Abstracts
The diversity of bacterial isolates from soil in response to different plants (control, Brachiaria ruziziensis and Cajanus cajan), fertilization (control, simple superphosphate and rock phosphate) and liming (with and without lime) was evaluated. Phenotypic and physiological characteristics of the isolates were recorded and organized in a file to identify the bacteria. Among the isolates, 95% were Gram-positive and 5% Gram-negative rods. Soil cultivated with B. ruziziensis favored the nonsporing Gram-positive and Gram-negative rods compared to soils with C. cajan or uncultivated. Number of spore-forming Gram-positive rods were higher in plots with superphosphate than in unfertilized soil or soil fertilized with rock phosphate. In unfertilized plots, larger number of Gram-positive cocci and Gram-negative rods was obtained than in fertilized plots. Unlimed plots favored spore-forming Gram-positive rods, Gram-positive cocci and Gram-negative rods, while with liming a larger proportion of nonsporing Gram-positive rods was found. From 7 to 86% of the total isolates utilized different carbohydrates. The recording data used in this experiment was effective in the isolates identification, and might be useful for diagnosis of soil bacteria. Bacillus, Cellulomonas, Rhodococcus, Enterobacter, Flavobacterium, Micrococcus and Arthrobacter were the genera more commonly found. Bacterial diversity was enhanced in limed, unfertilized and plant cultivated plots.
bacteria; phosphate; Brachiaria ruziziensis; Cajanus cajan; liming
Este estudo avaliou a diversidade de bactérias isoladas do solo em resposta a diferentes plantas (controle, Brachiaria ruziziensis e Cajanus cajan), fertilizantes (controle, superfosfato simples e fosfato de rocha) e calagem (com e sem calcário). As características fenotípicas e fisiológicas dos isolados foram registradas e organizadas em um arquivo para identificar as bactérias. Entre os isolados, 95% foram de bactérias Gram positivas e 5% bacilos Gram negativos. B. ruziziensis favoreceu mais os bacilos Gram positivos não esporulados e bacilos Gram negativos que o solo com C. cajan ou não cultivado. O número de bacilos Gram positivos esporulados foi superior nas parcelas fertilizadas com fosfato do que nas não adubadas ou fertilizadas com fosfato de rocha. Nas parcelas não fertilizadas, maior número de cocos Gram positivos e bacilos Gram negativos foi obtido do que nas parcelas fertilizadas. A ausência de calagem favoreceu os bacilos Gram positivos esporulados, cocos Gram positivos e os bacilos Gram negativos, enquanto que, com calagem foram encontradas proporções maiores de bacilos Gram positivos não esporulados. De 7 a 86% do total de isolados utilizaram diferentes carboidratos. O registro dos dados utilizado neste experimento foi efetivo na identificação dos isolados e pode ser útil para o diagnóstico das bactérias do solo. Os gêneros mais freqüentes foram Bacillus, Cellulomonas, Rhodococcus, Enterobacter, Flavobacterium, Micrococcus e Arthrobacter. A diversidade bacteriana foi aumentada nas parcelas com calagem, não fertilizadas e cultivadas com plantas.
bactéria; fosfato; Brachiaria ruziziensis; Cajanus cajan; calagem
Bacterial diversity in soil in response to different plans, phosphate fertilizers and liming
Diversidade bacteriana do solo em resposta a diferentes plantas, fertilizantes fosfatados e calagem
Paulo da SilvaI; Ely NahasII
IInstituto Adolfo Lutz, Laboratório I, Ribeirão Preto, SP, Brasil
IIDepartamento de Produção Vegetal, Faculdade de Ciências Agrárias e Veterinárias, Universidade Estadual Paulista, Jaboticabal, SP, Brasil
Correspondence to Correspondence Departamento de Produção Vegetal, FCAV/UNESP Rodovia Prof. Paulo Donato Castellane, s/n. 14870-000, Jaboticabal, SP, Brasil E-mail: enahas@fcav.unesp.br
ABSTRACT
The diversity of bacterial isolates from soil in response to different plants (control, Brachiaria ruziziensis and Cajanus cajan), fertilization (control, simple superphosphate and rock phosphate) and liming (with and without lime) was evaluated. Phenotypic and physiological characteristics of the isolates were recorded and organized in a file to identify the bacteria. Among the isolates, 95% were Gram-positive and 5% Gram-negative rods. Soil cultivated with B. ruziziensis favored the nonsporing Gram-positive and Gram-negative rods compared to soils with C. cajan or uncultivated. Number of spore-forming Gram-positive rods were higher in plots with superphosphate than in unfertilized soil or soil fertilized with rock phosphate. In unfertilized plots, larger number of Gram-positive cocci and Gram-negative rods was obtained than in fertilized plots. Unlimed plots favored spore-forming Gram-positive rods, Gram-positive cocci and Gram-negative rods, while with liming a larger proportion of nonsporing Gram-positive rods was found. From 7 to 86% of the total isolates utilized different carbohydrates. The recording data used in this experiment was effective in the isolates identification, and might be useful for diagnosis of soil bacteria. Bacillus, Cellulomonas, Rhodococcus, Enterobacter, Flavobacterium, Micrococcus and Arthrobacter were the genera more commonly found. Bacterial diversity was enhanced in limed, unfertilized and plant cultivated plots.
