Abstracts
Hydroxyapatite (HA) has been used in orthopedic, dental, and maxillofacial surgery as a bone substitute. The aim of this investigation was to study the effect of surface topography produced by the presence of microporosity on cell response, evaluating: cell attachment, cell morphology, cell proliferation, total protein content, and alkaline phosphatase (ALP) activity. HA discs with different percentages of microporosity (< 5%, 15%, and 30%) were confected by means of the combination of uniaxial powder pressing and different sintering conditions. ROS17/2.8 cells were cultured on HA discs. For the evaluation of attachment, cells were cultured for two hours. Cell morphology was evaluated after seven days. After seven and fourteen days, cell proliferation, total protein content, and ALP activity were measured. Data were compared by means of ANOVA and Duncan’s multiple range test, when appropriate. Cell attachment (p = 0.11) and total protein content (p = 0.31) were not affected by surface topography. Proliferation after 7 and 14 days (p = 0.0007 and p = 0.003, respectively), and ALP activity (p = 0.0007) were both significantly decreased by the most irregular surface (HA30). These results suggest that initial cell events were not affected by surface topography, while surfaces with more regular topography, as those present in HA with 15% or less of microporosity, favored intermediary and final events such as cell proliferation and ALP activity.
Hydroxyapatite; Porosity; Surface topography; ROS17/2.8 cells
A hidroxiapatita (HA) tem sido utilizada como revestimento de implantes e para substituição de tecido ósseo. O objetivo deste estudo foi avaliar o efeito da topografia de superfície da HA, resultante da presença de microporosidade, sobre a adesão, a morfologia e proliferação celulares, a medida de proteína total e a atividade de fosfatase alcalina. Discos de HA com diferentes porcentagens de microporosidade (< 5%, 15% e 30%) foram fabricados por uma combinação das técnicas de pressão uniaxial e sinterização. Células ROS17/2.8 foram cultivadas sobre os discos de HA. Para a adesão, as células foram cultivadas por duas horas. A morfologia foi avaliada após sete dias. A proliferação, medida de proteína total e atividade de ALP foram avaliadas após sete e quatorze dias. Os dados foram comparados por ANOVA e teste de Duncan quando apropriado. A adesão (p = 0,11) e a medida de proteína total (p = 0,31) não foram afetadas pela topografia de superfície. A proliferação após sete e quatorze dias (p = 0,0007 e p = 0,003, respectivamente), e a atividade de ALP (p = 0,0007) foram significantemente menores na superfície irregular (HA30). Esses resultados sugerem que eventos iniciais não são afetados pela topografia, enquanto superfícies com topografias mais regulares (microporosidade de 15% ou menos) favoreceram eventos intermediários e finais, como proliferação e atividade de ALP.
Hidroxiapatita; Porosidade; Topografia de superfície; Células ROS17/2.8
Implantodontia
Surface topography of hydroxyapatite affects ROS17/2.8 cells response
A topografia de superfície da hidroxiapatita afeta a resposta de células ROS17/2.8
Adalberto Luiz Rosa* * Department of Surgery, Maxillofacial Traumatology and Periodontology, School of Dentistry of Ribeirão Preto, University of São Paulo.
Márcio Mateus Beloti* * Department of Surgery, Maxillofacial Traumatology and Periodontology, School of Dentistry of Ribeirão Preto, University of São Paulo.
Richard van Noort** * Department of Surgery, Maxillofacial Traumatology and Periodontology, School of Dentistry of Ribeirão Preto, University of São Paulo.
Paul Vincent Hatton** * Department of Surgery, Maxillofacial Traumatology and Periodontology, School of Dentistry of Ribeirão Preto, University of São Paulo.
Anne Jane Devlin** * Department of Surgery, Maxillofacial Traumatology and Periodontology, School of Dentistry of Ribeirão Preto, University of São Paulo.
