Abstract
Our investigation of integrated biological control (IBC) started with an assay testing activity of the predacious yeast Saccharomycopsis crataegensis UFMG-DC19.2 against Penicillium digitatum LCP 4354, a very aggressive fungus that causes postharvest decay in oranges. Under unfavourable environmental conditions, the yeast showed a high potential for control (39.9% disease severity reduction) of this fungus. This result was decisive for the next step, in which S. crataegensis was tested in association with sodium bicarbonate salt, a generally regarded as safe (GRAS) substance. The yeast was able to survive at different concentrations of the salt (1%, 2% and 5%), and continued to grow for a week at the wound site, remaining viable at high population for 14 days on the fruit surface. The yeast alone reduced the severity of decay by 41.7% and sodium bicarbonate alone reduced severity of decay by 19.8%, whereas the application of both led to a delay in the development of symptoms from 2 to 10 days. Ingredients of the formulations were not aggressive to fruits since no lesions were produced in control experiments.
biological control; Saccharomycopsis; Penicillium; postharvest disease; orange
ENVIRONMENTAL MICROBIOLOGY
Integrated control of Penicillium digitatum by the predacious yeast Saccharomycopsis crataegensis and sodium bicarbonate on oranges
R. S. PimentaI,* * Corresponding Author. Mailing address: Laboratório de Microbiologia Ambiental e Biotecnologia, C. P. 114, Universidade Federal do Tocantins, Palmas TO, 77.020-210, Brazil.; Phone/fax: 55+63+32328007.; E-mail: pimentars@uft.edu.br ; J. F. M. SilvaI; C. M. CoelhoI; P. B. MoraisI; C. A. RosaII; A. Corrêa JrII
ILaboratório de Microbiologia Ambiental e Biotecnologia, Universidade Federal do Tocantins, Palmas TO, Brasil
IIDepartamento de Microbiologia, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil
ABSTRACT
Our investigation of integrated biological control (IBC) started with an assay testing activity of the predacious yeast Saccharomycopsis crataegensis UFMG-DC19.2 against Penicillium digitatum LCP 4354, a very aggressive fungus that causes postharvest decay in oranges. Under unfavourable environmental conditions, the yeast showed a high potential for control (39.9% disease severity reduction) of this fungus. This result was decisive for the next step, in which S. crataegensis was tested in association with sodium bicarbonate salt, a generally regarded as safe (GRAS) substance. The yeast was able to survive at different concentrations of the salt (1%, 2% and 5%), and continued to grow for a week at the wound site, remaining viable at high population for 14 days on the fruit surface. The yeast alone reduced the severity of decay by 41.7% and sodium bicarbonate alone reduced severity of decay by 19.8%, whereas the application of both led to a delay in the development of symptoms from 2 to 10 days. Ingredients of the formulations were not aggressive to fruits since no lesions were produced in control experiments.
Key words: biological control, Saccharomycopsis, Penicillium, postharvest disease, orange.
INTRODUCTION
Biological control of postharvest diseases has been used as an alternative for synthetic fungicides mainly against fruit decays (15, 18). The mechanism of this biological control is based on ecological interactions, such as competition for space and nutrients, mycoparasitism, antibiosis, predation or induction of plant defenses (15, 19, 20, 23). Traditionally, yeasts are the organism used most frequently as biocontrol agents due to their fast colonization of plant surfaces and their production of extracellular polysaccharides that enhance their survival and to restrain both colonization sites and the flow of germination cues to pathogen propagules (13). They use available nutrients to rapidly proliferate and to stay viable under different environmental conditions, seldom produce chemical antagonistic substances and are less affected by pesticides than filamentous (4, 9, 10, 14, 16, 23). However, for the most effective control of decays, biological control can be integrated with other alternative methods. This approach includes the utilization of a biological control agent in combination with substances Generally Regarded as Safe (GRAS) or other methods. Sodium carbonate, sodium bicarbonate, calcium chloride, and ethyl alcohol are GRAS substances frequently utilized in IBC programs (15). Other strategies utilize physical methods, such as UV irradiation, hot or cold water treatments, and modified atmosphere packaging (3, 18, 29). Some types of pathogens are satisfactorily controlled only when the additive or synergistic effect of yeast and GRAS substances take place. Primary pathogens, like Penicillium, Aspergillus and Rhizophus species, are non-specific and very aggressive, and because of this they are difficult to control. Penicillium digitatum grows saprophytically in soil and it is commonly isolated from plant material. It causes green mold of oranges, and is one of the most important disease of citrus fruit (25). The objectives of the present work was to study control of green mold of citrus after harvest using Saccharomycopsis species, determining population dynamics of S. crataegensis in fruit wounds, and evaluating efficacy of combination of the yeasts with sodium bicarbonate in controlling green mold caused by P. digitatum on oranges at room temperature.
