Abstract
Mamastrovirus 5 (MAstV5), belonging to the Astroviridae (AstV) family, previously known as canine astrovirus or astrovirus-like particles, has been reported in several countries to be associated with viral enteric disease in dogs since the 1980s. Astroviruses have been detected in fecal samples from a wide variety of mammals and birds that are associated with gastroenteritis and extra enteric manifestations. In the present study, RT-PCR was used to investigate the presence of MAstV5 in 269 dog fecal samples. MAstV5 was detected in 26% (71/269) of the samples. Interestingly, all MAstV5-positive samples derived from dogs displaying clinical signs suggestive of gastroenteritis, other enteric viruses were simultaneously detected (canine parvovirus, canine distemper virus, canine coronavirus, canine adenovirus and canine rotavirus). Based on genomic sequence analysis of MAstV5 a novel classification of the species into four genotypes, MAstV5a-MAstV5d, is proposed. Phylogenetic analyses based on the ORF2 amino acid sequences, samples described herein grouped into the putative genotype ‘a' closed related with Chinese samples. Other studies are required to attempt the clinical and antigenic implications of these astrovirus genotypes in dogs.
Keywords:
Mamastrovirus 5; Canine astrovirus; Dog; Gastroenteritis; MAstV5
Introduction
Viruses belonging to the Astroviridae (AstV) family are spherical, non-enveloped, 28-30 nm in size, with a surface that forms a characteristic star-like structure.11 Madeley CR, Cosgrove BP. Letter: 28 nm particles in faeces in infantile gastroenteritis. Lancet. 1975;2(7932):451-452. The RNA genome of AstV ranges from 6.8 to 7.9-kb in size, polyadenylated at the 3′ end, and contains three ORFs designated as ORF1a, ORF1b and ORF2. ORF1a encodes a protease, ORF1b encodes an RNA-dependent RNA-polymerase,22 De Benedictis P, Schultz-Cherry S, Burnham A, Cattoli G. Astrovirus infections in humans and animals - molecular biology, genetic diversity, and interspecies transmissions. Infect Genet Evol. 2011;11(7):1529-1544.,33 Jiang B, Monroe SS, Koonin EV, Stine SE, Glass RI. RNA sequence of astrovirus: distinctive genomic organization and a putative retrovirus-like ribosomal frameshifting signal that directs the viral replicase synthesis. Proc Natl Acad Sci U S A. 1993;90(22):10539-10543. while ORF2 encodes the viral capsid structural polyprotein that is required for virion assembly.44 Monroe SS, Jiang B, Stine SE, Koopmans M, Glass RI. Subgenomic RNA sequence of human astrovirus supports classification of Astroviridae as a new family of RNA viruses. J Virol. 1993;67(6):3611-3614.
The viral classification was previously based on the host and consisted of two genera, Avastrovirus and Mamastrovirus. However, recent characterization of novel astroviruses has taken in consideration that isolates from different animal species can be genetically similar, while genetically diverse viruses can be isolated from the same animal species.22 De Benedictis P, Schultz-Cherry S, Burnham A, Cattoli G. Astrovirus infections in humans and animals - molecular biology, genetic diversity, and interspecies transmissions. Infect Genet Evol. 2011;11(7):1529-1544. Based on this analysis, the International Committee on Taxonomy of Viruses renamed canine astrovirus as Mamastrovirus 5 (MAstV5).55 Bosch A, Guix S, Krishna NK, et al. Family Astroviridae. In: King AMQ, Lefkowitz E, Adams MJ, Carstens EB, eds. Virus Taxonomy: Classification and Nomenclature of Viruses (Ninth Report of the International Committee on the Taxonomy of Viruses). 9th ed. 2011. New York.
