Abstract
Pectinolytic enzymes are greatly important in winemaking due to their ability to degrade pectic polymers from grape, contributing to enhance process efficiency and wine quality. This study aimed to analyze the occurrence of pectinolytic yeasts during spontaneous fermentation of Argentine Bonarda grape, to select yeasts that produce extracellular pectinases and to characterize their pectinolytic activity under wine-like conditions. Isolated yeasts were grouped using PCR-DGGE and identified by partial sequencing of 26S rRNA gene. Isolates comprised 7 genera, with Aureobasidium pullulans as the most predominant pectinolytic species, followed by Rhodotorula dairenensis and Cryptococcus saitoi. No pectinolytic activity was detected among ascomycetous yeasts isolated on grapes and during fermentation, suggesting a low occurrence of pectinolytic yeast species in wine fermentation ecosystem. This is the first study reporting R. dairenensis and Cr. saitoi species with pectinolytic activity. R. dairenensis GM-15 produced pectinases that proved to be highly active at grape pH, at 12 °C, and under ethanol and SO2 concentrations usually found in vinifications (pectinase activity around 1.1 U/mL). This strain also produced cellulase activity at 12 °C and pH 3.5, but did not produce β-glucosidase activity under these conditions. The strain showed encouraging enological properties for its potential use in low-temperature winemaking.
Aureobasidium pullulans ; Cryptococcus saitoi ; pectinolytic activity; Rhodotorula dairenensis ; winemaking
Introduction
Pectinolytic enzymes are polysaccharidases that degrade pectins present in middle lamella and primary cell walls of plants. This ability is widely used in winemaking as pectinases can help to improve liquefaction, juice yield, clarification, filterability, and to increase the release of color and flavor compounds entrapped in grape skins (Fleet, 2008Fleet GH (2008) Wine yeasts for the future. FEMS Yeast Res 8:979–995.; Alimardani-Theuil et al., 2011Alimardani-Theuil P, Gainvors-Claisse A, Duchiron F (2011) Yeasts: An attractive source of pectinases-From gene expression to potential applications: A review. Process Biochem 46:1525–1537.; Martín and Morata de Ambrosini, 2014Martín MC, Morata de Ambrosini VI (2014) Effect of a cold-active pectinolytic system on colour development of Malbec red wines elaborated at low temperature. Int J Food Sci Technol 49:1893–1901.).
Grape skin forms a physical barrier to diffusion of anthocyanins, tannins and aroma contained in the skin cells. The permeability of skin cell walls to these compounds can be increased by partial hydrolysis of their structural polysaccharides (pectins, cellulose and hemicelluloses), a process that can be facilitated by pectinolytic preparations. These enzymes are complex mixtures mainly comprised of pectinases, but also other desirable activities like cellulases, hemicellulases and acid proteases. Cellulases and hemicellulases are also responsible for an increase in the extraction of grape juice, and the improvement in the clarification of wines (Romero-Cascales et al., 2008Romero-Cascales I, Fernández-Fernández JI, Ros-García JM et al. (2008) Characterisation of the main enzymatic activities present in six commercial macerating enzymes and their effects on extracting colour during winemaking of Monastrell grapes. Int J Food Sci Technol 43:1295–1305.).
Nowadays, commercial pectinase preparations are obtained from fungi and apart from desirable activities, they generally contain undesirable enzymes like pectinesterase (Alimardani-Theuil et al., 2011Alimardani-Theuil P, Gainvors-Claisse A, Duchiron F (2011) Yeasts: An attractive source of pectinases-From gene expression to potential applications: A review. Process Biochem 46:1525–1537.) and β-glucosidase (Romero-Cascales et al., 2008Romero-Cascales I, Fernández-Fernández JI, Ros-García JM et al. (2008) Characterisation of the main enzymatic activities present in six commercial macerating enzymes and their effects on extracting colour during winemaking of Monastrell grapes. Int J Food Sci Technol 43:1295–1305.) that can negatively affect the quality of wines. Yeasts are known to produce mostly polygalacturonases (Alimardani-Theuil et al., 2011Alimardani-Theuil P, Gainvors-Claisse A, Duchiron F (2011) Yeasts: An attractive source of pectinases-From gene expression to potential applications: A review. Process Biochem 46:1525–1537.), and they could therefore be a preferable source of pectinases. Considering the key role of yeasts in winemaking, selection of pectinolytic yeasts for enology is an alternative approach to fungal pectinases. Although some pectinolytic wine yeasts have already been described (Fernández et al., 2000Fernández M, Úbeda JF, Briones AI (2000) Typing of non-Saccharomyces yeasts with enzymatic activities of interest in wine-making. Int J Food Microbiol 59:29–36.; Fernández-González et al., 2004Fernández-González M, Úbeda JF, Vasudevan TG et al.(2004) Evaluation of polygalacturonase activity in Saccharomyces cerevisiae wine strains. FEMS Microbiol Lett 237:261–266.), further studies are needed.
“San Rafael” Designation of Origin (DO), an area in central-west Argentina, represents an important wine region in South America. Despite its long viticulture and enology history, very little is known about the microorganisms involved in spontaneous fermentations. Currently, Argentine Bonarda is one of the red grape varieties with increasing enological interest in the country.
Cold-active enzymes are attractive for usage in food industry since low-temperature conditions favor conservation of sensory and nutritional properties in the product (Sahay et al., 2013Sahay S, Hamid B, Singh P et al. (2013) Evaluation of pectinolytic activities for oenological uses from psychrotrophic yeasts. Lett Appl Microbiol 57:115–121.). A previous study reported cold-active pectinase activity by yeasts isolated from wine grapes (Merín et al., 2011Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148.). However, the presence of pectinolytic yeasts during fermentation and their enzymatic activities under winemaking conditions have not been described.
This study reports the occurrence of pectinolytic strains among representative yeasts isolated at different stages during spontaneous fermentation of Argentine Bonarda grape. And it focuses on selection and characterization of their enzymatic behavior under wine-like conditions (pH 3.5, 12 and 28 °C, ethanol and SO2) for their potential use in winemaking.
Materials and Methods
Study area, sampling and fermentation
Grape samples (Vitis vinifera L.) of cv. Argentine Bonarda were collected from Cuadro Benegas viticultural region (lat. 34.62° S, long. 68.45° W) in San Rafael DO, Mendoza, Argentina, during the 2009 vintage.
Representative bunches of healthy grapes were aseptically hand harvested, transported to the laboratory and kept cold until their study. The collected grapes were crushed to conduct the spontaneous fermentation in 1 L Erlenmeyer flasks containing 800 mL of must (reducing sugars 220 g/L; titratable acidity 4.6 g/L; yeast assimilable nitrogen (YAN) 310 mg/L; pH 4.2). Progress of the alcoholic fermentation was monitored daily by measuring weight loss using flasks with stoppers containing a Müller valve that allows only CO2 to escape from the system. Fermentations were carried out in duplicate at 25 ± 1 °C until constant weight for two consecutive days.