MATERIALS AND METHODS
Pathogen
P. digitatum (LCP 004354) was obtained as a gracious gift from the Laboratoire Cryptogamie - Paris and maintained on Potato Dextrose Agar (PDA) slants at 4ºC under mineral oil. To prepare conidial suspension the culture was grown on PDA for 7 -14 days and washed with 10 mL of GY broth (glucose 1% and yeast extract 0.01%) added with 20 µL of Tween 20 to harvest conidia and concentration of the conidia was adjusted with haemocytometer resulting in five ml of the suspension containing 104 conidia/ml. This conidia suspension was used to inoculate orange fruits.
Antagonistic yeast
The antagonistic yeast S. crataegensis UFMG-DC19.2 was isolated from a tropical fruit (Acrocomia aculeata) collected in the Ecological Reserve of the Universidade Federal de Minas Gerais, Belo Horizonte, Brazil. The cultures were purified on YM agar (0.3% yeast extract, 0.3% malt extract, 0.5% peptone, 1% glucose and 2% agar) and maintained on GYMP broth (2% glucose, 0.5 yeast extract, 1% malt Extract Control of P. Digitatum in oranges and 0.2% Na2PO4) supplemented with 20% glycerol frozen at 80ºC. The yeasts were grown at 25ºC for 48 h on YM agar, collected with a bacteriological loop and suspended in 10 ml of GY broth supplemented with 500 µl of Tween 20. Cell concentration was determined with a haemocytometer and adjusted to108 cells/ml (23).
Fruits
Ripe oranges [Citrus sinensis (L.) Osbeck variety Pêra Rio] of approximately 20 cm in circumference were acquired from a local commercial orchard and selected for lack of lesions or injuries. The fruits were superficially disinfected by immersion in 1% sodium hypochlorite for 3 min, rinsed with sterile water and dried in a sterile chamber. When dry, a wound was made around the entire equatorial region of each orange with a sterile blade. The wound was approximately 3 mm wide and 1 mm deep. The pathogen and/or the antagonistic yeast were inoculated into these wounds.
Biological control assay
Aliquots of 200 µl of pathogen suspension were deposited with a sterile pipette on the orange wound immediately after wounding and allowed to dry under aseptic conditions. After pathogen inoculation, 200 µl of a suspension of biocontrol agent was also deposited on the wound. The inoculated fruits were incubated in a 100% RH humidity chamber at 25ºC for 10 days to simulate the best conditions for the growth of the pathogen. Oranges inoculated with the pathogen alone served as a positive control. Negative controls were performed by inoculation of GY without yeast or pathogenic fungus, and the yeast alone. Four fruits arranged in a randomized block design were used per treatment, the treatment being the combined inoculation of pathogen and the yeast strain. All assays were designed in random blocks, with three repetitions. Tukey's test with a 5% confidence interval was used to asses the reduction in disease severity.
Maximum temperature of growth
The yeast was allowed to grow in glass test tubes with YM broth and was incubated at 25, 35, 36 and 37ºC. The growth was verified by turbidity of medium after 48 h of incubation. The experiment was repeated two times.
