Abstracts
The purpose of this study was to determine the prevalence of enteric bacteria and yeasts in biofilm of 80 HIV-positive patients with plaque-associated gingivitis or necrotizing periodontitis. Patients were subjected to extra, intra oral and radiographic examinations. The oral hygiene, bleeding on probing, gingival conditions, and attachment loss were evaluated. Clinical specimens were collected from gingival crevices or periodontal pockets, transferred to VMGA III, diluted and transferred to Sabouraud Dextrose agar with 100 µg/ml of chloramphenicol, peptone water, EVA broth, EMB agar, SS agar, Bile esculin agar and Brilliant green agar. Isolation of yeasts was carried out at room temperature, for 3-7 days; and for the isolation of enteric microorganisms plates were incubated at 37ºC, for 24-48 h. The yeasts identification was performed according to the carbon and nitrogen assimilation, fermentation of carbohydrates and germ tube formation. Bacteria were identified according to their colonial and cellular morphologies and biochemical tests. Yeasts were identified as Candida albicans and its occurrence was more common in patients with CD4+ below 200/mm³ and was affected by the extension of periodontal involvement (P = 0.0345). Enteric bacteria recovered from clinical specimens were identified as Enterobacter sakazakii, Enterobacter cloacae, Serratia liquefaciens, Klebsiella oxytoca and Enterococcus sp. Enterobacteriaceae and enterococci were detected in 32.5% of clinical samples from patients with necrotizing periodontitis. In conclusion, non-oral pathogenic bacteria and C. albicans were more prevalent in periodontal sites of HIV-positive patients with necrotizing periodontitis and chronic gingivitis.
HIV patients; Candida albicans; enterococci; enterobacteria
O objetivo desse estudo foi avaliar a ocorrência de bactérias entéricas e leveduras no biofilme subgengival de pacientes HIV-positivos com gengivite crônica ou periodontite necrosante. Os pacientes foram submetidos a exame clínico e radiográfico e de higiene bucal, sangramento à sondagem, condições gengivais e a perda de inserção. Os espécimes clínicos de sulcos gengivais ou bolsas periodontais foram inoculados em ágar Sabouraud dextrose com 100 mg/ml de cloranfenicol, água peptonada, caldo EVA, ágar EMB, ágar SS, ágar Bile esculina e ágar verde brilhante. O cultivo de leveduras foi realizado à temperatura ambiente, de 3-7 dias; das enterobactérias a 37ºC de 24-48 h. A identificação das leveduras foi realizada pela assimilação de carbono e nitrogênio, fermentação de açucares e formação de tubo germinativo. As bactérias de acordo com a morfologia celular e colonial e testes bioquímicos. Foram identificadas Candida albicans e sua prevalência foi maior em pacientes com contagens de CD4+ < 200/mm³, e sua ocorrência foi afetada pela extensão da destruição periodontal (P = 0,0345). Enterobacteriaceae e enterococos foram detectados em 32,5% dos pacientes com periodontite necrosante. As enterobactérias foram Enterobacter sakazakii, E. cloacae, Serratia liquefaciens, Klebsiella oxytoca e Enterococcus sp. Concluiu-se que bactérias patogênicas exógenas à cavidade bucal e C. albicans podem ser detectadas no biofilme subgengival de pacientes HIV-positivos com periodontite necrosante e gengivite.
