Abstracts
Nowadays, there is a tendency of use of low temperature in laundry cleaning for both environmental and economical reasons, which makes the use of enzymes in detergent products indispensable. Since lipases are efficient catalyst both in solution and at the water-liquid interface, they are potentially suitable for lipid stain removal applications in industrial laundry and household detergents. The effect of different commercial detergents and surfactants on enzymatic activity of lipase from Fusarium oxysporum was observed through p-nitrophenylpalmitate (pNPP) assay. The enzyme was compatible with various ionic and non-ionic surfactants as well as commercial detergents. Lipase activity was strongly inhibited by Sodium Dodecyl Sulphate (SDS), but not by Triton X-100 and Triton X-114. The best assay conditions observed for this lipase were pH 8.0 and 50ºC. The enzyme was stable at alkaline pH and remained 93% of residual activity during 1 h incubation at 60ºC. The highest lipase activity was measured with triglycerides of middle and long chain fatty acids (C8-C18). This enzyme showed a variable specifity/hydrolytic activity towards various fats and oils. All these properties and its resistance towards various surfactants and tolerance to commercial detergents make this lipase a potential additive for detergent formulation.
alkaline lipase; fusarium oxysporum; surfactants; detergents
Atualmente, há uma tendência de utilizar baixas temperaturas em limpeza de lavanderia por razões ambientais e econômicas, situações estas que tornam indispensáveis o uso de enzimas em produtos detergentes. Devido às lipases serem excelentes catalisadoras em soluções e em interface água-óleo, elas são potencialmente apropriadas para aplicação na remoção de manchas de gorduras em lavanderia industrial e detergentes domésticos. O efeito de diferentes detergentes comerciais e surfactantes sobre a atividade enzimática da lipase de Fusarium oxysporum foi observado através de ensaios com p-nitrofenilpalmitato (pNPP). A enzima foi compatível com vários surfactantes iônicos e não-iônicos como também com detergentes comerciais. Atividade lipolítica foi fortemente inibida por Sulfato Dodecil de Sódio (SDS), mas não por Triton X-100 e Triton X-114. As melhores condições de ensaio observadas para esta lipase foram pH 8,0 e 50ºC. A enzima foi estável em pH alcalino e manteve 93% da atividade residual durante 1 h de incubação a 60ºC. A maior atividade lipolítica foi medida com triglicerídeos de ácidos graxos de cadeia média e longa (C8-C18). A especificidade hidrolítica da enzima em vários óleos e gorduras testados foi variável. Todas estas propriedades e sua resistência a vários surfactantes e tolerância a detergentes comerciais fazem desta lipase um aditivo potencial para formulação de detergentes.
Lipase alcalina; Fusarium oxysporum; surfactantes; detergentes
INDUSTRIAL MICROBIOLOGY
Characterization of alkaline lipase from Fusarium oxysporum and the effect of different surfactants and detergents on the enzyme activity
Caracterização de lipase alcalina de Fusarium oxysporum e o efeito de diferentes surfactantes e detergentes sobre a atividade enzimática
Janaina Nicanuzia dos Prazeres* * Corresponding Author. Mailing address: Rua Jorge Krug, 178, Apto. 45 Vila Itapura 13023-210 Campinas, São Paulo - Brasil. Tel.: (19) 3788-3887. E-mail: nicanuzia@yahoo.com ; Juliana Aparecida Bortollotti Cruz; Gláucia Maria Pastore
Departamento de Ciência de Alimentos, Faculdade de Engenharia de Alimentos, Unicamp, Campinas, São Paulo, Brazil
ABSTRACT
Nowadays, there is a tendency of use of low temperature in laundry cleaning for both environmental and economical reasons, which makes the use of enzymes in detergent products indispensable. Since lipases are efficient catalyst both in solution and at the water-liquid interface, they are potentially suitable for lipid stain removal applications in industrial laundry and household detergents. The effect of different commercial detergents and surfactants on enzymatic activity of lipase from Fusarium oxysporum was observed through p-nitrophenylpalmitate (pNPP) assay. The enzyme was compatible with various ionic and non-ionic surfactants as well as commercial detergents. Lipase activity was strongly inhibited by Sodium Dodecyl Sulphate (SDS), but not by Triton X-100 and Triton X-114. The best assay conditions observed for this lipase were pH 8.0 and 50ºC. The enzyme was stable at alkaline pH and remained 93% of residual activity during 1 h incubation at 60ºC. The highest lipase activity was measured with triglycerides of middle and long chain fatty acids (C8-C18). This enzyme showed a variable specifity/hydrolytic activity towards various fats and oils. All these properties and its resistance towards various surfactants and tolerance to commercial detergents make this lipase a potential additive for detergent formulation.
