ABSTRACT
Nanotubes with their unique properties have diversified mechanical and biological applications. Due to similarity of dimensions with extracellular matrix (ECM) elements, these materials are used in designing scaffolds. In this research, Carboxylated Single-Wall Carbon Nanotubes in optimization of decellularized scaffold of bovine articular cartilage was used. At first, the articular cartilage was decellularized. Then the scaffolds were analyzed in: (i) decellularized scaffolds, and (ii) scaffolds plunged into homogenous suspension of nanotubes in distilled water, were smeared with Carboxylated-SWCNT. The tissue rings derived from the rabbit's ear were assembled with reinforced scaffolds and they were placed in a culture media for 15 days. The scaffolds in two groups and the assembled scaffolds underwent histologic and electron microscopy. Scanning electron microscopy showed that the structure of ECM of articular cartilage has been maintained well after decellularization. Fourier transform infrared analysis showed that the contents of ECM have not been changed under treatment process. Atomic force microscopy analysis showed the difference in surface topography and roughness of group (ii) scaffolds in comparison with group (i). Transmission electron microscopy studies showed the Carboxylated-SWCNT bond with the surface of decellularized scaffold and no penetration of these compounds into the scaffold. The porosity percentage with median rate of 91.04 in group (i) scaffolds did not have significant difference with group (ii) scaffolds. The electron microscopy observations confirmed migration and penetration of the blastema cells into the group (ii) assembled scaffolds. This research presents a technique for provision of nanocomposite scaffolds for cartilage engineering applications.
Key words:
Articular cartilage; Carboxylated-SWCNT; Extracellular matrix; Natural scaffold; Tissue engineering
INTRODUCTION
Cartilage damage is one of the most challenging medical issues concerning skeleton and muscle, since the tissue is of low capacity for repair and restoration. Despite the progress in orthopedic surgery, the lack of efficient and quality treatment for cartilage defects has shifted researches towards tissue engineering along with using chondrogenic cells, scaffolds, and environmental factors (Vinatier et al. 200954 Vinatier C, Mrugala D, Jorgensen C, Guicheux J, Noel D (2009) Cartilage engineering: a crucial combination of cells, biomaterials and biofactors. Trends Biotechnol 27:307-314. doi:10.1016/j.tibtech.2009.02.005
https://doi.org/10.1016/j.tibtech.2009.0...
). Synthesized and natural scaffolds are among the commonly used scaffolds in tissue engineering. Collagen matrix, fibrin glue, agarose, alginate, hyaluronic acid, chitosan, and cellulose are natural matrices. Polylactic acid, Polyglycolic acid, carbon fibers, Dacron, Teflon, and hydrogel are examples of synthesized matrices (Danisovic et al. 201214 Danisovic L, Varga I, Zamborsky R, Bohmer D (2012) The tissue engineering of articular cartilage: cells, scaffolds and stimulating factors. Exp Biol Med 237:10-17. doi: 10.1258/ebm.2011.011229
https://doi.org/10.1258/ebm.2011.011229...
). Due to biomechanical power and special compounds, the synthesized materials can change according to the changes in the combination of the polymers. However, the main challenge for these materials is this that they are outside the body and lack tissue integrity and satisfactory differentiation. The natural biomaterials do not have these challenges because they are biocompatible and biodegradable. Despite the great progress in the science of material construction based on natural tissues, still very simple biomaterials are used for repairing and restoring cartilage tissue (Benders et al. 20136 Benders KEM, Weeren RV, Badylak SF, Saris DBF, Dhert WJA (2013) Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol 31(3):169-176. doi:10.1016/j.tibtech.2012.12.004
https://doi.org/10.1016/j.tibtech.2012.1...
).
The ideal properties of a matrix are biocompatibility to prevent the inflammatory reactions to protect the host tissue. The three-dimensional shape allows proliferation and cellular differentiation and porosity and eases migration of cells and diffusion of molecules, nutrients and oxygen. The matrix must also allow cell adhesion to facilitate the implantation of cells in the lesion and maintenance in the implant. It can also be bioactive and allow the homogeneous and controlled release of growth factors or morphogens (Nesic et al. 200634 Nesic D, Whiteside R, Brittberg M, Wendt D, Martin I, Mainil-Varlet P (2006) Cartilage tissue engineering for degenerative joint disease. Adv Drug Deliv Rev 58:300-322. doi:10.1016/j.addr.2006.01.012
https://doi.org/10.1016/j.addr.2006.01.0...
). Today, tissue engineering has focused on increasing use of biomaterials or advanced natural ECM -based scaffolds that is a very acceptable achievement for repairing the cartilage (Vinatier et al. 200954 Vinatier C, Mrugala D, Jorgensen C, Guicheux J, Noel D (2009) Cartilage engineering: a crucial combination of cells, biomaterials and biofactors. Trends Biotechnol 27:307-314. doi:10.1016/j.tibtech.2009.02.005
https://doi.org/10.1016/j.tibtech.2009.0...
). The decellularized scaffolds that aim at maintaining the microscopic architecture of the structure of natural cartilage may be the key to any effort for repair and restoration of cartilaginous joints (Ye et al. 201357 Ye K, Felimban R, Moulton S, Wallance GG, Bella CD, Traianedes K, Choong PF, Myers DE (2013) Bioengineering of articular cartilage: past, present and future. Regen Med 8:333-349. doi:10.2217/rme.13.28
https://doi.org/10.2217/rme.13.28...
).
During the recent decade, the regenerative medicine and nanotechnology have been extensively developed. In molecular level, using various substances in nanoscale in various fields of regenerative medicine including tissue engineering, cell-based therapy, diagnosis and delivery of genes and drugs have been studied (Peran et al. 201239 Peran M, Garcia MA, Lopez-Ruiz E, Bustamate M, Jimenez G, Madeddu R, Marchal JA (2012) Functionalized nanostructures with application in regenerative medicine, Int J Mol Sci 13:3847-3886. doi:10.3390/ijms13033847
https://doi.org/10.3390/ijms13033847...
). In the course of endless studies, applying nanostructure materials in repairing various tissues such as heart (Engel et al. 200816 Engel E, Michiardi A, Navarro M, Lacroix D, Planell JA (2008) Nanotechnology in regenerative medicine: The materials side. Trends Biotechnol 26:39-47. doi:10.1016/j.tibtech.2007.10.005
https://doi.org/10.1016/j.tibtech.2007.1...
), bone (Pina et al. 201540 Pina S, Oliveira JM, Reis RL (2015) Natural-Based Nanocomposites for Bone Tissue Engineering and Regenerative Medicine: A Review. Advanced Materials 27: 1143-1169. doi:10.1002/adma.201403354
https://doi.org/10.1002/adma.201403354...
), cartilage (Jeon et al. 201327 Jeon JE, Vaquette C, Klein TJ, Hutmacher DW (2013) Perspectives in Multiphasic Osteochondral Tissue Engineering. The Anatomical Record 297:26-35. doi: 10.1002/ar.22795
https://doi.org/10.1002/ar.22795...
), skin (Sundaramurthi et al. 201449 Sundaramurthi D, Krishnan UM, Sethuraman S (2014) Electrospun Nanofibers as Scaffolds for Skin Tissue Engineering. Polymer Reviews 54: 348-376. doi: 10.1080/15583724.2014.881374
https://doi.org/10.1080/15583724.2014.88...
), bladder (Chun et al. 201111 Chun YW, Lim H, Webster TJ, Haberstroh KM (2011) Nanostructured bladder tissue replacements. WIREs Nanomed Nanobiotechnol 3: 134-145. doi: 10.1002/wnan.89
https://doi.org/10.1002/wnan.89...
), nerves (Yim et al. 200758 Yim EK, Pang SW, Leong KW (2007) Synthetic nanostructures inducing differentiation of human mesenchymal stem cells into neuronal lineage. Exp Cell Res 313:1820-1829. doi:10.1016/j.yexcr.2007.02.031
https://doi.org/10.1016/j.yexcr.2007.02....
), and veins (Li et al. 201429 Li S, Sengupta D, Chien S (2014) Vascular tissue engineering: from in vitro to in situ. WIREs Syst Biol Med 6:61-76. doi: 10.1002/wsbm.1246
https://doi.org/10.1002/wsbm.1246...
) have been reviewed. The majority of chemical and physical methods in nanoscale for nanopatterned geometric engineering have been developed on biocompatible scaffolds (Park and Im 201438 Park S and Im G (2014) Stem cell responses to nanotopography. Journal of Biomedical Materials Research 103: 1238-1245. doi: 10.1002/jbm.a.35236
https://doi.org/10.1002/jbm.a.35236...
). One of the most promising nanometer-sized cylinders that could imitate nanofibers present in native ECM, are carbon nanotubes (Ng et al. 201235 Ng R, Zang R, Yang KK, Liu N, Yang ST (2012) Three-dimensional fibrous scaffolds with microstructures and nanotextures for tissue engineering. RSC Advances 2:10110-10124. doi: 10.1039/C2RA21085A
https://doi.org/10.1039/C2RA21085A...
). Holmes et al. (201422 Holmes B, Fang X, Zarate A, Keidar M, Zhang LG (2014) Enhanced Human Bone Marrow Mesenchymal Stem Cell Chondrogenic Differentiation in Electrospun Constructs with Carbon Nanomaterials. Carbon 97:1-13. doi:10.1016/j.carbon.2014.12.035
https://doi.org/10.1016/j.carbon.2014.12...