HIV; gengivite; periodontite necrozante; Candida albicans; enterococos; enterobacterias
MEDICAL MICROBIOLOGY
Occurrence of yeasts, enterococci and other enteric bacteria in subgingival biofilm of HIV-positive patients with chronic gingivitis and necrotizing periodontitis
Ocorrência de leveduras, enterococos e outras bactérias entéricas no biofilme subgengival de pacientes HIV-positivos com gengivite crônica e periodontite necrosante
Elerson Gaetti-Jardim JúniorI; Viviane NakanoII; Thais C. WahasuguiII; Fátima C. CabralII; Rosa GambaII; Mario Julio Avila-CamposII
ILaboratório de Patologia, Faculdade de Odontologia de Araçatuba, Universidade Estadual de São Paulo, Araçatuba, SP, Brasil
IILaboratório de Anaeróbios, Departamento de Microbiologia, Instituto de Ciências Biomédicas, Universidade de São Paulo, São Paulo, SP, Brasil
ABSTRACT
The purpose of this study was to determine the prevalence of enteric bacteria and yeasts in biofilm of 80 HIV-positive patients with plaque-associated gingivitis or necrotizing periodontitis. Patients were subjected to extra, intra oral and radiographic examinations. The oral hygiene, bleeding on probing, gingival conditions, and attachment loss were evaluated. Clinical specimens were collected from gingival crevices or periodontal pockets, transferred to VMGA III, diluted and transferred to Sabouraud Dextrose agar with 100 µg/ml of chloramphenicol, peptone water, EVA broth, EMB agar, SS agar, Bile esculin agar and Brilliant green agar. Isolation of yeasts was carried out at room temperature, for 3-7 days; and for the isolation of enteric microorganisms plates were incubated at 37ºC, for 24-48 h. The yeasts identification was performed according to the carbon and nitrogen assimilation, fermentation of carbohydrates and germ tube formation. Bacteria were identified according to their colonial and cellular morphologies and biochemical tests. Yeasts were identified as Candida albicans and its occurrence was more common in patients with CD4+ below 200/mm3 and was affected by the extension of periodontal involvement (P = 0.0345). Enteric bacteria recovered from clinical specimens were identified as Enterobacter sakazakii, Enterobacter cloacae, Serratia liquefaciens, Klebsiella oxytoca and Enterococcus sp. Enterobacteriaceae and enterococci were detected in 32.5% of clinical samples from patients with necrotizing periodontitis. In conclusion, non-oral pathogenic bacteria and C. albicans were more prevalent in periodontal sites of HIV-positive patients with necrotizing periodontitis and chronic gingivitis.
Key-words: HIV patients, Candida albicans, enterococci, enterobacteria.
RESUMO
O objetivo desse estudo foi avaliar a ocorrência de bactérias entéricas e leveduras no biofilme subgengival de pacientes HIV-positivos com gengivite crônica ou periodontite necrosante. Os pacientes foram submetidos a exame clínico e radiográfico e de higiene bucal, sangramento à sondagem, condições gengivais e a perda de inserção. Os espécimes clínicos de sulcos gengivais ou bolsas periodontais foram inoculados em ágar Sabouraud dextrose com 100 mg/ml de cloranfenicol, água peptonada, caldo EVA, ágar EMB, ágar SS, ágar Bile esculina e ágar verde brilhante. O cultivo de leveduras foi realizado à temperatura ambiente, de 3-7 dias; das enterobactérias a 37ºC de 24-48 h. A identificação das leveduras foi realizada pela assimilação de carbono e nitrogênio, fermentação de açucares e formação de tubo germinativo. As bactérias de acordo com a morfologia celular e colonial e testes bioquímicos. Foram identificadas Candida albicans e sua prevalência foi maior em pacientes com contagens de CD4+ < 200/mm3, e sua ocorrência foi afetada pela extensão da destruição periodontal (P = 0,0345). Enterobacteriaceae e enterococos foram detectados em 32,5% dos pacientes com periodontite necrosante. As enterobactérias foram Enterobacter sakazakii, E. cloacae, Serratia liquefaciens, Klebsiella oxytoca e Enterococcus sp. Concluiu-se que bactérias patogênicas exógenas à cavidade bucal e C. albicans podem ser detectadas no biofilme subgengival de pacientes HIV-positivos com periodontite necrosante e gengivite.
Palavras-chave: HIV, gengivite, periodontite necrozante, Candida albicans, enterococos, enterobacterias.
INTRODUCTION
In AIDS patients the firsts clinical opportunistic pathologies are observed in oral cavity, particularly involving periodontal tissues (15). Clinical characteristics of periodontal disease in HIV-patients are closely related to the immunological status. Hence, in symptomatic HIV-patients, whose CD4+ cell counts are below 200/mm3 in the blood, necrotizing and acute involvements of the periodontal tissues are common, frequently followed by intense malodor, bleeding and pain (15), besides a rapid loss of attachment (3).
Previous studies have shown that microbiology of gingivitis and periodontitis in HIV patients may differ significantly in comparison to these periodontal pathologies in immune-competent individuals (1,16). These peculiarities are not related to the distribution of the strictly anaerobic (14), but probably it can involve superinfecting microorganisms and other pathogenic agents normally not involved in periodontal infections, such as fungi and enteric organisms (1,17).
Most of the HIV patients receive medicaments to control the viral infection and opportunistic diseases, this however produces a severe selective process on subgingival biofilm, leading to the elimination of members of the indigenous microbiota and giving conditions to the periodontal colonization by minor components of oral microbiota and superinfecting organisms (16,17).
