Abstracts
Antibiotic susceptibility of thirty Moraxella bovis strains recovered from outbreaks of Infectious Bovine Keratoconjunctivitis (IBK) in Argentina, Brazil and Uruguay between 1974 and 2001 was determined using the Kirby-Bauer and the Minimal Inhibitory Concentration (MIC) methods. Our results suggest that most strains were susceptible to the antibiotics used in the treatment of IBK, and that the antibiotic susceptibility of M. bovis varied with the geographical region and period of recovery.
Moraxella bovis; antibiotics; MIC; Infectious Bovine Keratoconjunctivitis; IBK
A susceptibilidade a antimicrobianos de trinta cepas de Moraxella bovis recuperadas entre 1974 e 2001 em surtos de Ceratoconjuntivite Infecciosa Bovina (CIB) ocorridos na Argentina, Brasil e Uruguai foi determinada pelos métodos de Kirby-Bauer e Concentração Inibitória Mínima. Nossos resultados indicam que a maioria das cepas é susceptível aos antibióticos utilizados no tratamento da CIB e que a susceptibilidade antimicrobiana da M. bovis variou conforme a região geográfica e período de recuperação.
Moraxella bovis; antibióticos; MIC; Ceratoconjuntivite Infecciosa Bovina; CIB
VETERINARY MICROBIOLOGY
SHORT COMMUNICATION
Antibiotic susceptibility of Moraxella bovis recovered from outbreaks of Infectious Bovine Keratoconjunctivitis in Argentina, Brazil and Uruguay between 1974 and 2001
Susceptibilidade a antimicrobianos de cepas de Moraxella bovis recuperadas em surtos de Ceratoconjuntivite Infecciosa Bovina ocorridos na Argentina, Brasil e Uruguai entre 1974 e 2001
Fabricio Rochedo ConceiçãoI; Diane Maria BertoncelliI; Otávio Brod StorchI; Fernando PaolicchiII; Ana Lia CoboIII; Carlos Gil-TurnesI
IUniversidade Federal de Pelotas, Centro de Biotecnologia, Faculdade de Veterinária, Pelotas, RS, Brasil
IIEstación Experimental Agropecuária INTA-Balcarce, Argentina
IIILadivet, Uruguay
Correspondence Correspondence to Carlos Gil-Turnes Universidade Federal de Pelotas, Centro de Biotecnologia Faculdade de Veterinária, Caixa Postal 354, 96010-900 Pelotas, RS, Brasil Tel.: (+5553) 275-7350; Fax (+5553) 275-7353 E-mail: gil@ufpel.tche.br
ABSTRACT
Antibiotic susceptibility of thirty Moraxella bovis strains recovered from outbreaks of Infectious Bovine Keratoconjunctivitis (IBK) in Argentina, Brazil and Uruguay between 1974 and 2001 was determined using the Kirby-Bauer and the Minimal Inhibitory Concentration (MIC) methods. Our results suggest that most strains were susceptible to the antibiotics used in the treatment of IBK, and that the antibiotic susceptibility of M. bovis varied with the geographical region and period of recovery.
Key words:Moraxella bovis, antibiotics, MIC, Infectious Bovine Keratoconjunctivitis, IBK
RESUMO
A susceptibilidade a antimicrobianos de trinta cepas de Moraxella bovis recuperadas entre 1974 e 2001 em surtos de Ceratoconjuntivite Infecciosa Bovina (CIB) ocorridos na Argentina, Brasil e Uruguai foi determinada pelos métodos de Kirby-Bauer e Concentração Inibitória Mínima. Nossos resultados indicam que a maioria das cepas é susceptível aos antibióticos utilizados no tratamento da CIB e que a susceptibilidade antimicrobiana da M. bovis variou conforme a região geográfica e período de recuperação.
Palavras-chave: Moraxella bovis, antibióticos, MIC, Ceratoconjuntivite Infecciosa Bovina, CIB
Infectious Bovine Keratoconjunctivitis (IBK) caused by fimbriated Moraxella bovis (6) is the most important disease of the eyes of cattle (1). Due to the economic impact of the disease in the MERCOSUR area, animal health authorities from Argentina, Brazil and Uruguay included it among the eight diseases of cattle to be studied by a multinational co-operative project.
Antibiotics are used all over the world to treat animals with IBK and to prevent dissemination of bacteria within a herd. Although Brown et al. (3) found that this bacterium is frequently susceptible to gentamicin, cephalosporin, trimethroprim, nitrofurans and tetracyclines, M. bovis recovered from different herds (5) or from different animals of the same herd (8) have shown variable response to antibiotics. Due to these variations in susceptibility, it is recommended to perform antibiograms of strains involved in an outbreak before the massive use of a specific antibiotic (14).
Vaccines containing adhesins as antigens were introduced in the Mercosur countries in 1983 (9). Since 1990 outbreaks in herds routinely vaccinated have been frequently reported, suggesting a modification in the genotypic and phenotypic characteristics, including their sensitivity to antibiotics.