) in their research project showed that the mechanical properties of polycaprolactone synthesized scaffold resulting from electrospinning method are improved in the presence of Carboxylated-SWCNT and chondrogenic adhesion, proliferation and differentiation of the stem cells of bone marrow in such scaffold are increased in comparison with polycaprolactone control scaffolds lacking Carboxylated-SWCNT. Ge et al. (201218 Ge Z, Li C, Heng BC, Cao G, Yang Z (2012) Functional biomaterials for cartilage regeneration. J Biomed Mater Res A 100:2526-2536. doi: 10.1002/jbm.a.34147
https://doi.org/10.1002/jbm.a.34147...
) too, have carried out comprehensive research into functional biomaterials in connection with cartilage tissue. They claim that carbon nanotubes shall be placed in this group because they are able to reinforce mechanical capacities of polymeric scaffolds. Efforts for expansion of the application of these biocompatible structures in tissue engineering techniques meant to study the interaction between living cells and montaged nanotubes in the scaffolds and their impacts on cellular behavior including reinforcement of adhesion, proliferation, migration and the performance of cell differentiation are all on the rise (Veetil and Ye 200953 Veetil JV and Ye k (2009) Tailored carbon nanotubes for tissue engineering application. Biotechnol Prog 25:709-721. doi: 10.1002/btpr.165
https://doi.org/10.1002/btpr.165...
).
The regeneration of rabbit’s ear is an epimorphic regeneration which is based on the blastema tissue. This regeneration takes place in the appendices of vertebrates. The blastema tissues is a group of dedifferentiated cells which can divide and differentiate into other cells. The progenitor cells derived from the blastema cells tend to have osteogenic, adipogenic and chondrogenic differentiation potential (Baghaban Eslaminejad and Bordbar 20133 Baghaban Eslaminejad MR and Bordbar S (2013) Isolation and Characterization of the Progenitor Cells from the Blastema Tissue Formed at Experimentally-Created Rabbit Ear Hole. Iran J Basic Med Sci 16:109-115). The blastema ring model of the rabbit ear is a simple and effective model which can be used for tissue engineering studies (Hashemzadeh et al. 201521 Hashemzadeh MR, Mahdavi-Shahri N, Bahrami AR, Kheirabadi M, Naseri F, Atighi M (2015) Use of an in vitro model in tissue engineering to study wound repair and differentiation of blastema tissue from rabbit pinna. In Vitro Cell Dev Biol Animal 51:680-689. doi10.1007/s11626-015-9868-0
https://doi.org/10.1007/s11626-015-9868-...
; Simkin et al. 201548 Simkin J, Sammarco MC, Dawson LA, Schanes PP, Yu L, Muneoka K (2015) The mammalian blastema: regeneration at our fingertips. Regeneration 2:93-105).
With respect to the above findings, the hypothesis of using Carboxylated-SWCNT in improving the capacity of three-dimensional scaffolding of decellularized cartilage for tissue engineering application was introduced and then these reinforced scaffold were assembled with blastema ring to investigate interaction between these scaffolds and the living cells so that they can be used in the studies of cartilage tissues engineering. Therefore, in this study we introduce a technique for montaging Carboxylated-SWCNT with the three-dimensional scaffolds resulting from decellularized bovine articular cartilage (Holstein male calves) and characterization of scaffold.
MATERIALS AND METHODS
Suspension of Carboxylated-SWCNT preparation. Carboxylated-SWCNT coded US4112 were purchased from US Research Nanomaterials Inc., located in Houston, the United States. The purity of the raw material was 99+% and containing carboxyl of 2.73%. The median diameter of the nanotubes was 1.1 nanometer with a length of 5-30 nanometer in black. The Carboxylated-SWCNT solution was prepared by adding 100 microgram/ml of nanotube deionized in water. The resulting solution underwent sonication for 10 minutes by electrosonicator (Soniprep 150 Plus, MSC Company, UK) and then centrifuged (Sigma, Germany) ( Chen and Hsiue 20139 Chen YS and Hsiue GH (2013) Directing neural differentiation of mesenchymal stem cells by carboxylated multiwalled carbon nanotubes. Biomaterials 34:4936-4944. doi:10.1016/j.biomaterials.2013.03.063
https://doi.org/10.1016/j.biomaterials.2...
) at 5000 r/m for 10 minutes.
Articular cartilage preparation and decellularization. The joint bovine articular cartilage (6-8 month old, Holstein male calves) were taken immediately after slaughtering. Then 2 x 2 mm cylindrical pieces were separated by metal puncher (Fig. 1). In physical phase, the samples were rinsed for several times by phosphate buffer (pH=7.2) and placed for 2 minutes in liquid nitrogen, followed by frosting samples in distilled water and then phosphate buffer was melted at room at 20-25cº for 10 minutes. The last step was repeated for 5 times. In chemical phase, the samples were treated at an Sodium Dodecyl Sulfate (SDS) density of 2.5% for 3 hours at room at 20-25cº (Tavassoli et al. 201250 Tavassoli A, Mahdavi-Shahri N, Matin MM, Fereidoni M, Shahabipour F (2012) Bovine articular cartilage decellularized matrix as a scaffold for use in cartilage tissue engineering. Iranian Journal of Veterinary Science and Technology 4:1-8). Finally, the cartilage pieces were rinsed with phosphate buffer for 30 minutes. Then, in order to eliminate SDS and maintain the remaining water in the samples, the cartilage pieces were placed in Sterile Buchner Funnel (Ilmabop, Germany) and rinsed with ethanol 75%, distilled water, and phosphate buffer respectively.
Decellularized Scaffolds Reinforced by Carboxylated-SWCNT. The decellularized scaffolds of the bovine articular cartilage were grouped in two: group (i) comprised of decellularized scaffolds without Carboxylated-SWCNT, and group (ii) comprised of scaffolds floated inside a 100 ml Erlenmeyer Flask of Carboxylated-SWCNT homogenous suspension with a density of 100 microgram/ml and then were placed for 24 hours inside an incubator shaker of 150 r/m at a temperature of 37 degrees centigrade (New Brunswick Scientific Co., Inc. USA). The carboxylated carbon layers floating homogenously in the suspension had stuck to the decellularized scaffolds and the surfaces were fully covered by carbon nanotubes (Fig. 1).
Punch-hole procedure. Five male New Zealand white rabbits were provided from Razi Institute of Mashhad, each one weighing 2500±100 g and aged about 2-3 month. The organ of focus in this study was the pinna. Special punching devices were used for punch. The hairs of the area were shaved and disinfected by ethanol (70%). The rabbits then, were anesthetized by intramuscular injection of anesthetics including a mixture of ketamine 10% (Alfasan, Woerden, Holland) (1ml/kg body weight) and xylazine 2% (Alfasan, Woerden, Holland) (0.5 ml/kg body weight). After that, the punch of the pinna was performed in the medial region of the ear, between the central arteries and peripheral veins, each hole with a diameter of 4 mm. Two days after the primary punch, the tissue around the punched holes was biopsied by a special gadget with a diameter of 5 mm.
Fabrication of Reinforced scaffolds and blastema ring. The biopsy materials (rings) were washed seven times with sterile normal saline and immediately were fabricated by reinforced scaffolds under strile conditions (Fig. 2).The assembled specimens were resuspended in culture medium containing DMEM (Gibco,Waltham, MA) supplemented with 15% fetal bovine serum (Sigma, St. Louis,MO), penicillin/streptomycin (1 μg/ml) and fungizone (0.06 μl/ml). In this way, the Blastema rings were cultured for 15 days. The medium was refreshed three times a week and the specimens were reseeded in the six-well plates.
Histological Studies. The scaffolds with and without nanotubes were fixed in Bouin,s fixative, dehydrated in an ethanol (Merk, Darmstadt, Germany) ascending mode and placed in paraffin. Then, widthwise slices with a thickness of 7 micrometers were provided by microtome (Leits, Austria). Then the slices were de-paraffined by xylene (Merck Company) and next hydrated, stained with Hematoxylin & Eosin, toluidine blue and DAPI were carried out according to standard protocol of cartilage tissue (Schmitz et al. 201044 Schmitz N, Laverty S, Kraus VB, Aigner T (2010) Basic methods in histopathology of joint tissues. Osteoarthritis and Cartilage 18:S113-S116. doi:10.1016/j.joca.2010.05.026
https://doi.org/10.1016/j.joca.2010.05.0...
).
Scanning electron microscopy. Scaffolds with and without nanotubes and assembled specimens were fixed for 24 hours with glutaraldehydes 2.5% (TAAB Laboratories, UK). Then they were rinsed in sodium cacodylate buffer with molar ratio of 0.1 (PH=7.2, TAAB Laboratories, UK). Then the samples were treated with 1% osmium tetroxide, rinsed for an hour more in 0.1 molar sodium cacodylate buffer and dehydrated in descending degrees of absolute ethanol (Merck Company)( Shahabipour et al. 201345 Shahabipour F, Mahdavi- Shahri N, Matin MM, Tavassoli A, Zebarjad SM (2013) Scaffolds derived from cancellous bovine bone support mesenchymal stem cells' maintenance and growth. In Vitro Cell Dev Biol Anim 49:440-448. doi:10.1007/s11626-013-9591-7
https://doi.org/10.1007/s11626-013-9591-...