Astroviruses have been detected in fecal samples from a wide variety of mammals and birds that are associated with gastroenteritis.22 De Benedictis P, Schultz-Cherry S, Burnham A, Cattoli G. Astrovirus infections in humans and animals - molecular biology, genetic diversity, and interspecies transmissions. Infect Genet Evol. 2011;11(7):1529-1544. In children, AstVs are the second most common cause of gastroenteritis after rotaviruses.22 De Benedictis P, Schultz-Cherry S, Burnham A, Cattoli G. Astrovirus infections in humans and animals - molecular biology, genetic diversity, and interspecies transmissions. Infect Genet Evol. 2011;11(7):1529-1544.,66 Finkbeiner SR, Li Y, Ruone S, et al. Identification of a novel astrovirus (astrovirus VA1) associated with an outbreak of acute gastroenteritis. J Virol. 2009;83(20):10836-10839. Human AstVs can also cause significant disease in the elderly77 Lewis DC, Lightfoot NF, Cubitt WD, Wilson SA. Outbreaks of astrovirus type 1 and rotavirus gastroenteritis in a geriatric in-patient population. J Hosp Infect. 1989;14(1):9-14. and immune-compromised patients.88 Wunderli W, Meerbach A, Güngör T, et al. Astrovirus infection in hospitalized infants with severe combined immunodeficiency after allogeneic hematopoietic stem cell transplantation. PLoS ONE. 2011;6(11):e27483.,99 Gallimore CI, Taylor C, Gennery AR, et al. Use of a heminested reverse transcriptase PCR assay for detection of astrovirus in environmental swabs from an outbreak of gastroenteritis in a pediatric primary immunodeficiency unit. J Clin Microbiol. 2005;43(8):3890-3894. In addition to enteric manifestations, AstVs have been associated with fatal hepatitis in ducks,1010 Fu Y, Pan M, Wang X, et al. Complete sequence of a duck astrovirus associated with fatal hepatitis in ducklings. J Gen Virol. 2009;90(5):1104-1108. interstitial nephritis in young chickens,1111 Imada T, Yamaguchi S, Mase M, Tsukamoto K, Kubo M, Morooka A. Avian nephritis virus (ANV) as a new member of the family Astroviridae and construction of infectious ANV cDNA. J Virol. 2000;74(18):8487-8493. stunting and pre hatching mortality in duck and goose embryos,1212 Biđin M, Lojkić I, Tišljar M, Biđin Z, Majnarić D. Astroviruses associated with stunting and pre-hatching mortality in duck and goose embryos. Avian Pathol. 2012;41(1):91-97. as well as shaking mink syndrome1313 Blomström AL, Widén F, Hammer AS, Belák S, Berg M. Detection of a novel astrovirus in brain tissue of mink suffering from shaking mink syndrome by use of viral metagenomics. J Clin Microbiol. 2010;48(12):4392-4396.. Recently, an AstV was also hypothesized to be the causative agent of nonsuppurative encephalitis in cattle.1414 Bouzalas IG, Wuthrich D, Walland J, et al. Neurotropic astrovirus in cattle with nonsuppurative encephalitis in Europe. J Clin Microbiol. 2014;52(9):3318-3324.
Since the 1980s, astrovirus-like particles have been reported in dogs with and without diarrhea.1515 Williams FP. Astrovirus-like, coronavirus-like, and parvovirus-like particles detected in the diarrheal stools of beagle pups. Arch Virol. 1980;66(3):215-226.
16 Vieler E, Herbst W. Electron microscopic demonstration of viruses in feces of dogs with diarrhea. Tierarztl Prax. 1995;23(1):66-69.-1717 Marshall JA, Healey DS, Studdert MJ, et al. Viruses and virus-like particles in the faeces of dogs with and without diarrhoea. Aust Vet J. 1984;61(2):33-38. To date, canine astroviruses or astrovirus-like particles infecting dogs have been reported in several countries.1515 Williams FP. Astrovirus-like, coronavirus-like, and parvovirus-like particles detected in the diarrheal stools of beagle pups. Arch Virol. 1980;66(3):215-226.
16 Vieler E, Herbst W. Electron microscopic demonstration of viruses in feces of dogs with diarrhea. Tierarztl Prax. 1995;23(1):66-69.
17 Marshall JA, Healey DS, Studdert MJ, et al. Viruses and virus-like particles in the faeces of dogs with and without diarrhoea. Aust Vet J. 1984;61(2):33-38.
18 Toffan A, Jonassen CM, De Battisti C, et al. Genetic characterization of a new astrovirus detected in dogs suffering from diarrhoea. Vet Microbiol. 2009;139(1-2):147-152.
19 Zhu a L, Zhao W, Yin H, et al. Isolation and characterization of canine astrovirus in China. Arch Virol. 2011;156(9):1671-1675.
20 Castro TX, Cubel Garcia RCN, Costa EM, Leal RM, Xavier MDPT, Leite JPG. Molecular characterisation of calicivirus and astrovirus in puppies with enteritis. Vet Rec. 2013;172(21):557.
21 Grellet A, De Battisti C, Feugier A, et al. Prevalence and risk factors of astrovirus infection in puppies from French breeding kennels. Vet Microbiol. 2012;157(1-2):214-219.