Compatibility of biocontrol agent with sodium bicarbonate solution
In this experiment one ml of a 10X concentrated sodium bicarbonate solution (SBC) was transferred to glass test tubes with 9 ml of S. crataegensis at a concentration of 106 cells/ml. The final concentrations of SBC were 1, 2 and 5%. The tubes were incubated at 25ºC for 0, 1, 2, 5 and 7 days on a rotary shaker at 50 rpm, after which 100 µl of the culture was plated on YM agar. After incubation for 48 h, the colonies were counted and the results were expressed in colony forming units (cfu)/ml. The experiment was repeated three times.
Integrated Biological Control
The biocontrol tests on fruit were performed by inoculating the pathogen alone or in combination with the yeast, or both. Aliquots of 200 µl of pathogen suspension were deposited with a sterile pipette onto the orange wound immediately after wounding, allowed to dry, and 200 µl of a suspension of S. crataegensis and/or sodium bicarbonate at 5% was also deposited onto the wound. The inoculated fruits were incubated in a 100% humidity chamber at 25ºC for 10 days. The controls included inoculation with pathogen alone, GY without the yeast or pathogenic fungus, the yeast alone, sodium bicarbonate at 5%, and the yeast and sodium bicarbonate at 5% together. There were three replicates of four fruits arranged in a randomized block design per treatment.
Assessment of lesion
The percentage of disease severity reduction (DSR%) was calculated by the equation:
Where DSc = lesion area on the positive control (pathogen alone) and DSt = lesion area on the treated fruit (1). Only lesion originating from the mechanically wounding were used in the disease assessment.
Antagonist recovery
To assess the ability of S. crataegensis to survive on the surface of oranges, wounds of fruits wounded as described above were treated with 200 µl of GY broth supplemented with 5% sodium bicarbonate and 2x108 cells/ml of the yeast. The survival of the yeast was determined immediately after application and after 4, 7 and 14 days of incubation at 25ºC in a 100% RH. To recover the yeast from fruits, portions of wounds from the treated oranges were removed with a 1.0 cm2 diameter cork borer, transferred to tubes containing GY broth, agitated in a vortex and serially diluted to 10-2, 10-4 and 10-6, followed by plating 100 µl of the suspension in triplicate on YM agar supplemented with 200 mg/l of chloramphenicol. The colonies were counted after 48h incubation at 25ºC.
RESULTS AND DISCUSSION
The Pera Rio orange variety has been shown to be sensitive to infection caused by P. digitatum (8). Our results showed that the yeast significantly reduce the disease severity (DSR%). The biocontrol efficacy of S. crataegensis was 60.1% (Fig. 1). The positive control containing only P. digitatum showed 100% of disease incidence. These results can be considered satisfactory, since incubation conditions (25ºC in high humidity) and the time of inoculation (inoculation of P. digitatum before the yeast) strongly favored the pathogen. Because some of the post-harvest pathogens that affect fruits initiate their disease cycle even before harvest this antagonist may have potential for controlling these type infections. The experiments were designed to simulate the post-harvest environmental situation and showed that S. crataegensis has potential for use in IBC under commercial conditions, since very few studies with yeast antagonists were conducted under such adverse conditions. Usually, the combination of bicarbonate with biological control agents has been observed to be synergistic (11). However, some studies have reported an inhibitory effect of sodium bicarbonate not only on pathogens but also on biocontrol agents (11, 17, 27). Most of the experiments about the combination of sodium bicarbonate and yeast utilize concentrations under 2% of the salt. The survival of S. crataegensis in formulations with 1, 2, and 5% sodium bicarbonate can be considered very satisfactory, since the yeast remained viable at all these concentrations (Fig. 2). After one week of incubation, the populations of the yeast were very similar to those at the beginning of the experiment. Strong resistance to SBC is an important characteristic of this yeast, because it allows for the use of SBC at higher concentrations, which may have stronger effect on reduction of fruit decay.