). Then the scaffolds were fixed on metal stubs and coated with gold-palladium by sputtering (Sputter Coater, SC7620, East Sussex, UK) and were studied by scanning electron microscopy (LEO 145oup, Germany).
Transmission electron microscopy. Scaffolds with and without nanotubes were immediately fixed in glutaraldehyde 2.5 percent solution for 30 minutes at room at 20-25cº and then kept for 24 hours in the refrigerator. Then the samples were rinsed with phosphate buffer (pH=7.2) for 18 minutes. Then, fixation in 2% osmium tetroxide was carried out for two hours at room at 20-25cº . The samples were immediately rinsed for once in phosphate buffer and two times in deionized water. The dehydration stages were carried out for three times completely at different densities of ethanol solution (Merk, Darmstadt, Germany) 70% (5 to 7 minutes), 95% (7 to 10 minutes), and 100% three times (each time 5 minutes)( Hashemzadeh et al. 201521 Hashemzadeh MR, Mahdavi-Shahri N, Bahrami AR, Kheirabadi M, Naseri F, Atighi M (2015) Use of an in vitro model in tissue engineering to study wound repair and differentiation of blastema tissue from rabbit pinna. In Vitro Cell Dev Biol Animal 51:680-689. doi10.1007/s11626-015-9868-0
https://doi.org/10.1007/s11626-015-9868-...
). After full passage of the samples and their placement in resin in 60 degrees centigrade for two days, the blocks were first trimmed and sectioning was then carried out by Ultra microtome. Then the samples were stained with 5% lead citrate and 3% uranyl acetate and prepared for electron microscopy studies (LEO 912AB).
Atomic force microscopy. Surface topography and roughness (Sq) a sample scaffolds with and without nanotubes and the intact cartilage were stationed on leg of the unit and were studied by atomic force microscopy (AFM model, Ara Research Company, Iran).
Fourier transform infrared Analysis. The raw Carboxylated-SWCNT powder and the powder resulting from the scaffolds with and without carbon nanotubes that were dried up at room at 20-25cº were mixed with potassium bromide (Merck Company, Germany) by a proportion of 1:100 (Chen and Hsiue 20139 Chen YS and Hsiue GH (2013) Directing neural differentiation of mesenchymal stem cells by carboxylated multiwalled carbon nanotubes. Biomaterials 34:4936-4944. doi:10.1016/j.biomaterials.2013.03.063
https://doi.org/10.1016/j.biomaterials.2...
) and scanned by spectrometer (Thermo Nicolet Model Avatar 370, USA).
Analysis of Porosity, Pore Size and Lacuna Area on the Scaffold. The porosity percentage was calculated in the scaffolds of the two groups by displacing fluid technique (Moradi et al. 201432 Moradi A, Pramanik S, Ataollahi F, Kamarul T, Pingguan-murphy B ( 2014) Archimedes revisited: computer assisted microvolumetric modification of liquid displacement method for porosity measurement of highly porous light materials. Anal Methods 6:4396-4401. doi: 10.1039/C4AY00666F
https://doi.org/10.1039/C4AY00666F...
). The data on porosity percentage, size of the pores and the lacuna area on the scaffolds of both groups were calculated based on Image J software (NIH, Bethesda, Maryland).
Statistical Analysis. Statistical analysis was carried out by SPSS Version 23. In order to evaluate a significant difference between the average porosity rate, size of the pores and the lacuna area on the scaffolds of both groups with and without nanotubes in comparison with each other, we used Tukey’s Test, considering minimum significance level of p<0.05.
RESULTS
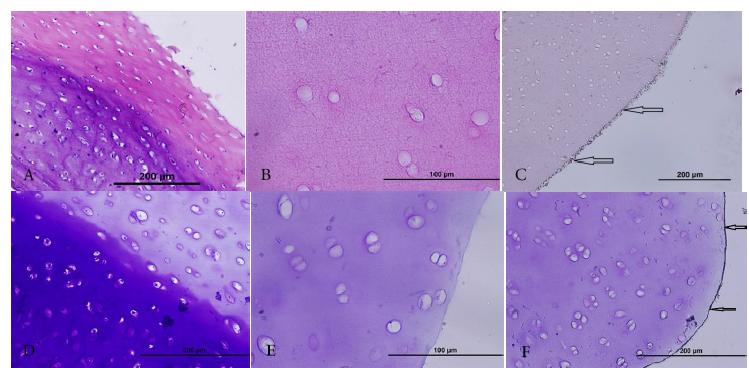
Fluorescent and Optical Microscopic observations. In order to study the full process of decellularization in scaffolds of the groups with and without nanotubes, and also for microscopic observation of carbon nanotubes staining with DAPI, H&A and toluidine blue was carried out. In staining with DAPI, in the healthy cartilage structure, due to its bond with a narrow line of DNA, two double-stranded DNA arrays in rich area of A-T in the presence of UV light with fluorescent microscope were expected to show DNA in chondrocyte nucleus (Fig. 3 A). Lack of these bright spots in the image indicates full decellularization in the scaffolds (Fig. 3 B). In H&E and toluidine blue staining, too, lacunas lacking cells in the scaffolds of both groups with and without nanotubes were observed (Fig. 4). In the scaffolds of both groups the adhesion of carbon nanotubes to the surface of the scaffolds were seen but penetration of nanotubes into the scaffolds was negative (Fig. 4).
Displaying decellularization from cartilage tissue with DAPI fluorescent stain, (A) Nucleus in the cartilage tissue with DAPI fluorescent is shown in bright spots and it is an indicator of chondrocytes in cartilage matrix (magnified x100). (B) Indicates scaffold of the decellularized cartilage in black due to lack of nucleus (magnified x100)
H&E staining (A to C) and toluidine blue (D to F). (A) and (D) are intact articular cartilage where chondrocytes are seen; (B) and (E) are decellularized scaffolds, (C) and (F) are decellularized scaffolds smeared with carbon nanotubes. Black Arrows show adhesion of carbon nanotubes to the surface of the scaffold.
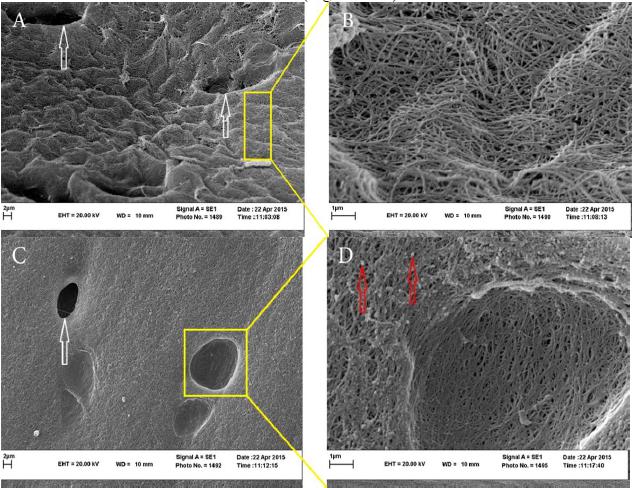
Scanning electron microscopy observations. In the scaffolds without carbon nanotubes, cell-free lacunal and regular dispersion of collagen strands on the surface of scaffolds are seen (Fig. 5 A&B). In the scaffolds with carbon nanotubes regular dispersion of carbon nanotubes on the surface of scaffolds are observable (Fig. 5 C&D).
Studying decellularized scaffolds in the two groups with SEM showed white arrows as lacunal free from cells in (A) group (i) scaffolds and (C) in group (ii) scaffolds. (B) Shows regular dispersion of collagen strands in group (i) and (D) Shows red arrows depicting carbon nanotubes scattered on the surface of the scaffold.
belastema cell migration, attachment and viability.
SEM microscopic imaging was conducted on the assembled specimens of fifteenth day of the culture. In these investigation were observed the migration, attachment and viability of the blastema cells (Fig. 6)
Blastema cells in different phases of development in fifteenth day of culture. (1) Isolated blastema cells in the vicinity of the reinforced scaffold (2) The formation of cytoplasmic filopodia during the migration and adhesion of cells to the surface of the reinforced scaffold (3) The attachment of the cells into the surface of the reinforced scaffold .
Transmission electron microscopy observations. TEM observation showed the adhesion of Carboxylated-SWCNT to the surface of decellularized group (ii) scaffolds. In this study, the penetration of Carboxylated-SWCNT into the ECM structure was not observed. In these observations the structural elements of cartilage matrix such as proteoglycan were observable (Fig. 7).
Studying decellularized scaffold smeared with carbon nanotubes with TEM. Red Arrows show adhesion of nanotubes to the surface of the scaffold (scale bar = 640 nm)
Atomic Force Microscopy observations. The topographic changes and roughness were compared between the intact cartilage and the scaffolds with and without carbon nanotubes. The dispersion of carbon nanotubes on the surface of scaffolds with nanotubes changed surface topography and roughness. In the intact cartilage and the scaffolds of a group with nanotubes, more surface incongruity was observed in comparison with scaffolds lacking carbon nanotubes (Fig. 8). The mean roughness value (Ra) which was calculated with the help of AFM images is shown in Table 1. The intact cartilage and the scaffolds with carbon nanotubes exhibited a higher degree of roughness than the scaffolds without carbon nanotubes.
