Abstract
Crude extracts and fractions of five species of Polygala - P. campestris, P. cyparissias, P. paniculata, P. pulchella and P. sabulosa - were investigated for their in vitro antifungal activity against opportunistic Candida species, Cryptococcus gattii and Sporothrix schenckii with bioautographic and microdilution assays. In the bioautographic assays, the major extracts were active against the fungi tested. In the minimal concentration inhibitory (MIC) assay, the hexane extract of P. paniculata and EtOAc fraction of P. sabulosa showed the best antifungal activity, with MIC values of 60 and 30 µg/mL, respectively, against C. tropicalis, C. gattii and S. schenckii. The compounds isolated from P. sabulosa prenyloxycoumarin and 1,2,3,4,5,6-hexanehexol displayed antifungal activity against S. schenckii (with MICs of 125 µg/mL and 250 µg/mL, respectively) and C. gattii (both with MICs of 250 µg/mL). Rutin and aurapten isolated from P. paniculata showed antifungal activity against C. gattii with MIC values of 60 and 250 µg/mL, respectively. In the antifungal screening, few of the isolated compounds showed good antifungal inhibition. The compound α-spinasterol showed broad activity against the species tested, while rutin had the best activity with the lowest MIC values for the microorganisms tested. These two compounds may be chemically modified by the introduction of a substitute group that would alter several physico-chemical properties of the molecule, such as hydrophobicity, electronic density and steric strain.
Polygalaceae; Polygala species; antifungal activity; rutin; α-spinasterol
MEDICAL MICROBIOLOGY
Antifungal activity of five species of Polygala
Susana JohannI; Beatriz G. MendesII; Fabiana C. MissauII; Maria A. de ResendeI; Moacir G. PizzolattiII,* * Corresponding Author. Mailing address: Department of Microbiology, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brazil.; Tel/Fax.: +55 48 3721 6844 FAX: +55 48 3721 6888.; E-mail: mgpizzo@qmc.ufsc.br
IDepartamento de Microbiologia, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil
IIDepartamento de Química, Universidade Federal de Santa Catarina, Florianópolis, SC, Brasil
ABSTRACT
Crude extracts and fractions of five species of Polygala - P. campestris, P. cyparissias, P. paniculata, P. pulchella and P. sabulosa - were investigated for their in vitro antifungal activity against opportunistic Candida species, Cryptococcus gattii and Sporothrix schenckii with bioautographic and microdilution assays. In the bioautographic assays, the major extracts were active against the fungi tested. In the minimal concentration inhibitory (MIC) assay, the hexane extract of P. paniculata and EtOAc fraction of P. sabulosa showed the best antifungal activity, with MIC values of 60 and 30 µg/mL, respectively, against C. tropicalis, C. gattii and S. schenckii. The compounds isolated from P. sabulosa prenyloxycoumarin and 1,2,3,4,5,6-hexanehexol displayed antifungal activity against S. schenckii (with MICs of 125 µg/mL and 250 µg/mL, respectively) and C. gattii (both with MICs of 250 µg/mL). Rutin and aurapten isolated from P. paniculata showed antifungal activity against C. gattii with MIC values of 60 and 250 µg/mL, respectively. In the antifungal screening, few of the isolated compounds showed good antifungal inhibition. The compound α-spinasterol showed broad activity against the species tested, while rutin had the best activity with the lowest MIC values for the microorganisms tested. These two compounds may be chemically modified by the introduction of a substitute group that would alter several physico-chemical properties of the molecule, such as hydrophobicity, electronic density and steric strain.
Key words: Polygalaceae, Polygala species, antifungal activity, rutin, α-spinasterol
INTRODUCTION
In recent years, fungal infections have emerged as a major cause of disease and mortality, largely because of the growing population of immunocompromised patients. In patients affected by AIDS, opportunistic yeasts of the genus Candida, such as C. albicans, have accounted for approximately 60% of clinical isolates. The appearance of other species of this genus as agents of candidosis may reflect the selection of species that are less susceptible to the action of antifungal agents (39). Approximately 90% of AIDS patients present with oropharyngeal candidosis at least once in the course of the disease, and two-thirds of these patients display the same characteristic at the onset of the symptoms (4).
On the other hand, Cryptococcus neoformans, the major cause of meningitis in AIDS patients, has been identified as the fourth most common cause of life-threatening infection in AIDS patients (36), affecting a more limited percentage (6-8%) (4). Cryptococcus gattii primarily infects otherwise healthy individuals, but serotype C isolates of C. gattii were found to be implicated in HIV-associated infections in California, Botswana and Malawi (6). Sporothrix schenckii is the etiological agent of sporotrichosis, a subcutaneous mycosis with a worldwide distribution that can affect humans and other animals (37).
Human mycoses are often not successfully treated because the available antifungal drugs are either ineffective, produce many adverse effects, fail to prevent recurrence of infection, or lead to the development of drug-resistant strains (11). Therefore, there is a distinct need for the discovery of new, safer and more-effective antifungal agents. Natural products from higher plants may provide a new source of antimicrobial agents with possibly novel mechanisms of action (27). In recent years, antimicrobial properties of medicinal plants have been increasingly reported from different parts of the world (17, 18, 24).
The Polygala genus belongs to the family Polygalaceae, which contains 600 species (11), 19 of which are found in the state of Santa Catarina, Brazil. A chemical investigation of the genus showed the occurrence of a variety of secondary metabolites, such as xanthones (5, 6, 22, 30), saponins (16, 25, 45), oligosaccharides (15, 21), flavonoids (35, 38), coumarins (12, 13, 32, 33) and styrylpyrones (31, 32).
Various pharmacological activities have been reported for some species of Polygala, such as anti-inflammatory activity in P. japonica and P. cyparissias (10, 19), anxiolytic-like effects in P. sabulosa (8, 9) trypanocidal activity in P. sabulosa and P. cyparissias (34, 35), antinociceptive effects in P. sabulosa (40) and P. cyparissias (34, 35), antibacterial activities in P. myrtifolia (20) and so on. Although the biological activities of various Polygala species have been investigated, their antifungal properties have not. Few studies have reported the antifungal activities of species belonging to the genus Polygala, such as P. myrtifolia (26), P. gazensis (3) and P. nyikensis (23).
In this paper, we report an evaluation of the antifungal activities of extracts, fractions and isolated compounds from different species of Polygala, all of them found in Santa Catarina State, Brazil. Extracts and fractions of Polygala cyparissias A. St. - Hil. & Moq., P. campestris Gardner., P. paniculata Lam., P. pulchella A. St. - Hil. & Moq. and P. sabulosa A.W. Bennett were tested against human opportunistic pathogenic fungi using a bioautographic assay. The minimal inhibitory concentrations (MICs) of the most active extracts and isolated compounds were then evaluated with the broth dilution method.
MATERIAL AND METHODS
Plant material
The species P. cyparissias (856 g) and P. paniculata (3500 g) (deposited at the Herbarium FLOR - UFSC, under number 22744, and Herbarium of the Department of Botany - UFPR, under number 26027, respectively) were collected from Daniela beach, Florianópolis in March 2004. The species P. pulchella (1000 g) and P. sabulosa (1700 g) (deposited in the Herbarium of the Department of Botany - UFPR, under numbers 28555 and 19640, respectively) were collected in Rancho Queimado in October 2004. P. campestris (700 g) (a voucher specimen was deposited in the Herbarium of the Department of Botany - UFPR, under number 7393) was collected in November 2003, also in Rancho Queimado. The plants were identified by Dr. Olavo de Araújo Guimarães, Department of Botany, Universidade Federal do Paraná (UFPR) and Dr. Leila da Graça Amaral, Department of Botany, Universidade Federal de Santa Catarina (UFSC).
