Abstracts
The present study aimed at evaluating the in vitro antimicrobial activity of methanolic extracts of some medicinal plants against Escherichia coli, Salmonella Typhimurium, Staphylococcus aureus and Enterococcus sp. The methanolic extract of Caryophyllus aromaticus presented the highest anti-S. aureus activity and was effective against all bacterial strains tested.
medicinal plants; antibacterial activity; Minimal Inhibitory Concentration
Avaliou-se a atividade antimicrobiana in vitro de extratos metanólicos de algumas plantas medicinais frente a Escherichia coli, Salmonella Typhimurium, Staphylococcus aureus e Enterococcus sp. O extrato metanólico de Caryophyllus aromaticus foi o mais eficaz para todas as bactérias testadas e apresentou a melhor atividade anti-S. aureus.
plantas medicinais; atividade antibacteriana; concentração inibitória mínima
GENERAL MICROBIOLOGY
SHORT COMMUNICATION
Antibacterial activity of medicinal plant extracts
Atividade antibacteriana de extratos de plantas medicinais
Priscila Ikeda UshimaruI; Mariama Tomaz Nogueira da SilvaI; Luiz Claudio Di StasiII; Luciano BarbosaIII; Ary Fernandes JuniorI* * Corresponding Author. Mailing address: Departamento de Microbiologia e Imunologia, IBB/UNESP, Botucatu, SP, Brasil. Tel.: (14) 3811-6058 FAX (14) 3815-3744. E-mail: ary@ibb.unesp.br
IDepartamento de Microbiologia e Imunologia, Instituto de Biociências de Botucatu, Universidade Estadual Paulista, Botucatu, SP, Brasil
IIDepartamento de Farmacologia, Instituto de Biociências de Botucatu, Universidade Estadual Paulista, Botucatu, SP, Brasil
IIIDepartamento de Bioestatística, Instituto de Biociências de Botucatu, Universidade Estadual Paulista, Botucatu, SP, Brasil
ABSTRACT
The present study aimed at evaluating the in vitro antimicrobial activity of methanolic extracts of some medicinal plants against Escherichia coli, Salmonella Typhimurium, Staphylococcus aureus and Enterococcus sp. The methanolic extract of Caryophyllus aromaticus presented the highest anti-S. aureus activity and was effective against all bacterial strains tested.
Key-words: medicinal plants, antibacterial activity, Minimal Inhibitory Concentration
RESUMO
Avaliou-se a atividade antimicrobiana in vitro de extratos metanólicos de algumas plantas medicinais frente a Escherichia coli, Salmonella Typhimurium, Staphylococcus aureus e Enterococcus sp. O extrato metanólico de Caryophyllus aromaticus foi o mais eficaz para todas as bactérias testadas e apresentou a melhor atividade anti-S. aureus.
Palavras-chave: plantas medicinais, atividade antibacteriana, concentração inibitória mínima
Further acquaintance with different ethnic groups has contributed to the development of research on natural products, to the increase in knowledge about the close relationship between the chemical structure of a certain compound and its biological properties, and to the understanding of the animal/insect-plant interrelation (8). For these reasons, medicinal plants are important substances for the study of their traditional uses through the verification of pharmacological effects and can be natural composite sources that act as new anti-infectious agents.
The present study aimed at evaluating the in vitro antimicrobial activity of plant (Allium sativum, Zingiber officinale, Caryophyllus aromaticus, Cymbopogon citratus, Mikania glomerata and Psidium guajava) extracts against Gram-positive and Gram-negative bacterial strains isolated from human infections.
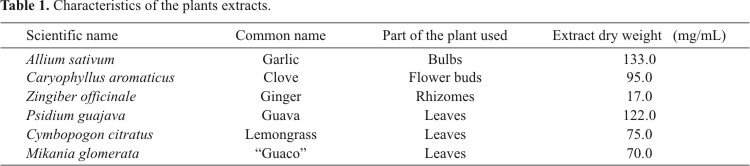
For the preparation of plant extracts, samples of A. sativum (bulbs), Z. officinale (rhizomes) and C. aromaticus (flower buds) were obtained at the local commerce in April 2006 and used in natura. Cymbopogon citratus (leaves) and Psidium guajava (leaves) were collected in May 2006 from an experimental field of the School of Agronomical Sciences, Unesp, Botucatu, São Paulo, Brazil. Mikania glomerata (leaves) was collected around Demétria farm, Botucatu, São Paulo, during the same period. The leaves were dried at approximately 50ºC and triturated in a mechanical mill. The voucher specimens were deposited in the Herbarium of the Department of Botany, Institute of Biosciences, Unesp, Botucatu-SP.
The determined plant parts (200g) were ground, extracted with 70% methanol and filtered after 48hs. The plant residue was re-extracted by adding 70% methanol and filtered again after 48hs. Such procedure was repeated every 72hs, completing three filtration processes. The filtrate was concentrated on a rotary evaporator at 45ºC for methanol elimination, and the extracts were kept in sterile bottles under refrigerated conditions until use. The dry weight of the extracts was obtained by allowing the solvent to evaporate and was used to determine concentration in mg/mL. (Methodology based on Betoni et al. (3); Table 1).
