Abstracts
A mineral phosphate solubilizing bacterium, Burkholderia cepacia DA23 has been isolated from cultivated soils. Phosphate-solubilizing activities of the strain against three types of insoluble phosphate were quantitatively determined. When 3% of glucose concentration was used for carbon source, the strain had a marked mineral phosphate-solubilizing activity. Mineral phosphate solubilization was directly related to the pH drop by the strain. Analysis of the culture medium by high pressure liquid chromatography identified gluconic acid as the main organic acid released by Burkholderia cepacia DA23. Gluconic acid production was apparently the result of the glucose dehydrogenase activity and glucose dehydrogenase was affected by phosphate regulation.
insoluble phosphate; gluconic acid; Burkholderia cepacia; glucose dehydrogenase
Uma bactéria capaz de solubilizar fosfato mineral, Burkholderia cepacea DA23, foi isolada de solo cultivado. A capacidade dessa bactéria solubilizar o fosfato de três tipos de fosfato insolúvel foi quantificada. Quando foi utilizada glicose a 3% como fonte de carbono, a bactéria apresentou uma intensa atividade solubilizante de fosfato, sendo a solubilização diretamente relacionada com a queda de pH causada pela bactéria. A análise do meio de cultura por cromatografia líquida de alta pressão indicou o ácido glicônico como principal ácido produzido por Burkholderia cepacea DA23. Aparentemente, a produção de ácido glicônico foi causada pela atividade da glicose desidrogenase. A enzima foi afetada pela regulação do fosfato.
fosfato insolúvel; ácido glicônico; Burkholderia cepacea; glicose desidrogenase
ENVIRONMENTAL MICROBIOLOGY
Solubilization of insoluble inorganic phosphate by Burkholderia cepacia DA23 isolated from cultivated soil
Solubilização de fosfato inorgânico insolúvel por Burkholderia cepacea DA23 isolada de solo cultivado
Ok-Ryul SongI; Seung-Jin LeeI; Yong-Seok LeeI; Sang-Cheol LeeI; Keun-Ki KimII; Yong-Lark ChoiI* * Corresponding Author. Mailing address: Department of Biotechnology, Faculty of Natural Resources and Life Science, Dong-A University, Busan, 604-714, Korea. Phone: +82-51-200-7585: Fax: +82-51-200-7505. E-mail: ylchoi@dau.ac.kr
IDepartment of Biotechnology, Faculty of Natural Resources and Life Science, Dong-A University, Busan, Korea
IIDepartment of Biotechnology, College of Natural Resources and Life Science, Pusan National University, Miryang, Korea
ABSTRACT
A mineral phosphate solubilizing bacterium, Burkholderia cepacia DA23 has been isolated from cultivated soils. Phosphate-solubilizing activities of the strain against three types of insoluble phosphate were quantitatively determined. When 3% of glucose concentration was used for carbon source, the strain had a marked mineral phosphate-solubilizing activity. Mineral phosphate solubilization was directly related to the pH drop by the strain. Analysis of the culture medium by high pressure liquid chromatography identified gluconic acid as the main organic acid released by Burkholderia cepacia DA23. Gluconic acid production was apparently the result of the glucose dehydrogenase activity and glucose dehydrogenase was affected by phosphate regulation.
Key-words: insoluble phosphate, gluconic acid, Burkholderia cepacia, glucose dehydrogenase.
RESUMO
Uma bactéria capaz de solubilizar fosfato mineral, Burkholderia cepacea DA23, foi isolada de solo cultivado. A capacidade dessa bactéria solubilizar o fosfato de três tipos de fosfato insolúvel foi quantificada. Quando foi utilizada glicose a 3% como fonte de carbono, a bactéria apresentou uma intensa atividade solubilizante de fosfato, sendo a solubilização diretamente relacionada com a queda de pH causada pela bactéria. A análise do meio de cultura por cromatografia líquida de alta pressão indicou o ácido glicônico como principal ácido produzido por Burkholderia cepacea DA23. Aparentemente, a produção de ácido glicônico foi causada pela atividade da glicose desidrogenase. A enzima foi afetada pela regulação do fosfato.
