ABSTRACT
In vitro rhizome production, encapsulation and cold storage of Acorus calamus were attempted for its propagation and ‘true-to-type’ conservation. Shoot cultures were initiated using underground rhizome buds, on 6-benzyladenine (BA) containing Murashige and Skoog (MS) medium. Maximum microrhizome production was observed in presence of 33.3 µM BA, on modified MS medium containing 6% sucrose, 100 mg/L citric acid and 1 g/L polyvinyl pyrrolidone-40. Synthetic seeds were produced from regenerated microtubers by encapsulation in calcium alginate beads. These synthetic seeds were stored in complete darkness at 100C temperature for different durations for mid-term conservation. After cold storage, synthetic seeds were re-cultured in vitro, 100% survival was recorded after the storage of 1, 3 or 6 months; and 80% survival was observed after the storage of 12 months. The microrhizomes were produced roots in 4.9 µM indole-3-butyric acid containing half strength MS medium. All the regenerated plantlets were successfully transferred to field after acclimatization. It is the first report on successful one year in vitro cold storage of A. calamus.
Key words:
Aromatic plant; Encapsulation; Sweet Flag; Synthetic seeds; Plant tissue culture
INTRODUCTION
Acorus calamus L. or Sweet Flag or Bach (family- Acoraceae) is a semi aquatic perennial herb distributed in Asia, North America and Europe. Plant may be indigenous to India, distributed in muddy places up to 1800 m height at 10 to 380C temperature range11. Sharma V, Singh I, Chaudhary P. Acorus calamus (The Healing Plant): a review on its medicinal potential, micropropagation and conservation. Nat Prod Res. 2014; DOI: 10.1080/14786419.2014.915827. Plant becomes endangered in many Indian states- Madhya Pradesh, Kerala, Andhra Pradesh and Chhattisgarh; also reported vulnerable in Tamil Nadu22. FRLHT (Foundation for Revitalisation of Local Health Traditions), Bangalore, India. Updated in 4th May 2016. Accessed in 13th May 2016. Available from http://envis.frlht.org/medicinal-plants-conservation-concern-species.php
http://envis.frlht.org/medicinal-plants-...
, due to rigorous collections from natural habitat because of its economic and medicinal importance. It is a good source of β-asarone33. Mazza G. Gas chromatographic and mass spectrometric studies of the constituents of the rhizome of calamus: the volatile constituents of the essential oil. J Chromatogr A. 1985; 328:179-194.. A. calamus is comprehensively used in the traditional medicines as sedative, anti-diarrheic, carminative, tonic, stimulant44. McGaw LJ, Jager AK, Staden J. Isolation of β-asarone, an antibacterial and anti-helminthic compound from Acorus calamus in South Africa. S Afr J Bot. 2002; 61:31-35.. The essential oil extracted from rhizome of A. calamus used in perfumes and beverages55. Motley TJ. The ethnobotany of sweet flag, Acorus Calamus (Araceae). Econ Bot. 1994; 48:397-412.. Seed production is scanty in this herb; hence, rhizomes are mainly used as propagule for its traditional cultivation, which has a slow rate of propagation66. Devi NS, Kishir R, Sharma GJ. Microrhizome induction in Acorus calamus Linn. - An important medicinal and aromatic plant. Hortic Environ Biote. 2012; 53(5):410-414.. Therefore, in vitro techniques may provide faster mass propagation for production of its disease free quality planting material as well as means of germplasm conservation. Reports are there on micropropagation of A. calamus77. Harikrishnan KN, Hariharan M. In vitro propagation of sweet flag (Acorus calamus L.) - a medicinal plant. In: Kishore PBK, Editor. Plant Tissue Culture and Biotechnology: Emerging Trends Hyderabad (India): Universities Press; 1999. p. 220-223.,88. Rani AS, Subhadra VV, Reddy VD. In vitro propagation of Acorus calamus Linn. - A medicinal plant. Indian J Exp Biol. 2000; 38:730-732.,99. Anu A, Babu KN, John CZ, Peter KV. In vitro clonal multiplication of Acorus calamus. J Plant Biochem Biot. 2001; 10:53-55.,1010. Sandhyarani N, Kishor R, Sharma GJ. Clonal propagation of triploid Acorus calamus Linn. using dual-phase culture system. J Crop Sci Biotech. 