Bacteria such as Porphyromonas gingivalis, Prevotella intermedia, Aggregatibacter (Actinobacillus) actinomyce-temcomitans, Fusobacterium nucleatum, Fusobacterium necrophorum, Fusobacterium varium, spirochetes, Peptostreptococcus micros, Campylobacter rectus, Bacteroides fragilis, Collinsella (Eubacterium) aerofaciens, fungi and enterobacteria, may be involved in the pathogenesis of periodontitis in HIV-patients (2,14). Enteric microorganisms frequently act as reservoirs for resistance genes to antimicrobials and may spread them to microbial population in nosocomial infections, since these bacterial groups are commonly involved in systemic infections (5).
In this study, the occurrence of yeasts, enterococci and enterobacteria in clinical samples of subgingival biofilm from chronic gingivitis or necrotizing periodontitis from HIV-positive patients was evaluated.
MATERIAL AND METHODS
1. Patients
Four hundred two HIV-positive patients from the Centro de Atendimento a Pacientes Especiais (CAPE), visiting the clinics of dentistry of the University of São Paulo, SP, Brazil, were initially subjected to intra and extra oral examinations. However, 80 patients, 59 males and 21 females, who presented clinical and radiographic features characteristics of chronic gingivitis (n = 40) or necrotizing periodontitis (n = 40) were selected. HIV-positive patients were confirmed by serologic examination for the HIV specific antibody (ELISA) and Western blot, and all of them were aged between 20 to 43 years old (Mean age 31.5). Medical staff of the CAPE provided data on the systemic conditions and medications used by the patients, as well as the CD4+ counts.
2. Clinical parameters
The medical and dental histories of the patients were analyzed and patients were submitted to extra and intra oral examinations. The oral hygiene was evaluated by mean of the plaque index of O'Leary. Bleeding on probing and attachment loss were also recorded. Complete radiographic examinations using the parallelism technique with a Kodak film (Ektaspeed plus) were performed. The criteria to determine the presence of periodontal disease were those previously established by Machtei et al. (10). Patients showing more than 5% gingival bleeding, but without clinical attachment loss higher than 6 mm in two or more sites and without one or more sites with probing depth (≥ 5 mm), were diagnosed as having chronic gingivitis. Finally, for the clinical definition of patients who positively and unequivocally exhibited periodontitis, patients showing more than 5% gingival bleeding, with clinical attachment loss higher than 6 mm in two or more sites and with one or more sites with probing depth ≥ 5mm were diagnosed as having periodontitis. Necrotizing periodontitis was identified by the presence necrosis of hard and gingival tissues in the periodontium and rapid tooth loss.
3. Microbiological parameters
3.1. Clinical Specimens
After probing, clinical specimens were collected from three periodontal sites: from two deepest periodontal sites and from area with the most significant attachment loss among the lower central incisors. Before sample collection, the supragingival biofilm was removed using a scaling (Trinity Ind. Co. Ltd., São Paulo, SP, Brazil). Then, three sterilized paper points were introduced into the apical region of the periodontal pocket or inflamed gingival crevice for 60 seconds. The paper points were then transferred to 2.0 ml of VMGA III (11) and processed within 2 h.
3.2. Microorganisms isolation and identification
Specimens were serially diluted and 0.1 ml of each dilution was transferred to Sabouraud dextrose agar with 100 mg/ml of chloramphenicol, peptone water and ethyl violet azide broth (EVA broth). Plates and tubes were incubated at room temperature, for 3 or 7 days. The bacterial growth in peptone water was transferred (0.1 ml) to eosin methylene blue agar (EMB), Salmonella-Shigella agar (SS) and brilliant green (BG) agar. Bacterial growth in EVA broth was transferred (0.1 ml) to bile esculin agar. Plates were incubated at 37ºC, for 24-48 h.
The yeast identification was performed by the carbon and nitrogen assimilation. Fermentation of dextrose, maltose and sucrose and germ tube formation at 37ºC and at 39ºC was also performed. Colonial morphology on CHROM agar Candida (MastDiagnostica, Paris, France), and growth at 37ºC and 42ºC were also evaluated. Bacterial species were identified according to their colonial characteristics, Gram-staining, growth at 10% sodium chloride, and biochemical tests using the PI-20E commercial kit (bioMèrieux).