Key words: alkaline lipase, fusarium oxysporum, surfactants, detergents
RESUMO
Atualmente, há uma tendência de utilizar baixas temperaturas em limpeza de lavanderia por razões ambientais e econômicas, situações estas que tornam indispensáveis o uso de enzimas em produtos detergentes. Devido às lipases serem excelentes catalisadoras em soluções e em interface água-óleo, elas são potencialmente apropriadas para aplicação na remoção de manchas de gorduras em lavanderia industrial e detergentes domésticos. O efeito de diferentes detergentes comerciais e surfactantes sobre a atividade enzimática da lipase de Fusarium oxysporum foi observado através de ensaios com p-nitrofenilpalmitato (pNPP). A enzima foi compatível com vários surfactantes iônicos e não-iônicos como também com detergentes comerciais. Atividade lipolítica foi fortemente inibida por Sulfato Dodecil de Sódio (SDS), mas não por Triton X-100 e Triton X-114. As melhores condições de ensaio observadas para esta lipase foram pH 8,0 e 50ºC. A enzima foi estável em pH alcalino e manteve 93% da atividade residual durante 1 h de incubação a 60ºC. A maior atividade lipolítica foi medida com triglicerídeos de ácidos graxos de cadeia média e longa (C8-C18). A especificidade hidrolítica da enzima em vários óleos e gorduras testados foi variável. Todas estas propriedades e sua resistência a vários surfactantes e tolerância a detergentes comerciais fazem desta lipase um aditivo potencial para formulação de detergentes.
Palavras-chaves: Lipase alcalina, Fusarium oxysporum, surfactantes, detergentes
INTRODUCTION
Lipases (triacylglycerol acylhidrolases, EC 3.1.1.3) hydrolyze tri-, di- and monoglycerides at an oil-water interface where the lipases activities are greatly increased by a mechanism of interfacial activation (5,8). The majority of lipases exhibit a high activity toward lipids with fatty acid residues of C8 to C18 chain length (15). The lipase used in each application is selected based on its substrate specificity such as fatty acid, alcohol, position (regio-) and stereospecificity, as well as temperature and pH stability (17).
Microbial lipases have already established their vast potential regarding to their usage in different industries. The interest in microbial lipase production has increased in the last decades, because of its large potential in industrial applications as additives for foods (flavor modification), fine chemicals (synthesis of esters), waste water treatment (decomposition and removal of oil substances), cosmetics (removal of lipids), pharmaceuticals (digestion of oil and fats in foods), leather (removal of lipids from animal skins) and medical (blood triglyceride assay) (2,4,9,13). However, the biggest market of their use is in the detergent formulation (7,16). Regarding detergents applications, new challenges for lipases producers can be pointed (7,18,19): (1) the high variation in the triglyceride content of fat stains, requiring lipases with low substrate specificity; (2) the relatively harsh washing conditions (pH 10-11 and 30-60ºC), requiring stable enzymes; and (3) the effects of chemical denaturation and/or proteolytic degradation caused by detergents additives such as surfactant linear alkyl benzene sulfonate (LAS) and proteases. The functional importance of lipases in the detergent industry is related to the removal of fatty residues in laundry, dishwashers as well as for cleaning of clogged drains.
The purposes of the present study were to characterize the alkaline lipase from Fusarium oxysporum with respect to optimal pH and temperature as well as stability, and evaluate the effect of several detergents and surfactants on the enzyme activity.
MATERIAL AND METHODS
Strain
Fusarium oxysporum was isolated from Brazilian Northeast fruits (Maceió, AL, and Aracaju, SE).
Chemicals
Olive oil used for lipase assay was from Azeite "Gallo", Victor Guedes, Ind. e Com., S.A., (Portugal). All analytical chemicals and media components used were of highest purity grade available commercially. All other oils and commercial detergents used were purchased locally.
Lipase production
Fusarium oxysporum was cultivated for 96 h, at 30ºC in a medium consisting of (w/v): glucose 1.0%; peptone 0.3%; yeast extract 0.2%; K2HPO4 0.2%; MgSO4.7H2O 0.1%; Na2CO3 0.1%; and agar 2.0%. After that, it was transferred to a liquid medium contained (w/v): olive oil 1.0%; peptone 1.5%; yeast extract 0.5%; KH2PO4 0.3%; MgSO4.7H2O 0.04%, pH 6.0. Fermentation was carried out at 30ºC under 160 rpm in shaker. After 24 h, 1 mL of the culture medium was transferred to a fresh medium and incubated for more 48 h. The fermented medium was chilled rapidly to 4ºC and the cells were removed by filtration. The supernatant was fractionated with ammonium sulfate to 70% saturation. After centrifugation, the precipitated was dialyzed against water at 4ºC and freeze-dried (Lin et al., modified (10)).
