Abstract
Ten bacterial strains that utilize cyanide (CN) as a nitrogen source were isolated from cassava factory wastewater after enrichment in a liquid media containing sodium cyanide (1 mM) and glucose (0.2% w/v). The strains could tolerate and grow in cyanide concentrations of up to 5 mM. Increased cyanide levels in the media caused an extension of lag phase in the bacterial growth indicating that they need some period of acclimatisation. The rate of cyanide removal by the strains depends on the initial cyanide and glucose concentrations. When initial cyanide and glucose concentrations were increased up to 5 mM, cyanide removal rate increased up to 63 and 61 per cent by Bacillus pumilus and Pseudomonas putida. Metabolic products such as ammonia and formate were detected in culture supernatants, suggesting a direct hydrolytic pathway without an intermediate formamide. The study clearly demonstrates the potential of aerobic treatment with cyanide degrading bacteria for cyanide removal in cassava factory wastewaters.
cassava factory wastewater; aerobic degradation; cyanide degrading bacteria
Introduction
The cassava processing industries, producing starch from raw cassava tubers are regarded as highly polluting agro industries in South India as they let out large quantities of wastewater, which is highly toxic and organic in nature causing a serious threat to the environment and aquatic life in the receiving waters. Unfortunately, during processing of cassava large amounts of natural cyanoglucosides released from their roots are enzymatically hydrolysed to cyanide often found in the wastewater discharges (Balagopalan and Rajalekshmy, 1998Balagopalan C, Rajalekshmy L (1998) Cyanogen accumulation in environment during processing of cassava (Manihot esculentaCrantz) for starch and sago. Water Air Soil Pollut 102:407–413.). The detected cyanide level in cassava wastewater ranged between 10.4 and 274 mg L−1 depending upon the cyanoglycoside content of the cassava varieties (Kaewkannetra et al., 2009Kaewkannetra P, Imai T, Garcia FJ et al. (2009) Cyanide removal from cassava mill wastewater using Azotobactor vinelandii TISTR 1094 with mixed microorganisms in activated sludge treatment system. J Hazard Mater 172:224–228.).
In India, Central Pollution Control Board has set a minimal national standard limit for cyanide in wastewater as 0.2 mg L−1. Current wastewater treatments for cyanide removal employ physical and chemical methods which are often expensive and involve the use of additional hazardous reagents (chlorine and sodium hypochlorite) (Watanabe et al., 1998Watanabe A, Yano K, Ikebukuro K et al. (1998) Cyanide hydrolysis in cyanide degrading bacterium, Pseudomonas stutzeri AK61, by cyanidase. Microbiology 144:1677–1682., Patil and Patnikar, 2000Patil YB, Paknikar KM (2000) Development of a process for biodetoxification of metal cyanide from wastewater. Process Biochem 35:1139–1151.). Thus, there is an urgent need for an alternative treatment process capable of achieving high degradation efficiency at low costs. Despite cyanide’s toxicity to living organisms, biodegradation of cyanide which typically relies upon the acclimatization and enhancement of indigenous microorganisms are feasible alternatives, which are potentially inexpensive and environmental friendly (Dash et al., 2009Dash RR, Gaur A, Balomajumder C (2009) Cyanide in industrial wastewaters and its removal: A review on biotreatment. J Hazard Mater 152:387–396.). Recently, Park et al., 2008Park D, Lee DS, Kim YM et al. (2008) Bioaugmentation of cyanide-degrading microorganisms in a full-scale cokes wastewater treatment facility. Bioresour Technol 99:2092–2096.reported the microbial destruction of cyanide using bacteria that possess enzymes to metabolize cyanide into non-toxic products. This enzymatic cyanide removing property can be exploited for the detoxification of cyanide rich cassava wastewater and industrial residues (Akcil et al., 2003Akcil A, Karahan AG, Ciftci H et al. (2003) Biological treatment cyanide by natural isolated bacteria (Pseudomonas sp.). Miner Eng 16:643–649.).
