus and amount of MUFAs have beenshown intwo microalgae Phaeodactylum tricornutum and Dunaliella tertiolecta4848. Siron R, Giusti G, Berland B. Changes in the fatty acid composition of Phaeodactylum tricornutum and Dunaliella tertiolecta during growth and under phosphorus deficiency. Mar Ecol-Prog Ser. 1989; 55: 95-100. and the yellow-green alga, Monodus subterraneus4949. Khozin-Goldberg I, Cohen Z. The effect of phosphate starvation on the lipid and fatty acid composition of the freshwater eustigmatophyte Monodus subterraneus. Phytochemistry . 2006; 67(7): 696-701., in which reduced phosphorus in medium had impacted MUFAs. By analyzing the changes in fatty acids composition of several algal species at different concentrations of nitrogen and silica, Shifrin and Chisholm 5050. Shifrin NS, Chisholm SW. Phytoplankton lipids: interspecific differences and effects of nitrate, silicate and light-dark cycles. J Phycol. 1981; 17: 374-384 showed increase in MUFAs as the result of nitrogen limitation. In our experiment, the most important factors correlated with high amount of MUFAs were also nitrogen and phosphorus, followed by potassium and magnesium to a lesser extent. Limitation of these parameters caused growth delay and an accumulation of fatty acids forming MUFAs especially elaidic acid.
CONCLUSIONS
Based on the results of the present study, it can be concluded that the same algal strain may be a source of different fatty acids, depending on the nutritive composition of the culture medium. Sodium, iron, cobalt and molybdenum impacted on the growth rate, SFAs in particular palmitic acid, and, as the result, the total lipid content in the green alga S. obliquus. The highest amount of PUFAs and oleic acid were related to potassium and magnesium and less nitrogen and phosphorus. The highest amount of MUFAs, especially ALA were obtained at limitation of nitrogen and phosphorus caused growth delay and an accumulation of fatty acids forming MUFAs especially elaidic acid. Thus, the knowledge about stimulation of the synthesis of different fatty acids may be used for algal cultivation with the different purposes.
ACKNOWLEDGMENTS
The authors express their gratitude to Tarbiat Modares University for providing funding and research facilities. We also thank laboratory assistant of Faculty of Marine Sciences for help.
REFERENCES
-
1Renaud SM, Parry DL, Luong-Van T, Kuo C, Padovan A, Sammy N. Effect of light intensity on the proximate biochemical and fatty acid composition of Isochrysis sp. and Nannochloropsis oculata for use in tropical aquaculture. J Appl Phycol. 1991; 3: 43-53.
-
2Richmond A, editor. Handbook of microalgal culture: Biotechnology and Applied Phycology. Wiley-Blackwell; 2003.
-
3Gopinathan CP. Differential growth rates of microalgae in various culture media. Indian J Fish. 1986; 33: 450-456.
-
4Wen ZY, Chen F. Heterotrophic production of eicosapentaenoic acid by microalgae. Biotechnol Adv. 2003; 21 (4): 273-294.
-
5Napolitano GE. Fatty acids as trophic and chemical markers in freshwater ecosystems. In: Arts MT, Wainman BC, editors. Lipids in Freshwater Ecosystems. Springer-Verlag, New York; 1999, p. 21-44.
-
6Bigogno Ch, Khozin-Goldberga I, Boussibaa S, Vonshaka A, Cohen Z. Lipid and fatty acid composition of the green oleaginous alga Parietochloris incisa, the richest plant source of arachidonic acid. Phytochemistry. 2002; 60: 497-503.
-
7Patil V, Källqvist T, Olsen E, Vogt G, Gislerød HR. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquacult Int. 2007; 15:1-9. doi:10.1007/s10499-006-9060-3.
» https://doi.org/10.1007/s10499-006-9060-3 -
8Nomura M, Kamogawa H, Susanto E, Kawagoe C, Yasui H, Saga N, Hosokawa M, Miyashita K. Seasonal variations of total lipids, fatty acid composition, and fucoxanthin contents of Sargassum horneri (Turner) and Cystoseira hakodatensis (Yendo) from the northern seashore of Japan. J Appl Phycol . 2013; 25(4): 1159-1169.
-
9Le HD, Meisel JA, De Meijer VE, Gura KM, Puder M. The essentiality of arachidonic acid and docosahexaenoic acid. Prostag Leukotr Ess. 2009; 81(2): 165-170.
-
10Deng X, Li Y and Fei X. Microalgae: A promising feedstock for biodiesel. Afr J Microbiol Res. 2009; 3 (13): 1008-1014.
-
11Mata TM, Martinsc AA, Caetano NS. Microalgae for biodiesel production and other applications: A review. Renew Sust Energ Rev. 2010; 14: 217-232.