Palavras-chave: fosfato insolúvel, ácido glicônico, Burkholderia cepacea, glicose desidrogenase.
INTRODUCTION
Phosphorus is an essential nutrient for biological growth and development of plants (14). The process of traditional P fertilizer production is environmentally undesirable, not least because of the release of contaminants into the main product, gas stream and by-products. These contaminants must be dealt with as potential air and water environmental pollutants (27). For over a century agricultural microbiologists and microbial ecologists have been interested in the ability of some bacteria to dissolve poorly soluble mineral phosphates such as tri-calcium phosphate or hydroxy-apatite (9,10), because most of the phosphorus in soils is present in insoluble form (1). Therefore, many researchers have tried to development biofertilizer for P-supply by means of phosphate-solubilizing microorganisms (PSMs). The PMS render insoluble phosphate into soluble form through the process of acidification, chelation and exchange reaction in soils (4-6,23,25).
Some Burkholderia sp. strains have been studied as PSMs. Burkholderia cepacia CC-A174 was able to solubilize tricalcium phosphate due to their production of organic acids. Mullan et al. reported Burkholderia cepacia involved in the removal of phosphate and accumulation of polyphosphate under the low pH conditions (8,15,21,24).
Many insoluble forms of calcium, iron and aluminum phosphate occur in soil, however, few studies are reported related to the solubilization of aluminum and iron phosphate (5,12,13). The ability of PSMs to release soluble orthophosphate (Pi) from rock phosphate ore makes this phenotype of great potential importance for the development of phosphate fertilizer technologies for agriculture (9,10).
Alexander (2) reported that phosphate solubilization was due to the production of organic acid and pH drop by PSMs. A number of organic acids have been considered due to its effect in pH lowering by microorganisms (7,22). Moghimi et al. (20) reported that solubilization was associated with a fraction containing large amounts of 2-ketogluconic acids. Many workers reported PSMs using the direct oxidation pathway could release significant amounts of Pi from calcium phosphates via this acidification of their extracellular environment (3,16).
In general, highly efficacious MPS bacteria utilize the direct oxidation pathway to produce gluconic and 2-ketogluconic acids, the direct oxidation pathway occurs on the outer face of the cytoplasmic membrane. The first step is the oxidation of glucose to gluconic acid by the glucose dehydrogenase (3).
Therefore, the objective of this study was to isolate and examine the solubilization of insoluble phosphate and biochemical analysis of the pH lowering in phosphate-solubilizing by Burkholderia cepacia DA23.
MATERIAL AND METHODS
Bacterial strains and growth conditions
Burkholderia cepacia DA23 (26) was isolated from Gimhae area soils, exposed to high salt and superfluity treatment of phosphate, in Korea. When the strain's mineral phosphate-solubilizing activity measured, the strain was cultured in the medium (28) containing 10 g·L-1 glucose, 5 g·L-1 tri-calcium phosphate, 0.27 g·L-1 NH4·NO3, 0.2 g·L-1 KCl, 0.1 g·L-1 MgSO4·7H2O, 1 mg·L-1 MnSO4·6H2O, 1 mg·L-1 FeSO4·7H2O, 0.1 g·L-1 Yeast extract and incubated on a rotary shaker (180rpm) at 26º. For liquid media mineral phosphate (tri-calcium phosphate, hydroxylapatite and aluminum phosphate) were sterilized separately and then mixed with the autoclaved medium for above.
When gluconic acid content and glucose-dehydrogenase activity in the culture medium measured, the strain was pre-cultured in LB medium (10 g·L-1 polypeptone, 5 g·L-1 NaCl, 5 g·L-1 Yeast extract, 1 g·L-1 glucose). The pre-culture was washed twice with 10 mM potassium phosphate buffer (pH 7.5) and resuspended in the same buffer at the same concentration as the original pre-culture. The suspension (inoculum size 5%, v/v) was then transferred to 100 ml glucose minimal medium containing 0.4% glucose and 21 mM potassium phosphate buffer (pH 6.8) and incubated on a rotary shaker (180rpm) at 30º.