ABSTRACT: Hydroxyapatite (HA) has been used in orthopedic, dental, and maxillofacial surgery as a bone substitute. The aim of this investigation was to study the effect of surface topography produced by the presence of microporosity on cell response, evaluating: cell attachment, cell morphology, cell proliferation, total protein content, and alkaline phosphatase (ALP) activity. HA discs with different percentages of microporosity (< 5%, 15%, and 30%) were confected by means of the combination of uniaxial powder pressing and different sintering conditions. ROS17/2.8 cells were cultured on HA discs. For the evaluation of attachment, cells were cultured for two hours. Cell morphology was evaluated after seven days. After seven and fourteen days, cell proliferation, total protein content, and ALP activity were measured. Data were compared by means of ANOVA and Duncans multiple range test, when appropriate. Cell attachment (p = 0.11) and total protein content (p = 0.31) were not affected by surface topography. Proliferation after 7 and 14 days (p = 0.0007 and p = 0.003, respectively), and ALP activity (p = 0.0007) were both significantly decreased by the most irregular surface (HA30). These results suggest that initial cell events were not affected by surface topography, while surfaces with more regular topography, as those present in HA with 15% or less of microporosity, favored intermediary and final events such as cell proliferation and ALP activity.
UNITERMS: Hydroxyapatite; Porosity; Surface topography; ROS17/2.8 cells.
RESUMO: A hidroxiapatita (HA) tem sido utilizada como revestimento de implantes e para substituição de tecido ósseo. O objetivo deste estudo foi avaliar o efeito da topografia de superfície da HA, resultante da presença de microporosidade, sobre a adesão, a morfologia e proliferação celulares, a medida de proteína total e a atividade de fosfatase alcalina. Discos de HA com diferentes porcentagens de microporosidade (< 5%, 15% e 30%) foram fabricados por uma combinação das técnicas de pressão uniaxial e sinterização. Células ROS17/2.8 foram cultivadas sobre os discos de HA. Para a adesão, as células foram cultivadas por duas horas. A morfologia foi avaliada após sete dias. A proliferação, medida de proteína total e atividade de ALP foram avaliadas após sete e quatorze dias. Os dados foram comparados por ANOVA e teste de Duncan quando apropriado. A adesão (p = 0,11) e a medida de proteína total (p = 0,31) não foram afetadas pela topografia de superfície. A proliferação após sete e quatorze dias (p = 0,0007 e p = 0,003, respectivamente), e a atividade de ALP (p = 0,0007) foram significantemente menores na superfície irregular (HA30). Esses resultados sugerem que eventos iniciais não são afetados pela topografia, enquanto superfícies com topografias mais regulares (microporosidade de 15% ou menos) favoreceram eventos intermediários e finais, como proliferação e atividade de ALP.
UNITERMOS: Hidroxiapatita; Porosidade; Topografia de superfície; Células ROS17/2.8.
INTRODUCTION
Hydroxyapatite (HA) has received considerable attention as a material for implants and bone augmentation procedures, since it bonds chemically directly to bone after implantation6. Synthetic HA for bone substitution can be produced by a combination of a powder pressing technique and sintering. Powder pressing techniques allow the obtainment of "green bodies", which are then sintered at high temperatures to produce the fusion of powder particles. Changes in the powder compaction and sintering conditions can result in a variety of chemical and porosity characteristics of HA16,20. Porosity can be classified, according to the diameter of pores, in macroporosity (> 10 mm) and microporosity (< 10 mm). It has been suggested that these structural modifications of HA influence cellular activity16,20.
Dziedzic et al.8 (1996) reported that HA samples with different microporosities result in different bone-HA interfaces after implantation in rats. Preliminary results from in vitro studies using a primary culture of rat bone marrow cells and a rat osteosarcoma cell line have demonstrated that an increase in micropore percentage negatively influences HA biocompatibility18,19.
Variations of micropore percentage result in different surface topographies3,6 and authors have agreed that topography must be considered in terms of amplitude and organization13. Anselme et al.1 (1997) observed a lower proliferation of human osteoblasts cultured on less organized Ti surfaces.
Considering that the surface characteristics of HA necessary for optimal osseointegration are still not fully understood, the aim of this study was to carry out an in vitro evaluation of the effect of varying the surface topography of HA on the response of osteoblast-like cells. For this, HA samples with less than 5%, 15%, and 30% of microporosity were evaluated using osteoblast-like cells derived from rat osteosarcoma (ROS17/2.8). These cells were used because they express numerous osteoblastic characteristics, including high levels of alkaline phosphatase (ALP) activity, synthesis of type I collagen, and osteocalcin10. The following parameters were evaluated: cell attachment, cell morphology, cell proliferation, total protein content, and ALP activity.