Assay of Lipase
All the experiments were performed in triplicate and the standard derivation calculated.
Assay using olive oil as substrate: Lipase activity was determined by titrimetry using olive oil emulsion which was prepared by mixing 25 mL of olive oil and 75 mL of 7% arabic gum solution in a homogenizer for 2 min. The reaction mixture containing 5 mL of olive oil emulsion, 4 mL of 50 mM Tris-HCl buffer, pH 8.0, 1 mL of 110 mM CaCl2 and 1 mL of enzyme (5 mg/mL) was incubated at 50ºC for 30 min under orbital shaking at 160 rpm. The reaction was immediately stopped after the incubation period by the addition of 15 mL acetone:ethanol mixture (1:1 v/v), and the released free fatty acids were titrated with 50 mM NaOH. One unit (U) of lipase activity was defined as the amount that released 1 µmol of fatty acid per min [11].
Assay using different fats and oils as substrates: 2% (w/v) of fats and oils (olive oil, soybean oil, sunflower oil, corn oil, chicken oil and milk cream) and 100 mM of triglycerides (triacetin, tributyrin, tricaprylin, trilaurin and triolein) were individually added to the reaction mixture containing 4 mL of 50 mM Tris-HCl buffer, pH 8.0, 1 mL of 110 mM CaCl2 and 1 mL of enzyme (5 mg/mL). After 30 min of incubation at 50ºC with orbital shaking at 160 rpm, the reaction was immediately stopped by the addition of 15 mL acetone:ethanol mixture (1:1 v/v), and the released free fatty acids were titrated with 50 mM NaOH. One unit (U) of lipase activity was defined as the amount that released 1 µmol of fatty acid per min.
Assay using p-nitrophenylpalmitate (pNPP) as substrate: The activity was assayed with reaction mixture, in a final volume of 1 mL, containing 40 mM Tris-HCl buffer (pH 8.0), 20 mM pNPP, as substrate, and 25 µL of enzyme (5 mg/mL). After 10 min of incubation at 40ºC, the reaction was stopped by the addition of 2 mL of ethanol 96%, and the p-nitrophenol released was monitored spectrophotometrically at 420 nm, using a standard curve. One lipase unit (U) was defined as the amount of enzyme that released 1 µmol p-nitrophenol per minute.
Effect of pH on activity and stability
The pH effect on activity and stability profiles were studied by olive oil assay in a pH range of 3.5-11.0 using different buffers at 50 mM concentration. For stability, 1 mL of enzyme (5 mg/mL) was mixed with 1 mL of respective buffer and incubated for 24 h at 4ºC. After that, the assay was performed as described for olive oil assay.
Effect of temperature on activity and stability
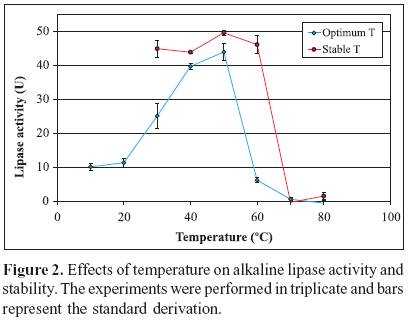
Temperature effect on lipase activity was studied by carrying out the enzyme reaction at different temperatures in the range of 10-70ºC at pH 8.0 using Tris-HCl buffer (50 mM). The thermostability of lipase was tested by pre-incubating the enzyme (5 mg/mL) at different temperatures ranging from 30 to 80ºC for 1 h. After that, the assay was performed as described for olive oil assay.
Effects of surfactants and commercial detergents on enzyme stability
Effects of surfactants (Tween 20, Tween 40, Tween 80, Triton X-100, Triton X-114, Sodium Dodecyl Sulphate (SDS) and surfactin from Bacillus subtilis (12)) and commercial detergents (Omo, Vida Plus, Tixan, Ace, Ariel, Brilhante, Surf and Revel) on enzyme stability were determined toward p-nitrophenylpalmitate (pNPP) assay. 50 µL of enzyme sample (5 mg/mL) were incubated in 50 µL of surfactants and commercial detergents (solution of 0.1, 1.0 and 10.0% w/v) for 1 h at room temperature (30 ± 2ºC) and the residual activity was determined at pH 8.0 and 40ºC for 10 min, using p-NPP as substrate as described for p-nitrophenypamitate assay.