4. Statistical analysis
The design of the study and statistical analysis were carried out using the SPSS-PC/Windows version 12.0 software package (SPSS Inc., Chicago, IL, USA) and the parameters such as sex, periodontal parameters, age and occurrence of the pathogens in the microbiota were evaluated. The relationship between the occurrence of yeasts and enteric bacteria and periodontal clinical parameters (plaque index, gingival index, bleeding on probing, attachment loss) was determined to all patients. The statistical analyses have included descriptive analysis (median and standard deviation). The Kruskal-Wallis, Mann-Whitney and Chi-square tests were employed to determine the presence of significant differences and correlations between sex, age, periodontal parameters and presence of fungi and enteric bacteria. Statistical significance was defined as P < 0.05
RESULTS
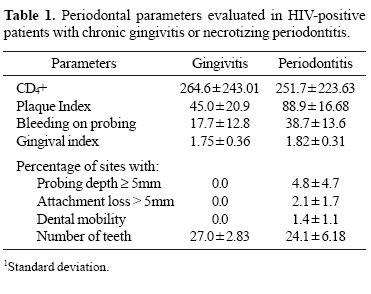
The results of this study show an evident homogeneity among analyzed groups particularly to the age (Mann-Whitney test, P = 0.105). Clinical parameters and the CD4+ counts of the patients are showed in Table 1. Gingivitis and periodontitis patients displayed similar scores of CD4+. Patients with necrotizing periodontitis showed statistically significant differences in comparison to patients with gingivitis, particularly with a high deterioration of the periodontal attachment and oral hygiene (Kruskal-Wallis test, P = 0.046), probing depth (Chi-square test, P = 0.041), gingival index (Kruskal-Wallis test, P < 0.001) and bleeding on probing (Mann-Whitney test, P = 0.031).
All the yeasts were identified as Candida albicans but this yeast was isolated in low number from patients with CD4+ > 200/mm3 when compared with HIV-patients with CD4+ < 200/mm3 (Chi-square test, P = 0.0279). The extension of the periodontal damage seems to be statistically related with the occurrence and levels of C. albicans in subgingival samples (Chi-square test, P = 0.034) (Table 2). This yeast was recovered in higher values from periodontitis (40%) than gingivitis (7.5%) patients. On the other hand, sex (P = 0.259) or age (P = 0.781) did not influence the occurrence of C. albicans. Moreover, the number of C. albicans isolated from gingivitis was 1.3 x 10 ± 1.3 x 10 CFU, while from necrotizing periodontitis was 3.8 x 10 ± 7.8 x 10 CFU.
Enterobacteriaceae and enterococci were isolated only from HIV-patients with necrotizing periodontitis (P < 0.001), regardless their immunological status (CD4+ counts). Enteric bacteria such as Enterobacter sakazakii, Enterobacter cloacae, Serratia liquefaciens, Klebsiella oxytoca and Enterococcus sp. (Table 2) were recovered in 32.5% necrotizing periodontitis samples.
DISCUSSION
Severe periodontal diseases have been associated to alterations in the host' immune system (1,20), which can predispose to gingivitis and periodontitis development. Moreover, the relevance of immune system in the periodontal tissues protection is documented and the impairment of this system could aggravate periodontal status (6). Associations between periodontal status and presence of the opportunistic infections have not been established, and both gingivitis and periodontitis are often found in symptomatic HIV-patients, where may acquire necrotizing or aggressive characteristics. This can be reflects of the role that CD4+ cells play in the HIV-infection progression, which are scarce in peripheral-blood of these patients.
The presence of the anaerobic bacteria, enterobacteria and superinfecting microorganisms may be relevant in the gingivitis and periodontitis etiology, especially in immunodeficent patients (1,14,16). However, the role of the enteric bacteria plays in the periodontal diseases etiology remains unclear, and it must be an alert to clinicians who use systemic antibiotics, such as ciprofloxacin as an adjunct in the periodontitis treatment in such patients (18).
In HIV-positive patients, the occurrence of enteric bacteria and other superinfecting microorganisms and the C. albicans proliferation may also be related to opportunistic viral infections, such as herpesvirus, which are common in these patients producing immunosuppressive factors and cytokines, leading to overgrowth of pathogenic bacteria and fungi (6).
Certainly, the peculiarities and differences among microbiota associated to gingivitis and necrotizing periodontitis of the HIV-positive patients can be influenced by medications, which are employed in the treatment of several opportunistic infections during the HIV-infection. Since all the patients received daily anti-retroviral or antibiotic therapy, this gives opportunity to colonization and dissemination of superinfecting microorganisms and fungi (1). Moreover, these data can represent an intense selective pressure of drugs on the resident microbiota and host immune response. Antimicrobial drugs and the immune deficiency are a way to the periodontal tissues colonization by non-oral organisms such as enteric rods. Moreover, they also give environmental conditions to the oral opportunistic microbiota, such as C. albicans (8).