Yeast isolation
Before the grape crushing, 10 berries were placed in a flask containing 10 mL of sterile peptone-water (0.1%, w/v) and were shaken at 165 rpm during 1 h at room temperature. Aliquots of adequate dilutions of both berry washing solution and grape must, sampled at 0, 3, 5, 8 and 12 days of fermentation, were plated onto WL Nutrient Agar. This medium allows presumptive discrimination among yeast species by colony morphology and color (Pallmann et al., 2001Pallmann C, Brown JA, Olineka TL et al. (2001) Use of WL medium to profile native flora fermentations. Am J Enol Vitic 52:198–203.). Plates were incubated at 25 °C for 5 days for colony development. A representative number of each colony type was recovered. Isolates were purified by streak plating and subcultured onto Yeast extract Peptone Dextrose (YPD: yeast extract, 10 g/L; peptone, 20 g/L; dextrose, 20 g/L; agar, 20 g/L) for subsequent identification.
Molecular yeast identification
Yeast isolates were examined with PCR-DGGE to group them according to their DGGE mobility and representative strains of each group were subsequently sequenced as described by Rantsiou et al.(2013)Rantsiou K, Campolongo S, Alessandria V et al.(2013) Yeast populations associated with grapes during withering and their fate during alcoholic fermentation of high-sugar must. Aust J Grape Wine Res 19:40–46.. Isolates were subjected to DNA extraction and a DNA fragment from the D1–D2 loop region of the 26S rRNA gene was amplified by PCR using primers NL1GC/LS2 following the protocol proposed by Cocolin et al. (2000)Cocolin L, Bisson LF, Mills DA (2000) Direct profiling of the yeast dynamics in wine fermentations. FEMS Microbiol Lett 189:81–87..
The DCodeTM Universal Mutation Detection System (Biorad, USA) was used for DGGE analysis according to Cocolin et al. (2000)Cocolin L, Bisson LF, Mills DA (2000) Direct profiling of the yeast dynamics in wine fermentations. FEMS Microbiol Lett 189:81–87. with minor modifications in the denaturing gradient (30 to 50% urea-formamide) and the electrophoresis conditions (constant voltage of 130 V for 4 h). To obtain species identification, PCR amplification of representative colonies from each migration-specific group was conducted by using primers NL1/NL4 (Kurtzman and Robnett, 1998Kurtzman CP, Robnett CJ (1998) Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie Van Leeuwenhoek 73:331–371.). PCR products were purified using the QIAquick® PCR Purification Kit (Qiagen, Germany) and sequencing was carried out at MWG Biotech (Germany) and CERELA (Argentina). Strains were identified by searching the GenBank database with the BLAST program (http://www.ncbi.nlm.nih.gov).
Screening of pectinolytic activity among isolated yeasts
The ability of isolated yeasts to hydrolyze pectin was assayed using the Petri dish method according to Merín et al. (2011)Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148.. The isolates were point-inoculated onto a mineral medium containing 0.2% (w/v) citric pectin as carbon source, at pH 4.5, and incubated at 28 °C for 48–72 h. Enzyme activity was evidenced by clear halos around the colonies against a purple-brown background after addition of Lugol’s solution.
Enzyme assays
Production of extracellular enzymatic extracts
For enzyme production, yeasts showing pectinolytic activity on plates were inoculated in a basal liquid medium (containing per L of 50 mM citric-citrate buffer: dextrose, 20 g; soy peptone, 10 g; meat peptone, 10 g; yeast extract, 10 g) at pH 3.5, proximate to wine pH. The cultures were incubated under shaking conditions (100 rpm) at 12 and 28 °C for 5 or 3 days, respectively. Cells were removed by centrifugation (5000× g, 15 min at 4 °C) and supernatants were filtered (0.22 μm) to obtain cell-free supernatants (enzymatic extracts) on which all the enzymatic activities were assayed throughout this study.
The assessment of enzymatic activities in the present work was always carried out at the same temperature of the enzymatic production, at 12 or 28 °C, as appropriate.
Pectinolytic activity
Pectinolytic activity was assayed by measuring the amount of reducing sugars released from a pectin dispersion (0.25% pectin in 50 mM citric-citrate buffer, pH 3.5) using 3,5-dinitrosalicylic acid (DNS) reagent (Miller, 1959Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428.). Galacturonic acid was used as standard (Sigma, USA). The reaction mixtures (enzymatic extract/substrate 1/10) were incubated at 12 or 28 °C, at the corresponding enzyme production temperature, for 30 min as previously described (Merín et al., 2011Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148.). One pectinase unit (U) is defined as the enzymatic activity that releases 1 μmol of reducing sugar per min under the assay conditions.
Other cold-active hydrolytic activities of enological interest
Cellulase and xylanase activities were assayed in enzymatic extracts obtained at 12 °C as described above by measuring the reducing sugars according to Miller (1959)Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428.. Cellulase was measured using microgranular cellulose (Whatman CC41) and xylanase using birchwood xylan (Sigma, USA) as substrates at a concentration of 0.25% (w/v) in 50 mM citric-citrate buffer (pH 3.5). The enzymatic reactions were carried out at 12 °C for 30 min. One cellulase or xylanase unit is defined as the enzymatic activity that releases 1 μmol of reducing sugar (as glucose or xylose, respectively) per min under the assay conditions.
β-Glucosidase activity was assayed by incubating 100 μL of enzymatic extract with 100 μL of 15 mM D-(+)-cellobiose (Fluka, USA) solution in citric-citrate buffer (pH 3.5) at 12 °C for 30 min. Glucose produced was quantified using the enzymatic colorimetric test (GOD-POD) (Arévalo-Villena et al., 2007Arévalo-Villena M, Úbeda Iranzo JF, Briones Pérez MI (2007) β-Glucosidase activity in wine yeasts: Application in enology. Enzyme Microb Technol 40:420–425.). One β-glucosidase unit is defined as the enzymatic activity that releases 2 μmol of glucose from cellobiose per min under the assay conditions.
Extracellular protease activity was assayed qualitatively by point-inoculation of yeasts on plates of skim milk agar and gelatin agar at pH 4.5, according to the method described by Charoenchai et al. (1997)Charoenchai C, Fleet GH, Henschke PA et al. (1997) Screening of non- Saccharomyces wine yeasts for the presence of extracellular hydrolytic enzymes. Aust J Grape Wine Res 3:2–8.. Skim milk agar plates were directly examined for clear zones surrounding yeast growth after incubation at 12 °C for 3–5 days, whereas gelatin agar plates were flooded with 10 mL acetic acid (50 g/L) before examination for clear zones around the yeast cells.
Appropriate enzyme and substrate blanks, as well as calibration curves, were included in all quantitative enzymatic assays.
T. delbrueckii BTd259 (Maturano et al., 2012Maturano YP, Rodríguez LA, Toro ME et al. (2012) Multi-enzyme production by pure and mixed cultures of Saccharomyces and non-Saccharomyces yeasts during wine fermentation. Int J Food Microbiol 155:43–50.) was used as positive control for pectinolytic and other hydrolytic activities.