Surface topography with AFM technique. (A) Surface topography of intact cartilage, (B) Surface topography of decellularized scaffold (without nanotube), (c) Surface topography of decellularized scaffold smeared with carbon nanotubes
FT-IR analysis. FT-IR analysis was carried out in the case of raw Carboxylated-SWCNT powder and the scaffolds of groups with and without carbon nanotubes. FT-IR analysis showed the presence of carboxylic acid groups with peak presence in absorption area of 1311cm-1 in comparison with FT-IR analysis of pure Single-wall carbon nanotube (Abjameh et al. 20141 Abjameh R, Moradi O, Amini J (2014) The study of synthesis and functionalized single-walled carbon nanotubes with amide group. Int Nano Lett 4:97-103. doi:10.1007/s40089-014-0097-4
https://doi.org/10.1007/s40089-014-0097-...
) (Fig. 9 A). The scaffolds of group without nanotubes had similar peaks with intact cartilage that shows the presence of collagen strands and proteoglycan in decellularized scaffolds. The scaffolds of the group with nanotubes had more absorption in comparison with the group without nanotubes and the created peak in areas with wavelength of 1236 and 1451 in the curve related to the scaffolds with nanotubes can show the presence of adhesion between the carbon nanotubes and the constituent elements of decellularized scaffolds (Fig. 9 B).
FT-IR absorption of raw Carboxylated-SWCNT powder, intact cartilage and the scaffold of the groups with and without carbon nanotubes. (A) FT-IR absorption of raw Carboxylated-SWCNT powder (CNT), black arrow shows the peak resulting from the presence of carboxylic acid. (B) Defined wavelengths in blue and red show collagen and proteoglycans presence respectively. Two sharp peaks in wavelengths of 1236 and 1451 can indicate establishment of a bond between carbon nanotubes and decellularized scaffold.
Porosity Rate, Pore Size and Lacuna Area on the Scaffold. The average porosity rate, size of pores and the lacuna area in the scaffolds with and without nanotubes have been presented in Table 2. After statistical analysis, using SPSS software and comparing the median data by using Tukey’s Test and considering the minimum significance level of p≤0.5 it was made clear that the porosity rate, pore size and lacuna area in the scaffolds with and without carbon nanotubes had no significant difference.
Average and Standard Deviation from Median Variables in Scaffolds with and without Carbon Nanotubes
DISCUSSION
The increasing demand by organizations and shortage of appropriate implants have led to the many developments in tissue engineering and regenerative medicine, particularly in scaffold design (Hopley et al. 201423 Hopley EL, Salmasi S, Kalaskar DM, Seifalian AM (2014) Carbon nanotubes leading the way forward in new generation 3D tissue engineering. Biotechnology Advances 32 : 1000-1014. doi:10.1016/j.biotechadv.2014.05.003
https://doi.org/10.1016/j.biotechadv.201...
). Recently, nonmaterial's have become innovative candidates for producing scaffolds that are able to copy natural ECM nanostructures for effective replacement of cartilage defect (Zhao et al. 201364 Zhao C, Tan A, Pastorin G, Ho KH (2013) Nanomaterial scaffolds for stem cell proliferation and differentiation in tissue engineering. Biotechnology Advances 31:654-668. doi:10.1016/j.biotechadv.2012.08.001
https://doi.org/10.1016/j.biotechadv.201...
). Nanotubes are reliable additives for the synthetic scaffolds that have entered into the regenerative medicine (Hopley et al. 2014). Compared to other studies dealing with characterization, synthesis and applications of nanotubes, to date, only a few reports have been investigated the different CNTs effects on the cell morphology and the protein structure, and these reports seem to be conflicting (Zeinabad et al. 201659 Zeinabad HA, Zarrabian A, Saboury AA, Alizadeh AM, Falahati M (2016) Interaction of single and multi wall carbon nanotubes with the biological systems: tau protein and PC12 cells as targets. Sci Rep 6:26508. doi: 10.1038/srep26508
https://doi.org/10.1038/srep26508...
). In contrast, many investigations reported CNT stimulated negligible risk of cytotoxicity (Huczko and Lange 200124 Huczko A. & Lange H (2001) Carbon nanotubes: experimental evidence for a null risk of skin irritation and allergy. Fullerene Science and Technology 9: 247-250. doi: 10.1081/FST-100102972
https://doi.org/10.1081/FST-100102972...
, Shi Kam et al. 200447 Shi Kam NW, Jessop TC, Wender PA, Dai H (2004) Nanotube molecular transporters: internalization of carbon nanotube-protein conjugates into mammalian cells. J Am Chem Soc 126:6850-6851. doi: 10.1021/ja0486059
https://doi.org/10.1021/ja0486059...
, Fiorito et al. 200617 Fiorito S, Serafino A, Andreola F, Bernier P (2006) Effects of fullerenes and single-wall carbon nanotubes on murine and human macrophages. Carbon 44:1100-1105., Nagai et al. 201333 Nagai H, Okazaki Y, Chew SH, Misawa N, Miyata Y, Shinohara H, Toyokuni S (2013) Intraperitoneal administration of tangled multiwalled carbon nanotubes of 15 nm in diameter does not induce mesothelial carcinogenesis in rats. Pathol Int 63:457-462. doi: 10.1111/pin.12093
https://doi.org/10.1111/pin.12093...
, Toyokuni et al. 201551 Toyokuni S, Jiang LI, Kitaura R, Shinohara H (2015) Minimal inflammogenicity of pristine single-wall carbon nanotubes. Nagoya J Med Sci 77:195-202.). Nanotubes have already been used in endless structures such as collagen, polystyrene, polycarbonate urethane, poly-l-lactide. Previous studies showed that although the scaffolds made only with these polymers are porous, they lack required quality and compound for implantation (Meng et al. 200931 Meng J, Kong H, Han Z, Wang C, Zhu G, Xie S, Xu H (2009) Enhancement of nanofibrous scaffold of multiwalled carbon nanotubes/polyurethane composite to the fibroblasts growth and biosynthesis. J Biomed Mater Res 88:105-16. doi: 10.1002/jbm.a.31862
https://doi.org/10.1002/jbm.a.31862...
). Carbon nanotubes have been also used in polymer scaffolds and hydrogels (Hopley et al. 2014). Also synthetic solid scaffolds, despite superiority in chemical and mechanical properties, absorbability in comparison with hydrogel scaffolds, due to reduced cell interaction and biological interactions of the scaffold-cell that ends in constant changes of polymer scaffolds during various tests, has been limited (Izadifar et al. 201225 Izadifar Z, Chen X, Kulyk W (2012) Strategic Design and Fabrication of Engineered Scaffolds for Articular Cartilage Repair. J Funct Biomater 3:799-838. doi:10.3390/jfb3040799
https://doi.org/10.3390/jfb3040799...
).
In this research work, the decellularized scaffold of bovine articular cartilage was smeared with Carboxylated-SWCNT. The successful clinical applications of decellularized tissues in the case of surgeries for intestinal and cardiovascular reconstruction have been reported (Crapo et al. 201112 Crapo PM, Gilbert TW, Badylak SF (2011) An overview of tissue and whole organ decellularization processes. Biomaterials 32:3233-3243. doi:10.1016/j.biomaterials.2011.01.057
https://doi.org/10.1016/j.biomaterials.2...
). Badylak (20072 Badylak SF (2007) The extracellular matrix as a biologic scaffold material. Biomaterials 28:3587-3593. doi:10.1016/j.biomaterials.2007.04.043
https://doi.org/10.1016/j.biomaterials.2...
) had studies on optimistic applications of extracellular decellularized matrix for osteochondral repair. Despite the success in decellularization of the cartilage tissue, during decellularization, the mechanical components of ECM change (Cheng et al. 201410 Cheng CW, Solorio LD, Alsberg E (2014) Decellularized tissue and cell-derived extracellular matrices as scaffolds for orthopaedic tissue engineering. Biotechnology Advances 32: 462-484. doi:10.1016/j.biotechadv.2013.12.012
https://doi.org/10.1016/j.biotechadv.201...
). Gilbert et al. (200619 Gilbert TW, Sellaro TL, Badylak SF (2006) Decellularization of tissues and organs. Biomaterials 27:3675-3683. doi:10.1016/j.biomaterials.2006.02.014
https://doi.org/10.1016/j.biomaterials.2...
) and Wiegand et al. (200956 Wiegand C, Abel M, Ruth P, Wilhelms T, Schulze D, Norgauer J, Hipler UC (2009) Effect of the sterilization method on the performance of collagen type I on chronic wound parameters in vitro. J Biomed Mater Res B Appl Biomater 90: 710-719. doi: 10.1002/jbm.b.31338
https://doi.org/10.1002/jbm.b.31338...
) have reported the destructive impact of decellularization and sterilization methods on tissue structure. Nanotubes can boost mechanical strength of the scaffolds and better adhesion or attachment of the cells for growth (Zhao et al. 201364 Zhao C, Tan A, Pastorin G, Ho KH (2013) Nanomaterial scaffolds for stem cell proliferation and differentiation in tissue engineering. Biotechnology Advances 31:654-668. doi:10.1016/j.biotechadv.2012.08.001
https://doi.org/10.1016/j.biotechadv.201...