The occurrence of yeasts and enterobacteria associated to periodontal diseases in HIV-patients (Table 2) is in accordance with Moore et al. (12). However, Dios et al. (7) isolated yeasts and enterobacteria in 68% of the oral smooth mucosa. A relationship between occurrence of Candida spp. and inflammation of the periodontal tissues was observed by Wang et al. (21). It is early to associate the presence of C. albicans in periodontal HIV-positive patients with gingival or periodontal inflammation, since in these patients, factors such as immune suppression, use of antibiotics and illegal intravenous drugs, may induce the periodontal tissues colonization for this yeast.
In our study, it was verified a close relationship between C. albicans, necrotizing periodontitis and CD4+ counts. Of the 19 Candida-positive patients, 14 were HIV-patients with CD4+ < 200 cells/mm3 and 16 of them displayed necrotizing periodontitis. Of the 16 necrotizing periodontits patients and harboring C. albicans, 11 showed CD4+ < 200 cells/mm3. The inflammation of the periodontal tissues colonized by C. albicans can be associated to the hyphae development invading the periodontal connective tissues and producing several degrees of inflammation (9).
In addition, the presence of the enteric organisms in low numbers, in oral cavity, is relevant since the periodontium may act as a reservoir to opportunistic microorganisms, especially in patients with immunodeficiency. In this study, the occurrence of enterobacteria was high in HIV-patients with necrotizing periodontitis and displaying CD4+ < 200/mm3. The role of the superinfecting microorganisms in patients with necrotizing periodontitis, remains unclear. Our data showed a correlation between the presence of opportunistic pathogens, attachment loss and presence of necrosis in periodontal tissues (P = 0.031).
Botero et al. (4) detected enteric microorganisms in higher numbers in HIV-positive patients with periodontitis than in HIV-negative patients. However, no ecological relationship between these microbial groups has been established. Since Enterobacteriaceae are not considered as important members of the oral microbiota, it is possible that disequilibrium of the oral microbiota collaborate to an establishment in oral biofilm (17).
In HIV patients with periodontitis is often observed an aggressive and necrotizing character, producing bad breath, pain and rapid conjunctive attachment loss (13). Data in literature show that enteric rods and cocci may play a role in the development of these pathologies (4,19), due to their wide scope of virulence factors that confer the ability to adhere and invade the host's tissues, besides the induction of local inflammatory processes in periodontal tissue (13).
ACKNOWLEDGEMENTS
Authors thank Dr. Nwaokorie Francisca Obiageri for the English review and Mrs. Zulmira Alves de Souza for her technical support. This study was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP).
Submitted: September 20, 2007; Returned to authors for corrections: February 08, 2008; Approved: May 04, 2008.
* Corresponding Author. Mailing address: Anaerobe Laboratory, Department of Microbiology, University of São Paulo. Av. Prof. Lineu Prestes 1374, 05508-900, São Paulo, SP, Brazil. Phone/fax: +55-11-3091-7344/3091-7354. E-mail: mariojac@usp.br
- 1. Aas, J.A.; Barbuto, S.M.; Alpagot, T.; Olsen, I.; Dewhirst, F.E.; Paster, B.J. (2007). Subgingival plaque microbiota in HIV positive patients. J. Clin. Periodontol, 34 (3), 189-195.
- 2. Alpagot, T.; Duzgunes, N.; Wolf, L.F.; Lee, A. (2004). Risk factors for periodontitis in HIV patients. J. Periodontal. Res, 39 (3), 149-157.
- 3. Alves, M.; Mulligan, M; Passaro, D.; Gawell, S.; Navazesh, M.; Phelan, J.; Greenspan, D.; Greenspan, J.S. (2006). Longitudinal evaluation of loss of attachment in HIV-infected women compared to HIV-uninfected women. J. Periodont, 77 (5), 773-779.
- 4. Botero, J.E.; Arce, R.M.; Escudero, M.; Betancourth, M.; Jamarillo, H.; Contreras, A. (2007). Frequency of detection of periodontopathic and superinfecting bacteria in HIV-positive patients with periodontitis. J. Int. Acad. Periodontol, 9 (1), 13-18.
