Abstract
Xylanolytic enzymes produced by Lentinula edodes UFV70, cultivated in eucalyptus sawdust/rice bran medium, were stable at 50, 60 and 65ºC for 21 hours, losing only 15-25% activity. Fungus incubation at 50ºC for 12 hours and at 65ºC for 24 hours increased the amount of xylose produced.
xylanase; thermostability; Lentinula edodes; xylose
INDUSTRIAL MICROBIOLOGY
Thermostability of xylanolytic enzymes produced by Lentinula edodes UFV70
Liliane Fraga Costa RibeiroI; Marcelo Gomes Marçal Vieira VazII; Virgínia Maria Chaves-AlvesIII; Maria Cristina Dantas VanettiIII; Maria Catarina Megumi KasuyaII; Flávia Maria Lopes PassosIII; Antônio Galvão do NascimentoIII
IDepartamento de Bioquímica e Imunologia, Faculdade de Medicina de Ribeirão Preto, Ribeirão Preto, SP, Brasil
IIUniversidade de São Paulo, Centro de Energia Molecular na Agricultura, Laboratório de Ecologia Molecular de Cianobactérias, Piracicaba, SP Brasil
IIIDepartamento de Microbiologia, Universidade Federal de Viçosa, Viçosa, MG, Brasil
ABSTRACT
Xylanolytic enzymes produced by Lentinula edodes UFV70, cultivated in eucalyptus sawdust/rice bran medium, were stable at 50, 60 and 65ºC for 21 hours, losing only 15-25% activity. Fungus incubation at 50ºC for 12 hours and at 65ºC for 24 hours increased the amount of xylose produced.
Key words: xylanase, thermostability, Lentinula edodes, xylose.
The increased expansion of agro-industrial activity has led to heavy accumulations of lignocellulosic residues that often contaminate the soil. Lignocellulose is the most abundant renewable biomass available on the planet, which consists of three major groups: cellulose, hemicellulose, and lignin (14). Hemicellulose classification is based on the main sugar unit, where a hydrolyzed polymer that yields xylose is considered a xylan (12).
Lentinula edodes is a white rot fungus that grows on lignocellulosic residues, such as Eucalyptus sawdust and rice bran (3). Its enzymes can be used in biotechnological applications, i.e., to create xylose for xylitol production (15), lignolytic enzyme production (2, 4), and applications in other biotechnological processes, such as bioethanol production (6), and to increase the final brightness ceiling in bleached pulp (13).
The thermostability of xylanolytic enzymes is an important characteristic that allows for their application in industrial processes (13). These enzymes must be stable at high temperatures for use in pulp bleaching, for example (13). It is known that thermophilic fungi produce thermostable β-xylanase, which retains its catalytic activity for many hours at high temperatures ranging from 80100 ˚C (5, 9, 16). However, mesophilic microorganisms produce enzymes with lower thermostability (7, 8, 11).
This study aimed to evaluate agro-industrial residue incubation conditions to maximize the amount of xylose produced, which is released by the enzymes from Lentinula edodes grown in Eucalyptus sawdust/rice bran medium. It was hypothesized that if lignocellulosic material, colonized by the fungus, was incubated at a temperature at which the fungus was unable to grow, but its xylanolytic enzymes were still able to hydrolyze the polysaccharides in the growth medium, then xylose concentrations would increase in the hydrolysates.
For this reason, L. edodes UFV70 was grown in potato dextrose agar (PDA) for 15 days until complete colonization of the PDA medium occurred. Two 9 mm discs from the colonized medium were transferred to 125 mL Erlenmeyers flasks containing 25 g of semi-solid sterilized medium, composed of 51.4% (w/w) eucalyptus sawdust, 12.4% (w/w) rice bran, 1.2% (w/w) mixture of CaCO3 and CaSO4 (1:4, respectively), and 35% water. The cultures were incubated at 25 ºC for 7 weeks. Samples were collected every week and stored at -20 ºC. Enzymatic extracts were made by mixing 15 mL of Na-acetate buffer (50 mM, pH 5.0) with 10 g of semi-solid medium colonized by the fungus. Initially, the extract was filtered using gauze and Whatman nº1 filter paper. Xylanolytic complex activity was determined using ´Birchwood` xylan as a substrate. The assay mixture contained 100 µL of 1% xylan and 300 µL of crude enzymatic extract. It was incubated at 40 ºC for 40 minutes, except when mentioned otherwise. The reaction was stopped by adding 1 mL of DNS (dinitrosalicylic acid) (10) and boiling for 5 minutes. The amount of reduced sugar released was quantified using xylose as a standard. One unit (U) of activity was defined as the number of µmoles of xylose released per minute under the assay conditions. Apparent specific activity was obtained by dividing the activity value (U) by the protein quantity (mg). Proteins were quantified using the Bradford method (1976) (1), in which serum albumin was used as a standard.