). The entry of nanotubes inside the scaffold causes structural and chemical reinforcement of scaffold such as flexibility and solidity, improved compatibility, reduced multiplication of cancerous cells and induction of angiogenesis (Hopley et al. 201423 Hopley EL, Salmasi S, Kalaskar DM, Seifalian AM (2014) Carbon nanotubes leading the way forward in new generation 3D tissue engineering. Biotechnology Advances 32 : 1000-1014. doi:10.1016/j.biotechadv.2014.05.003
https://doi.org/10.1016/j.biotechadv.201...
). In this project, after decellularization of the scaffolds the general structure of the tissue and collagen fiber array had been maintained well. SEM studies demonstrated the microstructure of the decellularized scaffold with an interconnected composition. This structural composition facilitated the cellular migration, nutrients transportation and the disposal of waste material during culture period (Fig. 5A,B). FT-IR analysis showed that in the decellularized scaffold of articular cartilage the major components sach as collagen 2 and proteoglycan were maintained (Fig. 9B). After immersion of decellularized scaffold in homogenous suspension of carbon nanotubes, these compounds were expected to penetrate into the structure of decellularized cartilage, but TEM analysis showed that they were placed on the surface of the decellularized scaffold. The intertwined status of collagen strands and concentration of negative loads on the structure of cartilage tissue can be one reason for the above lodgement. Changing the technique of smearing the decellularized scaffold with carbon nanotubes or functionalization of the nanotubes with other functional groups may improve their penetration, although it needs more studies. However, attachment of the carbon nanotubes to the surface could provide bio-nanocomposite scaffold that can be used in vitro studies on adhesion and cellular performance. According to other research Carbon nanotubes can become adhesive when their surface chemistry is modified(Rose et al. 201342 Rose S, Prevoteau A, Elzière P, Hourdet D, Marcellan A, Leibler L (2013) Nanoparticle solutions as adhesives for gels and biological tissues. Nature 505:382-5. doi: 10.1038/nature12806
https://doi.org/10.1038/nature12806...
). The nanotubes Can affect the cells and the cell morphology alter through the nanostructured surface. thus CNT-based surfaces provide a very good scaffold that can be used to stimulate the formation of cartilage tissue (Sutherland et al. 201546 Sutherland AJ, Beck EC, Dennis SC, Converse GL, Hopkins RA, Berkland CJ, Detamore MS (2015) Decellularized cartilage may be a chondroinductive material for osteochondral tissue engineering. PLoS One 10:e0121966. doi: 10.1371/journal.pone.0121966
https://doi.org/10.1371/journal.pone.012...
and Trzeciak et al. 201652 Trzeciak T, Rybka JD, Akinoglu EM, Richter M, Kaczmarczyk J, Giersig Michael (2016) In Vitro Evaluation of Carbon Nanotube-Based Scaffolds for Cartilage Tissue Engineering. J Nanosci Nanotechnol 16:9022-9025. doi: http://dx.doi.org/10.1166/jnn.2016.12733
https://doi.org/10.1166/jnn.2016.12733...
). We tried to keep natural structure of decellularized cartilage intact and put it close to nanotubes in such an environment.
Carboxylated-SWCNT in comparison with multi-wall nanotubes have more power to help proliferation of chondrocytes (Hopley et al. 201423 Hopley EL, Salmasi S, Kalaskar DM, Seifalian AM (2014) Carbon nanotubes leading the way forward in new generation 3D tissue engineering. Biotechnology Advances 32 : 1000-1014. doi:10.1016/j.biotechadv.2014.05.003
https://doi.org/10.1016/j.biotechadv.201...
). In this project, we used Carboxylated-SWCNT. The penetration of the carbon nanotubes into polymers is not free from complexity, because nanotubes are naturally neutral chemically and have weak dispersion in various solutions. Functionalization of nanotubes prevents their aggregation and allows penetration into polymers (Balasubramanian and Burghard 20055 Balasubramanian K and Burghard M (2005) Chemically functionalized carbon nanotubes. Small 1:180-192. doi: 10.1002/smll.200400118
https://doi.org/10.1002/smll.200400118...
; Harrison and Atala 200720 Harrison BS and Atala A (2007) Carbon nanotube applications for tissue engineering. Biomaterials 28:344-353. doi:10.1016/j.biomaterials.2006.07.044
https://doi.org/10.1016/j.biomaterials.2...
). Functionalization of the surface of nanotubes with bioactive molecules helps biocompatibility and bioactivity of the scaffolds (Hopley et al. 2014). Chahine et al. (20148 Chahine NO, Collette N, Thomas C, Genetos DC, Loots GG (2014) Nanocomposite scaffold for chondrocyte growth and cartilage tissue engineering: effects of carbon nanotube surface functionalisation. Tissue Eng Part A 20: 2305-2315. doi:10.1089/ten.tea.2013.0328
https://doi.org/10.1089/ten.tea.2013.032...
) in a study found out that the presence of carboxyl group in Carboxylated-SWCNT leads to reinforcement of the growth of chondrocytes and ECM protein. FT-IR analysis showed that attachment has been made between Carboxylated-SWCNT and the decellularized scaffolds. There is the possibility of carboxyl groups’ involvement in this attachment. Zhang et al. (201063 Zhang Y, Bai Y, Yan B (2010) Functionalized carbon nanotubes for potential medicinal applications. Drug Discovery Today 15:428-435. doi:10.1016/j.drudis.2010.04.005
https://doi.org/10.1016/j.drudis.2010.04...
) studied potentials of carbon nanotubes in the interaction with collagen fibrils as scaffold in engineering of various tissues. Collagen, as one of the major components of ECM causes durability against tension and proportion of load on the tissue (Zelenski et al. 2015). It seems that juxtaposition of carbon nanotubes with collagen has the potential to improve the electrical and mechanical properties of this biomaterial. Creating structural changes in the nanotubes and various covalent bond with various matrix elements were studied by Veetil and Ye (200953 Veetil JV and Ye k (2009) Tailored carbon nanotubes for tissue engineering application. Biotechnol Prog 25:709-721. doi: 10.1002/btpr.165
https://doi.org/10.1002/btpr.165...
) for discovering the closest synthesized structure to the natural tissue. With the creation of covalent bond between nanotubes and ECM elements such as collagen, creates tissue correlation that is influential in the durability of the scaffold. Carbon nanotubes with high level and hardness create a surface like collagen strands that are available in natural tissues (Zhang et al. 2010). Due to the capability of nanotubes that structurally copy collagen fibers, it has been proven that they are useful in boosting cell attachment, proliferation and differentiation (Zhang et al. 2008; Ross et al. 201243 Ross AM, Jiang Z, Bastmeyer M, Lahann J (2012) Physical aspects of cell culture substrates:topography, roughness, and elasticity. Small 8:336-355. doi: 10.1002/smll.201100934
https://doi.org/10.1002/smll.201100934...
). In tissue engineering the three-dimensional scaffolds usually exert biological and mechanical impacts on the cells (Zhao et al. 201364 Zhao C, Tan A, Pastorin G, Ho KH (2013) Nanomaterial scaffolds for stem cell proliferation and differentiation in tissue engineering. Biotechnology Advances 31:654-668. doi:10.1016/j.biotechadv.2012.08.001
https://doi.org/10.1016/j.biotechadv.201...
). During their tests, Lin et al. (201130 Lin C, Wang Y, Lai Y, Yang W, Jiao F, Zhang H, Ye S, Zhang Q (2011) Incorporation of carboxylation multiwalled carbon nanotubes into biodegradable poly(lactic-co-glycolic acid)for bone tissue engineering. Colloids Surf B Biointerfaces 83:367-375. doi:10.1016/j.colsurfb.2010.12.011
https://doi.org/10.1016/j.colsurfb.2010....
) found out that mesenchymal stem cells of mice on nanocomposite scaffolds of PLGA/C-MWCNT showed higher adhesion and viability in comparison with polyester scaffolds in 21 days of culture. Lee et al. (201528 Lee JH, Shim W, Choolakadavil Khalid N, Kang WS, Lee M, Kim HS, Choi J, Lee G, Kim JH (2015) Random Networks of Single-Walled Carbon Nanotubes Promote Mesenchymal Stem Cell’s Proliferation and Differentiation. ACS Appl Mater Interfaces 7:1560−1567. doi: 10.1021/am506833q
https://doi.org/10.1021/am506833q...
) in a study proved that synthetic scaffolds based on Carboxylated-SWCNT showed no toxicity while these scaffolds boost proliferation and differentiation of the mesenchymal stem cells taken from the rats.
Under in vivo conditions the celled are placed in the three-dimensional micro-environments surrounded by other cells and extracellular matrices. Elements such as collagen, elastin and laminine that have been organized in nanostructures, regulate the homeostasis of the cells by bioactive motifs (Li et al. 201429 Li S, Sengupta D, Chien S (2014) Vascular tissue engineering: from in vitro to in situ. WIREs Syst Biol Med 6:61-76. doi: 10.1002/wsbm.1246
https://doi.org/10.1002/wsbm.1246...
). Das and Zouani (2014) carried out extensive studies on the powerful influence of nanoscale topography on cellular behavior such as cellular bonds, dispersion or cytoskeleton organization and even regulating gene expression. Bettinger et al. (20097 Bettinger CJ, Langer R, Borenstein JT (2009) Engineering substrate topography at the microand nanoscale to control cell function. Angew Chem Int Ed Engl 48:5406-5415. doi: 10.1002/anie.200805179
https://doi.org/10.1002/anie.200805179...