RESULTS AND DISCUSSION
In the present work alkaline lipase was most active in pH range between 7.0 and 9.5. This enzyme was found to be stable in pH 7.0 to 9.0, and it kept around 45% activity after 24 h incubation in buffer of pH 11.0, at 4ºC (Fig. 1). The lipase from F. oxysporum f. sp. lini was stable in pH range 6.0-7.0 and had optimum activity at pH 7.0 (6), while this enzyme showed to be more stable in alkaline pH. On another hand, some lipases from Baccilus thermoleovorans were found to be most active at pH 9.0-10.0 and stable in a broad range of pH values 5.0-11.0, retaining more than 80% of activity after 26h at 30ºC (3).
The optimum reaction temperature was 50ºC. Thermal stability was investigated by incubating the enzyme at various temperatures for 1h. The enzyme showed great stability up to 60ºC (Fig. 2). Compared to higher thermal stability at 50ºC, this lipase kept 93% activity at 60ºC. These results show that this enzyme is more stable than lipase from F. oxysporum f. sp. lini which activity was reduced by about 50% at 60ºC when the enzyme solution was incubated for 1h (6).
Fig. 3 shows lipolytic hydrolysis toward various triglycerides. The order of hydrolysis rate was as follows: triolein > tricaprylin > trilaurin > triacetin > tricaproin > tributyrin. Higher lipolytic activity was observed when triglycerides of middle (C8-C12) and long (C18) chain fatty acids were used as substrates. This enzyme specificity towards lipids with fatty acid residues of C8-C18 chain length strongly suggest that the enzyme used in this study showed a true lipase activity (1,15). These results were similar to those presented by lipase from Fusarium oxysporum f. sp. lini (6). On another hand, Rapp (15) found different properties for lipase from Fusarium oxysporum f. sp. vasinfectum, which presented higher hydrolysis activity toward trilaurin at pH 5.0 and 45ºC, and it was not very thermostable.
The enzyme was found to be active on a wide range of natural substrates of either vegetable or animal origin. It hydrolyzed various natural lipids at different rates. This lipase showed major activity on vegetable oil and higher activity on corn oil 125% compared to olive oil. Lipolytic activity in milk cream was 30% lower than in olive oil (Fig. 4).
The effects of various surfactants and commercial detergents on the alkaline lipase activity were tested at 10% and 0.1% (w/v). Table 1 shows that the enzyme is stable in both ionic and non-ionic surfactants. Tween 40, Tween 80 and surfactin inhibited up to 30% lipase activity after 1h of incubation at 30ºC. On another hand, Triton X-100 and Triton X-114 showed an activating effect, as showed to lipase from F. oxysporum f. sp. lini (6) using Triton X-100. SDS was a strong inhibitor of Aspergillus caneus (17) and Bacillus thermoleovorans CCR11 (3) lipases, causing almost total inhibition of enzyme activity. Pocalyko and Tallman (14) have studied the adverse effect of SDS on the activity and stability of cutinase. They suggested that SDS causes local conformation changes in the active site that result in inhibition, partial reversible unfolding, and subsequent inactivation. Lipases are diverse in their sensitivity to solvents, but there is general agreement that polar water immiscible solvents are more destabilizing than water immiscible solvents (3).
The effects of commercial detergents showed different rates of lipase inhibition at the same conditions as the surfactants, probably because of commercial detergents composition. Rathi et al. (16) studied the effect of commercial detergents as Ariel, Wheel, Nirma, Fena, Surf Ultra, Rin Supreme on lipase activity from Burkholderia cepacia comparing with Lipolase® (Novo Nordisk, Denmark). They found that the lipase from B. cepacia exhibited better resistance to commercial detergents (57-80% residual activity) than Lipolase® (40-80% residual activity) after 1h of incubation at 37ºC and pH 11.0. In the present results, the lipase presented 20-52% residual activity, less than presented by Lipolase®.
CONCLUSION
In conclusion, the lipase from F. oxysporum has several properties of significant industrial importance, in particular, pH and temperature stability, C8-C18 wide specificity. All features presented by this lipase, activity in alkaline pH, high temperature, resistance to many surfactants, and tolerance to commercial detergents, make this enzyme a potential additive for detergent application. Further work to establish what kind of commercial detergent this lipase can be applied and better application conditions are necessary to be carried out.
ACKNOWLEDGEMENTS
This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil and Redes Cooperativas de Pesquisa/Financiadora de Estudos e Projetos (Recope/FINEP), Brazil.