MATERIAL AND METHODS
Preparation of HA samples
HA discs (with the diameter of 12 mm and height of 2 mm) with different degrees of microporosity (< 10 mm), as shown in Table 1, were confected by means of a combination of uniaxial powder pressing and different sintering conditions. This procedure allows the confection of HA with identical chemical composition and crystallinity but with different porosities20,21. The discs were made of a high purity and crystalline HA powder (Plasma Biotal, Tideswell, UK). The HA discs utilized in this study were divided in three groups: HA5, with less than 5% of microporosity, HA15, with 15% of microporosity, and HA30, with 30% of microporosity, as calculated from the actual density compared with the theoretical density of HA. The HA surface was evaluated by scanning electron microscopy (SEM - JSM-5410, Jeol, MA, USA). Surface roughness (Ra) was measured using a surface profilometer (Prazis Rug-03, São Paulo, Brazil), and an average was calculated for each disc, taking into account three measurements. The average Ra of each microporosity group was determined based on measurements of three discs (n = 3). All discs were cleaned in an ultrasonic bath and autoclaved before utilization in the cell culture experiments.
Cell culture
ROS17/2.8 cells were cultured in HAMS F12 (Gibco - Life Technologies, Grand Island, NY, USA) supplemented with 8% fetal bovine serum, 50 mg/ml penicillin-streptomycin (Gibco), glutamine 5.4 ´ 107 M (Gibco), and 0.3 mg/ml amphotericin B (Gibco). Cells were cultured at a concentration of 0.825 ´ 104 cells/well on HA discs in 24-well culture plates (Falcon, Franklin Lakes, NJ, USA). During the culture period, cells were incubated at 37ºC in a humidified atmosphere of 5% CO2 and 95% air, and the medium was changed every forty-eight hours. In each plate, empty wells were only used as a control of culture conditions.
Cell attachment
For the evaluation of attachment, cells were cultured for two hours on HA discs (n = 5, for each microporosity). The culture medium was removed, and the wells were washed three times with PBS at 37ºC to eliminate unattached cells. The adherent cells were then enzymatically released (1 mM EDTA + 0.25% trypsin - Gibco) from the HA discs, and counted using a hemacytometer. Cell attachment was expressed as a percentage of the initial number of cells.
Cell morphology
For the evaluation of morphology, cells were cultured for seven days on HA discs and processed for SEM. First, cells were fixed with 1.5% glutaraldehyde (Sigma, St. Louis, MO, USA) buffered in 0.1 M sodium cacodylate (Sigma), and stained in 1% osmium tetroxide (Sigma). Then, cells were dehydrated through a graded series of alcohol, followed by critical point drying at 32ºC and 1,300 psi for one hour. After that, samples were sputter-coated with gold (10 nm) before examination under a SEM.
Cell proliferation
For the evaluation of proliferation, cells were cultured for seven and fourteen days on HA discs (n = 5 for each day and each microporosity). The cells were released and counted as described in cell attachment.
Total protein content
Total protein content after seven and fourteen days in culture on HA discs (n = 5 for each microporosity) was calculated according to the modified Lowry et al.14 (1951) method. The culture medium was removed, the wells were washed three times with PBS at 37ºC and were filled with 2 ml of 0.1% sodium lauryl sulfate (Sigma). After thirty minutes, 1 ml of the solution from each well was mixed with 1 ml of Lowry solution (Sigma) and left for twenty minutes at room temperature. After this period, it was added to 0.5 ml of the solution of phenol reagent of Folin and Ciocalteau (Sigma). It stood for thirty minutes at room temperature to allow color development, and absorbance was then spectrophotometrically measured (CE3021 - Cecil, Cambridge, UK) at 680 nm. The total protein content was calculated from a standard curve of bovine albumin and expressed as µg/ml. These data were normalized by the number of cells counted after seven and fourteen days, respectively.