) proved that collagen-rich nanostructure bone and hydroxyapatite natural ECM take part in cell matrix signaling. Topographic signs in micro or Nano-dimension are among the preliminary mediators for surface absorption of proteins from the surrounding environment that mediate the process of cell attachment (Oliveira et al. 201436 Oliveira MB, Ribeiro MP, Miguel SP, Neto AI, Coutinho P, Correia IJ, Mano JF (2014) In vivo high-content evaluation of three-dimensional scaffolds biocompatibility. Tissue Eng Part C Methods 20: 851-864. doi:10.1089/ten.tec.2013.0738
https://doi.org/10.1089/ten.tec.2013.073...
). Controlling these properties can improve cell behavior. Designing special patterns and surface fissures can associate structural cellular model or arraying that changes the cytoskeleton contraction capacity, that is a key to cellular differentiation. Designing surface components and surface engineering techniques have been studied extensively (Oliveira et al. 2015). Therefore, with respect to the importance of surface topographical changes in the scaffolds in cellular behavior, AFM was carried out. AFM analysis showed surface topographic change in the scaffolds with nanotubes in comparison with scaffolds without nanotubes (Fig. 8). Surface roughness was found to be higher in the intact cartilage and the scaffolds with nanotubes in comparison to scaffolds without nanotubes (Table 1). In the natural cartilage, the surface layer of articular cartilage was very incongruous in terms of microscopy and this structure was due to special dispersion of the collagen fibers. On the other hand, the surface topography of viable articular cartilage too, is flexible and changes under stress or pressure. In the damaged cartilage this topography changes completely and affects the way of dispersion of collagen fibers (Wen et al. 201255 Wen CY, Wu CB, Tang B, Wang T, Yan CH, Lu WW, Pan H, Hu Y, Chiu KY (2012) Collagen fibril stiffening in osteoarthritic cartilage of human beings revealed by atomic force microscopy. Osteoarthritis and Cartilage 20:916-922. doi:10.1016/j.joca.2012.04.018
https://doi.org/10.1016/j.joca.2012.04.0...
). According to the results of AFM analysis it seems that attachment of the carbon nanotubes to the surface is effective is simulation of this surface as a viable articular cartilage and accordingly boosting its resistance against stress.
The porosity rate, pore size and lacuna area despite dispersion of carbon nanotubes on the surface of the scaffolds in two groups did not show significant meaning. All three factors are important in tissue engineering of the cartilages. The porosity rate in the scaffolds of group (ii) has been maintained in high level due to carbon nanotubes. Despite excellent mechanical, thermal and conductive properties carbon nanotubes are very porous and light and the pores in SWCNT are usually less than one nanometer in diameter (Jagtoyen et al. 200026 Jagtoyen M, Pardue J, Rantell T, Derbyshire F (2000) Porosity of carbon nanotubes. Adsorpt Sci Technol 17:289-293. doi: 10.1142/9789812793331_0058
https://doi.org/10.1142/9789812793331_00...
). Therefore, the presence of these compounds has not reduced the porosity rate in viable decellularized scaffold. This can lead us to access to a scaffold with ideal specifications. Various parameters on designing scaffold include size and geometry of pores, their distribution, as well as the diameter and distance of fibers. The size of fibers in the scaffolds affect cellular activity and thicker fibers impose negative impact on cellular activity due to creating more two-dimensional level for each cell (Izadifar et al. 201225 Izadifar Z, Chen X, Kulyk W (2012) Strategic Design and Fabrication of Engineered Scaffolds for Articular Cartilage Repair. J Funct Biomater 3:799-838. doi:10.3390/jfb3040799
https://doi.org/10.3390/jfb3040799...
). In the scaffold resulting from decellularization of articular cartilage, we have a viable ECM that their number, geometry of pores and diameter of fibers, distance and their state of positioning along with many important parameters that are considered in designing synthetic scaffolds have the maximum compatibility with the viable tissue of cartilage and this can help us in access to the goal of optimized tissue engineering of cartilage.
The cartilage of rabbit's ear has been used to study the regeneration process of perichondrium, skin and cartilage (Ten Koppel et al. 2001; Brockes and Kumar 2002). In rabbit's injured ear, blastema cells emerge through dedifferentiation process (Corcoran and Ferretti 1999). There are various theories about the origin of these like stem cells including perichondrium and cartilage cells (Hashemzadeh et al. 201521 Hashemzadeh MR, Mahdavi-Shahri N, Bahrami AR, Kheirabadi M, Naseri F, Atighi M (2015) Use of an in vitro model in tissue engineering to study wound repair and differentiation of blastema tissue from rabbit pinna. In Vitro Cell Dev Biol Animal 51:680-689. doi10.1007/s11626-015-9868-0
https://doi.org/10.1007/s11626-015-9868-...
; Simkin et al. 201548 Simkin J, Sammarco MC, Dawson LA, Schanes PP, Yu L, Muneoka K (2015) The mammalian blastema: regeneration at our fingertips. Regeneration 2:93-105). Due to their unique characteristics and the chondrogenic tendency of blastema cells as an in vitro model this research project has investigated the interaction between the reinforced scaffold and the blastema cells. In order to maximize the adhesion of the scaffold to the ring, we assembled the decellularized scaffold on to the blastema ring to optimize the scaffold attachment to the ring (Fig. 2). The assembled scaffold which had been in culture media for fifteen days was evaluated through SEM electronic microscope. According to SEM microscopic analyses, it was observed that there were cells which were migrating in different phases such as isolated round cells, some cells with cytoplasmic filopodia and Spindle shaped cells which were attached to the scaffold surface reveal phenotypic manifestation to the chondrocyte (Fig. 6). These cells eventually were influenced by the elements within the decellularized scaffolds. They also displayed the morphologic traits of chondrocytes. This phenomenon confirmed the regulatory and instructional role of the decellularized reinforced scaffold on cellular behavior and biocompatibility of carbon nanotubes which has received attention in other researches (Place et al. 200941 Place E S, Evans ND, Stevens MM (2009) Complexity in biomaterials for tissue engineering. Nat Mater 8:457-470. doi: 10.1038/nmat2441
https://doi.org/10.1038/nmat2441...
, Cross et al. 201613 Cross LM, Thakur A, Jalili NA, Detamore M, Gaharwar AK (2016) Nanoengineered biomaterials for repair and regeneration of orthopedic tissue interfaces. Acta Biomater. S1742-7061(16)30300-2. doi: 10.1016/j.actbio.2016.06.023
https://doi.org/10.1016/j.actbio.2016.06...
, Trzeciak et al. 201652 Trzeciak T, Rybka JD, Akinoglu EM, Richter M, Kaczmarczyk J, Giersig Michael (2016) In Vitro Evaluation of Carbon Nanotube-Based Scaffolds for Cartilage Tissue Engineering. J Nanosci Nanotechnol 16:9022-9025. doi: http://dx.doi.org/10.1166/jnn.2016.12733
https://doi.org/10.1166/jnn.2016.12733...
).
CONCLUSION
This project introduced a technique for montage of Carboxylated-SWCNT on decellularized scaffold of articular cartilage for engineering applications of the cartilage tissue. Histological and microscopic surveys showed that in the decellularized scaffold of articular cartilage the main components sach as collagen 2 and proteoglycans were preserved. This composition can be used as a useful model in fundamental researches of regenerative medicine. This reinforced scaffold can support the adhesion and survival of blastema cells.
Apparently, the blastema cells which are attached to this decellularized reinforced scaffold can be used in engineering researches of cartilage tissue. Further studies are recommended to perform gene expression analysis about cells penetrating to scaffold. Further supplementary studies in in vivo circumstances are also recommended. In this project, nanotubes as useful additives for decellularized scaffolds, could affect the surface topography of the scaffold.
Scaffold designing for optimization of the complicated tissue is still under study and exploration. This is, more than anything else, due to complexity of the scaffolds since they have to copy the viable ECM. Studying mechanical properties of the scaffolds with and without Carboxylated-SWCNT such as their resistance against pressure, tension is proposed. More studies are need to find out the impact of carbon nanotubes on the regenerative and restorative capacity of the scaffolds in animal models.