Key words: bacteria, phosphate, Brachiaria ruziziensis, Cajanus cajan, liming
RESUMO
Este estudo avaliou a diversidade de bactérias isoladas do solo em resposta a diferentes plantas (controle, Brachiaria ruziziensis e Cajanus cajan), fertilizantes (controle, superfosfato simples e fosfato de rocha) e calagem (com e sem calcário). As características fenotípicas e fisiológicas dos isolados foram registradas e organizadas em um arquivo para identificar as bactérias. Entre os isolados, 95% foram de bactérias Gram positivas e 5% bacilos Gram negativos. B. ruziziensis favoreceu mais os bacilos Gram positivos não esporulados e bacilos Gram negativos que o solo com C. cajan ou não cultivado. O número de bacilos Gram positivos esporulados foi superior nas parcelas fertilizadas com fosfato do que nas não adubadas ou fertilizadas com fosfato de rocha. Nas parcelas não fertilizadas, maior número de cocos Gram positivos e bacilos Gram negativos foi obtido do que nas parcelas fertilizadas. A ausência de calagem favoreceu os bacilos Gram positivos esporulados, cocos Gram positivos e os bacilos Gram negativos, enquanto que, com calagem foram encontradas proporções maiores de bacilos Gram positivos não esporulados. De 7 a 86% do total de isolados utilizaram diferentes carboidratos. O registro dos dados utilizado neste experimento foi efetivo na identificação dos isolados e pode ser útil para o diagnóstico das bactérias do solo. Os gêneros mais freqüentes foram Bacillus, Cellulomonas, Rhodococcus, Enterobacter, Flavobacterium, Micrococcus e Arthrobacter. A diversidade bacteriana foi aumentada nas parcelas com calagem, não fertilizadas e cultivadas com plantas.
Palavras-chave: bactéria, fosfato, Brachiaria ruziziensis, Cajanus cajan, calagem
INTRODUCTION
Knowledge of factors that influence the composition of soil bacterial populations is important for the development of more rational methods to diagnose problems, improve crop productivity and develop bioremediation strategies (37). Among these factors, phosphorus is one of the most critical elements for plant nutrition, because it is not only a necessary nutrient itself, but it can also influence the absorption of other nutrients. As phosphorus is poorly available in many soils (27), plants and soil microorganisms compete for the limited quantities of this mineral through the processes of precipitation-solubilization and adsorption-desorption (35), and therefore, fertilizers must be applied to achieve acceptable crop production.
The effects of fertilizers on soil bacteria have been described in literature. Kanazawa et al. (17) observed a variation in the total number of bacteria and fungi isolated from soils treated with chemical or organic fertilizers. The proportion of Gram-negative bacteria in the soil was found to be 7%, and 20% in the rhizosphere (15). Hasebe et al. (13) observed a predominance of cocci over the rods. The addition of superphosphate decreased significantly the bacteria and fungi counts in soil cultivated with corn (28).
Lawley et al. (22) found that the total number of bacteria did not vary in the rhizosphere of several grasses, but the bacterial composition was affected by the crop species, which influenced the number of chromogenic species, endospore-forming bacteria and Pseudomonas spp. Similar results were obtained by Miller et al. (26), who showed a variation in the number of coryneform bacteria and Pseudomonas spp. Bacteria isolated from the soil and roots of fruit trees were identified as endospore-forming Gram-positive rods and Gram-negative rods belonging to the genera Agrobacterium, Pseudomonas, and Xanthomonas (40).