Considering the diverse range of cyanide compounds and their recalcitrance to biodegradation, it is still necessary to screen more cyanide-degrading microorganisms in the environment. Most organisms capable of degrading cyanide are sensitive to cyanide concentration, with biodegradation and/or growth rate decreasing above specific thresholds for each organism. As industrial wastewater can have concentrations above the maximum range for most organisms, continuous investigations are going on for testing the degrading abilities of new microbes for a variety of cyanide compounds, both under laboratory and field conditions. However, field applications of cyanide degradation utilized only naturally occurring isolates obtained by enrichment culture developed in the laboratory or by natural selection in the field. As scanty information is available on the ability of indigenous organisms to degrade and remove cyanide from cassava wastewater, the present study was concerned with three different aspects: (a) to study the toxicity of cyanide and acclimatisation of the autochthonous bacteria from cassava discharges, (ii) aerobic degradation of cyanide, and (iii) the metabolic end products of cyanide degradation. To our knowledge, there has been no investigation on the cyanide removal and degradation pathway of autochthonous microorganisms from cassava wastewater. This work would throw light on our fundamental understanding of biological removal of cyanide from cassava factory wastewaters.
Materials and Methods
Sampling and enrichment culture
Samples were collected from wastewater discharges of the cassava processing industries located in Salem, Tamil Nadu, India. Bacterial communities were obtained by the subculture of enrichment cultures containing cyanide as sole nitrogen source. The enrichment medium consisted of the following (g/L): K2HPO4.2H2O 1.0, MgSO4.7H2O 0.2, CaCl2.2H2O 0.01, NaCl 0.01, MnSO4.4H2O 0.2, CuSO4.5H2O 0.2 and ZnSO4.7H2O 0.2 with pH 7.0. Cyanide (1 mM NaCN) and glucose (2 g/L) were filter sterilized (0.2 μm pore size; Sartorius, Germany) before adding to the medium. The enrichment cultures were incubated at 37 °C and 125 rpm. The culture solution was transferred into fresh enrichment medium at every 5-day intervals. After three successive sub culturings as above, 0.1 mL aliquots of the culture were streaked onto agar plates containing 1 mM CN as a sole nitrogen source. Distinct colonies were purified and picked onto slants of nutrient agar and maintained at 4 °C.
Identification
Identification of the strains was carried out by using 16S rRNA gene sequencing method. The 16S rRNA gene was amplified and sequenced using universal primers forward MF: 5′-GAG TTT GAT CMT GGC TCA G-3′ and reverse MR: 5′-ACG GYT ACC TTG TTA CGA CTT-3′ (Weisburg et al., 1991Weisburg WG, Barns SM, Pelletier DA et al. (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703.) and the PCR products were purified using spin columns according to the manufacturer’s instructions (Qiagen, Germany). Sequencing reactions were performed using ABI prism terminator cycle sequencing ready reaction kit and electrophoresis of the products were carried out on an Applied Biosystems (Model 3100) automated sequencer.
Biodegradation assay of cyanide
All the bacterial isolates were grown individually in nutrient broth for 48-h was harvested by centrifugation (10000 rpm for 10 min at 4 °C) and washed twice in a 50 mM phosphate buffer (pH 7). Washed cells were again suspended in the same buffer (A660 = 1.0) and 0.5 mL of the suspension inoculated into 50 mL of the mineral salts medium containing NaCN as sole nitrogen source and glucose as carbon source in a 150-mL Erlenmeyer flask. The cultures in triplicates were incubated at 30 °C with shaking and the samples were taken at regular intervals to measure optical density, cyanide, formamide, formic acid and ammonia.
In order to highlight the effect of cyanide concentration on the growth and cyanide degradation efficiency of the individual bacterial isolates, each isolate was grown in MSM containing increasing concentrations of CN (1 and 5 mM) as described above.
The effect of carbon (glucose) concentration on the growth and cyanide degradation efficiency by the bacterial isolates was also examined at two different glucose concentrations (1 mM and 5 mM) as described earlier.
Analytical methods
Growth determination
Cell growth was monitored by determining the optical density (O.D) of 1 mL culture at 660 nm by Spectrophotometry (GENESYS 10 UV-Vis Scanning, Thermo Scientific) and expressed as OD660 nm.
Cyanide determination
Cyanide content was determined by a modification of the picric acid method of Fisher and Brown (1952)Fisher FB, Brown JS (1952) Colorimetric determination of cyanide in stack gas and water. Analytical Chemistry 24:1440–1444.. A linear calibration curve was obtained with the standard cyanide solutions as follows: aliquots (0.05 mL) of cyanide solutions (after centrifugation at 15,000 g for 10 min at 4 °C) were added to 0.1 mL aliquots of a solution containing 0.5% (w/v) picric acid and 0.25 M Na2CO3. The resulting solutions were placed in a boiling water-bath for 5 min, diluted to 1 mL with 0.85 mL distilled water, and cooled in tap water for 30 min. The absorbance was read at 520 nm against a blank of distilled water and picric acid reagent.