ALP activity
ALP activity was assayed as the release of r-nitrophenol from r-nitrophenyl, using a commercial kit (Sigma), and the specific activity was calculated. From the same solutions used for calculating total protein content, 0.1 ml aliquots were assayed for measuring ALP activity. Absorbance was spectrophotometrically measured at 410 nm, and ALP activity was calculated from a standard curve obtained from different solutions of r-nitrophenol and sodium hydroxide 0.02 N. Results were calculated as µmol r-nitrophenol/hour, and data were expressed as ALP activity normalized by the total protein content and by the number of cells counted after seven and fourteen days, respectively.
Statistical analysis
Data were submitted to analysis of variance (ANOVA) and Duncans multiple range test, when appropriate. Differences at p £ 0.05 were considered statistically significant.
RESULTS
HA samples
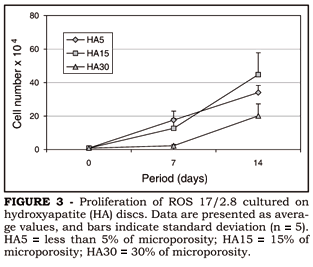
Ra measurements presented the following results: HA5 = 0.38 µm, HA15 = 0.48 µm and HA30 = 0.49 µm. ANOVA revealed that Ra was not affected by the manufacturing conditions of HA (F = 2.79; gl = 2; p = 0.14). It was possible to observe, from SEM micrographs, that the amount of microporosity produced surfaces with different topographies (Figure 1). Thus, HA5 and HA15 presented more regular surfaces while HA30 presented a very irregular surface topography (Figure 1).
Cell attachment
Cell attachment was not affected by HA surface topography (ANOVA: gl = 2; F = 2.65; p = 0.11). Data are presented in Table 2.
Cell morphology
HA5 e HA15 presented more cells than HA30. Cells observed on HA5 and HA15 presented a polygonal morphology with dorsal ruffles and a large number of filaments connecting distant cells (Figures 2A and 2B). On HA30 discs, cells did not cover all surfaces. In addition, the formation of a monolayer was observed, with cells predominantly presenting a more flattened morphology, showing almost no dorsal ruffles and filaments (Figure 2C).
Cell proliferation
The number of cells was affected by surface topography after both seven and fourteen days (ANOVA: gl = 2; F = 14.31; p = 0.0007 e gl = 2; F = 9.61; p = 0.0032, respectively) in the following order: HA5 = HA15 > HA30. Data are presented in Figure 3.
Total protein content
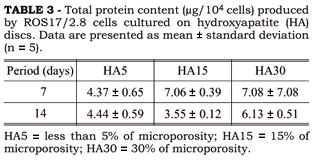
Total protein content after seven and fourteen days was not affected by both surface topography (ANOVA: gl = 2; F = 1.21; p = 0.31) and period of culture (ANOVA: gl = 1; F = 1.58; p = 0.22). Data are presented in Table 3.
ALP activity
ALP activity after seven and fourteen days was affected by surface topography (ANOVA: gl = 2; F = 9.69; p = 0.0007) in the following order: HA5 = HA15 > HA30. However, it was not affected by the period of culture (ANOVA: gl = 1; F = 0.21; p = 0.65). Data are presented in Table 4.
DISCUSSION
Early in vitro studies on the interaction between osteoblasts and biomaterials were essentially concerned with the effect of diverse range materials on cell response, without paying attention to the influence of surface characteristics23. However, it is now understood that the superficial properties of biomaterials play a critical role in the establishment of cell-biomaterial interfaces and that manufacturing conditions affect the superficial properties of HA1,20.
The present results revealed that different combinations of powder pressing and sintering produced HA samples with different microporosities and, consequently, different superficial appearances. Despite these differences, Ra, a factor known to influence cell response, was not significantly affected by the manufacturing process of the HA samples. Hence, any differences in cell response observed in this study would be a consequence of differences in the microporosity of HA samples.
Osteogenesis, induced by osteoblastic cells, is characterized by a sequence of events, involving cell attachment and cell proliferation, followed by the expression of osteoblastic phenotype6 . This study evaluated the response of ROS 17/2.8 osteoblast-like cells, originated from rat osteosarcoma, cultured on HA discs with different surface topographies. The present results revealed that all HA samples, regardless of surface topography, are biocompatible, considering that they allowed cell attachment, proliferation, and expression of markers of osteoblastic differentiation.