The composition of soils bacteria of tropical regions have been studied little and deserve more attention. Not only do they differ from soils of temperate regions, but the nature of their microbial populations may be influenced by environmental factors (12). The study of the biodiversity of the living microbial community from soils cultivated with different tropical crop species and fertilized with different phosphate sources may contribute to a better understanding of the dynamic of soil microbial communities.
MATERIALS AND METHODS
Soil Treatment
The study was conducted in a dark-red latosol as previously described (4), having been applied 400kg P2O5 ha-1 and, at the 30º day, 60kg N ha-1. The plants cultivated were the grass Brachiaria ruziziensis and the legume Cajanus cajan; the fertilizers studied were superphosphate and rock phosphate, both with or without liming. All treatments included plots without plants or fertilizers. Fifty well-isolated colonies were randomly chosen from each plot and streaked on potato dextrose yeast extract agar slants. To ensure culture purity, each isolate was re-streaked on nutrient agar plates then were transferred to nutrient agar slants.
Preliminary Tests
Colony morphology was determined after 2 to 4 days growth on nutrient agar plates incubated at 35-37ºC. Each isolate was submitted to Gram staining (12) and was examined for cellular morphology and arrangement. The isolates were also examined for spore formation according to the Wirtz-Conklin method (2). In addition, the strains were streaked on nutrient agar with 50 µg/ml crystal violet (15) and submitted to the fast method using 3% KOH, to distinguish Gram-negative and Gram-positive bacteria (8). To determine motility, strains were grown in tryptic soy broth and after 18 h, or as soon as growth was evident, a loopful of the culture was examined at 400 X magnification under dark-field microscopy (21).
Biochemical tests
Most of the biochemical tests were performed according to MacFaddin (25). Each isolated colony was streaked on nutrient agar slants and incubated at 35 -37ºC for up to 48 h to obtain abundant growth. The following tests were carried out for all 900 strains: oxidase and catalase production, methyl red; Voges-Proskauer; nitrate reduction; fermentation and oxidation, citrate utilization, lysine and ornithine decarboxylase, arginine dehydrolase, acid production in phenol red broth base containing 1% carbohydrate source (starch, L-arabinose, D-fructose, D-galactose, glycerol, D-glucose, lactose, maltose, mannitol, mannose, L-rhamnose, sucrose and D-sorbitol), gelatinase production (30), urea hydrolysis and indole production (29).
Additional tests
Aerobic, nonsporing, nonmotile, catalase-positive Gram-positive rods that exhibited filamentous morphology or that were fragmented into short rods or cocci, were classified in the genus Rhodococcus (5) and were submitted to the cAMP test along with Listeria ivanovii (7). Strains showing a positive cAMP test were classified as Rhodococcus equi. Gram-negative rods were streaked on McConkey agar and incubated according Bier (6), which provided a suitable medium for the presumptive identification of aerobic and anaerobic/facultative Gram-negative rods.
Recording the data
On the basis of the phenotypic characteristics, the isolates were classified into 6 groups (data not shown). The resulting data of 35 phenotypic (colony and cellular morphology not showed) and physiological characteristics of the species were verified using Bergey's Manual of Determinative Bacteriology (16), recorded, entered into a microcomputer, and also organized in a file using a commercially available database management program (Access; Microsoft, Calif.). The data for each isolate were checked against this file and the possible identities of strains that keyed out (certainty of identification, >90%) were noted.