Ammonia determination
Aliquot (2.5 mL) of the sample (after centrifugation at 15,000 rpm for 10 min at 4 °C) was added to 1.25 mL of Na-nitroprusside solution and vortexed briefly for a few seconds. To this, 500 μL of dichloroisocyanurate was added and the samples were incubated for 30 min in the dark. The absorbance was measured at 625 nm using ammonium chloride as standard.
HPLC analysis
Glucose and formate were assayed by HPLC (Sykam, Gilching, Germany), equipped with an HPX-87 P ion-exchange column Aminex (Bio-Rad Lab, USA). The mobile phase was 5 mM H2SO4 with the flow rate 0.8 mL/min. The column was maintained at room temperature and the absorbance was measured at 210 nm. All samples were filter-sterilized (0.2 μm pore size) to remove cells and other particles before analysis.
Degradation efficiency
The degradation efficiency (DE) of cyanide degrading bacterium was calculated as shown in following formula.
where, Ic = Initial concentration of cyanide (mg/L) and Rc = Residual concentration of cyanide (mg/L).
Results and Discussion
Isolation and identification of the bacteria
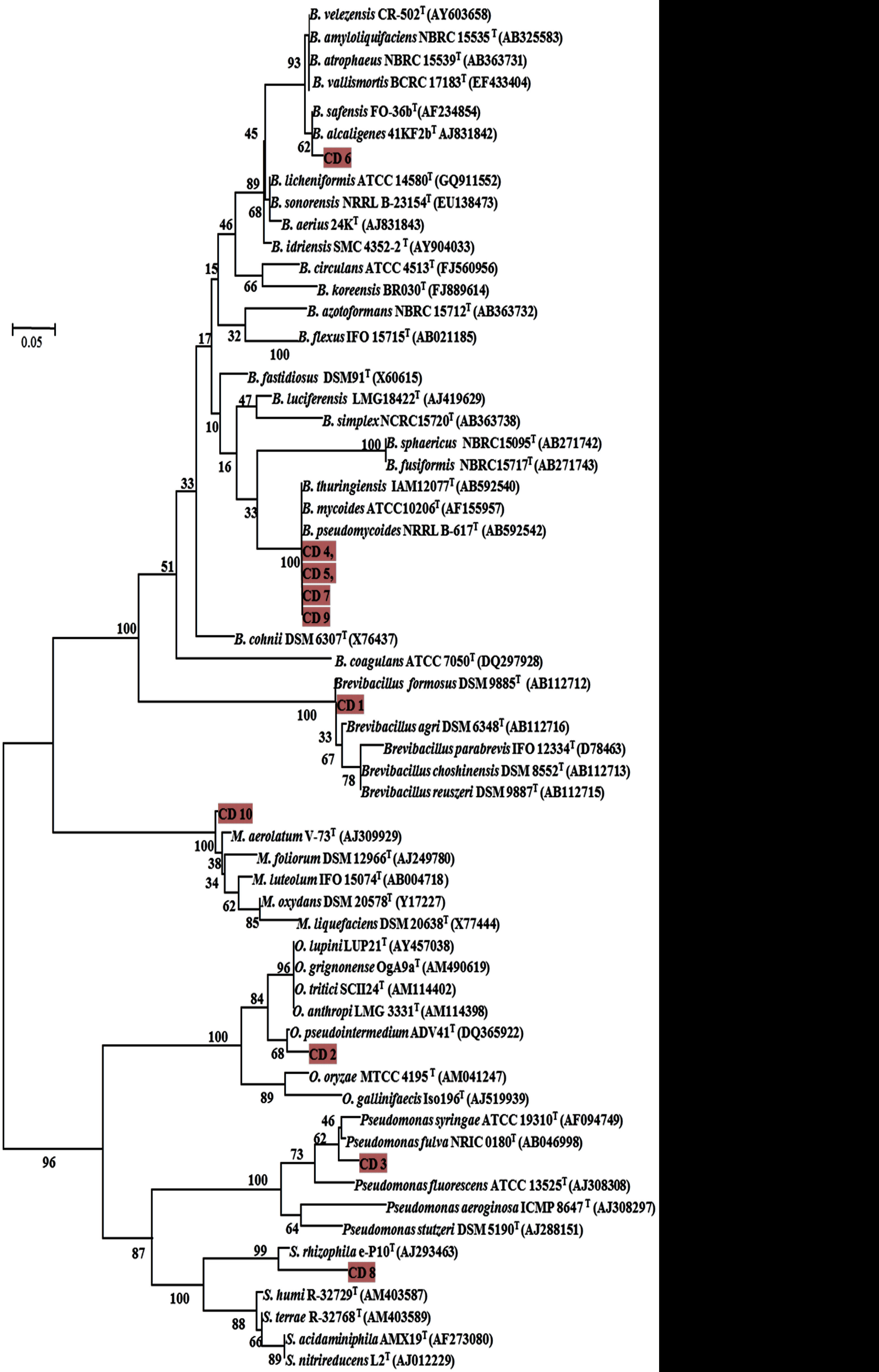
Selection of microorganisms from the autochthonous microflora of cyanide-contaminated wastewater using enrichment technique led to the isolation of seven gram positive and three gram negative bacteria. Molecular analysis based on 16S rRNA gene sequencing revealed that isolates CD 5 and CD 7 showed maximum similarity with Bacillus cereus to about 98 per cent each respectively (Figure 1).