Preparation of extracts and fractions
The dried and powdered whole plants were extracted, with different methods employed for the different species. P. paniculata was first extracted three times at room temperature with hexane (the "hexane extract"), then with 96% ethanol (EtOH extract), to avoid the formation of coumarinic artifacts such as those previously described by Pizzolatti et al. (33). P. pulchella and P. campestris were successively extracted three times each with dichloromethane (CH2Cl2 extract), ethyl acetate (EtOAc extract) and ethanol (EtOH extract). P. cyparissias and P. sabulosa were extracted exhaustively at room temperature with 96% ethanol (EtOH extract). The EtOH extract obtained from P. sabulosa was then partitioned into n-hexane, dichloromethane (CH2Cl2) and ethyl acetate (EtOAc) soluble fractions. Each extract or fraction was filtered, and the solvent was removed under reduced pressure to yield the respective extract.
Isolation of compounds
During evaporation of the ethanolic extract obtained from P. sabulosa (456.1 g), formation of a solid compound was observed. This compound was separated and washed with appropriate solvent (CH2Cl2 and acetone). The compound was analyzed by IV, 1H and 13C NMR and identified as 1,2,3,4,5,6-hexanehexol, previously described as occurring in this species by Pizzolatti et al. (31). The n-hexane fraction of P. sabulosa was subjected to column chromatography on silica gel eluted with n-hexane/EtOAc gradient. The fractions obtained were then monitored by TLC (Thin-layer chromatography, viewed by spraying with sulfuric vanillin reagent followed by heating at 110 ºC) and similar fractions (fr) were combined. The combined fr. 14-26 were recrystallized from acetone to give the sterol c-spinasterol (91 mg).
The CH2Cl2 soluble fraction was further subjected to silica gel column chromatography eluted with increasing amounts of ethyl acetate in n-hexane to give 95 fr. of 75 mL each. The combined fr. 21-22 were crystallized from hexane/EtOAc (3:1) to yield prenyloxycoumarin (319 g). The combined fr. 25-30 were processed by flash chromatography, yielding dihydrostyryl-2-pyrones 1 (19 mg) and 2 (47 mg). The combined fr. 34-39 were chromatographed on silica gel eluting with a hexane/EtOAc gradient to give the dihydrostyryl-2-pyrone 3 (23 mg) and the styryl-2-pyrone 4 (21 mg). Fraction 49 yielded a yellow powder containing the coumarin scopoletin (17 mg). Fraction 56 was purified by crystallization to yield the styryl-2-pyrone 5 (14 mg).
Detailed nuclear magnetic resonance spectroscopic analysis (1H and 13C) was performed and the physical and spectroscopic data compared to the confirmed structures of 6-methoxy-7-hydroxycoumarin (scopoletin), 6-methoxy-7-prenyloxycoumarin (prenyloxycoumarin), 4-methoxy-6-(11,12-methylenedioxydihydrostyryl)-2-pyrone (DST 1), 4-methoxy-6-(11,12-methylenedioxy-14-methoxy-dihydrostyryl)-2-pyrone (DST 2), 4-methoxy-6-(11,12-methylenedioxy-10,14-dimethoxy-dihydrostyryl)-2-pyrone (DST 3), 4-methoxy-6-(11,12-methylenedioxystyryl)-2-pyrone (STY 4) and 4-methoxy-6-(11,12-methylenedioxy-14-methoxystyryl)-2-pyrone (STY 5). All of these compounds have been previously found in the five Polygala species studied (31, 32).
Precipitation of an amorphous solid from the hexane extract of P. paniculata with partial evaporation of the solvent at controlled temperature was observed. This solid was separated and washed successively with n-hexane and yielded the compound phebalosin (2 g), identified by 1H and 13C NMR (33).
The EtOH extract was chromatographed in a silica gel column using a gradient of n-hexane/EtOAc/ethanol to give 120 fr. of 75 mL each. Fractions 13-16 and 18-20 were rechromatographed in a silica gel column eluting with an n-hexane/EtOAc gradient of increasing polarity to yield the compounds aurapten (87 mg) and  -spinasterol (55 mg), respectively. Fractions 68-75 precipitated a yellow solid, which was separated and washed with methanol to yield the compound rutin (430 mg). These compounds were previously described by Pizzolatti et al. (33) and identified by comparison with physical and spectroscopic data (1H and 13C NMR).
-spinasterol (55 mg), respectively. Fractions 68-75 precipitated a yellow solid, which was separated and washed with methanol to yield the compound rutin (430 mg). These compounds were previously described by Pizzolatti et al. (33) and identified by comparison with physical and spectroscopic data (1H and 13C NMR).
Microorganisms
For evaluation of antifungal activity, strains from the American Type Culture Collection (ATCC), Rochville, MD, USA were used: C. albicans ATCC 18804 C. krusei ATCC 20298, C. tropicalis ATCC 750, C. parapsilosis ATCC 22019, C. glabrata ATCC 2001, S. schenckii ATCC 20679 and C. gattii ATCC 32608. All fungal strains were maintained on Sabouraud dextrose agar (Oxoid, Basingstoke, UK).
Culture media and inoculum
Saboraud dextrose agar was used for the bioautographic tests. The synthetic medium RPMI (Sigma, St. Louis, MO, USA) with L-glutamine was buffered to pH 7.0 with 0.165 m morpholine propanesulfonic acid (MOPS; Sigma). The test was performed according to the CLSI M27-A standard (28) for determination of the Minimal Inhibitory Concentration (MIC). Inoculum of the fungal cultures of Candida spp. and C. gattii were prepared by picking five colonies of 21 mm in diameter from 48 h-old cultures grown at 35ºC. The chosen colonies were suspended in 5 ml of sterile 0.85% saline. For the susceptibility test, the resulting suspension was vortexed for 15 s and the cell density adjusted to 1.5 ± 1.0 x 103 cells/mL by spectrophotometric methods at 530 nm (29). To obtain Y cells of S. schenckii, the fungal culture was cultured on brain heart infusion agar containing 1% glucose (BHID) at 37ºC and maintained by continuous weekly passages. The cells were harvested 4 to 6 days after the last passage and packed into a small volume of sterile saline using a sterile cotton swab. The cells were then suspended in saline and thoroughly vortexed. If large aggregates existed, they were allowed to settle for several minutes, and the supernatant was collected. The suspension was then diluted in RPMI medium to obtain a final inoculum size of approximately a 1x105 - 5x105 cells/mL. For bioautographic tests, the inoculum suspension was appropriately diluted in Saboraud broth to a final concentration of approximately 105 cells/mL.
Antifungal assays
Bioautographic assay: Extracts and fractions were dissolved in dimethylsulfoxide (DMSO) at 100 µg/mL. 20 µL of the solution was applied to TLC plates (plates of silica gel of 60F254, Merck, Darmstadt, Germany) with graduated micropipettes. The plates were then submerged twice for five minutes in fungal suspensions and incubated at 35ºC in a hermetically sealed bell-jar for 48 h for the Candida species and 72 h for C. gattii and S. schenckii.
Subsequently, plates were sprayed with p-iodonitrotetrazolium violet (Sigma®) (INT) (5 mg/mL) and incubated again for 4 hours at 36 ± 1ºC. Inhibition zones were observed, and the diameter of inhibition in millimeters was measured. Solvents and DMSO were used as negative controls, while Amphotericin B (Sigma) was used as the positive control. Samples of the culture medium plus microorganisms were used as the growth control. Tests were performed in triplicate.