RESULTS AND DISCUSSION
A preferential stimulation of endospore-forming Gram-positive rods was found regarding other bacteria examined (
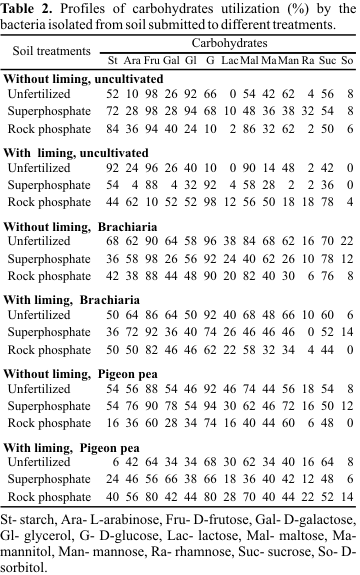
Table 1). Larger numbers of nonsporing, Gram-positive rods and Gram-positive cocci were found in plots cultivated with pigeon pea, while Gram-positive or Gram-negative rods were observed in uncultivated plots or plots cultivated with brachiaria grass. A larger proportion of nonsporing Gram-positive rods was found in soil fertilized with superphosphate compared to other treatments. Similarly, greater numbers of Gram-positive cocci and Gram-negative rods were observed in unfertilized plots. Unlimed plots favored the majority of bacteria, except the endospore-forming Gram-positive rods. Of the total isolates, 5.3% were Gram-negative and 94.7% Gram-positive, and among the latter, 76.3% were endospore-forming rods and 16.0% nonsporing rods. Gram-positive cocci were scarcely observed (2.4%), exhibiting along with Gram-negative rods a low occurrence in soils when compared to the number of Gram-positive rods. The results obtained were comparable to those from other reports. Cattelan et al. (10) verified the predominance of spore-forming and non-spore-forming Gram-positive bacteria in soil cultivated with soy beans. Sagardoy and Salerno (33) observed 26% Gram-negative bacteria and 74% Gram-positive bacteria, of which 21% were endospore-forming rods, 48% nonsporing rods and only 5% cocci. Conversely, Rouatt and Katznelson (31) found a larger number of Gram-negative than Gram-positive bacteria in soil cultivated with wheat; however, the number of Gram-positive cocci was proportionally smaller in agreement with our results.Carbohydrate fermentation by the isolates ranged from 7-20% for sorbitol, rhamnose and lactose, 40-56% for galactose, mannitol, mannose, arabinose, starch, glycerol and sucrose, and 60.8-86.1% for maltose, glucose and fructose (
Table 2). Similarly, Tate and Mills (36) demonstrated that lactose and sorbitol were utilized less than arabinose and starch. Contrasting results were obtained by Torvisk et al. (38), who found bacterial isolates with a lesser ability to ferment sucrose, maltose and starch. Arabinose was the most utilized pentose (4-76%), the largest percentage corresponding to the treatment pigeon pea-superphosphate-without liming. The hexoses most utilized were D-glucose (66-100%) and D-fructose (56-100%). Maltose (36-90%) was the disaccharide most utilized when compared to sucrose (36-78%), manitol (14-68%) and lactose (0-46%). The bacteria isolated from soils cultivated with brachiaria grass or pigeon pea utilized lactose better than did those from noncultivated soils. Among the sugar alcohols, glycerol (32-94%) was better utilized as a carbon source than manitol (14-68%) and sorbitol (0-22%). Starch was used in 6-92% of the isolates and the highest numbers were observed in uncultivated soil.The enzymatic reactions of the isolates are shown in
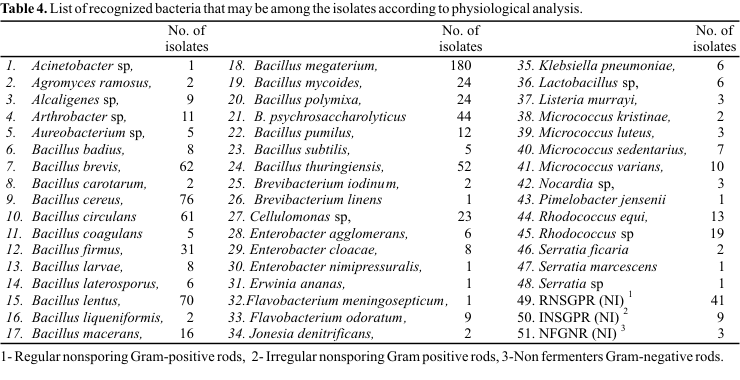
Table 3. Apparently, no factor studied influenced the diversity of the isolates. Isolates obtained in each treatment showed production of catalase (68-100%), gelatinase (62-100%), and oxidase, nitrate reductase and urease (4-86%). A low proportion of isolates showed indole production and positive reaction for the methyl red and Voges-Proskauer tests. The utilization of citrate as the sole carbon source was observed in 18-74% of the isolates. Only 14-86% of the isolates fermented glucose while 62-100% used it under aerobic conditions. Except for the phosphate fertilized plots, in absence of plants and liming, the other treatments showed a less than 50% proportion in the number of isolates that used L-arginine. The results of D-glucose utilization contrast with those obtained from Argentine soils where only 4% used D-glucose in oxidative metabolism and 33% in fermentative metabolism (33). However, similar results were obtained by Balkwill et al. (1) who obtained values of 82% and 4%, respectively.Twenty genera were identified and 53 strains that were not identified at the genus level were recorded (Table 4). The bacterial species found for each treatment