Neighbour-joining phylogenetic tree based on partial 16S rRNA gene of bacterial isolates from sago wastewater. The related species, strain name and accession number were collected from NCBI, GenBank database. The boot strap value of more than 50% (out of 1000) were denoted in each node of the tree. The scale bar indicates per cent changes in nucleotide position (5 out of 100 bp)
The isolates CD 2, CD 8 and CD 10 showed the highest similarity to Ochrobactrum intermedium (99 per cent), Stenotrophomonas maltophila (98 per cent) and Microbacterium paraoxydans (99 per cent) respectively. The isolates CD 1 and CD 3 was closely related to Brevibacillus brevis (98 per cent) and Pseudomonas putida (99 per cent). The isolates CD 4, CD 6 and CD 9 shared the highest similarity to Bacillus anthracis (98 per cent), Bacillus pumilus (98 per cent) and Bacillus weihenstephenensis (99 per cent).
Cyanide utilization by the bacterial isolates
Broth culture more closely mimics conditions of an aqueous sample and bioremediation plants typically involve inoculation of contaminated material using a liquid carrier solution. Therefore, evaluation of growth in broth culture for each bacterial group was carried out prior to inoculation experiments. Growth was scored as positive in broth culture, if the microorganisms showed an increase in optical density. Attempts to culture the bacterial isolates with CN as a sole carbon and nitrogen source were unsuccessful. Growth was observed when an external carbon source (glucose) was provided in the medium even at higher cyanide concentrations (5 mM). The toxicity of cyanide compounds restricts microbes for using them as primary carbon source for growth. Since the amount of nitrogen needed for growth is less than the requirement for carbon, it could be easier to utilize cyanide as a source of nitrogen in the presence of another source of carbon and energy. In the cyanide molecule, the oxidation state of C (+2, like that in CO) and N (−3, like that in NH4+) made this compound a bad C source but a good N source for bacterial growth (Dursun and Aksu, 2002Dursun AY, Aksu Z (2002) Biodegradation kinetics of ferrous (II) cyanide complex ions by immobilized Pseudomonas fluorescens in a packed bed column reactor. Process Biochem 35:615–622.).
Growth and cyanide degradation in flask scale
The growth and cyanide degradation efficiency of the individual bacterial isolates was determined in a complete mineral media supplemented with cyanide (1 mM and 5 mM) and glucose (1 mM and 5 mM). As growth occurred in the medium, levels of cyanide and glucose decreased in proportional to the incubation periods. The decrease of cyanide concentration in the medium was concomitant with an increase in cell density (bacterial population). The fact that the final cell density obtained was considerably high indicated the use of well acclimatised cyanide metabolizing culture. Higher microbial growth in terms of OD660 (Figure 2) was obtained with B. pumilus, P. putida followed by B. cereus C2 in media containing cyanide and glucose each at 5 mM concentrations. Even though, an increase in cell density was observed at higher cyanide concentration, at initial phase the cells needed some period of acclimatization. Particularly, it was observed that the period corresponding to initial microbial adaption phase before starting the cyanide degradation, significantly increased as the initial cyanide concentration is increased, and strongly suggesting a microbial inhibitor effect at higher concentrations (Panos and Bellini, 1999Panos NH, Bellini MR (1999) Microbial degradation of cyanides. Mine, water and environment 1:201–206.). In addition, cyanide concentration at 5 mM caused an extension of lag phase with the growth of the bacterial isolates at both the glucose concentrations. Extension of lag phase at higher cyanide concentrations in bacteria was already reported with P. putida (Chapatwala et al., 1998Chapatwala KD, Babu GRV, Vijaya OK et al. (1998) Biodegradation of cyanides, cyanates and thiocyanates to ammonia and carbon dioxide by immobilized cells of Pseudomonas putida. J Ind Microbiol Biotechnol 20:28–33.) and Zymomonas mobilis (Kalnenieks et al., 2000Kalnenieks U, Galinina N, Toma MM et al. (2000) Cyanide inhibits respiration yet stimulates aerobic growth of Zymomonas mobilis. Microbiol 146:1259–1266.).