-
12Mandal S, Mallick N. Biodiesel production by the green microalga Scenedesmus obliquus in a Recirculatory Aquaculture System. Appl. Environ. Microbiol. 2012; 78(16): 5929-5934. doi:10.1128/AEM.00610-12.
» https://doi.org/10.1128/AEM.00610-12 -
13Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M and Darzins A, Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J. 2008; 54: 621-639. doi: 10.1111/j.1365-313X.2008.03492.x.
» https://doi.org/10.1111/j.1365-313X.2008.03492.x. -
14Lürling M, De Lange HJ, Van Donk E. Changes in food quality of the green alga Scenedesmus induced by Daphnia infochemicals: biochemical composition and morphology. Freshwater Biol. 1997; 38: 619-628. doi: 10.1046/j.1365-2427.1997.00225.x.
» https://doi.org/10.1046/j.1365-2427.1997.00225.x. -
15Ferrigo D, Galla G, Sforza E, Morosinotto T, Barcaccia G, Berrini CC. Biochemical characterization and genetic identity of an oil-rich Acutodesmus obliquus isolate. J Appl Phycol . 2015; 27(1): 149-161. doi: 10.1007/s10811-014-0315-5.
» https://doi.org/10.1007/s10811-014-0315-5. -
16Goswami R, Kalita MK. Scenedesmus dimorphus and Scenedesmus quadricauda: Two potent indigenous microalgae strains for biomass production and CO mitigation a study on their growth behavior and lipid productivity under different concentration of urea as nitrogen source. J Algal Biomass Utilization. 2011; 2(4): 42-49.
-
17Guiry MD, Guiry GM. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway; (2015). Available from: http://www.algaebase.org
» http://www.algaebase.org -
18Zarei Darki B. Features of algae distribution of Order Sphaeropleales (Chlorophyceae) in different water bodies of Iran. Int J Algae. 2012; 14(4): 323-330.
-
19Gushina IA, Harwood JL. Algal lipids and effect on the environment on their biochemistry. In: Arts MT, Brett MT, Kainz M, editors. Lipids in Aquatic Ecosystems. Springer-Verlag, New York; 2009, p. 1-24. doi:10.1007/978-0-387-89366-2_1.
» https://doi.org/10.1007/978-0-387-89366-2_1 -
20Lynn SG, Kilham SS, Kreeger DA, Interlandi SJ. Effect of nutrient availability on the biochemical and elemental stoichiometry in the freshwater diatom Stephanodiscus minutulus (Bacillariophyceae). J Phycol. 2000; 36 (3): 510-522.
-
21Solovchenko AE. Physiological Role of Neutral Lipid Accumulation in Eukaryotic Microalgae under Stresses. Russ J Plant Physiol. 2012; 59 (2): 192-202.
-
22Xia L, Yang H, He Q, Hu Ch. Physiological responses of freshwater oleaginous microalgae Desmodesmus sp. NMX451 under nitrogen deficiency and alkaline pH-induced lipid accumulation. J Appl Phycol . 2014; 27(2): 649-659.
-
23Lopez Alonso D, Belarbi EH, Fernandez Sevilla JM, Ruiz RJ, Grima EM. Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum Phytochemistry . 2000; 54(5): 461-471.
-
24Chen M, Tang H, Ma H, Holland TC, Simon NKY, Salley SO. Effect of nutrients on growth and lipid accumulation in the green algae Dunaliella tertiolecta Bioresource Technol. 2011; 102: 1649-1655.
-
25Liu ZY, Wang C, Zhou BC. Effect of iron on growth and lipid accumulation in Chlorella vulgaris Bioresource Technol . 2008; 99 (11): 4717-4722.
-
26Gonzalez PM, Piloni NE, Puntarulo S. Iron overload and lipid peroxidation in biological systems. School of Pharmacy and Biochemistry, University of Buenos Aires; 2012, p. 89-108.
-
27Dou X, Lu XH, Lu MZ, Yu LS, Xue R, Ji JB. The effects of trace elements on the lipid productivity and fatty acid composition of Nannochloropis oculata J Renew Energy. 2013; 1-6. Available from: http://dx.doi.org/10.1155/2013 /671545
» http://dx.doi.org/10.1155/2013 /671545 -
28Mata TM, Almeida R, Caetano NS. Effect of the culture nutrients on the biomass and lipid productivities of microalgae Dunaliella tertiolecta Chem Eng Tranceact. 2013; 32: 973-978.
-
29Choi KJ, Nakhost Z, Barzana E, Karel M. Lipid content and fatty acid composition of green algae Scenedesmus obliquus grown in a constant cell density apparatus. Food Biotechnol. 1987; 1(1): 117-28.
-
30Makulla A. Fatty acid composition of Scenedesmus obliquus: Correlation to dilution rates. Limnologica. 2000; 30: 162-168.