Susceptibility test
Broth microdilution testing was performed in accordance with the guidelines in CLSI document M27-A (28). The susceptibility was determined with the microbroth dilution method, performed in sterile flat-bottom 96-well microplates (Difco Laboratories, Detroit, MI, USA).
The extracts and fractions were dissolved in DMSO after addition of RPMI. Later, serial dilutions were made with RPMI, maintaining a constant volume of 1000 µL in each tube. In this way, the extracts were tested at eight concentrations that varied from 7.8 to 1000 µg/mL. From each dilution, brackets of 100 µL were transferred to the microplates.
As a control for growth and sterility control, the RPMI was used without the addition of extract or solvent. The culture medium together with the solvent was used as a control for the toxicity of the solvent. Amphotericin B (Sigma) was included at a concentration of 0.03 at 4.0 µg/mL as the positive antifungal control.
After the assembly of the plates, each fungal strain was inoculated and the plates incubated at 35ºC for 48 hours for the Candida species and 72 hours for C. gattii and S. schenckii.
Tests were performed in triplicate. The endpoints were determined visually through comparison with the endpoints of the drug-free growth control well. The minimum inhibitory concentration (MIC), expressed in µg/mL, was defined as the lowest extract concentration at which the well was optically clear. Extracts, fractions and isolated compounds with MIC values < 1000 µg/mL were considered active.
RESULTS AND DISCUSSION
Physical and spectroscopic data of the compounds isolated from P. sabulosa and P. paniculata are presented bellow.
DST 1 (4-methoxy-6-(11,12-methylenedioxydihydrosty ryl)-2-pyrone): m.p. 137 - 140ºC; IR (KBr, ν cm-1): 3104, 2924, 1710, 1646, 1567, 1489, 1457, 1410, 1242, 1030, 925, 821. 1H NMR (400 MHz, CDCl3) δ (ppm): 6.72 (d, 1H, J=7.2 Hz, H-13), 6.65 (d, 1H, J=1.8 Hz, H-10), 6.60 (dd, 1H, J=7.2 Hz, 1.8 Hz, H-14), 5.92 (s, 2H, OCH2O at C-11 and C-12), 5.70 (sl, 1H, H-5), 5.40 (sl, 1H, H-3), 3.77 (s, 3H, 4-OCH3), 2.87 (t, 2H, H-8), 2.70 (t, 2H, H-7). 13C NMR (100 MHz, CDCl3) δ (ppm): 171.3 (C-2), 165.1 (C-4), 164.4 (C-6), 147.9 (C-11), 146.3 (C-12), 133.8 (C-9), 121.4 (C-14), 108.9 (C-10), 108.5 (C-13), 101.1 (OCH2O), 100.5 (C-5), 87.9 (C-3), 56.0 (4-OCH3), 35.9 (C-7), 32.8 (C-8).
DST 2 (4-methoxy-6-(11,12-methylenedioxy-14-methoxy - dihydrostyryl)-2-pyrone): m.p. 148 - 151ºC; 1H NMR (400 MHz, CDCl3) δ (ppm): 6.59 (s, 1H, H-10), 6.49 (s, 1H, H-13), 5.88 (s, 2H, OCH2O at C-11 and C-12), 5.71 (sl, 1H, H-5), 5.40 (sl, 1H, H-3), 3.77 (s, 3H, 4-OCH3), 3.74 (s, 3H, 14-OCH3), 2.85 (t, 2H, H-8), 2.65 (t, 2H, H-7). 13C NMR (50 MHz, CDCl3) δ (ppm): 171.1 (C-2), 164.9 (C-4), 164.1 (C-6), 152.2 (C-14), 146.5 (C-12), 140.6 (C-11), 120.1 (C-9), 109.5 (C-10), 100.9 (OCH2O), 99.5 (C-5), 94.5 (C-13), 87.5 (C-3), 56.1 (14-OCH3), 55.7 (4-OCH3), 34.0 (C-8), 27.6 (C-7).
DST 3 (4-methoxy-6-(11,12-methylenedioxy-10,14-dime thoxy-dihydrostyryl)-2-pyrone): m.p. 165 - 167ºC; IR (KBr, ν cm-1): 3089, 2951, 2849, 1727, 1646, 1572, 1508, 1474; 1H NMR (400 MHz, CDCl3) δ (ppm): 6.23 (s, 1H, H-13), 5.86 (s, 2H, OCH2O at C-11 and C-12), 5.71 (sl, 1H, H-5), 5.40 (sl, 1H, H-3), 3.96 (s, 3H, 10-OCH3), 3.78 (s, 3H, 4-OCH3), 3.71 (s, 3H, 14-OCH3), 2.90 (t, 2H, J=7,6 Hz, H-8), 2.58 (t, 2H, J=7,6 Hz, H-7).
STY 4 (4-methoxy-6-(11,12-methylenedioxystyryl)-2-py rone): m.p. 199 - 202ºC; 1H NMR (400 MHz, CDCl3) δ (ppm): 7.45 (d, 1H, J=16 Hz, H-8), 7.03 (s, 1H, H-10), 6.88 (s, 1H, H-14), 6.81 (s, 1H, H-13), 6.45 (d, 1H, J=16 Hz, H-7), 5.92 (s, 2H, OCH2O at C-11 and C-12), 5.70 (sl, 1H, H-5), 5.48 (sl, 1H, H-3), 3.89 (s, 3H, 4-OCH3).
STY 5 (4-methoxy-6-(11,12-methylenedioxy-14-metho xystyryl)-2-pyrone): m.p. 190 - 191ºC; 1H NMR (200 MHz, CDCl3) δ (ppm): 7.77 (d, 1H, J=16 Hz, H-8), 6.96 (s, 1H, H-10), 6.52 (s, 1H, H-13), 6.46 (d, 1H, J=16 Hz, H-7), 5.95 (s, 2H, OCH2O at C-11 at C-12), 5.84 (sl, 1H, H-5), 5.40 (sl, 1H, H-3), 3.87 (s, 3H, OCH3). 3.72 (s, 3H, OCH3).
Prenyloxycoumarin (6-methoxy-7-prenyloxycoumarin): m.p. 79 - 81ºC; IR (KBr, ν cm-1): 1713, 1612, 1561, 1513, 1381, 1280, 1248, 1140, 971, 812. 1H NMR (200 MHz, CDCl3) δ (ppm): 7.63 (d, 1H, J4,3=9.4 Hz, H-4), 6.86 (s, 1H, H-8), 6.82 (s, 1H, H-5), 6.26 (d, 1H, J3,4=9.4 Hz, H-3), 5.49 (t, 1H, [CH3]2-C=CH-CH2), 4.66 (d, 2H, [CH3]2-C=CH-CH2), 3.90 (s, 3H, 6-OCH3), 1.79 (s, 6H, [CH3]2-C=CH-CH2). 13C NMR (50 MHz, CDCl3) δ (ppm): 161.7 (C=O), 152.0 (C-7), 149.8 (C-10), 146.6 (C-6), 143.3 (C-4), 138.9 (C-3'), 118.6 (C-2'), 113.2 (C-3), 111.2 (C-9), 108.0 (C-5), 101.0 (C-8), 66.2 (C-1'), 56.3 (6-OCH3), 25.7 (C-4'), 18.3 (C-5').