Growth performance by the bacterial isolates in broth containing (a) each cyanide and glucose at 1 mM (b) cyanide and 5 mM glucose (c) 5 mM cyanide and 1 mM glucose (d) cyanide and glucose each at 5 mM
Interestingly, the rate of cyanide consumption upon subsequent time was dramatically faster than that observed upon initial inoculation, suggesting that adaptation to cyanide had occurred. When cyanide concentrations were increased in the media, the rate of cyanide removal also increased. The rate of cyanide removal seems to be dependent on the initial NaCN concentration. Azotobacter vinelandii reduced the cyanide levels to 56, 58 and 65.3 per cent corresponding to increased initial NaCN concentrations of 50, 100 and 150 mg/L respectively (Kaewkannetra et al., 2009Kaewkannetra P, Imai T, Garcia FJ et al. (2009) Cyanide removal from cassava mill wastewater using Azotobactor vinelandii TISTR 1094 with mixed microorganisms in activated sludge treatment system. J Hazard Mater 172:224–228.). In addition, increased levels of glucose in the media further improved the cyanide degrading activity of the bacterial isolates. Ezzi and Lynch (2005)Ezzi MI, Lynch JM (2005) Biodegradation of cyanide by Trichoderma spp. and Fusarium spp. Enzyme Microb Technol 36:849–854.noticed that the inclusion of glucose could improve degradation rate by three times in Trichoderma spp. Addition of higher glucose appears to act as a source of energy in sustaining the biomass so that the cyanide degrading capability is maintained during the stationary phase, although there is no increase in growth in the form of biomass. A maximum cyanide removal of 63 and 61 per cent was obtained with B. pumilus and P. putida in the media with cyanide and glucose concentration each at 5 mM (Figure 3).
Cyanide degrading efficiency of the bacterial isolates in mineral broth at different cyanide and glucose concentrations
Meanwhile, cyanide concentrations in the control system remained almost unchanged throughout the experimental process indicating that the depletion of cyanide is primarily due to biodegradation activity. The cyanide degrading activity of Bacillus anthracis and B. cereus C1 was very low compared to all other isolates, at both 1 mM and 5 mM cyanide concentration even with addition of glucose as co-metabolite. This may be due to the presence of a low level of constitutive synthesis of the cyanide degrading enzymes (Adjei and Ohta, 1999Adjei MD, Ohta Y (1999) Isolation and characterization of a cyanide-utilizing Burkholderia cepacia strain. World J Microbiol Biotechnol 15:699–704.).
Effect of carbon source on cyanide degradation
Glucose appears to act as a co-metabolite and there was a proportional increase of biomass in the glucose-inoculated experiments to serve this co-metabolism. Presence of glucose in the media remarkably increased the rate of cyanide degradation. As far as the requirement of carbon was concerned, it was observed that the minimum concentration of glucose required by the bacteria for degradation of 50 mg/L of SCN was 5 mM.
However, degradation efficiency dropped when < 5 mM glucose was incorporated in the medium (Yogesh, 2008Yogesh PB (2008) Degradation of thiocyanate from aqueous waste by a mixed bacterial community. Res J Chem Environ 12:69–75.). Similarly, the cyanide removal efficiency of all the bacterial isolates was tend to increase at 5 mM compared to 1 mM glucose concentration, at both the cyanide concentrations. The glucose utilization in the presence of cyanide, by the bacterial isolates is an indication of presence of metabolic activity of these strains and energy production by them (Ezzi and Lynch, 2005Ezzi MI, Lynch JM (2005) Biodegradation of cyanide by Trichoderma spp. and Fusarium spp. Enzyme Microb Technol 36:849–854.). An interesting observation recorded was that there is an apparent link between the decrease in cyanide concentration and the reduction of glucose levels in the medium. Higher glucose utilization of 86 per cent was observed with Bacillus pumilus, which reported maximum cyanide removal of 63 percent in media with 5 mM cyanide and glucose concentration each (Figure 4).