For determination of determination organic acid, the strain was cultured in GMS medium (24) containing 10 g·L-1 glucose, 2 g·L-1 (NH4)2·SO4, 0.3 g·L-1 MgCl2·6H2O, 1 mg·L-1 MnCl2·4H2O, 6 mg·L-1 FeSO4·7H2O and 6 mg·L-1 NaMoO4, thiamine(20µl·L-1) and tri-calcium phosphate (20 mg·mL-1) and incubated on a rotary shaker (180rpm) at 37º.
Mineral-phosphate-solubilizing activity of PSM
Cells were collected by centrifugation at different growth periods in order to record the change in pH and P-concentration in the medium. Samples were centrifuged 6,000 rpm for 10 min to receive clear solutions for analysis. The P-concentration and pH were determined from supernatants in each investigation. The P-concentration was estimated by the method of ammonium molybdate (23). The 50 ~ 200 ul of the culture filtrate was mixed with 900 ul of phosphorus regent solution (Sigma Co.). The mixed solution was incubated at room temperature for 10 min. The P concentration was measured spectrophotometrically at 340 nm. In order to observe the effect of cultural conditions for mineral phosphate solubilizing, bacterial strains were cultured at various insoluble phosphates (tri-calcium phosphate, hydroxy-apatite, and aluminum phosphate) and various concentrations of glucose conditions.
Determination of organic acid in the culture medium
Five ml of a precultured solution was inoculated in 50 ml of medium contained in a 250 ml Erlenmeyer flask. After inoculation, the flasks were placed on a shaker and the bacteria were grown at 37º for 24 h. The supernatant of each culture was obtained by centrifugation at 10,000 rpm for 10 min. For the experiment to determine gluconic acid, a 0.5 ml aliquot of the culture filtrate passed through 0.2 µm Whatman membrane filter. The organic acids in filtrates were identified by high-performance liquid chromatography with a Thermo hypersil C18 column (250 x 4.6mm). Organic acids were monitored using a UV detector at 220 nm. The mobile phase consisted of 50 mM sodium phosphate and 5 mM tetra-butyl-ammonium hydrogen sulfate, pH 6.5 (95%), plus acetonitrile (5%) and a flow rate of 0.25 ml·min-1 (24).
Enzyme assays and determination of gluconate content
Gluconate dehydrogenase (GDH) activity and gluconate content were measured spectrophotometrically at 25º using phenazine methosulfate (PMS) and 2,6-dichlorophenol indophenol (DCIP) as electron acceptors as described by Matsushita et al. (18). The assay of GDH activity was performed in 0.1 M potassium phosphate buffer (pH 6.0) and enzyme activity was calculated using and extinction coefficient of 13.2 mM-1 cm-1 for DCIP. The reaction mixture contained 33 ul of 10 mM DCIP, 33 ul of 3 mM PMS, 333 ul of 0.1 M potassium phosphate buffer (pH 6.0) and sample in 1 ml final volume. The reaction was initiated by addition of 100 ul of 1 M glucose. And gluconate content in the culture medium was determined using gluconate dehydrogenase (less than 0.1 unit), purified from Pseudomonas aeruginosa (17), which has a high specificity for gluconate (19).
RESULT AND DISCUSSION
Characterization of phosphate-solubilizing activities under various insoluble phosphate
The isolated bacterium DA23 had a marked insoluble phosphate solubilizing activity as visualized by the clear zone developed around the colony after 3 days incubation at 30º, We assayed for the mineral-phosphate solubilizing trait of Burkholderia cepacia DA23 in liquid cultures that contained known amounts of insoluble phosphate and analytically measured the levels of P in the medium at different points of the 3-day growth period at 26º (Fig. 1). The highest production of phosphorus during incubation was measured in the medium containing tri-calcium phosphate. The amount of released P in the medium showed a gradual increase and reached a concentration of 345.9 mg·L-1 (Fig. 1B). As shown in Fig. 1, the production of soluble P by Burkholderia cepacia DA23 with tri-calcium phosphate and hydroxyl apatite was higher compared to aluminum phosphate. The soluble P production was enhanced with increasing amount of these insoluble phosphates (data not shown). Concomitant with the P increase there was a drastic decrease in the pH of the medium with the bulk of the change occurring during the first 12 h of growth (Fig. 1A). The pH value dropped to 4.23 from an initial value of 7.0. Thus, there was significant difference in the pH of culture medium between the insoluble phosphates. This reduction in the pH value was inversely correlated with the phosphorus concentration. This observation is consistent with earlier reports, which have shown that the solubilization of mineral phosphate is accompanied by a decrease in pH (9).