Scopoletin (6-methoxy-7-hydroxycoumarin): m.p. 203 - 204ºC; IR (KBr, ν cm-1): 3340, 1709, 1267. 1H RMN (400 MHz, CDCl3) δ (ppm): 7.60 (d, 1H, J3,4=9.6 Hz, H-3), 6.91 (s, 1H, H-8), 6.85 (s, 1H, H-5), 6.27 (d, 1H, J4,3=9,6 Hz, H-4), 6.22 (s, 1H, OH), 3.95 (s, 3H, 6-OCH3). 13C NMR (CDCl3) δ (ppm): 161.8 (C=O), 150.4 (C-9), 149.9 (C-7), 144.0 (C-6), 143.5 (C-4), 113.6 (C-3), 111.7 (C-10), 107.7 (C-5), 103.4 (C-8), 56.6 (OCH3).
α-spinasterol (24α-ethyl-5α-cholesta-7,22E-dien-3β-ol): m.p. 153-154ºC. IR (KBr, ν cm-1): 3424, 2956, 2937, 2869. 1H NMR (200 MHz, CDCl3) δ (ppm): 5.14 (m, 3H, H-7, H-22 e H-23), 3.60 (m, H-3), 1.03 (d, J= 6.6, 21-CH3), 0.85 (d, J = 6.0, 26-CH3), 0.80 (s, 19-CH3), 0.80 (d, J = 6.3, 27-CH3), 0.79 (t, J = 6.2, 29-CH3), 0.55 (s, 18-CH3). 13C NMR (50 MHz, CDCl3) δ (ppm): 140.3 (C-8), 138.9 (C-22), 130.1 (C-23), 118.1 (C-7), 71.8 (C-3), 56.6 (C-17), 55.8 (C-14), 51.9 (C-24), 50.1 (C-9), 43.9 (C-13), 41.5 (C-20), 40.9 (C-5), 40.2 (C-12), 38.7 (C-4), 37.8 (C-1), 34.9 (C-10), 32.6 (C-25), 32.1 (C-2), 30.3 (C-6), 29.2 (C-16), 26.1 (C-28), 23.7 (C-15), 22.2 (C-11), 22.1 (C-21), 21.8 (C-26), 19.7 (C-27), 13.8 (C-19), 12.9 (C-29), 12.7 (C-18).
1,2,3,4,5,6-hexanehexol: m.p. 166 - 169ºC. 1H RMN (400 MHz, CDCl3) δ (ppm): 3.90 (td, 1H, J=5.2, J=3.4 Hz, H-2 and H-5), 3.80 (dd, 1H, J=3.4 Hz, H-3 and H-4), 3.50 (d, 2H, J=5.2, H-1 and H-6). All signs showed the 2:1:1 ratio.
Phebalosin (7-methoxy-8-(1',2'-epoxy-3'-methyl-3'-bute nyl)-coumarin): m.p. 124ºC. IR (KBr, ν cm-1): 3070, 1735, 1608. 1H NMR (400 MHz, CDCl3) δ (ppm): 6.26 (d, J=9.6, H-3), 7.62 (d, J=9.6, H-4), 7.42 (d, J=8.8, H-5), 6.87 (d, J=8.8, H-6), 3.99 (d, H-1'), 3.92 (d, J'=2.0, H-2'), 5.08 (s, H-4'), 5.30 (s, H-4'), 1.87 (H-5'), 3.97 (s, OCH3). 13C NMR (400 MHz, CD3OD) δ (ppm): 162.18 (C-2), 113.05 (C-3), 141.51 (C-4), 129.23 (C-5), 107.81 (C-6), 160.61 (C-7), unobserved (C-8), 154.09 (C-9), 112.76 (C-10), 52.00 (C-1'), 60.92 (C-2'), 113.78 (C-3'), 113.68 (C-4'), 17.68 (C-5'), 56.58 (OCH3).
Aurapten (7-geraniloxy coumarin): m.p. 65-66 ºC. IR (KBr, ν cm-1): 1729, 1592. 1H NMR (400 MHz, CDCl3) δ (ppm): 6.24 (d, J=9.5, H-3), 7.64 (d, J=9.5, H-4), 7.36 (d, J=8.2, H-5), 6.85 (d, J=8.2, H-6), 6.82 (s, H-8), 4.60 (d, J=6.5, H-1'), 5.47 (t, J=6.5, H-2'), 2.11 (H-4'), 2.11 (H-5'), 5.08 (sl, H-6'), 1.76 (s, H-8'), 1.67 (s, H-9'), 1.60 (s, H-10'). 13C NMR (400 MHz, CD3OD) δ (ppm): 126.12 (C-2), 112.94 (C-3), 143.42 (C-4), 128.64 (C-5), 113.21 (C-6), 161.27 (C-7), 101.57 (C-8), 155.85 (C-9), 112.39 (C-10), 65.46 (C-1'), 118.36 (C-2'), 142.34 (C-3'), 39.49 (C-4'), 26.20 (C-5'), 123.59 (C-6'), 131.94 (C-7'), 16.75 (C-8'), 25.63 (C-9'), 17.69 (C-10').
Rutin (Quercetin 3-rutinoside): m.p.: 188-190 ºC. IR (KBr, ν cm - 1): 3415, 1652, 1595, 1496. 13C NMR (400 MHz, CDCl3) δ (ppm): 157.27 (C-2), 134.79 (C-3), 178.33 (C-4), 158.04 (C-5), 97.99 (C-6), 166.21 (C-7), 92.07 (C-8), 167.50 (C-9), 105.37 (C-10), 122.97 (C-1'), 114.85 (C-2'), 145.74 (C-3'), 150.56 (C-4'), 116.23 (C-5'), 122.09 (C-6'), 101.19 (C-1G), 74.52 (C-2G), 76.93 (C-3G), 70.87 (C-4G), 76.02 (C-5G), 67.39 (C-6G), 103.10 (C-1R), 70.26 (C-2R), 71,05 (C-3R), 72.66 (C-4R), 68.54 (C-5R), 16.70 (C-6R). G glucose; R ramnose.
The results of the antifungal screening of Polygala spp. are shown in Table 1. The TLC bioassay (bioautography) used in the present study permitted an overview of the antifungal activity of the extracts and fractions of Polygala spp. and displayed clear zones on the TLC plates. The hexane fraction of P. sabulosa showed a broad spectrum of activity with antifungal properties for all microorganisms used in this study. However, the EtOH extract of P. sabulosa was not active against all of the microorganisms tested. All extracts and fractions of P. paniculata and P. pulchella presented activity against C. tropicalis. The other extracts and fractions showed activity for at least one of the fungal species tested.
Table 2 shows the Minimum Inhibitory Concentration (MIC) for the active extracts in bioautography assay. C. gattii was the fungal species most sensitive to the tested extracts. The EtOAc fraction of P. sabulosa inhibited C. gattii and S. schenckii, with the lowest active amount being 30 µg/mL. The hexane extract of P. paniculata also showed good antifungal activity, with MIC values of 60 µg/mL against C. tropicalis, 125 µg/mL against C. gattii and 250 µg/mL against C. glabrata and C. parapsilosis. However, the EtOH extract of P. paniculata was active only against C. tropicalis (250 µg/mL) and S. schenckii (1000 µg/mL).
The hexane and CH2Cl2 fractions from P. sabulosa had a wide spectrum of action, and the amounts required to inhibit the fungal growth varied between 250 and 1000 µg/mL. However, most of the fungal growth was inhibited at 500 and 1000 µg/mL. Only C. glabrata and S. schenckii were more susceptible at low concentration to the CH2Cl2 fraction of P. sabulosa, with an MIC of 250 µg/mL. The EtOH extract of P. cypariassias was active only against C. gattii (250 µg/mL).