Influence of enological parameters on pectinolytic activity
The effects of ethanol, sulfur dioxide (SO2) and a combination of ethanol and SO2 on pectinase activity were evaluated in cell-free supernatants (enzymatic extracts produced as described in previous section) under standard enzymatic assay conditions. The substrate was supplemented with ethanol and total SO2 at final concentrations of 15% (v/v) and 120 mg/L, respectively, and reactions were carried out at 12 and 28 °C, as appropriate. Reaction mixtures assayed under the same conditions at their respective temperatures but in absence of ethanol and SO2corresponded to the reference activity.
Statistical analysis
Analysis of variance (ANOVA) and Fisher’s LSD test (p < 0.05) were applied to all experimental data, using STATGRAPHICS Plus 5.1 (Manugistics, Rockville, MD, USA). Data normality and variance homogeneity in the residuals were verified by modified Shapiro-Wilks and Levene’s test, respectively.
Nucleotide sequence and yeast strain accession numbers
Partial sequence of the 26S rRNA gene of the most representative strains were submitted to the GenBank database available at NCBI under accession numbers: JF414133 (Saccharomyces cerevisiae), JF414134 (Candida zemplinina), JN637171 (Cryptococcus saitoi), JN637172 (Rhodotorula dairenensis).
The latter two strains were also deposited at the Banco Nacional de Microorganismos (BNM) Culture Collection (Buenos Aires, Argentina) under accession numbers: BNM 538 (Cryptococcus saitoi GM-4) and BNM 539 (Rhodotorula dairenensis GM-15).
Results and Discussion
Isolation and identification of representative yeasts from Argentine Bonarda grape and fermenting must
Alcoholic fermentation of Argentine Bonarda must was completed in 12 days. The red wine had a final pH of 4.1 and a final ethanol concentration of 11.7% (data not shown).
A total of 48 yeast colonies were isolated from grape surface, fresh must and spontaneous fermentation on WL medium. The 22 colonies isolated from grapes and must comprised 8 groups with different morphology. During the fermentation process, the diversity of the yeast colony morphology decreased and only 6 different types were observed. Consequently, the 48 colonies were classified into 14 morphological groups (Table 1), characterized by their specific color, consistency and surface, as previously described (Pallmann et al., 2001Pallmann C, Brown JA, Olineka TL et al. (2001) Use of WL medium to profile native flora fermentations. Am J Enol Vitic 52:198–203.; Urso et al., 2008Urso R, Rantsiou K, Dolci P et al. (2008) Yeast biodiversity and dynamics during sweet wine production as determined by molecular methods. FEMS Yeast Res 8:1053–1062.).
Identification at species level of morphological groups of yeasts isolated from grapes, fresh must and fermenting must, based on the migration of the bands obtained by PCR-DGGE
All the colonies of each morphological pattern were subjected to PCR-DGGE to establish species-specific migration groups. Profiles of each DGGE migration group are presented in Figure 1.
DGGE profiles of yeasts isolated from Argentine Bonarda grapes and must during spontaneous fermentation. Correlations between lane designations and colony groups as well as Roman numerals and DGGE patterns are indicated in Table 1. The bands common to all isolates are single stranded DNA artifacts that were not influenced differentially by the gradient (Cocolin et al., 2000Cocolin L, Bisson LF, Mills DA (2000) Direct profiling of the yeast dynamics in wine fermentations. FEMS Microbiol Lett 189:81–87.)
Seven different DGGE profiles were generated. Co-migrating DGGE bands were considered to belong to the same species. After sequencing, 7 yeast genera were identified, corresponding to A. pullulans, R. dairenensis, Cr. saitoi, C. zemplinina and S. cerevisiae species and Hanseniaspora/Kloeckera sp. and Metschnikowia sp. (Table 1). The identified yeasts are among the most frequently described on grape skins and fresh musts from diverse regions around the world (Merín et al., 2011Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148.; Barata et al., 2012Barata A, Malfeito-Ferreira M, Loureiro V (2012) The microbial ecology of wine grape berries. Int J Food Microbiol 153:243–259.; Rantsiou et al., 2013Rantsiou K, Campolongo S, Alessandria V et al.(2013) Yeast populations associated with grapes during withering and their fate during alcoholic fermentation of high-sugar must. Aust J Grape Wine Res 19:40–46.), and during wine fermentation (Fleet, 2008Fleet GH (2008) Wine yeasts for the future. FEMS Yeast Res 8:979–995.; Urso et al., 2008Urso R, Rantsiou K, Dolci P et al. (2008) Yeast biodiversity and dynamics during sweet wine production as determined by molecular methods. FEMS Yeast Res 8:1053–1062.; Rantsiou et al., 2013Rantsiou K, Campolongo S, Alessandria V et al.(2013) Yeast populations associated with grapes during withering and their fate during alcoholic fermentation of high-sugar must. Aust J Grape Wine Res 19:40–46.).
Occurrence of pectinolytic yeasts isolated during spontaneous fermentation of Argentine Bonarda grape
Out of 48 representative yeast isolates, 11 (23%) belonging to 3 species showed pectinolytic activity on agar plates (Figure 2). According to a recent review by Alimardani-Theuil et al. (2011)Alimardani-Theuil P, Gainvors-Claisse A, Duchiron F (2011) Yeasts: An attractive source of pectinases-From gene expression to potential applications: A review. Process Biochem 46:1525–1537., of approximately 700 yeast species identified to date, only very few produce pectinolytic enzymes, which is in accordance with our results.