The ability of antagonists to survive at sufficient populations on the fruit surface after their application is very important for efficacy and persistence of control (2). The ability of S. crataegensis to rapidly colonize and to stay viable in fruit wounds (the major points of entry for pathogens) at room temperature (25ºC) is a very important characteristic. Another important characteristic of S. crataegensis was the absence of growth at 37ºC, which eliminates this yeast as a potential human pathogen (6, 12). The wounds, which inevitably occur during harvest, transport, and handling, not only damage the fruit (22), but also provide pathways for pathogen invasion, especially for the primary pathogens (15). The yeast S. crataegensis was re-isolated from treated oranges throughout the 2 week experiment (Fig. 3). However, similar to other yeasts utilized in biological control of fruit decays that only work at high cell concentrations (21, 28), in this study the antagonistic yeast only produced an efficient control of molds when inoculated at a concentration of 108 cells/ml (data not shown). Despite high density, application of this yeast alone to oranges did not result in any necrosis, chlorosis or fruit decay which is another important characteristic of a biocontrol agent.
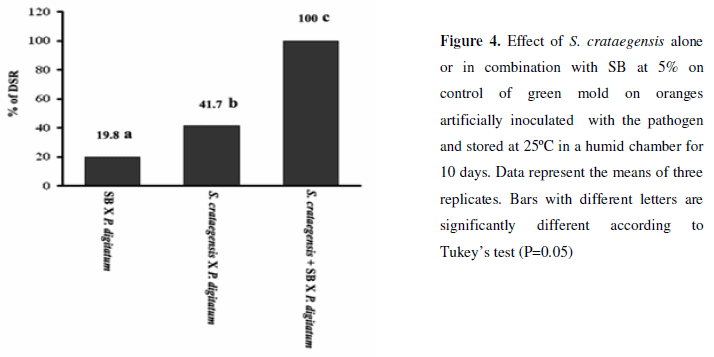
Results from the IBC experiment are very promising, especially under unfavorable environmental conditions such as room temperatures and high humidity. The pathogen alone infected all of the oranges within 4 days. The 5% sodium bicarbonate treatment alone reduced the disease severity by 19.8%, and the yeast alone reduced the disease severity in 41.7% (Fig. 4). When these treatments were combined initially, total control of the decay was achieved and the first symptoms started to appear only on the tenth day after inoculation. The development of a biological control strategy for citrus decay caused by wound invading P. digitatum is very difficult, mainly because Penicillium spp. are aggressive pathogens and are good competitors. Pilot test in citrus packinghouses indicated that antagonist alone cannot provide adequate control and must be combined with diluted fungicides or other methods of control (7).
In the biocontrol tests the yeast significantly reduced decay severity and incidence on mature oranges. Decays originating from natural infection were also observed and were quite variable but they diminished after treatment with S. crataegensis (data not shown).
An antagonist yeast is very promising if it can be combined with routine postharvest treatments (29). The best time for the postharvest application of a water-based treatment to the fruit should be during the hydrocooling, drenching or washing processes (17). Citrus fruits are usually treated with chlorinated water after harvest to reduce decays, but the use of chlorine on fruits and vegetables is banned in some countries due to deleterious effect on human health resulting from its reaction with organic matter that lead to the formation of carcinogenic compounds (5). Accumulation of these substances also causes environmental pollution (24, 26). The findings provide a powerful incentive for development of this yeast as an alternative to green mold control by P. digitatum in postharvest orange fruits.
ACKNOWLEDGMENTS
This work was funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico of Brazil (CNPq - PADCT process number 62.0477/98-9), Fundação de Amparo a Pesquisa de Minas Gerais (FAPEMIG process number CBB 1207/98). We acknowledge Rachel Basques Caligiorne for kindly providing the mold strains and Dr. W. Janisiewicz for kindly revising of the manuscript.