are listed in Table 5 (number corresponding to the species listed in the Table 4). The genera with more number of isolates were Bacillus, Rhodococcus, Micrococcus, Cellulomonas, Enterobacter, Flavobacterium, and Arthrobacter. Other genera observed included Alcaligenes, Lactobacillus, Klebsiella, Aureobacterium, Serratia, Nocardia, Listeria, Brevibacterium, Jonesia, Agromyces, Pimelobacter, Erwinia, and Acinetobacter. Similar observations have been reported for the composition of bacterial populations in the soil of different crops. Kloepper et al. (20) isolated bacteria of the genera Bacillus, Flavobacterium and Pseudomonas from peanut soil. Wheat soil was found to contain the genera Achromobacter, Arthrobacter, Bacillus, Cellulomonas, Flavobacterium, Micrococcus, Mycobacterium, Mycococcus, Mycoplana, Nocardia and Pseudomonas, with Arthrobacter and Pseudomonas being most predominant (33). Soybean soil presented greater proportions of Bacillus, Pseudomonas and Arthrobacter genera, with Bacillus being prevalent in the non-rhizosphere soil and Pseudomonas in the rhizosphere (10). Accordingly these results, B. megaterium was the predominant species.
Many studies have found treatment effects on microbial diversity or on the size of physiological groups of bacteria (9,11,18,39). However, these studies used different methods and systems, then it was impossible to directly compare to our studies. The bacterial diversity were analyzed by the fluctuations of species in the treatments. The number of species for the treatments varied from 9 to 22, demonstrating a strong influence on bacterial diversity. The largest number of species was found in limed plots (9 to 20). While the number of species ranged from 13 to 22 and 12 to 18 in plots cultivated with brachiaria grass and pigeon pea, respectively, this number in uncultivated plots varied from 9 to 14. Thus, 20 species were not found in the uncultivated soil. The differences in the bacterial composition may be due simply to the nature of the plants used (19). These results suggest that cultivation of soil increased the number of bacterial species probably due to the root excretions (31,32) that diffuse in the soil. The diversity of bacterial species in cultivated soil may be explained from a knowledge of the metabolic and growth characteristics (Tables 2-3) of the isolates (34). However, it was not possible to access how the physiological traits of the bacteria populations were affected by the cultivated soil.
In unfertilized soil were found 9 to 22 species, which corresponded to an increase from 15 to 21 % over fertilized plots, suggesting that phosphate fertilization had a negative effect on bacterial diversity. Accordingly these results, Lima et al. (23) found a higher number of bacteria in unfertilized soil compared to soil fertilized with superphosphate. Differently, Martyniuk and Wagner (24) found that microbial populations were greater in fertilized soil than in unfertilized soil. However, organic phosphorus decreased in the soil fertilized with superphosphate or rock phosphate when compared with the control (3). Therefore, it is possible that the limited content of organic phophorus had decreased the number of bacterial species in the soil fertilized with phosphates.
In conclusion, the results above indicated that the recording data used in this experiment were effective in the identification of soil bacteria, and might be useful for soil diagnosis of soil bacterial isolates. These results suggest that specific bacteria were found in soil and their diversity might fluctuate depending on plant type, phosphate fertilizer and liming.
ACKNOWLEDGEMENTS
The authors wish to acknowledge the support from FAPESP.
Submitted: March 12, 2002; Approved: December 05, 2002.
- 1. Balkwill, D.L.; Fredrickson, J.K.; Thomas, J.M. Vertical and horizontal variations in the physiological diversity of the aerobic chemoheterotrophic bacterial microflora in deep southeast coastal plain subsurface sediments. Appl. Environ. Microbiol, 55: 1058-1065, 1989.
- 2. Balows, A.; Hausler Jr., W.J.; Hermann, K.L.; Isenberg, H.D.; Shadomy, H.J. Manual of Clinical Microbiology 5ed. ASM, Washington, 1991, 1242p.