Some species of Polygala have previously been shown to have antifungal activity. From the species P. gazensis, the chromonocoumarin frutinones A and B and the lignans eudesmin, magnolin, yangambin and kobusin were isolated. Frutinone A showed antifungal activity against the pathogenic plant fungi Cladosporium cucumerinum and C. albicans, while eudesmin and kobusin were active against C. cucumerinum only. Frutinone B, magnolin and yangambin did not present antifungal activity (3). Two xanthones, 1,7-dihydroxy-4-methoxyxanthone and 1,7-dihydroxy-3,5,6-trimethoxyxan thone, isolated from the roots of P. nyikensis, exhibited antifungal activity against C. cucumerinum (23). In the P. myrtifolia species, the presence of saponins was related to activity against C. albicans (26).
Table 3 shows the antifungal activities (MIC values in µg/mL) of compounds isolated from two Polygala species, P. paniculata and P. sabulosa, along with the antifungal activities of the extracts from which they were isolated. The other three plant species did not show good activity; therefore, compounds were not isolated from these species. Scopoletin, DST 1, 2, 3 and STY 4, 5, isolated from the EtOAc extract of P. sabulosa, showed weak antifungal activity. In contrast, prenyloxycoumarin and 1,2,3,4,5,6-hexanehexol displayed antifungal activity against S. schenckii (with MICs of 125 µg/mL and 250 µg/mL, respectively) and C. gattii (both with an MIC of 250 µg/mL). The flavonoid rutin and the coumarin aurapten isolated from P. paniculata showed antifungal activity against C. gattii, with MIC values of 60 and 250 µg/mL, respectively. Phebalosin, another coumarin isolated from P. paniculata, did not show any antifungal activity at concentrations up to 1000 µg/mL. α-spinasterol, isolated from both Polygala species, showed antifungal activity between 125 and 500 µg/mL for all the fungal species tested.
The antifungal activity observed against C. gattii is significant due to the difficulties associated with the treatment of cryptococcosis and the resistance of this fungus to some antifungal drugs. In our study, the compounds prenyloxycoumarin, aurapten and 1,2,3,4,5,6-hexanehexol showed activity against C. gatii with an MIC of 250 µg/mL. The flavonoid rutin exhibited better activity against this fungus, with an MIC of 60 µg/mL.
Previous studies have shown the antifungal activity of some of these compounds. The α-spinasterol isolated from Solidago microglossa does not present activity against Staphylococcus aureus, S. epidermidis, Klebsiella pneumoniae, Escherichia coli, Salmonella setubal, Bacillus subtilis, Pseudomonas aeruginosa, Saccharomyces cerevisiae or C. albicans (29). In the present work, this compound was active against C. albicans with an MIC of 500 µg/mL. Rutin was already shown to be active against Phytophthora megasperma, Cylindrocarpon destructans (2) and C. albicans (MIC of 1000 µg/mL) (14). In the present study, rutin not was active against C. albicans, but it showed activity against C. krusei and C. gattii.
Regarding the lack of activity of the coumarins scopoletin and phebalosin, it has been reported that the pharmacological and biochemical properties of simple coumarins may depend on the substitution pattern of the benzene moiety. Sardari et al. (41) demonstrated that a free 6-OH is essential and that alkylated derivatives of 7-hydroxycoumarin may show both antifungal and antibacterial properties. Although the coumarins prenyloxycoumarin, phebalosin and aurapten do not possess a free 6-OH, they were not totally devoid of antifungal activity. Nevertheless, the observed lack of activity of scopoletin (MICs > 1000 µg/mL) is in conflict with the results of Sardari et al. (41) because this coumarin has a free 6-OH. There are also many studies that describe the antifungal activity of this coumarin against phytopathogens. There is a correlation between scopoletin accumulation and resistance to microbial pathogens in some plants showing important antifungal activity, scopoletin being considered a phytoalexin (42, 1, 43).
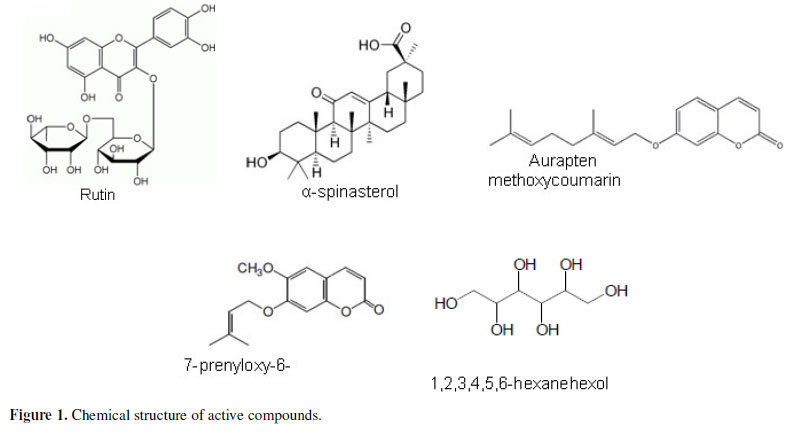
As is clear from Table 3, most of the isolated compounds were devoid of antifungal activity up to 1000 µg/mL, and only α-spinasterol, 1,2,3,4,5,6-hexanehexol, prenyloxycoumarin, rutin and aurapten (Figure 1) showed moderate activity toward some fungal species, albeit lower than that displayed by the original extracts or fractions. This interesting and unexpected result could be due to a synergistic action between the compounds of the extracts in the case of P. sabulosa and P. paniculata or could be ascribed to minor components of the extracts.
Of all the compounds screened, two of them showed antifungal inhibition, whereas eleven did not have significant activity. Rutin and aurapten isolated from P. paniculata showed antifungal activity against C. gattii with MIC values of 60 and 250 µg/mL, respectively. The compound α-spinasterol showed a wide activity against the species tested and rutin, with the lowest MIC for the microorganisms tested, had the best activity. Chemical modification may improve the activity of these two compounds.
ACKNOWLEDGMENTS
We thank the Brazilian government agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for financial support.
REFERENCES
1. Bayoumi, S.A.L.; Rowan, M.G.; Blagbrough, I.S.; Beeching, J.R. (2008). Biosynthesis of scopoletin and scopolin in cassava roots during post-harvest physiological deterioration: The E-Z-isomerisation stage. Phytochemistry 69,2928-2936.
2. Báidez, A.G.; Gómez, P.; Del Río, J.A.; Ortunõ, A. (2006). Antifungal capacity of major phenolic compounds of Olea europaea L. against Phytophthora megasperma Drechsler and Cylindrocarpon destructans (Zinssm.) Scholten. Physiol. Mol. Plant Path. 69,224-229.
3. Bergeron, C.; Marston, A.; Wolfender, J.L.; Mavi, S.; Roger, C.; Hostettmann, K. (1997). Isolation of polyphenols from Polygala gazensis and liquid chromatography-mass spectrometry of related African Polygala species. Phytochem. Anal. 8,32-36.
4. Casolari, C.; Rossi, T.; Baggio, G.; Coppi, A.; Zandomenegui, G.; Ruberto, A.I.; Farina, C.; Fabio, G.; Zanca, A.; Castelli, M. (2004). Interaction between saquinavir and antimycotic drugs on C. albicans and C. neoformans strains. Pharmacol. Res. 50,605-610.
5. Cristiano, R.; Pizzolatti, M.G.; Monache, F.D.; Rezende, C.M.; Branco, A. (2003). Two xanthones from Polygala paniculata and confirmation of the 1-hidroxy-2,3,5-trimetoxy-xanthone at trace level by HRGC-MS. Z. Naturforsch. 58,490-494.