Submitted: January 29, 2009; Returned to authors for corrections: May 08, 2009; Approved: November 07, 2009.
- 1. Assis, S.M.P.; Mariano, R.L.R.; Michereff, S.J.; Silva, G.; Maranhão, E.A.A. (1999). Antagonism of yeasts Xanthomonas campestris PV. Campestris on cabbage phylloplane in field. Rev. Microbiol. 30, 191-195.
- 2. Benbow, J.M.; Sugar, D. (1999). Fruit surface colonization and biological control of postharvest diseases of pear by preharvest yeast applications. Plant Dis. 83, 839-844.
- 3. Capdeville, G.; Souza, J.M.T.; Santos, J.R.P.; Miranda, S.P.; Caetano, A.R.; Torres, F.A.G. (2007) Selection and testing of epiphytic yeasts to control anthacnose in post-harvest of papaya fruit. Scien. Horticulturae 111, 179-185.
- 4. Cartwright, D.K.; Spurr, Jr.H.W. (1998). Biological control of Phytophthora parasitica var. nicotianae on tobacco seedlings with nonpathogenic binucleate Rhizoctonia fungi. Soil Biol. Biochem. 30, 1879-1884.
- 5. Chanchaichaovivat, A.; Ruenwongsa, P.; Panijpan, B. (2007). Screening and identification of yeast strains from fruits and vegetables: Potential for biological control of postharvest chilli anthracnose (Colletotrichum capsici). Biol. Control 42. 326-335.
- 6. Chand-Goyal, T.; Spotts, R.A. (1997). Biological control of postharvest diseases of apple and pear under semi-commercial and commercial conditions using three saprophytic yeasts. Biol. Control 10. 199-206.
- 7. Droby, S.; Cohen, L.; Daus, A.; Weiss, B.; Horev, B.; Chalutz, E.; Katz, H.; Keren-Tzur, M. & Shachnai, A. (1998). Commercial testing of aspire: a yeast preparation for the biological control of postharvest decay of citrus. Biol. Control 12. 97-101.
- 8. Droby, S.; Hofstein, R.; Wilson, C.L.; Wisniewski, M.; Fridlender, B.; Cohen, L.; Weiss, B.; Daus, A.; Timar, D.; Chalutz, E. (1993). Pilot testing of Pichia guilliermondii: a biocontrol agent of postharvest diseases of citrus fruit. Biol. Control 3. 47-52.
- 9. Droby, S.; Lischinski, S.; Cohen, L.; Weiss, B.; Daus, A.; Chand-Goyal, T.; Eckert, J.W.; Manulis, S.M. (1999). Characterization of an Epiphytic Yeast Population of Grapefruit Capable of Suppression of Green Mold Decay Caused by Penicillium digitatum. Biol. Control 16. 27-34.
- 10. Droby, S.; Vinokur, V.; Weiss, B.; Cohen, L.; Daus, A.; Gold-Schmidt, E.E.; Porat, R. (2002). Introduction of resistance to Penicillium digitatum in grapefruit by the yeast biocontrol agent Candida oleophila. Phytopathol. 92. 393-399.
- 11. Gamagae, S.U.; Sivakumara, D.; Wilson, R.S.; Wijesundera, R.L.C. (2003). Use of sodium bicarbonate and Candida oleophila to control anthracnose in papaya during storage. Crop Prot. 22. 775-779.
- 12. Hazen, K.C. (1995). New and emerging yeast pathogens. Clin. Microbiol. 8. 462-478
- 13. Janisiewicz, W.;. (1988). Biological control of diseases of fruit. In: Mukergi, K.G., Garg, K.L. (eds). Biocontrol of Plant Diseases II Boca Raton:,CRC Press; p. 153-165.
- 14. Janisiewicz, W.J. (1991). Control of postharvest diseases of fruits with biocontrol agents. In: Bay-Petersen, J. (eds). The Biological Control of Plant Diseases Taipei, Cent. Asian Pac. Reg; p. 56-68.