REFERENCES
-
1Abjameh R, Moradi O, Amini J (2014) The study of synthesis and functionalized single-walled carbon nanotubes with amide group. Int Nano Lett 4:97-103. doi:10.1007/s40089-014-0097-4
» https://doi.org/10.1007/s40089-014-0097-4 -
2Badylak SF (2007) The extracellular matrix as a biologic scaffold material. Biomaterials 28:3587-3593. doi:10.1016/j.biomaterials.2007.04.043
» https://doi.org/10.1016/j.biomaterials.2007.04.043 -
3Baghaban Eslaminejad MR and Bordbar S (2013) Isolation and Characterization of the Progenitor Cells from the Blastema Tissue Formed at Experimentally-Created Rabbit Ear Hole. Iran J Basic Med Sci 16:109-115
-
4Balakrishnan B and Banerjee R (2011) Biopolymer-based hydrogels for cartilage tissue engineering. Chem Rev 111:4453-4474. doi:10.1021/cr100123h
» https://doi.org/10.1021/cr100123h -
5Balasubramanian K and Burghard M (2005) Chemically functionalized carbon nanotubes. Small 1:180-192. doi: 10.1002/smll.200400118
» https://doi.org/10.1002/smll.200400118 -
6Benders KEM, Weeren RV, Badylak SF, Saris DBF, Dhert WJA (2013) Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol 31(3):169-176. doi:10.1016/j.tibtech.2012.12.004
» https://doi.org/10.1016/j.tibtech.2012.12.004 -
7Bettinger CJ, Langer R, Borenstein JT (2009) Engineering substrate topography at the microand nanoscale to control cell function. Angew Chem Int Ed Engl 48:5406-5415. doi: 10.1002/anie.200805179
» https://doi.org/10.1002/anie.200805179 -
8Chahine NO, Collette N, Thomas C, Genetos DC, Loots GG (2014) Nanocomposite scaffold for chondrocyte growth and cartilage tissue engineering: effects of carbon nanotube surface functionalisation. Tissue Eng Part A 20: 2305-2315. doi:10.1089/ten.tea.2013.0328
» https://doi.org/10.1089/ten.tea.2013.0328 -
9Chen YS and Hsiue GH (2013) Directing neural differentiation of mesenchymal stem cells by carboxylated multiwalled carbon nanotubes. Biomaterials 34:4936-4944. doi:10.1016/j.biomaterials.2013.03.063
» https://doi.org/10.1016/j.biomaterials.2013.03.063 -
10Cheng CW, Solorio LD, Alsberg E (2014) Decellularized tissue and cell-derived extracellular matrices as scaffolds for orthopaedic tissue engineering. Biotechnology Advances 32: 462-484. doi:10.1016/j.biotechadv.2013.12.012
» https://doi.org/10.1016/j.biotechadv.2013.12.012 -
11Chun YW, Lim H, Webster TJ, Haberstroh KM (2011) Nanostructured bladder tissue replacements. WIREs Nanomed Nanobiotechnol 3: 134-145. doi: 10.1002/wnan.89
» https://doi.org/10.1002/wnan.89 -
12Crapo PM, Gilbert TW, Badylak SF (2011) An overview of tissue and whole organ decellularization processes. Biomaterials 32:3233-3243. doi:10.1016/j.biomaterials.2011.01.057
» https://doi.org/10.1016/j.biomaterials.2011.01.057 -
13Cross LM, Thakur A, Jalili NA, Detamore M, Gaharwar AK (2016) Nanoengineered biomaterials for repair and regeneration of orthopedic tissue interfaces. Acta Biomater. S1742-7061(16)30300-2. doi: 10.1016/j.actbio.2016.06.023
» https://doi.org/10.1016/j.actbio.2016.06.023 -
14Danisovic L, Varga I, Zamborsky R, Bohmer D (2012) The tissue engineering of articular cartilage: cells, scaffolds and stimulating factors. Exp Biol Med 237:10-17. doi: 10.1258/ebm.2011.011229
» https://doi.org/10.1258/ebm.2011.011229 -
15Dos RK and Zouani OF (2014) A review of the effects of the cell environment physicochemical nanoarchitecture on stem cell commitment. Biomaterials 35: 5278-5293. doi:10.1016/j.biomaterials.2014.03.044
» https://doi.org/10.1016/j.biomaterials.2014.03.044 -
16Engel E, Michiardi A, Navarro M, Lacroix D, Planell JA (2008) Nanotechnology in regenerative medicine: The materials side. Trends Biotechnol 26:39-47. doi:10.1016/j.tibtech.2007.10.005
» https://doi.org/10.1016/j.tibtech.2007.10.005 -
17Fiorito S, Serafino A, Andreola F, Bernier P (2006) Effects of fullerenes and single-wall carbon nanotubes on murine and human macrophages. Carbon 44:1100-1105.
-
18Ge Z, Li C, Heng BC, Cao G, Yang Z (2012) Functional biomaterials for cartilage regeneration. J Biomed Mater Res A 100:2526-2536. doi: 10.1002/jbm.a.34147
» https://doi.org/10.1002/jbm.a.34147 -
19Gilbert TW, Sellaro TL, Badylak SF (2006) Decellularization of tissues and organs. Biomaterials 27:3675-3683. doi:10.1016/j.biomaterials.2006.02.014
» https://doi.org/10.1016/j.biomaterials.2006.02.014 -
20Harrison BS and Atala A (2007) Carbon nanotube applications for tissue engineering. Biomaterials 28:344-353. doi:10.1016/j.biomaterials.2006.07.044
» https://doi.org/10.1016/j.biomaterials.2006.07.044 -
21Hashemzadeh MR, Mahdavi-Shahri N, Bahrami AR, Kheirabadi M, Naseri F, Atighi M (2015) Use of an in vitro model in tissue engineering to study wound repair and differentiation of blastema tissue from rabbit pinna. In Vitro Cell Dev Biol Animal 51:680-689. doi10.1007/s11626-015-9868-0
» https://doi.org/10.1007/s11626-015-9868-0 -
22Holmes B, Fang X, Zarate A, Keidar M, Zhang LG (2014) Enhanced Human Bone Marrow Mesenchymal Stem Cell Chondrogenic Differentiation in Electrospun Constructs with Carbon Nanomaterials. Carbon 97:1-13. doi:10.1016/j.carbon.2014.12.035
» https://doi.org/10.1016/j.carbon.2014.12.035 -
23Hopley EL, Salmasi S, Kalaskar DM, Seifalian AM (2014) Carbon nanotubes leading the way forward in new generation 3D tissue engineering. Biotechnology Advances 32 : 1000-1014. doi:10.1016/j.biotechadv.2014.05.003
» https://doi.org/10.1016/j.biotechadv.2014.05.003 -
24Huczko A. & Lange H (2001) Carbon nanotubes: experimental evidence for a null risk of skin irritation and allergy. Fullerene Science and Technology 9: 247-250. doi: 10.1081/FST-100102972
» https://doi.org/10.1081/FST-100102972 -
25Izadifar Z, Chen X, Kulyk W (2012) Strategic Design and Fabrication of Engineered Scaffolds for Articular Cartilage Repair. J Funct Biomater 3:799-838. doi:10.3390/jfb3040799
» https://doi.org/10.3390/jfb3040799 -
26Jagtoyen M, Pardue J, Rantell T, Derbyshire F (2000) Porosity of carbon nanotubes. Adsorpt Sci Technol 17:289-293. doi: 10.1142/9789812793331_0058
» https://doi.org/10.1142/9789812793331_0058 -
27Jeon JE, Vaquette C, Klein TJ, Hutmacher DW (2013) Perspectives in Multiphasic Osteochondral Tissue Engineering. The Anatomical Record 297:26-35. doi: 10.1002/ar.22795
» https://doi.org/10.1002/ar.22795 -
28Lee JH, Shim W, Choolakadavil Khalid N, Kang WS, Lee M, Kim HS, Choi J, Lee G, Kim JH (2015) Random Networks of Single-Walled Carbon Nanotubes Promote Mesenchymal Stem Cell’s Proliferation and Differentiation. ACS Appl Mater Interfaces 7:1560−1567. doi: 10.1021/am506833q
» https://doi.org/10.1021/am506833q -
29Li S, Sengupta D, Chien S (2014) Vascular tissue engineering: from in vitro to in situ. WIREs Syst Biol Med 6:61-76. doi: 10.1002/wsbm.1246
» https://doi.org/10.1002/wsbm.1246 -
30Lin C, Wang Y, Lai Y, Yang W, Jiao F, Zhang H, Ye S, Zhang Q (2011) Incorporation of carboxylation multiwalled carbon nanotubes into biodegradable poly(lactic-co-glycolic acid)for bone tissue engineering. Colloids Surf B Biointerfaces 83:367-375. doi:10.1016/j.colsurfb.2010.12.011
» https://doi.org/10.1016/j.colsurfb.2010.12.011 -
31Meng J, Kong H, Han Z, Wang C, Zhu G, Xie S, Xu H (2009) Enhancement of nanofibrous scaffold of multiwalled carbon nanotubes/polyurethane composite to the fibroblasts growth and biosynthesis. J Biomed Mater Res 88:105-16. doi: 10.1002/jbm.a.31862
» https://doi.org/10.1002/jbm.a.31862 -
32Moradi A, Pramanik S, Ataollahi F, Kamarul T, Pingguan-murphy B ( 2014) Archimedes revisited: computer assisted microvolumetric modification of liquid displacement method for porosity measurement of highly porous light materials. Anal Methods 6:4396-4401. doi: 10.1039/C4AY00666F
» https://doi.org/10.1039/C4AY00666F -
33Nagai H, Okazaki Y, Chew SH, Misawa N, Miyata Y, Shinohara H, Toyokuni S (2013) Intraperitoneal administration of tangled multiwalled carbon nanotubes of 15 nm in diameter does not induce mesothelial carcinogenesis in rats. Pathol Int 63:457-462. doi: 10.1111/pin.12093