- 3. Barroti, G. Características microbiológicas e físico-químicas de solo adubado com fosfato natural em diferentes tipos de cultivos. Araraquara, 1998, 81p. (MS Thesis. Instituto de Química, UNESP).
- 4. Barroti, G.; Nahas, E. População microbiana total e solubilizadora de fosfato em solo submetido a diferentes sistemas de cultivo. Pesq. Agropec. Bras, 35: 2043 2050, 2000.
- 5. Beaman, B.L.; Saubolle, M.A.; Wallace, R.J. Nocardia, Rhodococcus, Streptomyces, Oerskovia, and other aerobic actinomycetes of medical importance. In: Murray, P.R.; Baron, E.J.; Pfaller, M.A.; Tenover, F.C.; Yolken, R.H. (eds.) Manual of Clinical Microbiology 6.ed. ASM, Washington, 1995. p.379-399.
- 6. Bier, O. Fisiologia bacteriana. In: Microbiologia e Imunologia 24.ed. Melhoramentos, São Paulo, 1985, p.43-77.
- 7. Bille, J.; Doyle, M. Listeria and Erysipelothrix In: Balows, A.; Hausler Jr., W.J.; Hermann, K.L.; Isenberg, H.D.; Shadomy, H.J. (eds.) Manual of Clinical Microbiology 5.ed. ASM, Washington, 1991, p.287-295.
- 8. Buck, J.D. Nonstaining (KOH) method for determination of Gram reactions of marine bacteria. Appl. Environ. Microbiol, 44: 992-993, 1982.
- 9. Buyer, J.S.; Kaufman, D.D. Microbial diversity in the rhizosphere of corn grown under conventional and low-input systems. Appl. Soil Ecol., 5: 21-27, 1996.
- 10. Cattelan, A.J.; Hartel, P.G.; Fuhrmann, J.J. Bacterial composition in the rhizosphere of nodulating and non-nodulating soybean. Soil Sci. Soc. Am. J, 62: 1549-1555, 1998.
- 11. Frostegard, A.; Baath, E.; Tunlid, A. Shifts in the structure of soil microbial communities in limed forest as revealed by phospholipid fatty acid analysis. Soil Biol. Biochem., 25: 723-730, 1993.
- 12. Giller, K.E.; Beare, M.H.; Lavelle, P.; Izac, A.-M.N.; Swift, M.J. Agricultural intensification, soil biodiversity and agroecosystem function. Appl. Soil Ecol., 6: 3-16, 1997.
- 13. Hasebe, A.; Kanazawa, S.; Takai, Y. Microbial biomass in paddy soil. I. Microbial biomass calculated from direct count using fluorescence microscope. Soil Sci. Plant Nutr., 30: 175-187, 1984.
- 14. Hendrickson, D.A. Reagents and stains. In: Lennette, E.H.; Balows, A.; Hauler Jr., W.J.; Truant, J. P. (eds.) Manual of Clinical Bacteriology 4th ed. ASM, Washington, 1985, p.1056-1149.
- 15. Holding, A.J. The properties and classification of the predominant Gram-negative bacteria occurring in soil. J. Appl. Bacteriol, 23: 515-525, 1960.
- 16. Holt, J.G.; Krieg, N.R.; Sneath, P.H.A.; Staley, J.T.; Williams, S.T. Bergey's manual of determinative bacteriology 9.ed. Williams & Wilkins, Baltimore, 1994, 787p.
- 17. Kanazawa, S.; Asakawa, S.; Takai, Y. Effect of fertilizer and manure application on microbial numbers, biomass and enzyme activities in volcanic ash soils. Soil Sci. Plant Nutr., 34: 429-439, 1988.
- 18. Kennedy, A.C. Bacterial diversity in agroecosystems. Agric. Ecosystems Environ., 74: 65-76, 1999.
- 19. Kirchner, M.J.; Wollum II, A.G. & King, L.D. Soil microbial populations and activities in reduced chemical input agroecosystems. Soil Sci. Soc. Amer. J., 57: 1289-1295, 1993.
- 20. Kloepper, J.W.; McInroy, J.A.; Bowen, K.L. Comparative identification by fatty acid analysis of soil, rhizosphere, and geocarposphere bacteria of peanut (Arachis hypogaea L.). Plant Soil, 139: 85-90, 1992.