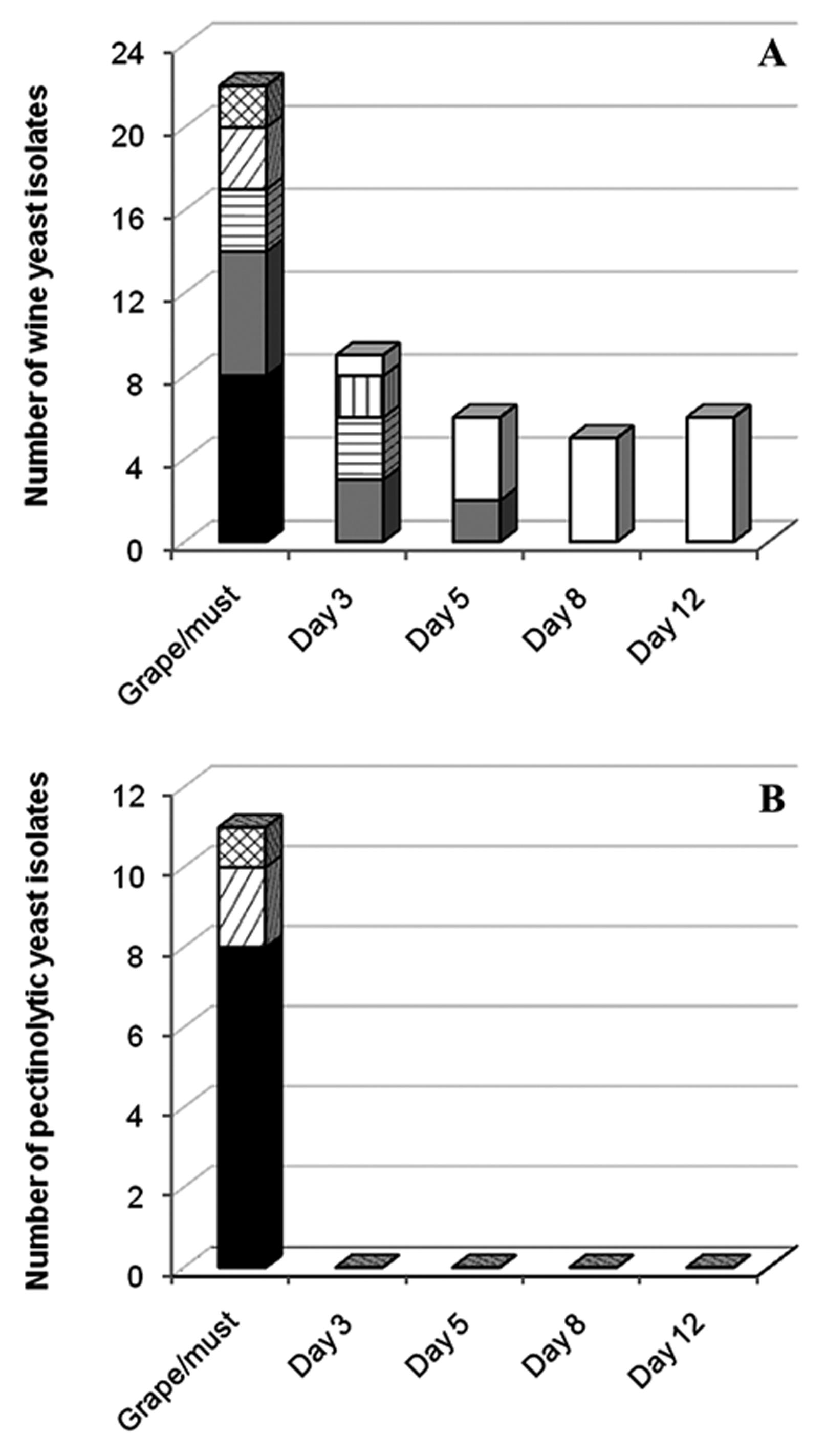
Occurrence of pectinolytic yeasts among representative yeasts isolated from grapes, fresh must and fermentation (sampling time: days 3 to 12) of cv. Argentine Bonarda. Number of isolates of yeast species (A) and of pectinolytic yeast species (B) found on grapes and during spontaneous fermentation. Aureobasidium pullulans, Hanseniaspora sp., Metschnikowia sp., Rhodotorula dairenensis, Cryptococcus saitoi, Candida zemplinina and Saccharomyces cerevisiae
The total number of representative yeasts isolated from grapes, fresh must and fermentations is shown in Figure 2A. Among the yeast isolated from grapes and must, A. pullulans was the most abundant species with 8 isolates, followed by Hanseniaspora sp. with 6, Metschnikowiasp. and R. dairenensis, both with 3, and Cr. saitoi with 2 isolates.
Figure 2B shows that pectinolytic yeasts were only isolated from grape berry surfaces and fresh must. A. pullulans was the predominant pectinolytic species, representing 73% of the total number of isolates able to degrade pectin. Besides, all A. pullulans isolates produced pectinases. Similar results have previously been observed in wine grapes, with A. pullulansas the most frequent cold-active-pectinase-producing species (Merín et al., 2011Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148., 2014Merín MG, Mendoza LM, Morata de Ambrosini VI (2014) Pectinolytic yeasts from viticultural and enological environments: novel finding of Filobasidium capsuligenum producing pectinases. J. Basic Microbiol 54:835–842.), and in tropical environments where this species represented the highest proportion (22%) of pectinolytic microorganisms (Buzzini and Martini, 2002Buzzini P, Martini A (2002) Extracellular enzymatic activity profiles in yeast and yeast-like strains isolated from tropical environments. J Appl Microbiol 93:1020–1025.). The results suggest that this pectinolytic microorganism is more prevalent in plant materials, soil and water.
The remaining pectinolytic species corresponded to the basidiomycetous yeasts R. dairenensis and Cr. saitoi, representing 18% and 9% of the total pectinolytic isolates, respectively (Figure 2B). Two thirds of the representative isolates of R. dairenensis and half of the Cr. saitoi isolates produced pectinases. Yeasts belonging to these two genera have previously been described as pectinolytic microorganisms (Federici, 1988Federici F (1988) Pectic enzyme production by Cryptococcus albidus var. albidus on olive vegetation waters enriched with sunflower calathide meal. Biol Wastes 25:291–301.; Nakagawa et al., 2004Nakagawa T, Nagaoka T, Taniguchi S et al. (2004) Isolation and characterization of psychrophilic yeasts producing cold-adapted pectinolytic enzymes. Lett Appl Microbiol 38:383–387.; Turchetti et al., 2008Turchetti B, Buzzini P, Goretti M et al. (2008) Psychrophilic yeasts in glacial environments of Alpine glaciers. FEMS Microbiol Ecol 63:73–83.), particularly the species R. mucilaginosa (Vaz et al., 2011Vaz AB, Rosa LH, Vieira ML et al. (2011) The diversity, extracellular enzymatic activities and photoprotective compounds of yeasts isolated in antarctica. Braz J Microbiol 42:937–947.; Sahay et al., 2013Sahay S, Hamid B, Singh P et al. (2013) Evaluation of pectinolytic activities for oenological uses from psychrotrophic yeasts. Lett Appl Microbiol 57:115–121.) and R. glutinis (Taskin, 2013Taskin M (2013) Co-production of tannase and pectinase by free and immobilized cells of the yeast Rhodotorula glutinis MP-10 isolated from tannin-rich persimmon (Diospyros kaki L.) fruits. Bioproc Biosyst Eng 36:165–172.), which are phylogenetically closely related to R. dairenensis. Nevertheless, to our knowledge, this is the first study reporting pectinase activity by R. dairenensis and Cr. saitoispecies.