2011; 14(3):213-217.. Another technique namely, in vitro storage organ production has many advantages over in vitro plantlet regeneration; for example micro-tubers/rhizomes can be easily acclimatized, easier to handle, storage and exchange of germplasm, etc. Thus, selected clones can be maintained safely by tissue culture technique under disease and pest free environment. However, under the normal culture conditions, continuous growth of cultures is there induced by exogenous plant growth regulators and cultures are also subjected to frequent subcultures for long durations; may induce off types via somaclonal variations1111. Bairu MW, Aremu AO, Staden JV. Somaclonal variation in plants: causes and detection methods. Plant Growth Regul. 2011; 63:147-173.,1212. Hrahsel L, Basu A, Sahoo L, Thangjam R. In vitro propagation and assessment of the genetic fidelity of Musa acuminata (AAA) cv. Vaibalhla derived from immature male flowers. Appl Biochem Biotech. 2014; 172:1530-1539.,1313. Devi SP, Kumaria S, Rao SR, Tandon P. Single primer amplification reaction (SPAR) methods reveal subsequent increase in genetic variations in micropropagated plants of Nepenthes khasiana Hook. f. maintained for three consecutive regenerations. Gene 2014; 538:23-29. Therefore, growth of in vitro cultures must be checked for storage of germplasm to overcome the risk of off type production and it is economic too in comparison to maintain fast growing cultures1414. Chauhan R, Keshavkant S, Jadhav SK, Quraishi Afaque. In vitro slow-growth storage of Chlorophytum borivilianum Sant. et Fernand: a critically endangered herb. In Vitro Cell Dev-Pl. 2016; 52:315-321.. Long term in vitro storage is carried out in liquid nitrogen that is a costly and highly technical method. In vitro cold storage technique is used for mid-term conservation has many plus points i.e., simple, economic, disease free with lower requirement of manpower and space. It is also suitable for commercial micropropagation units; as in vitro cold stored material can be multiplied according to demand. In vitro microrhizome production of A. calamus is reported by Devi et al.66. Devi NS, Kishir R, Sharma GJ. Microrhizome induction in Acorus calamus Linn. - An important medicinal and aromatic plant. Hortic Environ Biote. 2012; 53(5):410-414.. Encapsulation of microrhizomes (synthetic seeds) of this species and its in vitro conservation by synthetic seeds via storage in dark at low temperature is reported firstly here.
MATERIAL AND METHODS
Explant Establishment
Mother plants of A. calamus were collected from Chhattisgarh State Forest Departmental Nursery, Raipur (India). The underground rhizome segments with shoot buds were used as explants to start in vitro culture. The rhizomes were washed thoroughly in running tap water to remove soil and then divided into approximately 2 cm pieces each. Leaves and roots were also removed from explants. Then the explants were treated with 1% Tween-80 for 15 minutes. Next step was carried out under laminar airflow hood; explants were dipped in fungicide solution (0.1% Carbendazim and 0.25% Mancozeb) for 30 minutes, followed by rinsing with sterile distilled water for 3 - 4 times. Now the explants were quickly dipped in 70% ethanol and then finally were surface disinfested with 0.2% (w/v) aqueous mercuric chloride for 20 minutes and were rinsed 4 - 5 times with sterile distilled water. Again the explants were cut to final size approximately 1 cm long and then were transferred aseptically on to culture establishment medium.
Murashige and Skoog (MS) medium1515. Murashige T, Skoog F. A revised medium for rapid growth and bioassay with tobacco tissue culture. Physiol Plant. 1962; 15:473-497. supplemented with 3% sucrose and different concentration of 6-benzyladenine (BA) was used as culture medium. The media were adjusted to pH 5.7 with 1N NaOH and sterilized by autoclaving for 20 min at 1.05 kg/cm2 pressure at 1210C. The inoculation were carried out and the cultures was incubated at 250C ±2 under 16/8 (day/night) photoperiod with 40 μmol m−2 s−1 light intensity provided through white cool fluorescent tubes. Explant establishment and rooting experiments were carried out in 25×150 mm glass culture tubes containing 15 ml medium each, and 250 ml conical flasks containing 50 ml medium each were used for microrhizome production studies, respectively. The bud break percentage, number and length of shoots per explant were recorded after the 30 days of culture.