-
31Radmann EM, Costa JAV. Lipid content and fatty acids composition variation of microalgae exposed to CO2, SO2 and NO. Quím. Nova. 2008; 31(7). http://dx.doi.org/10.1590/S0100-40422008000700002
» http://dx.doi.org/10.1590/S0100-40422008000700002 -
32Salama ES, Kim HC, Abou-Shanab RAI, Ji MK, Oh YK, Kim SH, Jeon BH. Biomass, lipid content, and fatty acid composition of freshwater Chlamydomonas mexicana and Scenedesmus obliquus grown under salt stress. Bioprocess Biosystems Eng. 2013; 36 (6): 827-833.
-
33Trenkenshu RP, Borovkov AB, Lelekov AS. Standardized laboratory setting to study the lower phototrophs. Sevastopol; 2009. Russian.
-
34Guillard RRL. Division rates. In: Stein JR, editors. Handbook of phycological methods, Cambridge University Press, Cambridge; 1973, p. 289 - 312.
-
35Kilham SS, Kreeger DA, Lynn SG, Goulden CE. COMBO: a defined freshwater culture medium for algae and zooplankton. Hydrobiologia. 1998; 337(4): 147-159.
-
36Bligh EG, Dyer WJ. A rapid method for total lipid extraction and purification. Can J Biochem Physiol. 1959; 37: 911-917.
-
37Radwan SS. Sources of C20 polyunsaturated of fatty acids for use. Appl. Microbiol Biotechnol. 1978; 35: 421-430.
-
38Zhila NO, Kalacheva GS, Volova TG. Effect of salinity on the biochemical composition of the alga Botryococcus braunii Kütz IPPAS H-252. J Appl Phycol . 2011; 23(1): 47-52. doi: 10.1007/s10811-010-9532-8.
» https://doi.org/10.1007/s10811-010-9532-8. -
39Juneja A, Ruben Cm, Murthy Gs. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: A review. Energies. 2013; 6: 4607-4638. doi: 10.3390/en6094607.
» https://doi.org/10.3390/en6094607 -
40El-Kassas HY. Corrigendum to "Growth and fatty acid profile of the marine microalga Picochlorum sp. grown under nutrient stress conditions. Egyp J Aquatic Resear. 2014; 39: 233-239.
-
41Tholstrup T, Marckmann P, Jespersen J, Vessby B, Jart A and Sandström B. Effect on Blood Lipids, Coagulation and Fibrinolysis of a Fat High in Myristic Acid and a Fat High in Palmitic Acid. Am. J. Clin. Nutr. 1994; 59: 371-377.
-
42Napolitano GE, Ratnayake WMN, Ackman RG. Fatty acid components of larval Ostrea edulis (L.) importance of triacylglycerols as fatty acid reserve. Comp Biochem Physiol. 1988; 90B: 875-883.
-
43Brown MR, Jeffrey SW, Volkman JK, Dunstan GA. Nutritional properties of microalgae for mariculture. Aquaculture. 1997; 151: 315-331.
-
44Solovchenko AE, Khozin-Goldberg I, Didi-Cohen S, Cohen Z, Merzlyak MN. Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa J Appl Phycol . 2008; 20: 245-251. doi: 10.1007/s10811-007-9233-0.
» https://doi.org/10.1007/s10811-007-9233-0. -
45Naderi Farsani M, Meshkiniy S, Manaffar R, Asal Pishe Z. Response of growth, protein and fatty acid content of Desmodesmus cuneatus to the repletion and depletion of nitrogen. Biolog J Microorg. 2015; 3(12): 59-68.
-
46Ruiz-Domínguez MC, Vaquero I, Obregón V, De La Morena B, Vílchez C, Vega JM. Lipid accumulation and antioxidant activity in the eukaryotic acidophilic microalga Coccomyxa sp. (strain onubensis) under nutrient starvation. J Appl Phycol . 2015; 27(3): 1099-1108.
-
47Lawton CL, Delargry HJ, Brockman J, Simith RC and Blundell JE. The degree of saturation of fatty acids influences in post ingestive satiety. Brit J Nutr. 2000; 83 (5): 473 - 482.
-
48Siron R, Giusti G, Berland B. Changes in the fatty acid composition of Phaeodactylum tricornutum and Dunaliella tertiolecta during growth and under phosphorus deficiency. Mar Ecol-Prog Ser. 1989; 55: 95-100.
-
49Khozin-Goldberg I, Cohen Z. The effect of phosphate starvation on the lipid and fatty acid composition of the freshwater eustigmatophyte Monodus subterraneus Phytochemistry . 2006; 67(7): 696-701.
-
50Shifrin NS, Chisholm SW. Phytoplankton lipids: interspecific differences and effects of nitrate, silicate and light-dark cycles. J Phycol. 1981; 17: 374-384
Publication Dates
-
Publication in this collection
2017
History
-
Received
03 Feb 2016 -
Accepted
14 July 2016