6. Chaturvedi, S.; Dyavaiah, M.; Larsen, RA.; Chaturvedi, V. (2005). Cryptococcus gattii in AIDS patients, southern California. Emerg. Infect. Dis. 11,1686-1692.
7. Dall'Acqua, S.; Viola, G.; Cappelletti, E.M.; Innocenti, G. (2004). Xanthones from Polygala alpestris (Rchb). Z. Naturforsch. 59,335-338.
8. Duarte, F.S.; Duzzioni, M.; Mendes, B.G.; Pizzolatti, M.G.; Lima, T.C.M. (2007). Participation of dihydrostyryl-2-pyrones and styryl-2-pyrones in the central effects of Polygala sabulosa (Polygalaceae), a folk medicine topical anesthetic. Pharmacol. Biochem. Behav. 86,150-161.
9. Duarte, F.S.; Marder, M.; Hoeller, A.A.; Duzzioni, M.; Mendes, B.G.; Pizzolatti, M.G.; De Lima, T.C.M. (2008). Anticonvulsant and anxiolytic-like effects of compounds isolated from Polygala sabulosa (Polygalaceae) in rodents: in vitro and in vivo interactions with benzodiazepine binding sites. Psychopharmacology 197,351-360.
10. El Sayah, M.; Cechinel Filho, V.; Pinheiro, T.R.; Yunes, R.A.; Calixto, J.B. (1999). In vitro effect of the extract and the 1,7-dihydroxy-2,3-dimethoxy xanthone from Polygala cyparissias on the contractions induced by inflammatory mediators and albumin in normal and actively sensitised trachea from guinea pig. Inflam. Res. 48,218-223.
11. Fenner, R.; Sortino, M.; Rates, S.M.K.; Dall'Agnol, R.; Ferraz, A.; Bernardi, A.P.; Albring, D.; Nör, C.; Von Poser, G.; Schapoval, E.; Zacchino, S. (2005). Antifungal activity of some Brazilian Hypericum species Phytomedicine 12,236-240.
12. Hamburger, M.; Gupta, M.; Hostettmann, K. (1985). Coumarins from Polygala paniculata. Planta Med. 51,215-217.
13. Hamburger, M.; Hostettmann, K.; Stoeckli-Evans, H. (1984). A new pyranocoumarin diester from Polygala paniculata L. Helv. Chim. Acta 67,1729-1733.
14. Han, Y. (2009). Rutin has therapeutic effect on septic arthritis caused by Candida albicans. Int. Immunopharmacol. 9,207-211.
15. Ikeya, Y.; Takeda, S.; Tunakawa, M.; Karakida, H.; Toda, K.; Yamaguchi,T.; Aburada, M. (2004) Cognitive improving and cerebral protective effects of acylated oligosaccharides in Polygala tenuifolia. Biol. Pharm. Bull. 27,1081-1085.
16. Jia, H.; Jiang, Y.; Ruan, Y.; Zhang, Y.; Ma, X.; Zhang, J.; Beyreuther, K.; Tu, P.; Zhang, D. (2004). Tenuigenin treatment decreases secretion of the Alzheimer's disease amyloid beta-protein in cultured cells. Neurosci. Lett. 367,123-128.
17. Johann, S.; Pizzolatti, M.G.; Resende, M.A.; Donnici, C. (2007) Antifungal properties of plants used in Brazilian traditional medicine against clinically relevant fungal pathogens. Braz. J. Microbiol. 38,632-637.
18. Johann, S.; Silva, D.L.; Martins, C.V.B.; Pizzolatti, M.G.; Zani, C.L.; Resende, M.A. (2008). Inhibitory effect of extracts from Brazilian medicinal plants on the adhesion of Candida albicans to buccal epithelial cells. World J. Microbiol. Biotechnol. 24,2459-2464.
19. Kou, J.; Si, M.; Dai, G.; Lin, Y.; Zhu, D. (2006) Antiinflammatory activity of Polygala japonica extract. Fitoterapia 77,411-415.
20. Lall, N.; Meyer, J.J. (1999). In vitro inhibition of drug-resistant and drug-sentive strais of Mycobacterium tuberculosis by ethnobotanically selected South African plants. J. Ethnopharmacol. 66,347-354.
21. Li, J.; Jiang, Y.; Tu, P.F. (2005) Tricornoses A-L, oligosaccharide multi-esters from the roots of Polygala tricornis. J. Nat. Prod. 68,739-744.
22. Linn, L.L.; Huang, F.; Chen, S.B.; Yang, D.J.; Chen, S.L.; Yang, J.S.; Xiao, P.G. (2005). Xanthones from the roots of Polygala caudata and their antioxidation and vasodilatation activities in vitro. Planta Med. 71,372-375.
23. Marston, A.; Hamburger, M.; Sordat-Diserens, I.; Msonthi, J.D.; Hostettmann, K. (1993). Xanthones from Polygala nyikensis. Phytochemistry 33,809-812.
24. Masoko, P.; Picard, J.; Eloff, J.N. (2005). Antifungal activities of six South African Terminalia species (Combretaceae). J. Ethnopharmacol. 99,301-308.
25. Mitaine-Offer, A.C.; Miyamoto, T.; Laurens, V.; Delaude, C.; Lacaille-Dubois, M.A. (2003) New acylated triterpene saponins from Polygala arenaria. Helv. Chim. Acta 86,2404-2413.
26. Motsei, M.L.; Lindsey, K.L.; von Staden, J.; Jäger, A.K. (2003). Screening of traditionally used South African plants for antifungal activity against Candida albicans. J. Ethnopharmacol. 86,235-241.
27. Mothana, R.A.A.; Lindequist, U. (2005). Antimicrobial activity of some medicinal plants of the island Soqotra. J. Ethnopharmacol. 96,177-181.
28. (CLSI) National Committee for Clinical Laboratory Standards: Reference method for broth dilution antifungal susceptibility testing of yeasts (2002) Approved standard M27-A2. NCCLS, Villanova, PA, USA.
29. Pfaller, M.A.; Burmeister, L.; Bartlett, M.S.; Rinaldi, M.G. (1988). Multicenter evaluation of four methods of yeast inoculum preparation. J. Clin. Microbiol. 26,1437-1441.
30. Pinheiro, T.R.; Cechinel Filho, V.; Santos, A.R.S.; Calixto, J.B.; Delle Monache, F.; Pizzolatti, M.G.; Yunes, R.A. (1998) Three xanthones from Polygala cyparissias. Phytochemistry 48,725-728.
31. Pizzolatti, M.G.; Cunha, Jr.A.; Pereira, W.S.; Delle Monache, F. (2004). A new styryl-2-pyrone derivative from Polygala sabulosa (Polygalaceae). Biochem. Sys. Ecol. 32,603-606.
32. Pizzolatti, M.G.; Luciano, C.; Delle Monache, F. (2000) Styryl-and dihydrostyryl-2-pyrones derivatives from Polygala sabulosa. Phytochemistry 55,819-822.
33. Pizzolatti, M.G.; Cristiano, R.; Monache, F.D.; Branco, A. (2002) Artefatos cumarínicos isolados de Polygala paniculata L. (Polygalaceae). Brazilian. J. Pharmacog. 12,21-26.
34. Pizzolatti, M.G.; Koga, A.H.; Grisard, E.C.; Steindel, M. (2003). Trypanocidal activity of extracts from Brazilian Atlantic Rain Forest plant species. Phytomedicine 10,422-426.