Most yeasts present on wine grape at harvest time and during fermentation belong to ascomycetous species (Fleet, 2008Fleet GH (2008) Wine yeasts for the future. FEMS Yeast Res 8:979–995.; Urso et al., 2008Urso R, Rantsiou K, Dolci P et al. (2008) Yeast biodiversity and dynamics during sweet wine production as determined by molecular methods. FEMS Yeast Res 8:1053–1062.; Barata et al., 2012Barata A, Malfeito-Ferreira M, Loureiro V (2012) The microbial ecology of wine grape berries. Int J Food Microbiol 153:243–259.). Nevertheless, only a few ascomycetous species have been reported to produce pectinases in wine ecosystem such as Kluyveromyces, Candida, Metschnikowia and some S. cerevisiae strains (Fernández et al., 2000Fernández M, Úbeda JF, Briones AI (2000) Typing of non-Saccharomyces yeasts with enzymatic activities of interest in wine-making. Int J Food Microbiol 59:29–36.; Fernández-González et al., 2004Fernández-González M, Úbeda JF, Vasudevan TG et al.(2004) Evaluation of polygalacturonase activity in Saccharomyces cerevisiae wine strains. FEMS Microbiol Lett 237:261–266.). In this study, no pectinolytic activity was detected among ascomycetous yeasts, either non-Saccharomyces or S. cerevisiaeisolates (Figure 2). Comparable results have recently been obtained in studies of pectinolytic yeasts isolated from viticultural and enological environments (Merín et al., 2011Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148., 2014Merín MG, Mendoza LM, Morata de Ambrosini VI (2014) Pectinolytic yeasts from viticultural and enological environments: novel finding of Filobasidium capsuligenum producing pectinases. J. Basic Microbiol 54:835–842.), which are in agreement with findings reported by other authors. In an extensive screening survey, Buzzini and Martini (2002)Buzzini P, Martini A (2002) Extracellular enzymatic activity profiles in yeast and yeast-like strains isolated from tropical environments. J Appl Microbiol 93:1020–1025. demonstrated that pectinase activity was rarely found within ascomycetes in tropical environments, observing this ability in only 1.5% of studied ascomycetes. Charoenchai et al. (1997)Charoenchai C, Fleet GH, Henschke PA et al. (1997) Screening of non- Saccharomyces wine yeasts for the presence of extracellular hydrolytic enzymes. Aust J Grape Wine Res 3:2–8. did not detect pectinolytic activity in ascomycetous wine yeasts.
The fact that pectinolytic yeasts were only found on grape surfaces and must, whereas non-pectinolytic yeasts dominated the middle and last fermentation stages (Figure 2), suggests that a higher incidence of pectinolytic species may be observed in nutrient-poor environments like the grape surface and other plant surfaces, and in marine and glacial ecosystems (Nakagawa et al., 2004Nakagawa T, Nagaoka T, Taniguchi S et al. (2004) Isolation and characterization of psychrophilic yeasts producing cold-adapted pectinolytic enzymes. Lett Appl Microbiol 38:383–387.; Turchetti et al., 2008Turchetti B, Buzzini P, Goretti M et al. (2008) Psychrophilic yeasts in glacial environments of Alpine glaciers. FEMS Microbiol Ecol 63:73–83.). It seems that pectinolytic yeasts play an ecological role on the grape surface since they can utilize pectin from cell walls releasing intracellular sugars to surface. Conversely, microorganisms in musts do not require pectinolytic activity to acquire carbon sources because of the abundance of readily usable sugars. This is consistent with results reported by Barata et al. (2012)Barata A, Malfeito-Ferreira M, Loureiro V (2012) The microbial ecology of wine grape berries. Int J Food Microbiol 153:243–259., who observed that the microbiota of truly intact berries after véraison is dominated by basidiomycetous yeasts (e.g. Cryptococcus spp., Rhodotorula spp., Sporobolomyces spp.) and A. pullulans; while visually intact berries may bear microfissures and softens, increasing nutrient availability and explaining the possible dominance by the oxidative or weakly fermentative ascomycetous populations (e.g. Candida spp., Hanseniasporaspp., Metschnikowia spp.) approaching harvest time.
According to our results, the microfissures and softens on visually intact berries could be due to the action of pectinases produced by pectinolytic yeasts on grapes, like A. pullulans and basidiomycetous yeasts found in this study, which would release intracellular substrates from grape, thus sustaining the growth of non-pectinolytic ascomycetous yeasts on the grape berry surface.
Characterization of enzymatic activities
Hydrolytic activities under winemaking conditions
All yeasts showing extracellular pectinase activity on plate were also assayed in liquid medium at 28 °C and pH 3.5 to select the best pectinolytic yeasts under winemaking conditions (data not shown). Of the 11 pectinolytic yeasts, two A. pullulans strains (GM-1 and GM-2) and two basidiomycetous yeasts (R. dairenensis GM-15 and Cr. saitoi GM-4) produced the highest activities at grape pH (0.967, 1.325, 1.260, 0.930 U/mL, respectively). These activities are higher than those produced by other yeasts, such as Saccharomyces strains: 0.107–0.679 U/mL (Oliveira et al., 2006Oliveira KF, Malavolta L, Souza CS et al. (2006) Pectinolytic activity secreted by yeasts isolated from fermented citrus molasses. J Appl Microbiol 100:633–640.) and Zygoascus hellenicus strains: 0.025–0.220 U/mL (Ahansal et al., 2008Ahansal L, Ben Sassi A, Martini A et al. (2008) Biodiversity of yeasts isolated from the indigenous forest of Argan (Argania spinosa (L.) Skeels) in Morocco. World J Microbiol Biotechnol 24:777–782.). However, the pectinolytic activities cited were displayed at pH 5.0.
The studied strains exhibited similar pectinolytic activity to that previously reported for A. pullulans at pH 3.5 and 12 °C, which has already been characterized (Merín et al., 2011Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148.). Consequently, considering that this is the first study reporting pectinase production by R. dairenensis and Cr. saitoi, both strains (GM-15 and GM-4) were selected to characterize their enzymatic activity under wine-like conditions.
Because of the interest in cold-active enzymes, pectinolytic and other hydrolytic activities useful in vinification were further assayed in yeast enzymatic extracts produced at pH 3.5 and 12 °C. Pectinase activity in both strains was significantly higher than activity showed by T. delbrueckii BTd259 control strain, particularly in R. dairenensis GM-15 (1.104 U/mL) (Table 2), and also higher than pectinolytic activities produced by other yeasts, like Saccharomyces and Zygoascus hellenicus strains (0.100–0.679 U/mL), even at 30 or 50 °C (Oliveira et al., 2006Oliveira KF, Malavolta L, Souza CS et al. (2006) Pectinolytic activity secreted by yeasts isolated from fermented citrus molasses. J Appl Microbiol 100:633–640.; Ahansal et al., 2008Ahansal L, Ben Sassi A, Martini A et al. (2008) Biodiversity of yeasts isolated from the indigenous forest of Argan (Argania spinosa (L.) Skeels) in Morocco. World J Microbiol Biotechnol 24:777–782.).
Hydrolytic activities of enological interest of selected pectinolytic strains assessed at low temperature (12 °C) and grape pH (3.5)
Cellulase and hemicellulase enzymes degrade cellulose and hemicelluloses, respectively, present in grape cell walls. Consequently, they are responsible for an increase in the extraction of juice and color, and for improvement in the clarification of wines (Romero-Cascales et al., 2008Romero-Cascales I, Fernández-Fernández JI, Ros-García JM et al. (2008) Characterisation of the main enzymatic activities present in six commercial macerating enzymes and their effects on extracting colour during winemaking of Monastrell grapes. Int J Food Sci Technol 43:1295–1305.). Of the secondary enzymes assayed, only cellulase activity was detected in GM-15 enzymatic extract with a considerable activity (0.549 U/mL) at 12 °C (Table 2).