Microrhizome Induction
The in vitro elongated shoots were further used for protocol standardization of microrhizome induction. Two different levels of sucrose viz 6 and 9% were supplemented in MS medium. Along with sucrose three different concentrations of BA were also incorporated in the media. Activated charcoal (0.1%) was also evaluated for microrhizome induction along with 6 and 9% of sucrose supplemented medium. MS medium containing 6% sucrose without any PGR serves as control for this experiment. The number and length of microrhizomes produced from each culture were recorded after 30 days of culture.
Microrhizome Production
After 30 days, the shoots elongated from the explants were excised; leaves were trimmed and then used for microrhizome production. MS medium with best responded sucrose level was used for microrhizome production. This microrhizome production medium was also contained 100 mg/L citric acid and 1 g/L polyvinyl pyrrolidone-40 (PVP-40). The different concentrations of BA were also incorporated in to this modified MS medium for study the effect of microrhizome production. The number and length of microrhizomes produced from each culture were recorded after 30 days of culture.
Encapsulation and Cold Storage
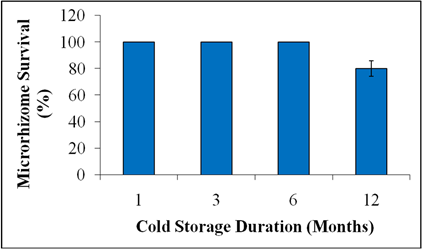
For synthetic seed production, in vitro produced microrhizomes were encapsulated into calcium alginate beads. The microrhizomes were suspended in sterilized 3% sodium alginate solution in ½ strength of MS medium; and were dispensed drop-wise into sterilized 3% calcium chloride solution to trap in calcium alginate gel. These encapsulated microrhizomes were washed with sterile distilled water and were sealed in petri-plates. Further, these synthetic seeds were stored at 100C in complete darkness for different durations i.e., 1, 3, 6 or 12 months. After different time periods, survival percentage of the cold stored synthetic seeds were analyzed by re-culture them on to the microrhizome production medium under the normal culture conditions.
Rooting and Acclimatization
In vitro grown microrhizomes were subjected to rooting in the presence of 4.9 µM indole-3-butyric acid (IBA) in ½ strength MS medium. For acclimatization, plantlets with well developed roots were removed from vessels, washed thoroughly in tap water to remove the nutrient medium and then dipped in fungicide solution (0.1% Carbendazim and 0.25% Mancozeb) for 30 minutes. The plantlets were then transplanted to pots containing coco-pit. These pots were placed at 300C ±2 and 90% relative humidity (RH) for one month. For secondary hardening, RH is gradually reduced in next two months; finally transferred to the field.
Data Analysis
All the experiments were of completely randomized design; contained ten replicates in each treatment, and each experiment was repeated thrice (total thirty replicates). The data were analyzed by the analysis of variance (ANOVA), means were separated by Duncan’s Multiple Range Test (DMRT), at 5% level; using SPSS-10 software1616. SPSS, Inc. SPSS 10.0 syntax reference guide. Chicago, IL; 1999..
RESULTS AND DISCUSSIONS
Explant Establishment
Underground rhizome bud explants of A. calamus were exhibited 100% bud break in growth regulator free basal MS medium (Table 1). However, shoot number as well as shoot length were highly influenced by concentration of BA, as probability (p) of ANOVA was <0.0001. Highest shoot number and elongation were recorded in the presence of 35.52 µM BA, as both the values were ranked ‘a’ by DMRT in comparison to other concentrations including control. Beneficial effect of BA on explant establishment of A. calamus was also observed previously88. Rani AS, Subhadra VV, Reddy VD. In vitro propagation of Acorus calamus Linn. - A medicinal plant. Indian J Exp Biol. 2000; 38:730-732..
Microrhizome Induction
Microrhizome induction was more influenced by enhanced level of sucrose along with combination of PGR (Table 2). Both the sucrose level (6 and 9%) in combination with slightly increased concentration of BA was found favorable for microrhizome induction. However, 6% sucrose level induced more numbers of in vitro rhizomes instead of 9% level. Moreover, black leaching was observed in all the rhizome inducing cultures. Thus, for microrhizome production 6% sucrose level was opted with various concentration of BA. In previous report also, 6% sucrose level was found suitable for induction of microrhizome in the same plant66. Devi NS, Kishir R, Sharma GJ. Microrhizome induction in Acorus calamus Linn. - An important medicinal and aromatic plant. Hortic Environ Biote. 2012; 53(5):410-414..