Effect of carbon sources on phosphorus concentration in culture medium
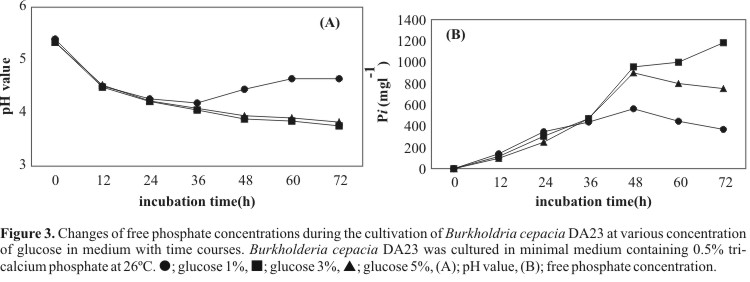
To elucidate the influence of C-source on the insoluble phosphate solubilization, the phosphate solubilization in Burkholderia cepacia DA23 was estimated in mineral medium contained glucose, sucrose or maltose. As shown in Fig. 2, the highest phosphorus concentrations indicated in all treatments at 48 h. The free phosphates solubilized from sucrose medium were 2 times less than that of glucose medium (Fig. 2B). The C-source which resulted in the most extensive solubilization of calcium phosphate was glucose between the tested sugars. To determine the effect of glucose concentration on the insoluble phosphate solubilization, various concentrations of glucose ranging from 1% to 5% (w/v) were added to the medium. Fig. 3 showed that the insoluble phosphate solubilization was enhanced with increasing amounts of glucose up to 3% (w/v), but decreased in the 5% of glucose. P-solubilization was more effective when the glucose concentration in the medium was raised from 1 to 37%. Soluble P production in the 3% glucose medium for 72 hr was around 1200 mg/l, which was approximately 3-fold higher than the yield in the 1% glucose. The ability of the strain to solubilize mineral phosphate in 3% glucose medium was increased at a high level up to 5 days (data not shown). These results are consistent with the earlier report which P-solubilizing ability was increased with increasing concentration of glucose in Pseudomonas sp. (4) and P. agglomerans R-42 (25).
Burkholderia cepacia DA23 releases gluconic acid in the culture medium
The mechanism, MPS microorganisms solubilize mineral phosphate, has been investigated by several workers (2,3). It is generally believed that the MPS trait is correlated with the production of organic acids. There were reports that MPS released many kinds of organic acids, such as citric, butyric, lactic, succinic, malic, gluconic, acetic, glyconic, fumaric, and 2-ketogluconic acids (23). The inverse correlation between pH value of the culture and the released P concentration indicates that organic acid production by Burkholderia cepacia DA23 may play a role in the solubilization of insoluble phosphate. In order to examine whether the MPS trait of Burkholderia cepacia DA23 is also associated with organic acid production, we employed reverse-phase HPLC to identify different organic acids present in the culture medium. A major peak was detected at 10 min elution time (Fig. 4B). This peak was identified as a gluconic acid based on the comparison of the elution time of a gluconic acid standard (Fig. 4A). Evidence from this study indicated that one of the solubilization mechanism associates with the production of gluconic acid and pH drop. In order to measure gluconic acid contents and GDH activity of Burkholderia cepacia DA23, we determined spectrophotometrically at 25º using PMS and DCIP by Matsushita et al. (18). On cell growing, the pH value dropped to 4.0 from an initial value of 7.0 (Fig. 5A), because gluconic acid was produced. Gluconic acid contents detected maximum 9.4 mM at 36 h. This result indicates that the phosphate solubilizing ability of this strain was caused by the modification of pH brought about by the release of organic acids. It is reported that Penicillium billaji secretes 10mM each of citric acid and oxalic acids (7) and Citrobacter. koseri and Bacillus coagulans secretes various organic acid in the range 1~5mM (11). However, the concentration of these acids required to reduce the pH of the soil was 20~50 times more. On gluconic acid contents increasing the GDH activity was higher (Fig. 5B). As shown in Table 1, JM109 did not detect the GDH activity, when grown in glucose minimal medium containing 0.4% tri-calcium phosphate whereas Burkholderia cepacia DA23 detected. But Burkholderia cepacia DA23 was affected by phosphate regulation.