Glucose utilization efficiency of the bacterial isolates in mineral broth at different cyanide and glucose concentrations
Identification of the metabolic end products of cyanide
Microorganisms can grow on substrates containing cyanide by anaerobic metabolism, or by using an aerobic respiration chain as an alternative pathway. Raybuck (1992)Raybuck SA (1992) Microbes and microbial enzymes for cyanide degradation. Biodegradation 3:3–18. and Ebbs (2004)Ebbs S (2004) Biological degradation of cyanide compounds. Curr Opin Biotechnol 15:231–236. demonstrated the existence of different metabolic ways that result in end-products such as formamide, formic acid or ammonia as an outcome of cyanide degradation by various microorganisms. Our results showing cyanide degradation to formate and ammonia are in line with the hydrolytic pathway. A significant formamide formation during biological cyanide degradation was excluded, because there was no evidence of formamide formation, in batch cultures. Thus, biological degradation of cyanide must have proceeded almost exclusively to formate and ammonia. Hydrolytic reactions are mainly characterized for the direct formation of the products formamide or formic acid and ammonium, which are less toxic than cyanide and may also serve for growth of the bacteria (Huertas et al., 2006Huertas MJ, Luque-Almagro VM, Martinez-Luque M et al. (2006) Cyanide metabolism of Pseudomonas pseudoalcaligens CECT5344: role of siderophores. Biochem Soc Trans 34:152–155.).
Ammonia was described as one of the end-products of the aerobic (Ingvorsen et al., 1991Ingvorsen K, Hojer-Pedersen B, Godtfredsen SE (1991) Novel cyanide-hydrolyzing enzyme from Alcaligenes xylosoxidans subsp. denitrificans. Appl Environ Microbiol 57:1783–1789.) and anaerobic (Fallon et al., 1991Fallon RD, Cooper DA, Speece R et al. (1991) Anaerobic biodegradation of cyanide under methanogenic conditions. Appl Environ Microbiol 57:1656–1662.) degradation of cyanide. Profiles in all the broth cultures, at both 1 and 5 mM cyanide concentrations generally showed that free cyanide disappearance was accompanied by a concomitant increase in ammonia. Rapid removal of cyanide and the simultaneous accumulation of ammonia were observed in the reaction mixtures. This is in agreement with Fisher et al. (2000)Fisher K, Dilworth MJ, Kim CH et al. (2000) Azotobacter vinelandii nitrogenises with substitutions in the FeMo-cofactor environment of the MoFe protein: effects of acetylene or ethylene on interactions with H+, HCN, and CN−. Biochem 39:10855–10865. who observed increased NH3 and CH4 at NaCN concentrations of up to 5 mM indicative of increased cyanide reduction. The pattern of ammonia production coincided with cell growth rather than cyanide disappearance. There are several reports on microbial production of ammonia from cyanide (Ingvorsen et al., 1991Ingvorsen K, Hojer-Pedersen B, Godtfredsen SE (1991) Novel cyanide-hydrolyzing enzyme from Alcaligenes xylosoxidans subsp. denitrificans. Appl Environ Microbiol 57:1783–1789.; Kao et al., 2003Kao CM, Liu JK, Lou HR et al. (2003) Biotransformation of cyanide to methane and ammonia by Klebsiella oxytoca. Chemosphere 50:1055–1061.), but the enzymatic pathways involved have not yet been elucidated. Production of ammonia from cyanide may occur via a number of different enzymatic pathways (Ebbs, 2004Ebbs S (2004) Biological degradation of cyanide compounds. Curr Opin Biotechnol 15:231–236.) and is in itself, therefore, no proof of the involvement of single enzyme. Several heterotrophic bacteria, a Pseudomonas stutzeri like bacterium, Arthrobacter and Methylobacterium thiocyanatum, degrade thiocyanate during growth and to use the ammonia released as a source of nitrogen (Yogesh, 2008Yogesh PB (2008) Degradation of thiocyanate from aqueous waste by a mixed bacterial community. Res J Chem Environ 12:69–75.). In the present study, the molar ratio of ammonia production from cyanide degraded was not in stoichiometric levels as that of formate suggesting that some of the nitrogen was assimilated into the cell biomass (White et al., 2000White DM, Pilon TA, Woolard C (2000) Biological treatment of cyanide containing wastewater. Water Res 34:2105–2109.). Similarly, Adjei and Ohta (1999)Adjei MD, Ohta Y (1999) Isolation and characterization of a cyanide-utilizing Burkholderia cepacia strain. World J Microbiol Biotechnol 15:699–704. observed the accumulation of ammonia during cyanide degradation by Burkholderia, which was apparently further metabolized, particularly under prolonged incubation periods, when cyanide was supplied at low (2 mM) concentrations.