Microrhizome Production
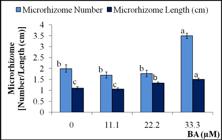
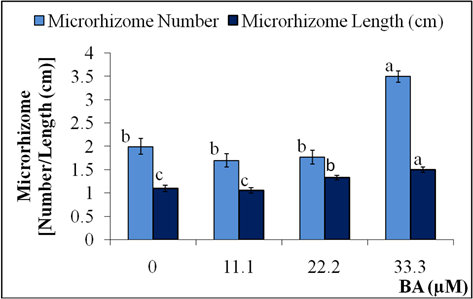
For microrhizome production, the shoots which were elongated from the explants were removed from vessels aseptically after one month of culture and each shoot was divided into nodes and cultured onto microrhizome production medium. At induction stage slight blackening and phenolic leaching were observed in cultures that may inversely affect the number of microrhizome (Table 2). To overcome this problem, citric acid (antioxidant) and PVP-40 (absorbent) were also incorporated into the production medium. The problem of blackening and leaching in Lagerstroemia parviflora cultures were also circumvent by the similar strategy1717. Quraishi A, Koche V, Mishra SK. Micropropagation of Lagerstroemia parviflora through axillary bud culture. Silvae Gene t. 1997; 46(4):242-245.. In vitro microrhizome production (number and length) was also influenced by BA level in the medium as p of ANOVA was highly significant (<0.0001) (Fig. 1 and Fig. 2). Maximum microrhizomes were produced in the presence of 33.3 µM BA, as both microrhizome number and length were ranked ‘a’ by DMRT. Higher level of sucrose in culture medium facilitates higher carbon energy to induce formation of storage organs in vitro1818. Tyagi RK, Bhat SR, Chandel KPS. In vitro conservation strategies for spices crop germplasm Zingiber, Curcuma and Piper species. In: Mathew NM, Kuruvila JC, Editors. Developments in plantation crop research. India: Rubber Research Institute of India; 1998. p. 77-82.,1919. Nayak S. In vitro multiplication and microrhizome induction in Curcuma aromatic Salisb. Plant Growth Regul. 2000; 32:41-47.. Presence of cytokinin in culture medium with high sucrose level positively influenced microrhizome formation, probably because microrhizome is a modified shoot. Therefore, the best microrhizome formation in high BA and high sucrose containing medium was also reported in Curcuma aromatica1919. Nayak S. In vitro multiplication and microrhizome induction in Curcuma aromatic Salisb. Plant Growth Regul. 2000; 32:41-47., Curcuma amada2020. Nayak S. High frequency in vitro production of microrhizomes of Curcuma amada. Indian J Exp Biol. 2002; 40:230-232., Kaempferia galanga2121. Chirangini P, Sinha SK, Sharma GJ. In vitro propagation and microrhizome induction in Kaempferia galanga Linn. and K. rotunda Linn. Indian J Biotechnol. 2005; 4:404-408..
Effect of BA concentration on in vitro microrhizome formation of Acorus calamus in MS medium supplemented with 60 g L-1 sucrose (6%), 100 mg L-1 citric acid and 1 g L-1 PVP-40. Observations were recorded after the 30 days of culture.
Each experiment consisted of 10 replicates and each experiment was repeated three times. Similar letters on mean values do not differ significantly at 5% level, by DMRT.
ANOVA: f F p
Shoot number: 3 33.347 <0.0001
Shoot Length: 3 12.016 <0.0001
In vitro microrhizome production of Acorus calamus, on MS medium supplemented with 33.3 µM BA, 60 g L-1 sucrose, 100 mg L-1 citric acid and 1 g L-1 PVP-40.