The effect of surface characteristics on cells may be the result of the surface characteristics themselves or the result of the reaction that occurs as the surface is conditioned by the medium and serum15. This initial interaction produces a layer of macromolecules that modify cell response. Fibronectin, a cell adhesion protein present in serum, has been shown to mediate cell attachment and spread on artificial substrates by interacting with glycosaminoglycans and with the cytoskeleton7. This study did not show any effect of surface topography on cell attachment. While it may occur, 2 hours which was the period used in this evaluation is a very short time to observe the development of a difference in protein adhesion that could affect cell attachment. Besides the period of incubation, it is also possible that cell attachment in tissue culture is not seriously influenced by any characteristic of the material, since it has been shown that materials with different degrees of biocompatibility produce little or no differences in cell attachment17.
In the present study, the amount of protein was normalized by the number of cells. Hence, these results evaluated the cell secretory activities that were not affected by surface topography. Similarly, Castellani et al.4 (1999) did not find significant differences in the production of protein by rat bone marrow cells cultured on titanium with different surface topographies. Thus, it is possible that the total protein production, regardless of the chemical composition of the biomaterial, is not affected by surface topography.
Cell proliferation and ALP activity were affected by surface topography. HA samples with more regular surfaces such as HA5 e HA15 allowed greater cell proliferation and ALP activity than HA samples with irregular surface (HA30). These results suggest that more regular surfaces would facilitate cell growth and the expression of osteoblast phenotype markers. Cell morphology confirmed these findings, since cells cultured on more regular surfaces presented a polygonal morphology with dorsal ruffles and a large number of filaments. This morphology has been described as osteoblast feature22. On the contrary, cells cultured on irregular surfaces presented a flattened morphology showing almost no dorsal ruffles and filopodia. Other studies pointed out low cell proliferation and ALP activity on less organized surfaces1,6, showing that the regularity and uniformity of the HA surface are important factors that affect cell proliferation and differentiation2,12. Therefore, differences in surface topography result in different degrees of HA biocompatibility, which confirms our previous results that revealed that HA30 presents, comparatively, low biocompatibility18,19.
An explanation for these findings is that the physicochemical properties of the ceramic surface can affect the reorganization of proteins on ceramic specimens and change the profile of adsorbed proteins8. The type of substrate determines which integrins and extracellular matrix proteins are expressed by osteoblasts, providing information about how implant materials may affect osteoblastic differentiation23. The differences in protein adsorption can result in very different initial cell responses23, and it is known that some proteins, such as fibronectin, have an important role in osteoblastic differentiation11. It is possible that organized surfaces favor the selective adsorption of proteins responsible for osteoblastic differentiation in serum. As a result, biomaterials that present more regular surfaces are more biocompatible.
CONCLUSIONS
-
Varied manufacturing conditions produced HA samples with different microporosities. Surface roughness was not affected by the presence of micropores. However, surfaces with smaller percentages of microporosity presented more regular surface topography.
-
All HA samples, regardless of surface topography, are biocompatible. However, samples with more regular surface topography were more biocompatible, which was evidenced by the greater cell proliferation and ALP activity observed on their surfaces, when compared with samples with irregular surface topography.
ACKNOWLEDGMENTS
This work was supported by the State of São Paulo Research Foundation (FAPESP), Grant no. 98/06851-0.
Recebido para publicação em 10/12/01
Enviado para reformulação em 06/06/02
Aceito para publicação em 25/06/02
** Centre for Biomaterials and Tissue Engineering, University of Sheffield, England.
-
11. Anselme K, Bigerrelle M, Noel B, Dufresne E, Judas D, Iost A, Hardouin P. Qualitative and quantitative study of human osteoblast adhesion on materials with various surface roughnesses. J Bimed Mater Res 1997;49(2):155-66.
-
22. Bowers K, Keller JC, Randolph B, Wick D, Michaels C. Optimization of surface micromorphology for enhanced osteoblast responses in vitro. Int J Oral Maxillofac Impl 1992;7(3):302-10.
-
33. Boyan DB, Hummet TW, Dean DD, Schwartz Z. Role of material surfaces in regulating bone and cartilage cell response. Biomaterials 1996;17(2):137-46.