- 15. Janisiewicz, W.J.; Korsten, L. (2002). Biological control of postharvest disease of fruits. Annu. Rev. Phytopathol 40. 411-41.
- 16. Janisiewicz, W.J.; Tworkoski, T.J.; Kurtzman, C.P. (2001). Biocontrol potential of Metchnikowia pulcherrima strains against blue mold of apple. Biol. Control 91. 1098-1108.
- 17. Karabulut, O.A.; Arslan, U.; Ilhan, K.; Kuruoglu, G. (2005). Integrated control of postharvest diseases of sweet cherry with yeast antagonists and sodium bicarbonate applications within a hydrocooler. Postharvest Biol. Technol 37. 135-141.
- 18. Karabulut, O.A.; Baykal, N. (2004). Integrated control of postharvest diseases of peaches with a yeast antagonist, hot water and modified atmosphere packaging. Crop Prot. 23. 431-435.
- 19. Kavková, M.; Curn, V. (2005). Paecilomyces fumosoroseus (Deuteromycotina: Hyphomycetes) as a potential mycoparasite on Sphaerotheca fuliginea (Ascomycotina: Erysiphales). Mycopathologia 159. 53-63.
- 20. Massart, S.; Jijakli, M.H. (2007). Use of molecular techniques to elucidate the mechanisms of action of fungal biocontrol agents: a review. J. Microbiol. Meth. 69. 229-241.
- 21. MCguire, R.G. (1994). Application of Candida guilliermondii in commercial citrus coatings for biological control of Penicillium digitatum on grapefruits. Biol. Control 4. 1-7.
- 22. Miller, A.R. (2003). Harvest and handing injury: physiology, biochemistry, and deletion. In: Bartz, J.; Brecht, J. (eds). Postharvest Physiology and Pathology of Vegetables New York, Marcel Dekker, p. 177-208.
- 23. Pimenta, R.S.; Silva, F.L.; Silva, J.F.M.; Morais, P.B.; Braga, D.T.; Rosa, C.A.; Corrêa-Jr, A. (2008). Biological control of Penicillium italicum, P. Digitatum and P. Expansum by the predacious yeast Saccharomycopsis schoenii on oranges. Braz. J. of Microbiol 39. 85-90.
- 24. Smith, M.D. (2001). A chlorine sunset? An analysis of the movement to phase out chlorine and chlorinated derivatives. American Inst. Chem. Eng 1-4.
- 25. Stange, R.R.; Midland, S.L.; Sims, J.J.; Mccollum, T.G. (2002). Differential effects of citrus peel extracts on growth of Penicillium digitatum, P. italicum, and P. expansum Physiol. Mol. Plant Pathol. 61. 303-311.
- 26. Stringer, R.; Johnston, P. (2002). Chlorine in the environment: an overview of the chlorine industry. Environ. Sci. Pollut. Res. 9. 356-357.
- 27. Yaoa, H.; Tian, S.; Wang, Y. (2004). Sodium bicarbonate enhances biocontrol efficacy of yeasts on fungal spoilage of pears. Int. J. Food Microbiol. 93. 297-304.
- 28. Zhang,, Hong-yin.; Zheng, Xiao-dong.; Xi, Yu-fang. (2005). Biological control of postharvest blue mold of oranges by Cryptococcus laurentii (Kufferath) Skinner. Biol. Control 50. 331-342.
- 29. Zhang, S.; Schisler, D.A.; Boehm, M.J.; Slininger, P.J. (2007). Utilization of chemical inducers of resistance and Cryptococcus flavescens OH 182.9 to reduce Fusarium head blight under greenhouse conditions. Biol. Control. 42. 308-315.
Publication Dates
-
Publication in this collection
16 Apr 2010 -
Date of issue
June 2010
History
-
Accepted
07 Nov 2009 -
Reviewed
08 May 2009 -
Received
29 Jan 2009