The effect of temperature on xylanolytic complex activity was evaluated in the range of 35 to 80 ºC. The thermostability was determined by pre-incubation of samples at temperatures ranging from 35 to 80 ºC, for three hours, and at 50, 60, and 65 ºC, for 21 hours. At various time intervals, aliquots were withdrawn, and the activity was measured at 40 ºC for 40 minutes, as previously mentioned.
The apparent specific activity of xylanolytic enzymes was detected after the second week, varying from 0.5 to 1.5 U mg-1 of protein (data not shown). The temperature for maximum apparent specific xylanolytic activity, 70 ºC (data not shown), was similar to thermophilic fungi, such as Thermomyces lanuginosus (16), but higher than that previously found in other L. edodes strains (5060 ºC) (7, 8, 11). Xylanolytic enzymes were stable in a temperature range of 3565 ºC for 3 hours. At 70 ºC, the activity was 11% lower than that of the control (data not shown). When the activity was measured after pre-incubation, it was 25, 20, and 15% lower after 21 hours of pre-incubation at 65, 60, and 50 ºC, respectively (Figure 1).
The maximum apparent activity occurred at a higher temperature (70 ºC) than previously reported (5060 ºC) (7, 8, 11). Furthermore, these enzymes were considerably stable when pre-incubated up to 65 ºC, for up to 21 hours (Figure 1). Compared to the reported 50% loss of β-xylanase activity at 60 ºC after 6 hours for this same species (7), this current study found these enzymes to be much more stable. These differences may be due to the use of crude enzymatic extracts in this study, instead of purified enzymes (7). Some substances present in the crude extracts may have had an influence on the thermostability of these enzymes. Also, the current study used a different strain, L. edodes UFV70. These results are promising, because the use of crude extracts is a less expensive process than using enzymes.
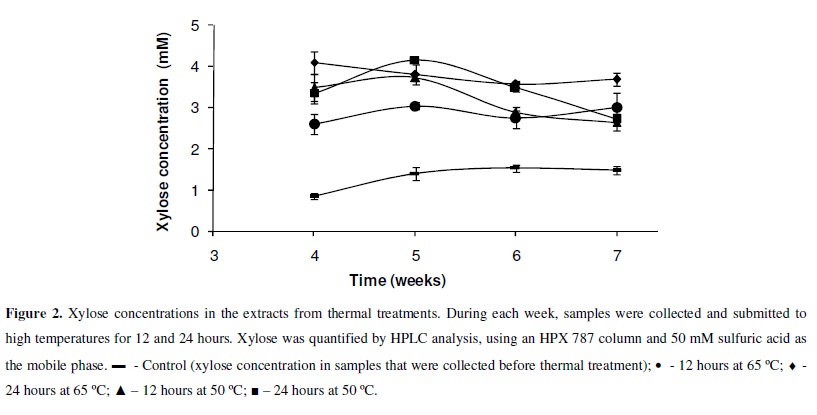
For thermal treatment of the cultures, L. edodes was cultured in semi-solid medium at 25 ºC for seven weeks. Every week, from the fourth to the seventh week, medium containing the mycelia were transferred to temperatures of 50 and 65 ºC, for 12 and 24 hours, respectively. At these temperatures, this fungus does not grow, but its enzymes retain considerable activity for at least 21 hours (this study). After the incubation intervals, 15 mL of Na-acetate buffer (50 mM, pH 5.0) was added to 10 g of semi-solid colonized medium. Xylose in the extract was quantified by HPLC analysis using an HPX 787 column and 50 mM sulfuric acid as the mobile phase. All experiments were carried out in triplicate (n=3). The averages of thermal treatment experiments data were compared using the "t" test, with p > 0.05.
In this experiment, the enzymes hydrolyzed xylan in medium at both temperatures tested, resulting in higher xylose concentrations, as compared with the respective controls (Figure 2) (p > 0.05). However, these xylose concentrations were not significantly different in all thermal treatments (50 ºC for 12 or 24 hours and 65 ºC for 12 or 24 hours) (p>0.05) (Figure 2). This error was probably a consequence of water loss during thermal treatments. Additional experiments are necessary to know exactly which factors limit the release of xylose and how to improve enzyme activity in this semi-solid medium, increasing monosaccharide sugar release.
These data indicate the possibility of incubating crude enzymatic extracts at high temperatures, at which activity is higher and persists longer. This would represent an advantage to industrial processes, as reported by Sandrim et al. (13).