The objective of this work was to compare the sensitivity of 30 strains of M. bovis recovered between 1974 and 2001 from diseased animals of Argentina, Brazil and Uruguay, to antimicrobials used in these countries to treat IBK.
The strains were recovered as already reported (7), characterised following Fraser and Gilmour (4) and lyophilised.
The sensitivity of the strains to the antimicrobials listed in Table 2 was detected by the Kirby-Bauer and Minimal Inhibitory Concentration tests.
The Kirby-Bauer method was performed following a slightly modified NCCLS (11) recommended protocol. Briefly, suspensions of 24 h cultures of each isolate, with a concentration equivalent to McFarland tube 1, were spread on Mueller-Hinton medium (MERCK, Darmstadt, Germany) supplemented with 6% ovine defibrinated blood. Once the inoculum was dry, disks were placed on the medium at a distance of 3 cm between each other. The plates were incubated at 37ºC for 20 h and the inhibition haloes measured. The strains were considered sensitive (S), intermediate (I) or resistant (R) following Barry and Thornsberry (2).
The agar plate dilution method was used to determine MICs (15). Tubes containing 1 ml of Brain Heart Infusion (BHI) (MERCK, Darmstadt, Germany) were inoculated with two colonies of the respective isolate grown on blood agar for 24 h at 37ºC, and then incubated at the same temperature in an orbital shaker with stirring of 200 rpm for 2 to 5 h. The turbidity of these cultures was adjusted to 0.5 of the McFarland scale and then diluted 1/20. Two mL of the diluted suspensions were dropped on BHI agar supplemented with 6% defibrinated ovine blood containing the antibiotic under test at appropriate dilutions. Each antibiotic was tested in at least five dilutions. The plates were then incubated at 37ºC for 20 h. MIC was the lowest concentration of antibiotic that inhibited growth, and MIC90 the lowest concentration that inhibited 90% of the strains. Plates containing medium without antibiotics sown with the same suspensions were used as controls. Means of MICs of strains recovered from each country, and before and after 1990, were compared, and the differences tested by the Student's t test.
The 30 strains tested were sensitive to ampicillin, gentamicin, cephalotin, neomycin, nitrofurantoin, rifampicin, tetracycline and streptomycin, by the Kirby-Bauer method. All the strains were considered intermediate and sensitive to erythromycin, but 57% and 77% of them were resistant to lincomycin and novobiocin, respectively. 63% and 75% of the Brazilian strains, and 58% and 100% of the Argentinean strains were resistant to lincomycin and novobiocin, respectively, while 50% of the Uruguayan strains were resistant to both. 61% and 53% of the strains recovered before and after 1990, respectively, were resistant to lincomycin, and 77% and 76% of the strains recovered before and after 1990, respectively, were resistant to novobiocin.
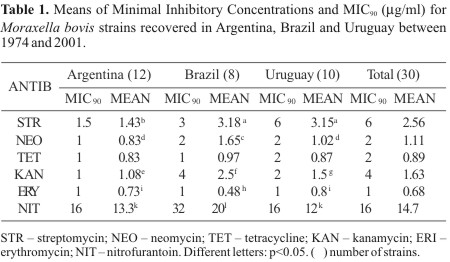
MIC90 and mean MICs for streptomycin, neomycin, tetracycline, kanamycin, erythromycin and nitrofurantoin of strains of the different countries differed (Table 1). With the exception of erythromycin, the mean MICs for Brazilian strains were higher than those from Uruguay, and these higher than those from Argentina (p<0.05). MIC90 and mean MICs for tetracycline and kanamycin of the strains recovered before 1990 were higher than those of strains recovered since 1990 (Table 2). Mean MIC for erythromycin was lower (p<0.05) with the strains recovered before 1990, while those of streptomycin, tetracycline and kanamycin were lower with the strains recovered since 1990 (Table 2).
The great majority of strains tested by the Kirby-Bauer method were susceptible to antibiotics routinely used in Mercosur countries. Resistance to lincomycin and novobiocin, antibiotics seldomly used to treat IBK, was detected in 55 and 77% of the strains, respectively. Sixty-two and 53% of the strains recovered before and after 1990 were resistant to lincomycin, showing a small decrease in resistance, while the rates of strains resistant to novobiocin remained unchanged. Both antibiotics are considered of reduced efficiency against aerobic bacteria (10).
Three strains recovered from the same animal showed different antibiotic susceptibility, suggesting that the microflora of the eye is heterogeneous (12). Strains recovered from animals of a single herd in Argentina during the course of an outbreak, also showed differences in antibiotic susceptibility, in agreement with results obtained in Brazil in 1984 (8).
Although data of MICs for M. bovis are very scarce, our results show that, with the exception of erythromycin, the strains we studied were susceptible to those we tested (NCCLS, 1994). Erythromycin showed MICs higher than 0.5 mg/ml for 64% of the strains, close to the rate of the isolates studied in the USA by Shryock et al. (13). Forty-six and 77 % of the strains recovered before and after 1990, respectively, were not susceptible to this antibiotic, indicating a decrease in susceptibility during that period (Table 2).