β-Glucosidase enzymes hydrolyze glycosilated complexes releasing volatile compounds that contribute to wine aroma (Rodríguez et al., 2007Rodríguez ME, Lopes C, Vlales S et al. (2007) Selection and preliminary characterization of β-glycosidases producer Patagonian wild yeasts. Enzyme Microb Technol 41:812–820.). However, commercial pectinases containing β-glucosidases may cause a loss of color in red wines because some of these enzymes are able to degrade anthocyanins, glycosilated polyphenols that are mainly responsible for the red color of wine (Romero-Cascales et al., 2008Romero-Cascales I, Fernández-Fernández JI, Ros-García JM et al. (2008) Characterisation of the main enzymatic activities present in six commercial macerating enzymes and their effects on extracting colour during winemaking of Monastrell grapes. Int J Food Sci Technol 43:1295–1305.). The fact that β-glucosidase was not detected in the enzymatic systems analyzed (Table 2) is a positive feature when the yeast enzymes are applied to red winemaking.
Protease activity was also assayed on plate at 12 °C. Neither of the strains secreted proteases into the medium, unlike the T. delbrueckii BTd259 control strain (Table 2).
Effect of ethanol and sulfur dioxide on pectinolytic activity
Wine is a complex system that presents a combination of factors such as pH, temperature, ethanol and SO2, among others. Since the sum of the responses of each of the single parameters does not necessarily predict the combined response of such parameters (Grimaldi et al., 2005Grimaldi A, Bartowsky E, Jiranek V (2005) A survey of glycosidase activities of commercial wine strains of Oenococcus oeni. Int J Food Microbiol 105:233–244.), the combined effect of ethanol and sulfur dioxide on enzymatic activity should be studied.
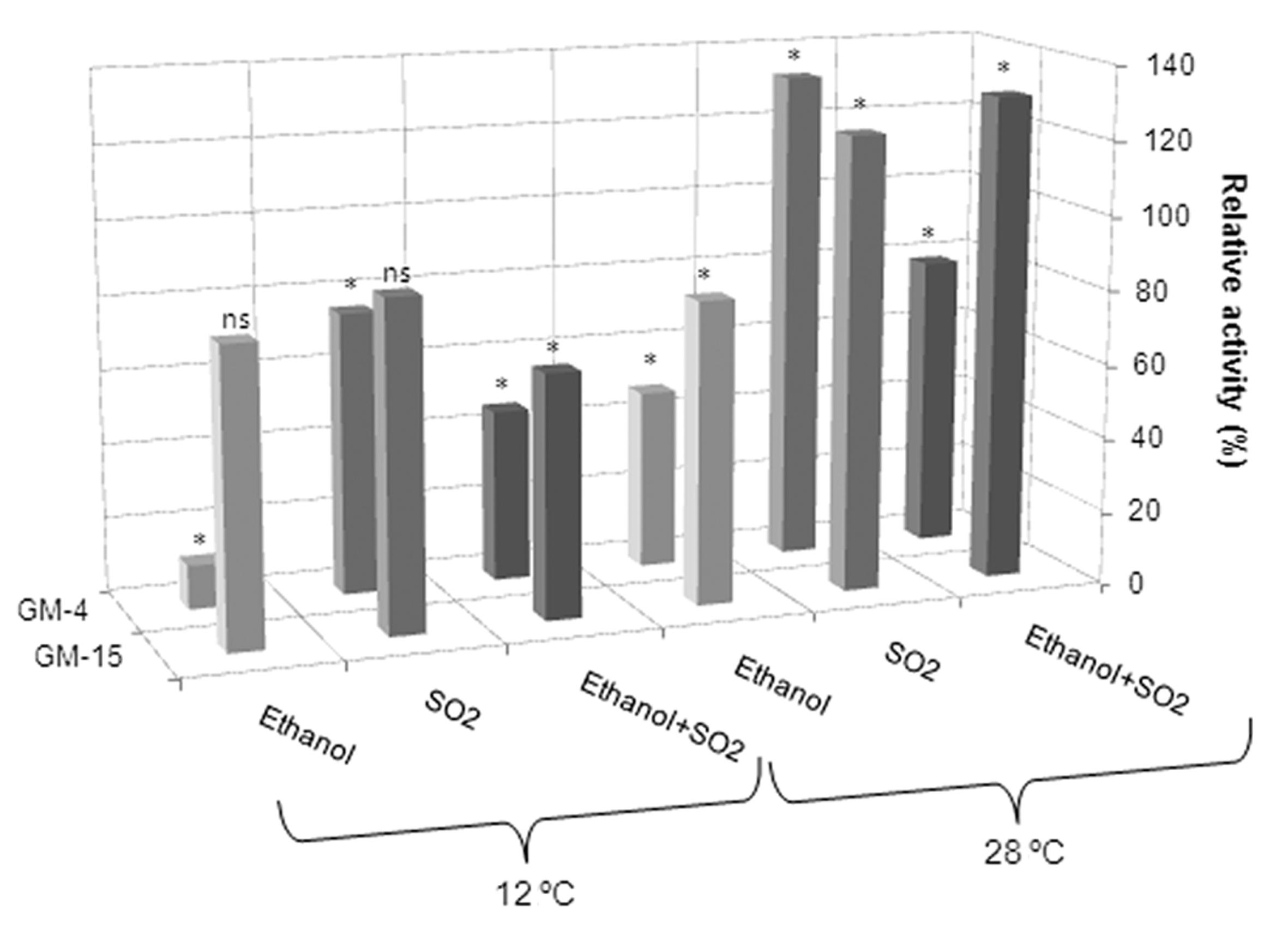
To acquire high quality products, the enology sector uses different technologies like low temperature vinification (Gómez-Míguez et al., 2007Gómez-Míguez M, González-Miret ML, Heredia FJ (2007) Evolution of colour and anthocyanin composition of Syrah wines elaborated with pre-fermentative cold maceration. J Food Eng 79:271–278.; Fleet, 2008Fleet GH (2008) Wine yeasts for the future. FEMS Yeast Res 8:979–995.) and due to the potential value of cold-active enzymes, research on these enzymes is increasing conspicuously in these years. Therefore, the individual and combined effect of 15% (v/v) ethanol and 120 mg/L SO2 on pectinolytic activity was assessed at 12 and 28 °C (Figure 3). At 12 °C, 15% (v/v) ethanol and 120 mg/L SO2 as individual parameters hardly affected pectinolytic activity (retaining around 80–90% of relative activity), except for GM-4 pectinase, which was greatly inhibited by ethanol (conserving only 12% of relative activity). The combination of ethanol and SO2 reduced pectinolytic activity to 50 and 70%, for GM-4 and GM-15, respectively.