35. Pizzolatti, M.G.; Mendes, B.G.; Cunha, Jr.A.; Soldi, C.; Koga, A.H.; Eger, I.; Grisard, E.C.; Steindel, M. (2008) Trypanocidal activity of coumarins and styryl-2-pyrones from Polygala sabulosa A.W. Bennett (Polygalaceae). Brazilian J. Pharmacog. 18,177-182.
36. Portillo, A.; Vila, R.; Freixa, B.; Adzet, T.; Cañigueral, S. (2001). Antifungal activity of Paraguayan plants used in traditional medicine. J. Ethnopharmacol. 76,93-98.
37. Ramos-e-Silva, M.; Vasconcellos, C.; Carneiro, S.; Cestari, T. (2007) Sporotrichosis. Clin. Dermatol. 25,181-187.
38. Rao, M,S.; Raman, N.V. (2004). A novel flavonoid from Polygala chinensis. Biochem Sis Ecol 32,447-448.
39. Resende, J.C.P.; Resende, M.A.; Saliba, J.L. (2002) Prevalence of Candida spp. in hospitalized patients and their risk factors. Mycoses 45,306-312.
40. Ribas, C.M.; Meotti, F.C.; Nascimento, F.P.; Jacques, A.V.; Dafre, A.L.; Rodrigues, A.L.S.; Farina, M.; Soldi, C.; Mendes, B.G.; Pizzolatti, M.G.; Santos, A.R.S. (2008). Antinociceptive effect of the Polygala sabulosa hydroalcoholic extract in mice: evidence for the involvement of glutamatergic receptors and cytokine pathways. Basic. Clin. Pharmacol. Toxicol. 103,43-47.
41. Sardari, S.; Mori, Y.; Horita, K.; Micetich, R.G.; Nishibe, S.; Daneshtalab, M. (1999). Synthesis and antifungal activity of coumarins and angular furanocoumarins. Bioorg. Med. Chem. 7,1933-1940.
42. Sharma, R.R.; Singh, D.; Singh, R. (2009). Biological control of postharvest diseases of fruits and vegetables by microbial antagonists: A review. Biol Control. 50,205-221.
43. Valle, T.; López, J.L.; Hernández, J.M.; Corchete, P. (1997) Antifungal activity of scopoletin and its differential accumulation in Ulmus pumila and Ulmus campestris cell suspension cultures infected with Ophiostoma ulmi spores. Plant. Sci. 125,97-101.
44. Wurdack, J.J.; Smith, L.B. (1971). Flora Ilustrada Catarinense. Polygalaceae. Tipografia e Livraria Blumenauense S.A., Itajaí .
45. Zhang, D.; Miyase, T.; Kuroyanagi, M.; Umehara, K.; Noguchi, H. (1997). Oligossacharide polyesters from roots of Polygala fallax. Phytochemistry 45,733-741.
Submitted: August 10, 2010; Returned to authors for corrections: October 13, 2010; Approved: May 16, 2011.
- 1. Bayoumi, S.A.L.; Rowan, M.G.; Blagbrough, I.S.; Beeching, J.R. (2008). Biosynthesis of scopoletin and scopolin in cassava roots during post-harvest physiological deterioration: The E-Z-isomerisation stage. Phytochemistry 69,2928-2936.
- 2. Báidez, A.G.; Gómez, P.; Del Río, J.A.; Ortunõ, A. (2006). Antifungal capacity of major phenolic compounds of Olea europaea L. against Phytophthora megasperma Drechsler and Cylindrocarpon destructans (Zinssm.) Scholten. Physiol. Mol. Plant Path 69,224-229.
- 3. Bergeron, C.; Marston, A.; Wolfender, J.L.; Mavi, S.; Roger, C.; Hostettmann, K. (1997). Isolation of polyphenols from Polygala gazensis and liquid chromatography-mass spectrometry of related African Polygala species. Phytochem. Anal. 8,32-36.
- 4. Casolari, C.; Rossi, T.; Baggio, G.; Coppi, A.; Zandomenegui, G.; Ruberto, A.I.; Farina, C.; Fabio, G.; Zanca, A.; Castelli, M. (2004). Interaction between saquinavir and antimycotic drugs on C. albicans and C. neoformans strains. Pharmacol. Res. 50,605-610.
- 5. Cristiano, R.; Pizzolatti, M.G.; Monache, F.D.; Rezende, C.M.; Branco, A. (2003). Two xanthones from Polygala paniculata and confirmation of the 1-hidroxy-2,3,5-trimetoxy-xanthone at trace level by HRGC-MS. Z. Naturforsch 58,490-494.
- 6. Chaturvedi, S.; Dyavaiah, M.; Larsen, RA.; Chaturvedi, V. (2005). Cryptococcus gattii in AIDS patients, southern California. Emerg. Infect. Dis. 11,1686-1692.
- 7. Dall'Acqua, S.; Viola, G.; Cappelletti, E.M.; Innocenti, G. (2004). Xanthones from Polygala alpestris (Rchb). Z. Naturforsch. 59,335-338.
- 8. Duarte, F.S.; Duzzioni, M.; Mendes, B.G.; Pizzolatti, M.G.; Lima, T.C.M. (2007). Participation of dihydrostyryl-2-pyrones and styryl-2-pyrones in the central effects of Polygala sabulosa (Polygalaceae), a folk medicine topical anesthetic. Pharmacol. Biochem. Behav. 86,150-161.
- 9. Duarte, F.S.; Marder, M.; Hoeller, A.A.; Duzzioni, M.; Mendes, B.G.; Pizzolatti, M.G.; De Lima, T.C.M. (2008). Anticonvulsant and anxiolytic-like effects of compounds isolated from Polygala sabulosa (Polygalaceae) in rodents: in vitro and in vivo interactions with benzodiazepine binding sites. Psychopharmacology 197,351-360.
- 10. El Sayah, M.; Cechinel Filho, V.; Pinheiro, T.R.; Yunes, R.A.; Calixto, J.B. (1999). In vitro effect of the extract and the 1,7-dihydroxy-2,3-dimethoxy xanthone from Polygala cyparissias on the contractions induced by inflammatory mediators and albumin in normal and actively sensitised trachea from guinea pig. Inflam. Res. 48,218-223.
- 11. Fenner, R.; Sortino, M.; Rates, S.M.K.; Dall'Agnol, R.; Ferraz, A.; Bernardi, A.P.; Albring, D.; Nör, C.; Von Poser, G.; Schapoval, E.; Zacchino, S. (2005). Antifungal activity of some Brazilian Hypericum species Phytomedicine 12,236-240.
- 12. Hamburger, M.; Gupta, M.; Hostettmann, K. (1985). Coumarins from Polygala paniculata. Planta Med 51,215-217.
- 13. Hamburger, M.; Hostettmann, K.; Stoeckli-Evans, H. (1984). A new pyranocoumarin diester from Polygala paniculata L. Helv. Chim. Acta 67,1729-1733.
- 14. Han, Y. (2009). Rutin has therapeutic effect on septic arthritis caused by Candida albicans. Int. Immunopharmacol 9,207-211.
- 15. Ikeya, Y.; Takeda, S.; Tunakawa, M.; Karakida, H.; Toda, K.; Yamaguchi,T.; Aburada, M. (2004) Cognitive improving and cerebral protective effects of acylated oligosaccharides in Polygala tenuifolia. Biol. Pharm. Bull 27,1081-1085.
- 16. Jia, H.; Jiang, Y.; Ruan, Y.; Zhang, Y.; Ma, X.; Zhang, J.; Beyreuther, K.; Tu, P.; Zhang, D. (2004). Tenuigenin treatment decreases secretion of the Alzheimer's disease amyloid beta-protein in cultured cells. Neurosci. Lett. 367,123-128.