Besides ammonia, formate was also found in the broth cultures in almost equimolar amounts to the degraded cyanide except with Brevibacillus brevis and Ochrobactrum intermedium due to the rapid conversion of formate into CO2 by both the bacterial isolates. Kwon et al. (2002)Kwon HK, Woo SH, Park JM (2002) Degradation of tetracyanonickelate(II) by Cryptococcus humicolus MCN2. FEMS Microbiol Lett 214:211–216.reported similar results from potassium cyanide degradation using Cryptococcus humicolus indicating that more than one enzymatic pathway could be involved in the cyanide degradation. Formamide could not be detected by HPLC indicating direct hydrolysis of cyanide to formate by the bacterial isolates (i.e., HCN + 2H2O → HCOOH + NH3). Similar results were already reported from bacterial isolates Alcaligenes xylosoxidans subsp. denitrificans (Ingvorsen et al., 1991Ingvorsen K, Hojer-Pedersen B, Godtfredsen SE (1991) Novel cyanide-hydrolyzing enzyme from Alcaligenes xylosoxidans subsp. denitrificans. Appl Environ Microbiol 57:1783–1789.), Pseudomonas stutzeri (Watanabe et al., 1998Watanabe A, Yano K, Ikebukuro K et al. (1998) Cyanide hydrolysis in cyanide degrading bacterium, Pseudomonas stutzeri AK61, by cyanidase. Microbiology 144:1677–1682.) and Burkholderia cepacia(Adjei and Ohta, 1999Adjei MD, Ohta Y (1999) Isolation and characterization of a cyanide-utilizing Burkholderia cepacia strain. World J Microbiol Biotechnol 15:699–704.) to degrade cyanide into ammonia and formate directly without any intermediate (formamide) formation by the enzyme cyanidase (cyanide dihydratase).
These results provide a good perspective for the aerobic degradation of cyanide in waste waters. Further research is required to establish the optimum conditions for microbial adaptation and cyanide degradation under aerobic conditions. Once these have been established, aerobic treatment may also be considered specifically for the removal of cyanide from waste streams.
Conclusions
The ultimate aim of this study was to investigate if the autochthonous cyanide utilizers of the cassava wastewater could serve as a biomaterial for CN removal from aqueous solutions and its potential could be increased by external carbon supply. The rate of cyanide removal increased at higher cyanide and glucose concentrations and a maximum of 63 per cent CN removal was observed with B. pumilus. These results showed that aerobic systems can be successfully applied for cyanide removal from aqueous solutions up to high CN concentrations. Our effort is contribution of these studies to a sufficiently economical and efficient method, which can be taken as a promising alternative technique offering an applicable solution to the problem. The continued development and application of biotechnologies for cyanide biodegradation is limited primarily by physical and economic factors. However, optimization of the use of such biological systems in wastewater is still required. Further, development of microbial processes that is efficient in extreme environmental conditions, such as low pH and contaminant toxicity, is required to ensure a technology that is competitive with the chemical and physical remediation strategies currently practiced to reduce cyanide pollution. This would go a long way in creating safe and friendly environment in communities where cassava tubers are processed into products.
Acknowledgments
The authors would like to gratefully acknowledge the support extended by the authorities of Justus Liebig University, Giessen in carrying out the research work in Department of Agrobiotechnology for providing lab facilities and DST and DAAD fellowship for their financial support.
References
- Adjei MD, Ohta Y (1999) Isolation and characterization of a cyanide-utilizing Burkholderia cepacia strain. World J Microbiol Biotechnol 15:699–704.
- Akcil A, Karahan AG, Ciftci H et al. (2003) Biological treatment cyanide by natural isolated bacteria (Pseudomonas sp.). Miner Eng 16:643–649.
- Balagopalan C, Rajalekshmy L (1998) Cyanogen accumulation in environment during processing of cassava (Manihot esculentaCrantz) for starch and sago. Water Air Soil Pollut 102:407–413.