ACKNOWLEDGMENTS
This work was partially supported by a grant from the Ministry of Agriculture and Forestry, Republic of Korea. Lee, S.C. and Lee, Y.S. was supported by the Brain Korea 21 project.
Submitted: May 27, 2007; Returned to authors for corrections: November 21, 2007; Approved: January 18, 2008.
- 1. Abd-Alla, M.H. (1994). Phosphates and the utilization of organic phosphorus by Rhizobium leguminosarum biovar viceae. Lett. Appl. Microbiol, 18, 294-296.
- 2. Alexander, M. (1997). Introduction to Soil Microbiology. 33-399. John Wiley and Sons, New York. Asea, P.E.A.
- 3. Babu-Khan, S.; Yeo, T.C.; Martin, W.L.; Duron, M.D.; Rogers, R.D.; Goldstein, A.H. (1995). Cloning of a mineral phosphate solubilizing gene from Pseudomonas cepacia Appl. Environ. Microbiol, 61, 972-978.
- 4. Banik, S. (1983). Variation in potentiality of phosphate solubilizing soil microorganisms with phosphate and energy source. Zentralbl. Mikrobiol., 138, 209-216
- 5. Barroso, C.V.; Pereira, G.T.; Nahas, E. (2006). Solubilization of CaHPO4 and AlPO4 by Aspergillus niger in culture media with different carbon and nitrogen sources. Braz. J. Microbiol., 37, 434-438.
- 6. Berthelin, F.; Leyval, C.; Laheurte, F.; De-Giudici, P. (1991). Involvements of roots and Rhizospere microflora in the chemical weathering of soil minerals. In Plant Root Growth; an Ecological Perspective(D. Atkinson, ED.). Special Publication series of the British Ecological Society, NO. 10. Black-well Scientific, Oxford, 1991.
- 7. Cunningahm, J.E.; Kuiack, C. (1992). Production of citric and oxalic acids and solubilization of calcium phosphate by Penicillium bilaji Appl. Environ. Microbiol, 58, 1451-1458.
- 8. Dubey, S.K.; Billore, S.D. (1992). Phosphate solubilizing microorganism(PSM) as inoculant their role in augmenting crop productivity India-A review. Crop Res. Hisar., 5, 11-17.
- 9. Goldstein, A.H.; Liu, S.T. (1987). Molecular cloning and regulation of a mineral phosphate solubilizing gene from Erwivia herbicola Bioresour. Technol, 5, 72-74.
- 10. Goldstein, A.H.; Rogers, R.D.; Mead, G. (1993). Separating phosphte from ores via bioprocessing. Bioresour. Technol, 11, 1250-1254.
- 11. Gyaneshwar, P.; Naresh, K.G.; Parekh, L.J. (1998). Effect of buffering on the phophate-solubilizing ability of microorganisms. World J. Microbiol. Biotechnol., 14, 669-673.
- 12. Illmer, P.; Schinner, F. (1992). Solubilization of inorganic phosphates by microorganisms isolated from forest soils. Soil Biol. Biochem., 24, 389-395.
- 13. Jones, D.; Smith, B.F.C.; Wilson, M.J.; Goodman, B.A. (1991). Phosphate solubilizing fungi in a Scottish upland soil. Mycol. Res, 95, 1090-1093.
- 14. Kucey, R.M.N. (1988). Effect of Penicillium bilagi on the solubility and uptake of P and micronutrients from soil by wheat. Can. J. Soil Sci., 68, 261-270.