Encapsulation and Cold Storage
The regenerated microrhizomes of A. calamus were encapsulated in calcium alginate gel using sodium alginate and calcium chloride solutions to produce synthetic seeds (Fig. 3A). Synthetic seeds were also produced by encapsulation of in vitro grown vegetative propagules in calcium alginate beads in many species i.e., Acacia hybrid2222. Nor Asmah H, Nor Hasnida H, Noraliza A, Nashatul Zaimah NA, Nadiah Salmi N. In vitro propagation of Acacia hybrid through alginate-encapsulated shoots and axillary buds. Afr J Biotechnol. 2012; 11(65):12814-12817., Cassia angustifolia2323. Bukhari NAW, Siddique I, Perveen K, Siddiqui I, Alwahibi MS. Synthetic seed production and physio-biochemical studies in Cassia angustifolia Vahl. - a medicinal plant. Acta Biol Hung. 2014; 65(3):355-367., etc. They can be used like real seeds for transport, storage and plant production. Therefore, synthetic seeds of A. calamus were further tested for cold storage for mid-term conservation. For the purpose, they were aseptically sealed in petri-dishes and were kept at 100C in complete darkness for different durations. Up to 6 months, the cold stored synthetic seeds were showed 100% survival when re-cultured onto the microrhizome production medium (Fig. 3B and Fig. 4). After the one year of cold storage, 80% synthetic seeds were survived. Thus, encapsulated microtubers as synthetic seeds were stored for one year without any subculture in economic manner. Although the microrhizome cold storage of A. calamus has not been reported previously, but the cold storage of in vitro raised storage organs has been reported in other plant species. In Zingiber microrhizomes, 50% survival was recorded up to 14 months under 16 h and 24 h light in normal culture condition; and only 20% survival under dark incubation2424. Tyagi RK, Anuradha A, Yusuf A. Conservation of Zingiber germplasm through in vitro rhizome formation. Sci Hortic-Amsterdam. 2006; 108:210-219.. Potato microtubers were stored in dark at 40C for 12 to 15 months2525. Mix-Wagner G. The conservation of potato cultivars. Potato Res. 1999; 42:427-436.. Nonetheless, in vitro culture storage provides conservation and safe exchanges of germplasm that can be bulked up again whenever required2626. Nayak S, Naik PK. Factors effecting in vitro microrhizome formation and growth in Curcuma longa L. and improved field performance of micropropagated plants. ScienceAsia. 2006; 32:31-37..
In vitro cold storage of Acorus calamus. A: Synthetic seeds produced by microrhizome encapsulation in calcium alginate beads. B: Re-growth of synthetic seed after one year of storage at 10oC in dark.
Survival percentage of in vitro cold stored (100C in dark) synthetic seeds of Acours calamus, after the re-culture. Each experiment consisted of 10 replicates and each experiment was repeated three times.
Rooting and Acclimatization
Each microrhizome was aseptically transferred to rooting medium for complete plantlet formation. These microrhizomes were easily rooted in presence of an auxin- 4.9 µM IBA in half strength MS medium. All the regenerated plantlets were survived after one month of acclimatization in greenhouse. Similar, high rate of survival in A. calamus microrhizomes after acclimatization was also recorded previously66. Devi NS, Kishir R, Sharma GJ. Microrhizome induction in Acorus calamus Linn. - An important medicinal and aromatic plant. Hortic Environ Biote. 2012; 53(5):410-414..
CONCLUSION
Thus, the developed protocol facilitates simple in vitro technique for synthetic seed production by encapsulation of microrhizomes and conservation via these synthetic seeds in complete dark at 100C up to one year; for this economically important medicinal and aromatic plant- A. calamus.
ACKNOWLEDGEMENTS
Authors are thankful to Head of School of Studies in Biotechnology, Pt. Ravishankar Shukla University, Raipur (India) for providing laboratory facilities and support.
REFERENCES
-
1Sharma V, Singh I, Chaudhary P. Acorus calamus (The Healing Plant): a review on its medicinal potential, micropropagation and conservation. Nat Prod Res 2014; DOI: 10.1080/14786419.2014.915827
-
2FRLHT (Foundation for Revitalisation of Local Health Traditions), Bangalore, India. Updated in 4th May 2016. Accessed in 13th May 2016. Available from http://envis.frlht.org/medicinal-plants-conservation-concern-species.php
» http://envis.frlht.org/medicinal-plants-conservation-concern-species.php -
3Mazza G. Gas chromatographic and mass spectrometric studies of the constituents of the rhizome of calamus: the volatile constituents of the essential oil. J Chromatogr A 1985; 328:179-194.
-
4McGaw LJ, Jager AK, Staden J. Isolation of β-asarone, an antibacterial and anti-helminthic compound from Acorus calamus in South Africa. S Afr J Bot. 2002; 61:31-35.
-
5Motley TJ. The ethnobotany of sweet flag, Acorus Calamus (Araceae). Econ Bot. 1994; 48:397-412.
-
6Devi NS, Kishir R, Sharma GJ. Microrhizome induction in Acorus calamus Linn. - An important medicinal and aromatic plant. Hortic Environ Biote 2012; 53(5):410-414.