Microbial susceptibility assays using the agar dilution (Mueller-Hinton Agar) method (%v/v and corresponding mg/mL values) and the Minimal Inhibitory Concentration (MIC) were carried out for fifteen Salmonella Typhimurium, S. aureus, Enterococcus sp and E. coli strains plus one ATCC strain of each bacterium. Overnight cultures (37ºC) in Brain Heart Infusion (BHI) were adjusted to 0.5 Mac Farland standard and inoculated on Petri plates by using a Steer's replicator. After 37ºC/24 hours, MIC values (4,6) were read and MIC 50% and 90% values calculated. Kruskal-Wallis test for significant analysis (p<0.05) and Dunn's Test for multiple comparisons were carried out. Then, the results mean values, which represent the inhibitory capacity of each plant extract against the bacteria tested, were obtained and expressed as %v/v and mg/mL (Table 2).
MIC 90% values were different among extracts. Garlic and ginger extracts showed high antimicrobial action against Gram-negative strains. Gram-positive bacteria were more susceptible to the other plant extracts. Clove extracts were highly effective against bacterial strains, especially S. aureus strain (Table 2).
The antimicrobial properties of medicinal plants has been explained by the chemical association of active substances; however, the activity of their extracts is not related to their respective dry weights, which can be proven when more effective extracts are considered, e.g. clove extract (95 mg/mL) which had relatively lower activity than guava (122 mg/mL) and garlic (133 mg/mL) extracts, and all these extracts showed similar antimicrobial activity patterns. Such results were different from data reported in literature. Samy (7) used methanolic extracts of ginger which did not present antimicrobial effect against S. aureus and E. coli. However, Indu et al. (5), using a different method of ginger extract preparation, verified an inhibitory action against E. coli as well as high antimicrobial activity of garlic extracts against E. coli and Salmonella.
Ahmad and Aqil (2) concluded that ethanolic extracts of garlic did not have anti-E. coli or anti-Shigella action. Using another methodology, Vuddhakul et al. (9) observed that garlic extracts inhibited the growth of V. parahaemolyticus, E. coli and S. aureus; however, lemongrass and ginger extracts did not show any antimicrobial activity. Such behavior of the antibacterial action was also verified by Adonizio et al. (1), who used lemongrass extracts and did not observe antibacterial effects.
Comparisons with pertinent data from literature indicate that, according to the methodology adopted in studies on antimicrobial activity, the most diverse results can be obtained. Plant extracts have shown inhibitory effect on the growth of the bacteria studied, although of distinct forms. It is therefore recommended that the nature and the number of the active antibacterial principles involved in each plant extract be studied in detail.
ACKNOWLEDGEMENTS
This work was supported by Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP, Brazil). The authors thank Dr. Lin Chau Ming (FCA/Unesp/Botucatu) for providing the plant specimens and Dr. Maria de Lourdes Ribeiro de Souza da Cunha (IBB/Unesp/Botucatu) for supplying S. aureus strains.
Submitted: February 14, 2007; Returned to authors for corrections: May 19, 2007; Approved: September 27, 2007.
- 1. Adonizio, A.L.; Downum, K.; Bennett, B.C.; Mathee, K. (2006). Anti-quorum sensing activity of medicinal plants in southern Florida. J. Ethnopharmacol., 105: 427-435.
- 2. Ahmad, I.; Aqil, F. (2007). In vitro efficacy of bioactive extracts of 15 medicinal plants against ESbL-producing multidrug-resistant enteric bacteria. Microbiol. Res., 162: 264-275.
- 3. Betoni, J.E.C.; Mantovani, R.P.P.; Barbosa, L.N.; Di Stasi, L.C.; Fernandes Junior, A. (2006). Synergism between plant extract and antimicrobial drugs used on Staphylococcus aureus diseases. Mem. Inst. Oswaldo Cruz, 101: 387-390.
-
4Clinical and Laboratory Standards Institute (CLSI). (2005). Performance standards for antimicrobial susceptibility testing. Fifteenth informational supplement. CLSI document M100-S15. Wayne, PA, USA.
- 5. Indu, M.N.; Hatha, A.A.M.; Abirosh, C.; Harsha, U.; Vivekanandan, G. (2006). Antimicrobial activity of some of the south-Indian spices against serotypes of Escherichia coli, Salmonella, Listeria monocytogenes and Aeromonas hydrophila Braz. J. Microbiol., 37: 153-158.
-
6National Commitee for Clinical Laboratory Standards (NCCLS). (2003). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; Approved Standard - Sixth Edition NCCLS document M7-A6. Wayne, PA, USA.
- 7. Samy, R.P. (2005). Antimicrobial activity of some medicinal plants from India. Fitoterapia, 76: 697-699.
- 8. Viegas, C.; Bolzani, V.S. (2006). Os produtos naturais e a química medicinal moderna. Quím. Nova, 29: 326-337.
- 9. Vuddhakul, V.; Bhooponga, P.; Hayeebilana, F.; Subhadhirasakulb, S. (2007). Inhibitory activity of Thai condiments on pandemic strain of Vibrio parahaemolyticus Food Microbiol., 24: 413-418.
Publication Dates
-
Publication in this collection
28 Jan 2008 -
Date of issue
Dec 2007
History
-
Accepted
27 Sept 2007 -
Received
14 Feb 2007 -
Reviewed
19 May 2007