- Baxter J, Cummings SP (2006) The current and future applications of microorganism in the bioremediation of cyanide contamination. Antonie Van Leeuwenhoek 90:1–17.
- Chapatwala KD, Babu GRV, Vijaya OK et al. (1998) Biodegradation of cyanides, cyanates and thiocyanates to ammonia and carbon dioxide by immobilized cells of Pseudomonas putida J Ind Microbiol Biotechnol 20:28–33.
- Dash RR, Gaur A, Balomajumder C (2009) Cyanide in industrial wastewaters and its removal: A review on biotreatment. J Hazard Mater 152:387–396.
- Dursun AY, Aksu Z (2002) Biodegradation kinetics of ferrous (II) cyanide complex ions by immobilized Pseudomonas fluorescens in a packed bed column reactor. Process Biochem 35:615–622.
- Ebbs S (2004) Biological degradation of cyanide compounds. Curr Opin Biotechnol 15:231–236.
- Ezzi MI, Lynch JM (2005) Biodegradation of cyanide by Trichoderma spp. and Fusarium spp. Enzyme Microb Technol 36:849–854.
- Fallon RD, Cooper DA, Speece R et al. (1991) Anaerobic biodegradation of cyanide under methanogenic conditions. Appl Environ Microbiol 57:1656–1662.
- Fisher FB, Brown JS (1952) Colorimetric determination of cyanide in stack gas and water. Analytical Chemistry 24:1440–1444.
- Fisher K, Dilworth MJ, Kim CH et al. (2000) Azotobacter vinelandii nitrogenises with substitutions in the FeMo-cofactor environment of the MoFe protein: effects of acetylene or ethylene on interactions with H+, HCN, and CN− Biochem 39:10855–10865.
- Huertas MJ, Luque-Almagro VM, Martinez-Luque M et al. (2006) Cyanide metabolism of Pseudomonas pseudoalcaligens CECT5344: role of siderophores. Biochem Soc Trans 34:152–155.
- Ingvorsen K, Hojer-Pedersen B, Godtfredsen SE (1991) Novel cyanide-hydrolyzing enzyme from Alcaligenes xylosoxidans subsp. denitrificans. Appl Environ Microbiol 57:1783–1789.
- Kaewkannetra P, Imai T, Garcia FJ et al. (2009) Cyanide removal from cassava mill wastewater using Azotobactor vinelandii TISTR 1094 with mixed microorganisms in activated sludge treatment system. J Hazard Mater 172:224–228.
- Kalnenieks U, Galinina N, Toma MM et al. (2000) Cyanide inhibits respiration yet stimulates aerobic growth of Zymomonas mobilis Microbiol 146:1259–1266.
- Kao CM, Liu JK, Lou HR et al. (2003) Biotransformation of cyanide to methane and ammonia by Klebsiella oxytoca Chemosphere 50:1055–1061.
- Kwon HK, Woo SH, Park JM (2002) Degradation of tetracyanonickelate(II) by Cryptococcus humicolus MCN2. FEMS Microbiol Lett 214:211–216.
- Panos NH, Bellini MR (1999) Microbial degradation of cyanides. Mine, water and environment 1:201–206.
- Park D, Lee DS, Kim YM et al. (2008) Bioaugmentation of cyanide-degrading microorganisms in a full-scale cokes wastewater treatment facility. Bioresour Technol 99:2092–2096.
- Patil YB, Paknikar KM (2000) Development of a process for biodetoxification of metal cyanide from wastewater. Process Biochem 35:1139–1151.
- Raybuck SA (1992) Microbes and microbial enzymes for cyanide degradation. Biodegradation 3:3–18.
- Watanabe A, Yano K, Ikebukuro K et al. (1998) Cyanide hydrolysis in cyanide degrading bacterium, Pseudomonas stutzeri AK61, by cyanidase. Microbiology 144:1677–1682.
- Weisburg WG, Barns SM, Pelletier DA et al. (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703.
- White DM, Pilon TA, Woolard C (2000) Biological treatment of cyanide containing wastewater. Water Res 34:2105–2109.
- Yogesh PB (2008) Degradation of thiocyanate from aqueous waste by a mixed bacterial community. Res J Chem Environ 12:69–75.
Publication Dates
-
Publication in this collection
Jul-Sep 2015
History
-
Received
12 May 2013 -
Accepted
16 Nov 2014