- 15. Lin, T.F.; Huang, H.I.; Shen, F.T.; Young C.C. (2006). The protons of gluconic aicd are the major factor responsible for the dissolution of tricalcium phosphate by Burkholderia cepacia CC-A174. Bioresour. Technol., 97, 957-960.
- 16. Liu, S.T.; Lee, L.Y.; Tai, C.Y.; Ilorng, C.; Chang, Y.S.; Wolfram, J.H.; Rogers, R.; Goldstein, A.H. (1992). Cloning of an Erwinia herbicola gene necessary for gluconic acid production and enhanced mineral phosphate solubilization in Escherichia coli HB101. J. Bacteriol., 174, 5814-5819.
- 17. Matsushita, K.; Shinagawa, E.; Adachi O.; Ameyama, M. (1979). Membrane bound D-gluconate dehydrogenase from Pseudomonas aeruginosa Purification and structure of cytochromebinding form. J. Biochem, 85, 1173-1181.
- 18. Matsushita, K.; Ohno, Y.; Shinagawa, E.; Adachi, O.; Ameyama, M. (1980). Membrane bound D-gluconate dehydrogenase from Pseudomonas sp: solubilization, purification and characterization. Agric. Biol. Chem, 44, 1505-1512.
- 19. Matsushita, K.; Shinagawa, E.; Ameyama, M. (1982). D-gluconate dehydrogenase from bacteria, 2-keto-D-gluconate yielding, membrane-bound. Meth. Enzymol, 89, 187-193.
- 20. Moghimi, A.; Tate, M.E.; Oades, J.M. (1978). Characterization of rhizophere products especially 2-ketogluconic acid. Soil Biol. Biochem., 10, 289-292.
- 21. Mullan, A.; Quinn, J.P.; McGrath, J.W. (2002). Enhanced phosphate uptake and polyphosphate accumulation in Burkholderia cepacia grown under low pH conditions. Microb. Ecol, 44, 69-77.
- 22. Nahas, E.; Banzatto, D.A.; Assis, L.C. (1990). Fluorapatite solubilization by Aspergillus niger in vinasse medium. Soil Biol. Biochem, 22, 1097-1110.
- 23. Rodriguez, H.; Gonzalez, T.; Selman, G. (2000). Expression of a mineral phosphate solubilizing gene from Erwinia herbicola in two rhizobacterial strains. J. Biotechnol, 84, 155-161.
- 24. Safura, B.K.; Yeo, T.C.; Martin, W.L.; Duron, M.R.; Robert, D.R.; Goldstein, A.H. (1995). Cloning of a mineral phosphate-solubilizing gene from Pseudomonas cepacia Appl. Environ. Microbiol, 61, 972-978.
- 25. Son, H.J.; Park, G.T.; Cha, M.S.; Heo, M.S. (2006). Solubilization of insoluble inorganic phosphate by a novel salt- and pH-tolerant Pantoea agglomerans R-42 isolated from soybean rhizosphere. Bioresour. Technol, 97, 204-210.
- 26. Song, O.R.; Lee, S.J.; Kim, S.H.; Chung, S.Y.; Cha, I.H.; Choi, Y.L. (2001). Isolation and cultural characteristics of a phosphate-solubilizing bacterium, Aeromonas hydrophila DA57. J. Korean Soc. Agric. Chem. Biotechnol., 44, 257-261.
- 27. Vassilev, N.; Vassileva, M. (2003). Biotechnological solubilization of rock phosphate on media containing agro-industrial wastes. Appl. Microbiol. Biotechnol., 61, 435-440.
- 28. Young, C.C. (1990). Effects of phosphorus-solubilizing bacteria and vesicular-arbuscular mycorrhizal fungi on the growth of tree species in subtropical-tropical soils. Soil Sci. Plant Nutr, 36, 225-231.
Publication Dates
-
Publication in this collection
11 Apr 2008 -
Date of issue
Mar 2008
History
-
Accepted
18 Jan 2008 -
Received
27 May 2007