Submitted: February 02, 2011; Approved: August 15, 2011.
*Corresponding Author. Mailing address: Biochemistry and Immunology Departament or Faculdade de Medicina de Ribeirão Preto, USP.; E-mail: lilianebqi@usp.br
- 1. Bradford, M.M. (1976) A dye binding assay for protein. Anal. Biochem. 72:248-9 254.
- 2. Cavallazzi, J.R.P.O.; Almeida, M.G.; Kasuya, M.C.M. (2004). Laccase production by Lepista sordida Braz. J. Microbiol. 35, 261-263.
- 3. Elisashvili, V.P., H.; Kachlishvili, E.; Chichua, D.; Bakradze, M.; Kohreidze, N. (2001). Ligninolytic activity of basidiomycetes grown under submerged and solid-state fermentation on plant raw material (sawdust of grapevine cuttings). Adv. Food. Sci 23, 117-123.
- 4. Elisashvili, V.; Penninckx, M.; Kachlishvili, E.; Tsiklauri, N.; Metreveli, E.; Kharziania, T.; Kvesitadze, G. (2007). Lentinus edodes and Pleurotus species lignocellulolytic enzymes activity in submerged and solid-state fermentation of lignocellulosic wastes of different composition. Bioresour. Technol. 99, 457-462.
- 5. Haki, G.D.; Rakshit, S.K. (2003). Developments in industrially important thermostable enzymes: a review. Bioresour. Technol. 89, 17-34.
- 6. Lakó, J.; Hancsók, J.; Yuzhakova, T.; Marton, G.; Utasi, A.; Rédey, A. (2008). Biomass - a Source of Chemicals and Energy for Sustainable Development. Environ. 24 Eng. Manage J. 7, 499-509.
- 7. Lee, J.W; Gwak, K.S.; Kim, SI; Kim, M.; Choi, D.H.; Choi, I.G. (2007). Characterization of Xylanase from Lentinus edodes M290 Cultured on Waste Mushroom Logs. J. Microbiol. Biotechnol. 17, 1811-1817.
- 8. Lee, T.H.; Lim, P.O.; Lee, Y.E. (2007). Cloning, characterization, and 4 expression of xylanase A gene from Paenibacillus sp. DG-22 in Escherichia coli J. Microbiol. Biotechnol. 17, 29-36.
- 9. Lucena-Neto, S.A.; Ferreira-Filho, E.X. (2004). Purification and Characterization of a new Xylanase from Humicola grisea var. thermoidea Braz. J.Microbiol 35, 86-90.
- 10. Miller, G. L. (1959). Use of dinitrosalicyIic acid reagent for determination of reducing sugar. Analytical Chemistry, vol.31, pp. 426-428.
- 11. Mishra, C.; Forrester, I.T.; Kelley, B.D.; Burgess, R.R.; Leatham, G.F. (1990). Characterization of a major xylanase purified from Lentinula edodes cultures grown on a commercial solid lignocellulosic substrate. Appl. Microbiol. Biotechnol. 33, 226-232.
- 12. Polizeli, M.L.T.M.; Rizzatti, A.C.S.; Monti, R.; Terenzi, H.F.; Jorge, J.A.; Amorim, D.S. (2005). Xylanases from fungi: properties and industrial applications. Appl. Microbiol. Biotechnol. 67, 577-591.
- 13. Sandrim, V.C.; Rizzatti, A.C.S.; Terenzi, H.F.; Jorge, J.A.; Milagres, A.M.F.; Polizeli, M.L.T.M. (2005). Purification and biochemical characterization of two xylanases produced by Aspergillus caespitosus and their potential for kraft pulp bleaching. Process Biochem. 40, 1823-1828.
- 14. Sánchez, C. (2009). Lignocellulosic residues: biodegradation and bioconversion by fungi. Biotechnol Adv. 27, 185-194.
- 15. Sampaio, F.C.; Mantovani, H.C.; Passos, F.J.V.; De Moraes, C.A.; Converti, A.; Passos, F.M.L. (2005). Bioconversion of D-xylose to xylitol by Debaryomyces hansenii UFV-170 : Product formation versus growth. Process Biochem. 40, 3600-3606.
- 16. Singh, S.; Pillay, B.; Prior, B.A. (2000). Thermal stability of β-xylanases produced by different Thermomyces lanuginosus strains. Enzyme Microb. Technol. 26, 502-508.
Publication Dates
-
Publication in this collection
02 May 2012 -
Date of issue
Mar 2012
History
-
Received
02 Feb 2011 -
Accepted
15 Aug 2011