-
44. Castellani R, De Ruijter JE, Renggli H, Jansen JA. Response of rat bone marrow cells to differently roughened titanium discs. Clin Oral Impl Res 1999;10(5):369-78.
-
55. Curtis ASG, Wilkinson CDW. Reactions of cells to topography. J Biomater Sci Polymer Edn 1998;12(9):1313-29.
-
66. Deligianni DD, Katsala ND, Koutsoukos PG, Missirlis YF. Effect of surface roughness of hydroxyapatite on human bone marrow cell adhesion, proliferation, differentiation and detachment strength. Biomaterials 2001;22(1):87-96.
-
77. Doillon CJ, Silver FH, Berg RA. Fibroblast growth on a porous collagen sponge containing hyaluronic acid and fibronectin. Biomaterials 1987;8(3):195-200.
-
88. Dziedzic DM, Sawa IH, Wilkinson DS, Davies JE. Osteoconduction on, and bonding to, calcium phosphate ceramic implants. Mater Res Soc Symp Proc 1996;414:147-56.
-
99. Frayssinet P, Rouquet N, Fages J, Durand M, Vidalain PO, Bonel G. The influence of sintering temperature on the proliferation of fibroblastic cells in contact with HA-bioceramics. J Biomed Mater Res 1997;35(3):337-47.
-
1010. Garcia AJ, Ducheyne P, Boettinger D. Effect of surface reaction stage on fibronectin-mediated adhesion of osteoblast-like cells to bioactive glass. J Biomed Mater Res 1998;40(1):48-56.
-
1111. Globus RK, Doty SB, Lull JC, Holmuhamedov E, Humphries MJ, Damsky CH. Fibronectin is a survival factor for differentiated osteoblasts. J Cell Sci 1998;111(pt10): 1385-93.
-
1212. Keller JC, Collins JG, Niederauer GG, McGee TD. In vitro attachment of osteoblast-like cells to osteoceramic materials. Dent Mater 1997;13(1):62-8.
-
1313. Lopez J, Hanseli G, Le Bosse JC, Mathia T. Caractérisation factale de la rugosité tridimensionnelle de surface. J Phys III France 1994;4:2201-19.
-
1414. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem 1951;193:265-75.
-
1515. Martin JY, Schawartz Z, Hummert TW, Schraub DM, Simpson J, Lankford JJr, et al. Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells (MG63). J Biomed Mater Res 1995;29(3):389-401.
-
1616. Ong JL, Hoppe CA, Cardenas HL, Cavin R, Carnes DL, Sogal A, et al. Osteoblast precursor cell activity on HA surfaces of different treatments. J Biomed Mater Res 1998;39(2):176-183.
-
1717. Puleo DA, Hollerman LA, Domerus RH, Bizios R. Osteoblast responses to orthopedic implant materials in vitro. J Biomed Mater Res 1991;25(6):11-23.
-
1818. Rosa AL, Hatton PV, Van Noort R. The influence of porosity on the in vitro biocompatibility of hydroxyapatite. J Dent Res 1997;76(5):1081.
-
1919. Rosa AL, Devlin JA, Hatton PV, Van Noort R. In vitro biocompatibility of microporous hydroxyapatite. J Dent Res 1999;78(5):409.
-
2020. Rosa AL, Shareef MY, Van Noort R. Efeito das condições de preparação e sinterização sobre a porosidade da hidroxiapatita. Braz Oral Res 2000;14(3):273-7.
-
2121. Shareef MY, Messer PF, Van Noort R. Fabrication, characterization and fracture study of a machinable hydroxyapatite ceramic. Biomaterials 1993;14(1):69-75.
-
2222. Vrouwenvelder WCH, Groot CG. Histological and biochemical evaluation of osteoblasts cultured on bioactive glass, hydroxyapatite, titanium alloy, and stainless steel. J Biomed Mater Res 1993;27(4):465-75.
-
2323. Zeng H, Chittur KK, Lacefield WR. Analysis of bovine serum albumin adsorption on calcium phosphate and titanium surfaces. Biomaterials 1999;20(4):377-84.
Publication Dates
-
Publication in this collection
16 Oct 2002 -
Date of issue
Sept 2002
History
-
Accepted
25 June 2002 -
Reviewed
06 June 2002 -
Received
10 Dec 2001