Relative pectinolytic activity of Cr. saitoiGM-4 and R. dairenensis GM-15 assayed at 12 and 28 °C in presence of 15% (v/v) ethanol or 120 mg/L SO2 or a combination of both compounds. Reference activity (100% of relative activity): 0.736 ± 0.071 U/mL at 12 °C and 1.349 ± 0.133 U/mL at 28 °C for Cr. saitoi GM-4, and 0.972 ± 0.083 U/mL at 12 °C and 1.483 ± 0.091 U/mL at 28 °C for R. dairenensis GM-15. (*): significant difference, (ns): not significant, according to the LSD test (n = 3, p < 0.05). In all cases SD was lower than 10%
At 28 °C, the ethanol concentration assayed reduced the pectinolytic activity of GM-4 by 50% and of GM-15 only by 20%. Nevertheless, emphasis must be laid in the fact that at the SO2 concentration assayed (120 mg/L, the highest concentration generally observed in must at the beginning of the fermentation), activity increased by 33% (GM-4) and 22% (GM-15), compared with the reference activity. The combination of ethanol and SO2assayed at 28 °C slightly reduced GM-4 activity by 20%, while it enhanced GM-15 activity by 30% (Figure 3).
With respect to GM-4 pectinase activity, the combined effect of the two parameters produced a relative activity intermediate between the response to ethanol and SO2 as single parameters at both temperatures. On the other hand, GM-15 pectinase behaved differently at the two temperatures assayed. Temperature seemed to affect the relative activity in presence of ethanol and SO2, since at 12 °C the activity was negatively affected, but unexpectedly, at 28 °C the enzymatic activity was slightly increased. At the higher temperature, SO2 probably counteracts the negative effect of ethanol on the relative activity. This could be explained by the unique ability of SO2 to act as an oxidizing or a reducing agent that enables it either to inactivate enzyme systems by splitting their disulfide linkages (Cecil and Wake, 1962Cecil R, Wake RG (1962) The reactions of inter- and intra-chain disulphide bonds in protein with sulphite. Biochem J 82:401.) or activate certain hydrolytic enzyme systems, probable by bringing about conformational changes (Malhotra and Hocking, 1976Malhotra SS, Hocking D (1976) Biochemical and cytological effects on sulphur dioxide on plant metabolism. New Phytol 76:227–237.).
Different responses of related hydrolytic activities to these compounds have been reported. Ethanol concentrations of 12% (v/v) have been found to decrease polygalacturonase activity from S. cerevisiae(Fernández-González et al., 2004Fernández-González M, Úbeda JF, Vasudevan TG et al.(2004) Evaluation of polygalacturonase activity in Saccharomyces cerevisiae wine strains. FEMS Microbiol Lett 237:261–266.), protease activity from Ananas comosus (Esti et al., 2011Esti M, Benucci I, Liburdi K et al. (2011) Effect of wine inhibitors on free pineapple stem bromelain activity in a model wine system. J Agric Food Chem 59:3391–3397.) and α-L-rhamnosidase activity from Aspergillus terreus (Gallego et al., 2001Gallego MV, Piñaga F, Ramón D et al. (2001) Purification and characterization of an α-L-rhamnosidase from Aspergillus terreus of interest in winemaking. Food Chem Toxicol 66:204–209.) to around 20–50%. Likewise, 15% (v/v) ethanol decreased the activity of β-glucosidases and β-xylosidases from wine yeasts to around 35% and 55%, respectively (Rodríguez et al., 2007Rodríguez ME, Lopes C, Vlales S et al. (2007) Selection and preliminary characterization of β-glycosidases producer Patagonian wild yeasts. Enzyme Microb Technol 41:812–820.). Conversely, ethanol concentrations of 15 to 20% (v/v) produced significant increases (150–500% relative activity) in β-glucosidase (Barbagallo et al., 2004Barbagallo RN, Spagna G, Palmeri R et al. (2004) Selection, characterization and comparison of β-glucosidase from mould and yeasts employable for enological application. Enzyme Microb Technol 35:58–66.) and β-xylosidase (Rodríguez et al., 2007Rodríguez ME, Lopes C, Vlales S et al. (2007) Selection and preliminary characterization of β-glycosidases producer Patagonian wild yeasts. Enzyme Microb Technol 41:812–820.) activities from wine yeasts.
SO2 has been reported to strongly inhibit protease activity from Ananas comosus (Esti et al., 2011Esti M, Benucci I, Liburdi K et al. (2011) Effect of wine inhibitors on free pineapple stem bromelain activity in a model wine system. J Agric Food Chem 59:3391–3397.). Nevertheless, polygalacturonase (Fernández-González et al., 2004Fernández-González M, Úbeda JF, Vasudevan TG et al.(2004) Evaluation of polygalacturonase activity in Saccharomyces cerevisiae wine strains. FEMS Microbiol Lett 237:261–266.) and α-L-rhamnosidase (Gallego et al., 2001Gallego MV, Piñaga F, Ramón D et al. (2001) Purification and characterization of an α-L-rhamnosidase from Aspergillus terreus of interest in winemaking. Food Chem Toxicol 66:204–209.) activities were not affected by 50 mg/L SO2. Rodríguez et al. (2007)Rodríguez ME, Lopes C, Vlales S et al. (2007) Selection and preliminary characterization of β-glycosidases producer Patagonian wild yeasts. Enzyme Microb Technol 41:812–820. observed that β-glucosidase activity from wine yeasts was not affected by 150 mg/L SO2, which agrees with our results that showed a slight increase in pectinolytic activity in presence of 120 mg/L SO2compared with the reference activity. However, it is important to mention that the authors cited did not determine the effect of a combination of ethanol and SO2 on the enzyme activities assayed.
These outcomes suggest that application of pectinase of R. dairenensis GM-15 in the production of red wine is preferably carried out at traditional temperatures (26–28 °C); although in vinifications at low temperature (12 °C) it would still retain high residual activity.
In conclusion, the current study has demonstrated that inoculation of pectinolytic yeasts or addition of pectinases in the vinification process is necessary since they do not naturally occur during wine fermentation. Our results also suggest that pectinolytic yeasts should be isolated from the grape surface. To our knowledge, this is the first report on pectinase production by Cr. saitoi and R. dairenensis species. The outcomes of the combined effect of ethanol and SO2 at two possible fermentation temperatures on the pectinolytic activity indicated that the studied enzymes, particularly GM-15 pectinase, performed satisfactorily under wine-like conditions: at low pH (3.5), at low (12 °C) and traditional (28 °C) temperature in red winemaking, and in the presence of potential enzymatic inhibitors like ethanol (15% [v/v]) and SO2 (120 mg/L). R. dairenensis GM-15 also produced cellulase activity at low temperature and at grape pH, and did not produce β-glucosidase activity avoiding risks of color loss when it is used in red wine-making. Further studies regarding effects of these enzymes on wine processing and quality are needed in order to propose them in enology.
Acknowledgments
This research was supported by grants from CONICET (PIP N∘ 11220110100823 project); SECTyP-UNCuyo (N∘ 06/L116 project) and I+D (UNCuyo) program, and ANPCyT-MINCYT (PICT 2010 N∘ 0847 project). The authors wish to thank Raúl Carrión for supplying grape samples, and Dr. Lucía Mendoza and Raúl Raya (PhD) for their assistance in yeast identification. This work is dedicated to the memory of Dr. Marta Farías.