- 17. Johann, S.; Pizzolatti, M.G.; Resende, M.A.; Donnici, C. (2007) Antifungal properties of plants used in Brazilian traditional medicine against clinically relevant fungal pathogens. Braz. J. Microbiol. 38,632-637.
- 18. Johann, S.; Silva, D.L.; Martins, C.V.B.; Pizzolatti, M.G.; Zani, C.L.; Resende, M.A. (2008). Inhibitory effect of extracts from Brazilian medicinal plants on the adhesion of Candida albicans to buccal epithelial cells. World J. Microbiol Biotechnol. 24,2459-2464.
- 19. Kou, J.; Si, M.; Dai, G.; Lin, Y.; Zhu, D. (2006) Antiinflammatory activity of Polygala japonica extract. Fitoterapia 77,411-415.
- 20. Lall, N.; Meyer, J.J. (1999). In vitro inhibition of drug-resistant and drug-sentive strais of Mycobacterium tuberculosis by ethnobotanically selected South African plants. J. Ethnopharmacol. 66,347-354.
- 21. Li, J.; Jiang, Y.; Tu, P.F. (2005) Tricornoses A-L, oligosaccharide multi-esters from the roots of Polygala tricornis. J. Nat. Prod. 68,739-744.
- 22. Linn, L.L.; Huang, F.; Chen, S.B.; Yang, D.J.; Chen, S.L.; Yang, J.S.; Xiao, P.G. (2005). Xanthones from the roots of Polygala caudata and their antioxidation and vasodilatation activities in vitro. Planta Med 71,372-375.
- 23. Marston, A.; Hamburger, M.; Sordat-Diserens, I.; Msonthi, J.D.; Hostettmann, K. (1993). Xanthones from Polygala nyikensis. Phytochemistry 33,809-812.
- 24. Masoko, P.; Picard, J.; Eloff, J.N. (2005). Antifungal activities of six South African Terminalia species (Combretaceae). J. Ethnopharmacol 99,301-308.
- 25. Mitaine-Offer, A.C.; Miyamoto, T.; Laurens, V.; Delaude, C.; Lacaille-Dubois, M.A. (2003) New acylated triterpene saponins from Polygala arenaria. Helv. Chim. Acta 86,2404-2413.
- 26. Motsei, M.L.; Lindsey, K.L.; von Staden, J.; Jäger, A.K. (2003). Screening of traditionally used South African plants for antifungal activity against Candida albicans. J. Ethnopharmacol. 86,235-241.
- 27. Mothana, R.A.A.; Lindequist, U. (2005). Antimicrobial activity of some medicinal plants of the island Soqotra. J. Ethnopharmacol 96,177-181.
- 28. (CLSI) National Committee for Clinical Laboratory Standards: Reference method for broth dilution antifungal susceptibility testing of yeasts (2002) Approved standard M27-A2. NCCLS, Villanova, PA, USA.
- 29. Pfaller, M.A.; Burmeister, L.; Bartlett, M.S.; Rinaldi, M.G. (1988). Multicenter evaluation of four methods of yeast inoculum preparation. J. Clin. Microbiol. 26,1437-1441.
- 30. Pinheiro, T.R.; Cechinel Filho, V.; Santos, A.R.S.; Calixto, J.B.; Delle Monache, F.; Pizzolatti, M.G.; Yunes, R.A. (1998) Three xanthones from Polygala cyparissias. Phytochemistry 48,725-728.
- 31. Pizzolatti, M.G.; Cunha, Jr.A.; Pereira, W.S.; Delle Monache, F. (2004). A new styryl-2-pyrone derivative from Polygala sabulosa (Polygalaceae). Biochem. Sys. Ecol 32,603-606.
- 32. Pizzolatti, M.G.; Luciano, C.; Delle Monache, F. (2000) Styryl-and dihydrostyryl-2-pyrones derivatives from Polygala sabulosa. Phytochemistry 55,819-822.
- 33. Pizzolatti, M.G.; Cristiano, R.; Monache, F.D.; Branco, A. (2002) Artefatos cumarínicos isolados de Polygala paniculata L. (Polygalaceae). Brazilian. J. Pharmacog 12,21-26.
- 34. Pizzolatti, M.G.; Koga, A.H.; Grisard, E.C.; Steindel, M. (2003). Trypanocidal activity of extracts from Brazilian Atlantic Rain Forest plant species. Phytomedicine 10,422-426.
- 35. Pizzolatti, M.G.; Mendes, B.G.; Cunha, Jr.A.; Soldi, C.; Koga, A.H.; Eger, I.; Grisard, E.C.; Steindel, M. (2008) Trypanocidal activity of coumarins and styryl-2-pyrones from Polygala sabulosa A.W. Bennett (Polygalaceae). Brazilian J. Pharmacog. 18,177-182.
- 36. Portillo, A.; Vila, R.; Freixa, B.; Adzet, T.; Cañigueral, S. (2001). Antifungal activity of Paraguayan plants used in traditional medicine. J. Ethnopharmacol. 76,93-98.
- 37. Ramos-e-Silva, M.; Vasconcellos, C.; Carneiro, S.; Cestari, T. (2007) Sporotrichosis. Clin. Dermatol 25,181-187.
- 38. Rao, M,S.; Raman, N.V. (2004). A novel flavonoid from Polygala chinensis. Biochem Sis Ecol 32,447-448.
- 39. Resende, J.C.P.; Resende, M.A.; Saliba, J.L. (2002) Prevalence of Candida spp. in hospitalized patients and their risk factors. Mycoses 45,306-312.
- 40. Ribas, C.M.; Meotti, F.C.; Nascimento, F.P.; Jacques, A.V.; Dafre, A.L.; Rodrigues, A.L.S.; Farina, M.; Soldi, C.; Mendes, B.G.; Pizzolatti, M.G.; Santos, A.R.S. (2008). Antinociceptive effect of the Polygala sabulosa hydroalcoholic extract in mice: evidence for the involvement of glutamatergic receptors and cytokine pathways. Basic. Clin. Pharmacol. Toxicol. 103,43-47.
- 41. Sardari, S.; Mori, Y.; Horita, K.; Micetich, R.G.; Nishibe, S.; Daneshtalab, M. (1999). Synthesis and antifungal activity of coumarins and angular furanocoumarins. Bioorg. Med. Chem. 7,1933-1940.
- 42. Sharma, R.R.; Singh, D.; Singh, R. (2009). Biological control of postharvest diseases of fruits and vegetables by microbial antagonists: A review. Biol Control. 50,205-221.
- 43. Valle, T.; López, J.L.; Hernández, J.M.; Corchete, P. (1997) Antifungal activity of scopoletin and its differential accumulation in Ulmus pumila and Ulmus campestris cell suspension cultures infected with Ophiostoma ulmi spores. Plant. Sci. 125,97-101.
- 44. Wurdack, J.J.; Smith, L.B. (1971). Flora Ilustrada Catarinense. Polygalaceae Tipografia e Livraria Blumenauense S.A., Itajaí
- 45. Zhang, D.; Miyase, T.; Kuroyanagi, M.; Umehara, K.; Noguchi, H. (1997). Oligossacharide polyesters from roots of Polygala fallax. Phytochemistry 45,733-741.
Publication Dates
-
Publication in this collection
21 Dec 2011 -
Date of issue
Sept 2011
History
-
Accepted
16 May 2011 -
Reviewed
13 Oct 2010 -
Received
10 Aug 2010