Submitted: March 08, 2006; Returned to authors for corrections: June 01, 2006; Approved: October 10, 2006
- 1. Arpigny, J.L.; Jaeger, K.-E. Bacterial lipolytic enzymes: classification and properties. Biochem. J., 343: 177-183, 1999.
- 2. Burkert, J.F.M.; Maugeri, F.; Rodrigues, M.I. Optimization of extracellular lipase production by Geotrichum sp. using factorial design. Bioresour. Technol., 91: 77-84, 2004.
- 3. Castro-Ochoa, L.D.; Rodríguez-Gómez, C.; Valerio-Alfaro, G.; Ros, R.O. Screening, purification and characterization of the thermoalkalophilic lipase produced by Bacillus thermoleovorans CCR11. Enzyme Microb. Tech., 37: 648-654, 2005.
- 4. Davranov, K. Microbial lipases in biotechnology (Review). Appl. Biochem. Microbiol., 30(4-5): 427-432, 1994.
- 5. Helistö, P.; Korpela, T. Effects of detergents on activity of microbial lipases as measured by the nitrophenyl alkanoate esters method. Enzyme Microb. Tech., 23: 113-117, 1998.
- 6. Hoshino, T.; Sasaki, T.; Watanabe, Y.; Nagasawa, T.; Yamane, T. Purification and some characteristics of extracellular lipase from Fusarium oxysporum f. sp. lini Biosci. Biotech. Biochem., 56(4): 660-64, 1992.
- 7. Jaeger, K.-E.; Reetz, M.T. Microbial lipases form versatile tools for biotechnology. TIBTECH, 16: 396-403, 1998.
- 8. Kulkarni, N.; Gadre, R.V. Production and properties of alkaline, thermophilic lipase from Pseudomonas fluorescens NS2W. J. Ind. Microbiol. Biotechnol., 28: 344-348. 2002.
- 9. Kumar, S.; Kikon, K.; Upadhyay, A.; Kanwar, S.S.; Gupta, R. Production, purification, and characterization of lipase from thermophilic and alkaliphilic Bacillus coagulans BTS-3. Protein Expr. Purif., 41: 38-44, 2005.
- 10. Lin, S-F.; Chiou, C.-M.; Tsai, Y.-C. Effect of Triton X-100 on alkaline lipase production by Pseudomonas pseudoalcaligenes F-111. Biotechnol. Lett., 17(9): 956-962, 1995.
- 11. Macedo, G.A.; Park, Y.K.; Pastore, G.M., Partial purification and characterization of na extracellular lipase from a newly isolated strain of Geotrichum sp. Braz. J. Microbiol., 39: 687-692, 1997.
- 12. Nitschke, M.; Pastore, G.M. Production and properties of a surfactant obtained from Bacillus subtilis grown on cassava wastewater. Biotechnol. Research, 97: 336-341, 2006.
- 13. Pandey, A.; Benjamin, S.; Soccol, C. R.; Nigam, P.; Kriger, N.; Soccol. V.T. The realm of microbial lipases in biotechnology. Biotechnol. Appl. Bioc, 29: 119-131, 1999.
- 14. Pocalyko, D.J.; Tallman, M. Effect of amphipaths on the activity and stability of Fusarium solani pisi cutinase. Enzyme Microb. Tech, 22: 647-651, 1998.
- 15. Rapp, P. Production, regulation, and some properties of lipase activity from Fusarium oxysporum f. sp. vasinfectum Enzyme Microb. Tech., 17: 832-838, 1995.
- 16. Rathi, P.; Saxena, R.K.; Gupta, R. A novel alkaline lipase from Burkholderia cepacia for detergent formulation. Process Biochem., 37: 187-192, 2001.
- 17. Saxena, R.K.; Davidson, W.S.; Sheoran, A.; Giri, B. Purification and characterization of alkaline thermostable lipase from Aspergillus carneus Process Biochem., 39: 239-247, 2003.
- 18. Sharma, R.; Chisti, Y.; Banerjee, U.C. Production, purification, characterization, and applications of lipases. Biotechnol. Adv., 19: 627-662, 2001.
- 19. Sharma, R.; Soni, S.K.; Vohra, R.M.; Gupta, L.K.; Gupta, J.K. Purification and characterization of a thermostable alkaline lipase from a new thermophilic Bacillus sp. RSJ-1. Process Biochem., 37: 1075-1084, 2002.
Publication Dates
-
Publication in this collection
01 Mar 2007 -
Date of issue
Dec 2006
History
-
Accepted
10 Oct 2006 -
Reviewed
01 June 2006 -
Received
08 Mar 2006