References
- Ahansal L, Ben Sassi A, Martini A et al. (2008) Biodiversity of yeasts isolated from the indigenous forest of Argan (Argania spinosa (L.) Skeels) in Morocco. World J Microbiol Biotechnol 24:777–782.
- Alimardani-Theuil P, Gainvors-Claisse A, Duchiron F (2011) Yeasts: An attractive source of pectinases-From gene expression to potential applications: A review. Process Biochem 46:1525–1537.
- Arévalo-Villena M, Úbeda Iranzo JF, Briones Pérez MI (2007) β-Glucosidase activity in wine yeasts: Application in enology. Enzyme Microb Technol 40:420–425.
- Barata A, Malfeito-Ferreira M, Loureiro V (2012) The microbial ecology of wine grape berries. Int J Food Microbiol 153:243–259.
- Barbagallo RN, Spagna G, Palmeri R et al. (2004) Selection, characterization and comparison of β-glucosidase from mould and yeasts employable for enological application. Enzyme Microb Technol 35:58–66.
- Buzzini P, Martini A (2002) Extracellular enzymatic activity profiles in yeast and yeast-like strains isolated from tropical environments. J Appl Microbiol 93:1020–1025.
- Cecil R, Wake RG (1962) The reactions of inter- and intra-chain disulphide bonds in protein with sulphite. Biochem J 82:401.
- Charoenchai C, Fleet GH, Henschke PA et al. (1997) Screening of non- Saccharomyces wine yeasts for the presence of extracellular hydrolytic enzymes. Aust J Grape Wine Res 3:2–8.
- Cocolin L, Bisson LF, Mills DA (2000) Direct profiling of the yeast dynamics in wine fermentations. FEMS Microbiol Lett 189:81–87.
- Esti M, Benucci I, Liburdi K et al. (2011) Effect of wine inhibitors on free pineapple stem bromelain activity in a model wine system. J Agric Food Chem 59:3391–3397.
- Federici F (1988) Pectic enzyme production by Cryptococcus albidus var. albidus on olive vegetation waters enriched with sunflower calathide meal. Biol Wastes 25:291–301.
- Fernández M, Úbeda JF, Briones AI (2000) Typing of non-Saccharomyces yeasts with enzymatic activities of interest in wine-making. Int J Food Microbiol 59:29–36.
- Fernández-González M, Úbeda JF, Vasudevan TG et al.(2004) Evaluation of polygalacturonase activity in Saccharomyces cerevisiae wine strains. FEMS Microbiol Lett 237:261–266.
- Fleet GH (2008) Wine yeasts for the future. FEMS Yeast Res 8:979–995.
- Gallego MV, Piñaga F, Ramón D et al. (2001) Purification and characterization of an α-L-rhamnosidase from Aspergillus terreus of interest in winemaking. Food Chem Toxicol 66:204–209.
- Gómez-Míguez M, González-Miret ML, Heredia FJ (2007) Evolution of colour and anthocyanin composition of Syrah wines elaborated with pre-fermentative cold maceration. J Food Eng 79:271–278.
- Grimaldi A, Bartowsky E, Jiranek V (2005) A survey of glycosidase activities of commercial wine strains of Oenococcus oeni Int J Food Microbiol 105:233–244.
- Kurtzman CP, Robnett CJ (1998) Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie Van Leeuwenhoek 73:331–371.
- Malhotra SS, Hocking D (1976) Biochemical and cytological effects on sulphur dioxide on plant metabolism. New Phytol 76:227–237.
- Martín MC, Morata de Ambrosini VI (2014) Effect of a cold-active pectinolytic system on colour development of Malbec red wines elaborated at low temperature. Int J Food Sci Technol 49:1893–1901.
- Maturano YP, Rodríguez LA, Toro ME et al. (2012) Multi-enzyme production by pure and mixed cultures of Saccharomyces and non-Saccharomyces yeasts during wine fermentation. Int J Food Microbiol 155:43–50.
- Merín MG, Mendoza LM, Farías ME et al. (2011) Isolation and selection of yeasts from wine grape ecosystem secreting cold-active pectinolytic activity. Int J Food Microbiol 147:144–148.
- Merín MG, Mendoza LM, Morata de Ambrosini VI (2014) Pectinolytic yeasts from viticultural and enological environments: novel finding of Filobasidium capsuligenum producing pectinases. J. Basic Microbiol 54:835–842.
- Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428.
- Nakagawa T, Nagaoka T, Taniguchi S et al. (2004) Isolation and characterization of psychrophilic yeasts producing cold-adapted pectinolytic enzymes. Lett Appl Microbiol 38:383–387.
- Oliveira KF, Malavolta L, Souza CS et al. (2006) Pectinolytic activity secreted by yeasts isolated from fermented citrus molasses. J Appl Microbiol 100:633–640.
- Pallmann C, Brown JA, Olineka TL et al. (2001) Use of WL medium to profile native flora fermentations. Am J Enol Vitic 52:198–203.
- Rantsiou K, Campolongo S, Alessandria V et al.(2013) Yeast populations associated with grapes during withering and their fate during alcoholic fermentation of high-sugar must. Aust J Grape Wine Res 19:40–46.
- Rodríguez ME, Lopes C, Vlales S et al. (2007) Selection and preliminary characterization of β-glycosidases producer Patagonian wild yeasts. Enzyme Microb Technol 41:812–820.
- Romero-Cascales I, Fernández-Fernández JI, Ros-García JM et al. (2008) Characterisation of the main enzymatic activities present in six commercial macerating enzymes and their effects on extracting colour during winemaking of Monastrell grapes. Int J Food Sci Technol 43:1295–1305.
- Sahay S, Hamid B, Singh P et al. (2013) Evaluation of pectinolytic activities for oenological uses from psychrotrophic yeasts. Lett Appl Microbiol 57:115–121.
- Taskin M (2013) Co-production of tannase and pectinase by free and immobilized cells of the yeast Rhodotorula glutinis MP-10 isolated from tannin-rich persimmon (Diospyros kaki L.) fruits. Bioproc Biosyst Eng 36:165–172.
- Turchetti B, Buzzini P, Goretti M et al. (2008) Psychrophilic yeasts in glacial environments of Alpine glaciers. FEMS Microbiol Ecol 63:73–83.
- Urso R, Rantsiou K, Dolci P et al. (2008) Yeast biodiversity and dynamics during sweet wine production as determined by molecular methods. FEMS Yeast Res 8:1053–1062.
- Vaz AB, Rosa LH, Vieira ML et al. (2011) The diversity, extracellular enzymatic activities and photoprotective compounds of yeasts isolated in antarctica. Braz J Microbiol 42:937–947.
Publication Dates
-
Publication in this collection
Jul-Sep 2015
History
-
Received
19 Feb 2014 -
Accepted
02 Feb 2015