- 5. Chintu, L.; Bhat, G.J.; Walker, A.S.; Mulenga, V.; Sinyinza, F.; Lishimpi, K.; Farrelly, L.; Naganson, N.; Zumla, A.; Gillespie, S.H.; Nunn, A.J.; Gibb, D.M. (2004). Co-trimoxazole as prophylaxis against opportunistic infections in HIV-infected Zambian children (CHAP): a double-blind randomized placebo-controlled trial. Lancet, 364 (9448), 1865-1871.
- 6. Cobb, C.M.; Ferguson, B.L.; Keselyak, M.T.; Holt, L.A.; MacNeill, S.R.; Rapley, J.W. (2003). A TEM/SEM study of the microbial plaque overlying the necrotic gingival papillae of HIV-seropositive necrotizing ulcerative periodontitis. J. Periodont. Res, 38 (2), 147-55.
- 7. Dios, P.D.; Feijoo, J.; Alvarez, F.J.; Castro, M.; Varela, J. (1993). Oral Enterobacteriaceae in HIV patients. I International Conference of AIDS, Berlin, Ger, p. 436.
- 8. Helovuo, H.; Hakkarainen, K.; Paunio, K. (1993). Changes in the prevalence of subgingival enteric rods, staphylococci and yeasts after treatment with penicillin and erythromycin. Oral Microbiol. Immunol., 8 (2), 75-9.
- 9. Järvensivu, A.; Hietanen, J.; Rautenaa, R.; Sorsa, T.; Richardson, M. (2004). Candida yeasts in chronic periodontitis tissues and subgingival, microbial biofilms in vivo. Oral Dis, 10 (2), 106-12.
- 10. Machtei, E.E.; Christersson, L.A.; Grossi, S.G.; Dunford, R.; Zambon, J.J.; Genco, R.J. (1992). Clinical criteria for the definition of established periodontitis. J. Periodontol, 63 (3), 207-215.
- 11. Möller, Å.J.R. (1966). Microbial examination of root canals and periapical tissues of human teeth. Odontologisk Tidskrift 74 (special issue), 1-380.
- 12. Moore, L.V.H.; Moore, W.E.C.; Riley, C.; Brooks, C.N.; Burmeister, J.A.; Smibert, R.M. (1993). Periodontal microflora of HIV positive subjects with gingivitis or adult periodontitis. J. Periodontol., 64 (1), 48-56.
- 13. Narani, N.; Epstein, J.B. (2001). Classifications of oral lesions in HIV infection. J. Clin. Periodontol., 28 (2), 137-45.
- 14. Patel, M.; Coogan, M.M.; Galpin, J.S. (2003). Periodontal pathogens in subgingival plaque of HIV-positive subjects with chronic periodontitis. Oral Microbiol Immunol, 18 (3), 199-201.
- 15. Ranganathan, K.; Hemalatha, R. (2006). Oral lesions in HIV infection in developing countries: an overview. Adv. Dent. Res, 19 (1), 63-68.
- 16. Robinson, P.G.; Adegboye, A.; Rowland, R.W.; Yeung, S.; Johnson, N.W. (2002). Periodontal diseases and HIV infection. Oral Dis., 8(suppl. 2), 144-50.
- 17. Sixou, M. (2003). Diagnostic testing as a supportive measure of treatment strategy. Oral Dis, 9 (suppl. 1), 54-62.
- 18. Slots, J.; Ting, M. (2002). Systemic antibiotics in the treatment of periodontal disease. Periodontol. 2000, 28 (1), 106-76.
- 19. Souto, R.; Andrade, A.F.B.; Uzeda, M.; Colombo, A.P.V. (2006). Prevalence of non-oral pathogenic bacteria in subgingival biofilm of subjects with chronic periodontitis. Braz. J. Microbiol., 37 (3), 208-15.
- 20. Walsh, M.C.; Kim, N.; Kadono, Y.; Rho, J.; Lee, S.Y.; Lorenzo, J.; Choi, J. (2006). Osteoimmunology: interplay between the immune system and bone metabolism. Annu Rev. Immunol., 23 (1), 33-63.
- 21. Wang, J.; Ohshima, T.; Yasunari, U.; Namikoshi, S.; Yoshihara, A.; Miyazaki, H.; Maeda, N. (2006). The carriage of Candida species on the dorsal surface of the tongue: the correlation with the dental, periodontal and prosthetic status in elderly subjects. Gerodontology, 23 (3), 157-63.
Publication Dates
-
Publication in this collection
31 July 2008 -
Date of issue
June 2008
History
-
Accepted
04 May 2008 -
Received
20 Sept 2007