- 21. Koneman, E.W.; Allen, S.D.; Dowell, V.R.; Sommer, H.M. Introdução à microbiologia médica. In: Diagnóstico microbiológico: texto e atlas colorido. 2.ed. Panamericana, São Paulo,1993, p.1- 60.
- 22. Lawley, R.A.; Campbell, R.; Newman, E.I. Composition of the bacterial flora of the rhizosphere of three grassland plants grown separately and in mixtures. Soil Biol. Biochem., 15: 605-607, 1983.
- 23. Lima, J.A.; Nahas, E.; Gomes, A.C. Microbial populations and activities in sewage sludge and phosphate fertilizer-amended soil. Appl. Soil Ecol., 4: 75-82. 1996.
- 24. Martyniuk, S.; Wagner, G.H. Quantitative and qualitative examination of soil microflora associated with different management systems. Soil Sci, 125: 343-350. 1978.
- 25. Mc Faddin, J.F. Pruebas bioquimicas para la identificacion de bacterias de importancia clinica Panamericana, Buenos Aires, 1984, p.11-301.
- 26. Miller, H.J.; Henken, G.; Veen, J.A. Variation and composition of bacterial populations in the rhizospheres of maize, wheat, and grass cultivars. Can. J. Microbiol., 35: 656-660, 1989.
- 27. Nahas, E.; Centurion, J.F. ; Assis, L.C. Efeito das características químicas dos solos sobre os microrganismos solubilizadores de fosfato e produtores de fosfatases. Rev. Bras. Ciência Solo, 18: 49-53, 1994.
- 28. Nahas, E.; Fornasieri, D.J.; Assis, L.C. Resposta à inoculação de fungo solubilizador de fósforo em milho. Scientia Agricola, 51: 463-469, 1994.
-
29Pasteur manual of microbiology and immunology, 3rd ed. Urea-indol (combined media), 1991, p.32-33.
- 30. Priest, F.G.; Goodfellow, M.; Todd, C. A numerical classification of the genus Bacillus J. Gen. Microbiol, 134: 1847-1882, 1988.
- 31. Rouatt, J.W.; Katznelson, H. A study of the bacteria on the root surface and in the rhizosphere soil of crop plants. J. Appl. Bacteriol 24: 164-171, 1961.
- 32. Rovira, A.D.; McDougall, B.M. Microbiological and biochemical aspects of the rhizosphere. In: A.D. McLaren; G.H. Peterson (eds). Soil biochemistry Marcel Dekker, New York, 1967. vol. 1, p.417-463.
- 33. Sagardoy, M.A.; Salerno, C.M. Number, distribution, and characterization of heterotrophic bacteria in some Argentine soils. An. Edafol. Agrobiol., 42: 2069-2081, 1983.
- 34. Scott, J.S.; Knudsen, G.R. Soil amendment effects of rape (Brassica napus) residues on pea rhizosphere bacteria. Soil Biol. Biochem., 31: 1435-1441, 1999.
- 35. Tate, K.R. The biological transformation of P in soil. Plant Soil, 76: 245-256, 1984.
- 36. Tate, K.R.; Mills, A.L. Cropping and the diversity and function of bacteria in Pahokee Muck. Soil Biol. Biochem., 15: 175-179, 1983.
- 37. Thomas, C.M. Bacterial diversity and the environment. Trends Biotech, 14: 327-329, 1996.
- 38. Torvisk, V.; Salte, K.; Sorheim, R.; Goksöyr, J. Comparison of phenotypic diversity and DNA heterogeneity in a population of soil bacteria. Appl. Environ. Microbiol, 56: 776-781, 1990.
- 39. Workneh, F.; van Bruggen, A.H.C. Microbial density, composition, and diversity in organically and conventionally managed rhizosphere soil in relation to suppression of corky root of tomatoes. Appl. Soil Ecol., 1: 219-230, 1994.
- 40. Yasuda, M.; Katoh, K. Characteristics of bacteria isolated from soil and roots of fruit trees. Soil Sci. Plant Nutr, 35: 501-508, 1989.
Publication Dates
-
Publication in this collection
18 June 2003 -
Date of issue
Dec 2002
History
-
Received
12 Mar 2002 -
Accepted
05 Dec 2002