-
7Harikrishnan KN, Hariharan M. In vitro propagation of sweet flag (Acorus calamus L.) - a medicinal plant. In: Kishore PBK, Editor. Plant Tissue Culture and Biotechnology: Emerging Trends Hyderabad (India): Universities Press; 1999. p. 220-223.
-
8Rani AS, Subhadra VV, Reddy VD. In vitro propagation of Acorus calamus Linn. - A medicinal plant. Indian J Exp Biol 2000; 38:730-732.
-
9Anu A, Babu KN, John CZ, Peter KV. In vitro clonal multiplication of Acorus calamus J Plant Biochem Biot 2001; 10:53-55.
-
10Sandhyarani N, Kishor R, Sharma GJ. Clonal propagation of triploid Acorus calamus Linn. using dual-phase culture system. J Crop Sci Biotech 2011; 14(3):213-217.
-
11Bairu MW, Aremu AO, Staden JV. Somaclonal variation in plants: causes and detection methods. Plant Growth Regul. 2011; 63:147-173.
-
12Hrahsel L, Basu A, Sahoo L, Thangjam R. In vitro propagation and assessment of the genetic fidelity of Musa acuminata (AAA) cv. Vaibalhla derived from immature male flowers. Appl Biochem Biotech. 2014; 172:1530-1539.
-
13Devi SP, Kumaria S, Rao SR, Tandon P. Single primer amplification reaction (SPAR) methods reveal subsequent increase in genetic variations in micropropagated plants of Nepenthes khasiana Hook. f. maintained for three consecutive regenerations. Gene 2014; 538:23-29
-
14Chauhan R, Keshavkant S, Jadhav SK, Quraishi Afaque. In vitro slow-growth storage of Chlorophytum borivilianum Sant. et Fernand: a critically endangered herb. In Vitro Cell Dev-Pl. 2016; 52:315-321.
-
15Murashige T, Skoog F. A revised medium for rapid growth and bioassay with tobacco tissue culture. Physiol Plant 1962; 15:473-497.
-
16SPSS, Inc. SPSS 10.0 syntax reference guide. Chicago, IL; 1999.
-
17Quraishi A, Koche V, Mishra SK. Micropropagation of Lagerstroemia parviflora through axillary bud culture. Silvae Gene t. 1997; 46(4):242-245.
-
18Tyagi RK, Bhat SR, Chandel KPS. In vitro conservation strategies for spices crop germplasm Zingiber, Curcuma and Piper species. In: Mathew NM, Kuruvila JC, Editors. Developments in plantation crop research. India: Rubber Research Institute of India; 1998. p. 77-82.
-
19Nayak S. In vitro multiplication and microrhizome induction in Curcuma aromatic Salisb. Plant Growth Regul. 2000; 32:41-47.
-
20Nayak S. High frequency in vitro production of microrhizomes of Curcuma amada Indian J Exp Biol 2002; 40:230-232.
-
21Chirangini P, Sinha SK, Sharma GJ. In vitro propagation and microrhizome induction in Kaempferia galanga Linn. and K. rotunda Linn. Indian J Biotechnol. 2005; 4:404-408.
-
22Nor Asmah H, Nor Hasnida H, Noraliza A, Nashatul Zaimah NA, Nadiah Salmi N. In vitro propagation of Acacia hybrid through alginate-encapsulated shoots and axillary buds. Afr J Biotechnol. 2012; 11(65):12814-12817.
-
23Bukhari NAW, Siddique I, Perveen K, Siddiqui I, Alwahibi MS. Synthetic seed production and physio-biochemical studies in Cassia angustifolia Vahl. - a medicinal plant. Acta Biol Hung. 2014; 65(3):355-367.
-
24Tyagi RK, Anuradha A, Yusuf A. Conservation of Zingiber germplasm through in vitro rhizome formation. Sci Hortic-Amsterdam. 2006; 108:210-219.
-
25Mix-Wagner G. The conservation of potato cultivars. Potato Res. 1999; 42:427-436.
-
26Nayak S, Naik PK. Factors effecting in vitro microrhizome formation and growth in Curcuma longa L. and improved field performance of micropropagated plants. ScienceAsia 2006; 32:31-37.
Publication Dates
-
Publication in this collection
2017
History
-
Received
03 Feb 2016 -
Accepted
14 July 2016