Rates of susceptibility to antibiotics frequently used to treat IBK differed among strains of different regions. All the strains tested by us were susceptible to tetracycline, whereas 26% of those studied by Shryock et al. (13) in USA, were resistant to oxytetracycline (13). In our study mean MICs and MIC90 of streptomycin, neomycin, tetracycline, kanamycin, erythromycin and nitrofurantoin differed among the strains of the different countries (Table 2).
The route of administration must be considered when choosing an antibiotic to treat IBK. Aminoglycosides do not reach therapeutic concentrations in tears after parenteral administration (10), but the MICs detected in our study suggest that they may inhibit the strains tested when used topically. Rifampicin, on the other hand, that is seldomly used to treat IBK, is easily distributed in body fluids due to its liposolubility, being an alternative for parenteral therapy of the disease. All the strains tested were susceptible to cephalotin, an antibiotic that has limited use in veterinary medicine because of its cost, but that may be used in special cases.
Our results show that M. bovis have patterns of antibiotic susceptibility that vary among countries and with the period of recovery, indicating the need to determine the effectiveness of the antibiotic of choice before initiating its therapeutical use.
AKNOWLEDGEMENTS
This work was financed by Fundação de Amparo à Pesquisa do Rio Grande do Sul (FAPERGS) grants Nº 99/1203, 00/1257.2 and 00/1993.8 and PROCISUR.
Submitted: March 26, 2004; Approved: December 20, 2004
- 1. Baptista, P.J.H.P. Infectious Bovine Keratoconjunctivitis: a review. Brit. Vet. J., 135:225-242, 1979.
- 2. Barry, A.L.; Thornsberry, C. Susceptibility Testing: Diffusion Test Procedures. In: Lennette, E.H. Manual of Clinical Microbiology 3Ş ed., ASM, Washington D.C, 1980, p.464.
- 3. Brown, M.H.; Brightman, A.H.; Fenwick, B.W.; Rider, M.A. Infectious Bovine Keratoconjunctivitis: A Review. J. Vet. Intern. Med., 12:259-266, 1998.
- 4. Fraser, J.; Gilmour, N.J.L. The identification of Moraxella bovis and Neisseria ovis from the eyes of cattle and sheep. Res. Vet. Sci., 27:127-128, 1979.
- 5. George, L.; Wilson, W.; Desmond, J.B.; Mihalyi, J.E. Antibiotic treatment of Moraxella bovis infection in cattle. J. Am. Vet. Med. Assoc, 185:1206-1209, 1984.
- 6. Gil Turnes, C. Hemagglutination, autoagglutination and pathogenicity of Moraxella bovis strains. Can. J. Comp. Med, 47:503-504, 1983.
- 7. Gil Turnes, C.; Araújo, F.L. Serological characterization of strains of Moraxella bovis using double immunodiffusion. Can. J. Comp. Med, 46:165-168, 1982.
- 8. Gil-Turnes, C.; Albuquerque, I.M.B. Serotypes and antibiotic sensitivity of Moraxella bovis isolated from an outbreak of infectious bovine keratoconjunctivitis. Can. J. Comp. Med, 46:428-430, 1984.
- 9. Gil Turnes, C.; Souza, R.S.M.; Araújo, F.L.; Reyes, J.C.S. Evaluation of a vaccine prepared with adherent Moraxella bovis for the control of Infectious Bovine Keratoconjunctivitis. Proc XIV World Congress on Diseases of Cattle, Dublin, Ireland 1986; 1233-1236.
- 10. Merck. Manual Merck de Veterinária., 7Ş ed. Roca, São Paulo, Brasil, 1996.
-
11NCCLS, 1994. National Committee for Clinical Laboratory Standards Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals; Proposed Standard. NCCLS document M31-P (ISBN 1-56238-258-6). NCCLS, 771 East Lancaster Avenue, Villanova, PA 19085, USA, 1994.
- 12. Schramm, R.C.; Araújo, F.L. Diferenças antigênicas de clones de Moraxella bovis Pesq. Vet. Bras., 14:75-78, 1994.
- 13. Shryock, T.R.; White, D.W.; Werner, C.S. Antimicrobial susceptibility of Moraxella bovis Vet. Microbiol, 61:305-309, 1998.
- 14. Smith, J.A.; George, L.W. Treatment of acute ocular Moraxella bovis infections in calves with a parenterally administered long-acting oxytetracycline formulation. Am. J. Vet. Res, 46:804-807, 1985.
- 15. Washington, J.A.; Sutter, V.L. Susceptibility test: Agar and macro-broth dilution procedures. In: Lennette, E.H. Manual of Clinical Microbiology 3Ş ed., ASM, Washington D.C., 1980, p.453-458.
Correspondence to
Publication Dates
-
Publication in this collection
23 May 2005 -
Date of issue
Dec 2004