» https://doi.org/10.1111/pin.12093 -
34Nesic D, Whiteside R, Brittberg M, Wendt D, Martin I, Mainil-Varlet P (2006) Cartilage tissue engineering for degenerative joint disease. Adv Drug Deliv Rev 58:300-322. doi:10.1016/j.addr.2006.01.012
» https://doi.org/10.1016/j.addr.2006.01.012 -
35Ng R, Zang R, Yang KK, Liu N, Yang ST (2012) Three-dimensional fibrous scaffolds with microstructures and nanotextures for tissue engineering. RSC Advances 2:10110-10124. doi: 10.1039/C2RA21085A
» https://doi.org/10.1039/C2RA21085A -
36Oliveira MB, Ribeiro MP, Miguel SP, Neto AI, Coutinho P, Correia IJ, Mano JF (2014) In vivo high-content evaluation of three-dimensional scaffolds biocompatibility. Tissue Eng Part C Methods 20: 851-864. doi:10.1089/ten.tec.2013.0738
» https://doi.org/10.1089/ten.tec.2013.0738 -
37Oliveira SM, Reis RL, Mano JF (2015) Towards the Design of 3D Multiscale Instructive Tissue Engineering Constructs: current approaches and trends. Biotechnology Advances 33:842-855. doi:10.1016/j.biotechadv.2015.05.007
» https://doi.org/10.1016/j.biotechadv.2015.05.007 -
38Park S and Im G (2014) Stem cell responses to nanotopography. Journal of Biomedical Materials Research 103: 1238-1245. doi: 10.1002/jbm.a.35236
» https://doi.org/10.1002/jbm.a.35236 -
39Peran M, Garcia MA, Lopez-Ruiz E, Bustamate M, Jimenez G, Madeddu R, Marchal JA (2012) Functionalized nanostructures with application in regenerative medicine, Int J Mol Sci 13:3847-3886. doi:10.3390/ijms13033847
» https://doi.org/10.3390/ijms13033847 -
40Pina S, Oliveira JM, Reis RL (2015) Natural-Based Nanocomposites for Bone Tissue Engineering and Regenerative Medicine: A Review. Advanced Materials 27: 1143-1169. doi:10.1002/adma.201403354
» https://doi.org/10.1002/adma.201403354 -
41Place E S, Evans ND, Stevens MM (2009) Complexity in biomaterials for tissue engineering. Nat Mater 8:457-470. doi: 10.1038/nmat2441
» https://doi.org/10.1038/nmat2441 -
42Rose S, Prevoteau A, Elzière P, Hourdet D, Marcellan A, Leibler L (2013) Nanoparticle solutions as adhesives for gels and biological tissues. Nature 505:382-5. doi: 10.1038/nature12806
» https://doi.org/10.1038/nature12806 -
43Ross AM, Jiang Z, Bastmeyer M, Lahann J (2012) Physical aspects of cell culture substrates:topography, roughness, and elasticity. Small 8:336-355. doi: 10.1002/smll.201100934
» https://doi.org/10.1002/smll.201100934 -
44Schmitz N, Laverty S, Kraus VB, Aigner T (2010) Basic methods in histopathology of joint tissues. Osteoarthritis and Cartilage 18:S113-S116. doi:10.1016/j.joca.2010.05.026
» https://doi.org/10.1016/j.joca.2010.05.026 -
45Shahabipour F, Mahdavi- Shahri N, Matin MM, Tavassoli A, Zebarjad SM (2013) Scaffolds derived from cancellous bovine bone support mesenchymal stem cells' maintenance and growth. In Vitro Cell Dev Biol Anim 49:440-448. doi:10.1007/s11626-013-9591-7
» https://doi.org/10.1007/s11626-013-9591-7 -
46Sutherland AJ, Beck EC, Dennis SC, Converse GL, Hopkins RA, Berkland CJ, Detamore MS (2015) Decellularized cartilage may be a chondroinductive material for osteochondral tissue engineering. PLoS One 10:e0121966. doi: 10.1371/journal.pone.0121966
» https://doi.org/10.1371/journal.pone.0121966 -
47Shi Kam NW, Jessop TC, Wender PA, Dai H (2004) Nanotube molecular transporters: internalization of carbon nanotube-protein conjugates into mammalian cells. J Am Chem Soc 126:6850-6851. doi: 10.1021/ja0486059
» https://doi.org/10.1021/ja0486059 -
48Simkin J, Sammarco MC, Dawson LA, Schanes PP, Yu L, Muneoka K (2015) The mammalian blastema: regeneration at our fingertips. Regeneration 2:93-105
-
49Sundaramurthi D, Krishnan UM, Sethuraman S (2014) Electrospun Nanofibers as Scaffolds for Skin Tissue Engineering. Polymer Reviews 54: 348-376. doi: 10.1080/15583724.2014.881374
» https://doi.org/10.1080/15583724.2014.881374 -
50Tavassoli A, Mahdavi-Shahri N, Matin MM, Fereidoni M, Shahabipour F (2012) Bovine articular cartilage decellularized matrix as a scaffold for use in cartilage tissue engineering. Iranian Journal of Veterinary Science and Technology 4:1-8
-
51Toyokuni S, Jiang LI, Kitaura R, Shinohara H (2015) Minimal inflammogenicity of pristine single-wall carbon nanotubes. Nagoya J Med Sci 77:195-202.
-
52Trzeciak T, Rybka JD, Akinoglu EM, Richter M, Kaczmarczyk J, Giersig Michael (2016) In Vitro Evaluation of Carbon Nanotube-Based Scaffolds for Cartilage Tissue Engineering. J Nanosci Nanotechnol 16:9022-9025. doi: http://dx.doi.org/10.1166/jnn.2016.12733
» https://doi.org/10.1166/jnn.2016.12733 -
53Veetil JV and Ye k (2009) Tailored carbon nanotubes for tissue engineering application. Biotechnol Prog 25:709-721. doi: 10.1002/btpr.165
» https://doi.org/10.1002/btpr.165 -
54Vinatier C, Mrugala D, Jorgensen C, Guicheux J, Noel D (2009) Cartilage engineering: a crucial combination of cells, biomaterials and biofactors. Trends Biotechnol 27:307-314. doi:10.1016/j.tibtech.2009.02.005
» https://doi.org/10.1016/j.tibtech.2009.02.005 -
55Wen CY, Wu CB, Tang B, Wang T, Yan CH, Lu WW, Pan H, Hu Y, Chiu KY (2012) Collagen fibril stiffening in osteoarthritic cartilage of human beings revealed by atomic force microscopy. Osteoarthritis and Cartilage 20:916-922. doi:10.1016/j.joca.2012.04.018
» https://doi.org/10.1016/j.joca.2012.04.018 -
56Wiegand C, Abel M, Ruth P, Wilhelms T, Schulze D, Norgauer J, Hipler UC (2009) Effect of the sterilization method on the performance of collagen type I on chronic wound parameters in vitro. J Biomed Mater Res B Appl Biomater 90: 710-719. doi: 10.1002/jbm.b.31338
» https://doi.org/10.1002/jbm.b.31338 -
57Ye K, Felimban R, Moulton S, Wallance GG, Bella CD, Traianedes K, Choong PF, Myers DE (2013) Bioengineering of articular cartilage: past, present and future. Regen Med 8:333-349. doi:10.2217/rme.13.28
» https://doi.org/10.2217/rme.13.28 -
58Yim EK, Pang SW, Leong KW (2007) Synthetic nanostructures inducing differentiation of human mesenchymal stem cells into neuronal lineage. Exp Cell Res 313:1820-1829. doi:10.1016/j.yexcr.2007.02.031
» https://doi.org/10.1016/j.yexcr.2007.02.031 -
59Zeinabad HA, Zarrabian A, Saboury AA, Alizadeh AM, Falahati M (2016) Interaction of single and multi wall carbon nanotubes with the biological systems: tau protein and PC12 cells as targets. Sci Rep 6:26508. doi: 10.1038/srep26508
» https://doi.org/10.1038/srep26508 -
60Zelenski N A, Leddy H A, Sanchez-Adams J, Zhang J, Bonaldo P, Liedtke W, Guilak F (2015) Type VI Collagen Regulates Pericellular Matrix Properties, Chondrocyte Swelling, and Mechanotransduction in Mouse Articular Cartilage. Arthritis & Rheumatology 67: 1286-1294. doi: 10.1002/art.39034
» https://doi.org/10.1002/art.39034 -
61Zhang D, Yi C, Qi S, Yao X, Yang M (2010) Effects of carbon nanotubes on the proliferation and differentiation of primary osteoblasts. Methods Mol Biol 625:41-53. doi: 10.1007/978-1-60761-579-8_5
» https://doi.org/10.1007/978-1-60761-579-8_5 -
62Zhang X, Wang X, Lu Q, Fu C (2008) Influence of carbon nanotube scaffolds on human cervical carcinoma HeLa cell viability and focal adhesion kinase expression. Carbon 46:453-60. doi:10.1016/j.carbon.2007.12.015
» https://doi.org/10.1016/j.carbon.2007.12.015 -
63Zhang Y, Bai Y, Yan B (2010) Functionalized carbon nanotubes for potential medicinal applications. Drug Discovery Today 15:428-435. doi:10.1016/j.drudis.2010.04.005
» https://doi.org/10.1016/j.drudis.2010.04.005 -
64Zhao C, Tan A, Pastorin G, Ho KH (2013) Nanomaterial scaffolds for stem cell proliferation and differentiation in tissue engineering. Biotechnology Advances 31:654-668. doi:10.1016/j.biotechadv.2012.08.001
» https://doi.org/10.1016/j.biotechadv.2012.08.001
Publication Dates
-
Publication in this collection
2017
History
-
Received
03 Feb 2016 -
Accepted
14 July 2016