22 Caddy SL, Goodfellow I. Complete genome sequence of canine astrovirus with molecular and epidemiological characterization of UK strains. Vet Microbiol. 2015;177(1-2):206-213.
23 Takano T, Takashina M, Doki T, Hohdatsu T. Detection of canine astrovirus in dogs with diarrhea in Japan. Arch Virol. 2015;160(6):1549-1553.
24 Choi S, Lim S-I, Kim YK, Cho Y-Y, Song J-Y, An D-J. Phylogenetic analysis of astrovirus and kobuvirus in Korean dogs. J Vet Med Sci. 2014;76(8):1141-1145.-2525 Mihalov-Kovács E, Martella V, Lanave G, et al. Genome analysis of canine astroviruses reveals genetic heterogeneity and suggests possible inter-species transmission. Virus Res. 2016, http://dx.doi.org/10.1016/j.virusres.2016.12.005.
http://dx.doi.org/10.1016/j.virusres.201...
Despite the detection of MAstV5 in association with gastroenteritis in dogs, which suggests a possible role for MAstV5 as a canine enteric pathogen, the association of MAstV5 with clinical disease remains obscure in such reports. Here, we investigated the presence of MAstV5 using RT-PCR in fecal samples from dogs of different ages with and without diarrhea. The partial genomes of selected MAstV5 RNA-positive samples were also sequenced to perform a phylogenetic analysis comparing them with the MAstV5 sequences described in the literature as the cause of enteric disease.1818 Toffan A, Jonassen CM, De Battisti C, et al. Genetic characterization of a new astrovirus detected in dogs suffering from diarrhoea. Vet Microbiol. 2009;139(1-2):147-152.,2020 Castro TX, Cubel Garcia RCN, Costa EM, Leal RM, Xavier MDPT, Leite JPG. Molecular characterisation of calicivirus and astrovirus in puppies with enteritis. Vet Rec. 2013;172(21):557.,2323 Takano T, Takashina M, Doki T, Hohdatsu T. Detection of canine astrovirus in dogs with diarrhea in Japan. Arch Virol. 2015;160(6):1549-1553.,2626 Martella V, Moschidou P, Catella C, et al. Enteric disease in dogs naturally infected by a novel canine astrovirus. J Clin Microbiol. 2012;50(3):1066-1069. Additionally, MAstV5 was proposed to be classified in four putative genotypes.
Materials and methods
Samples and nucleic acid extraction
A total of 269 dog fecal samples were collected between 2008 and 2014 in veterinary clinics and hospitals by convenience. These samples were obtained from eight Federal States of Brazil (Acre, Mato Grosso do Sul, Paraná, Rio Grande do Sul, Rio de Janeiro, Rondônia, Santa Catarina and São Paulo). The animal's age was recorded and ranked from puppy (equal or less than one-year-old) to adult dog (more than one-year-old); some samples from dogs of unknown age were included. Animals not presenting diarrhea at the time of sampling were considered asymptomatic and those presenting clinical signs of enteric disease diarrhea were classified as symptomatic. Samples were diluted to 20% (w/v) in phosphate buffered saline (PBS, pH 7.4) and stored at -80 °C for further analysis. Subsequently, viral DNA isolation from the supernatant was performed using a commercial kit (NewGene Preamp®, Simbios Biotecnologia, Brazil) based on guanidine isothiocyanate and silica.2727 Boom R, Sol CJ, Salimans MM, Jansen CL, Wertheim-van Dillen PM, van der Noordaa J. Rapid and simple method for purification of nucleic acids. J Clin Microbiol. 1990;28(3):495-503. Viral RNA was isolated using TRIzol® LS Reagent (Life Technologies™, USA) according to the manufacturer's instructions.
Oligonucleotides for MAstV5 detection and sequencing
An initial screening using RT-PCR to detect a larger number of Mamastrovirus species was achieved by amplifying 422 bp of the ORF1b fragment using oligonucleotides, as previously described.2828 Chu DKW, Poon LLM, Guan Y, Peiris JSM. Novel astroviruses in insectivorous bats. J Virol. 2008;82(18):9107-9114. For the specific detection of MAstV5, 92 nucleotide sequences of this species were retrieved from GenBank database (http://www.ncbi.nlm.nih.gov/nucleotide), and aligned using CLUSTAL W within Molecular Evolutionary Genetics Analysis version 6 (MEGA6).2929 Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. 2013;30:2725-2729. The MAstV5 specific RT-PCR was designed with a primer pair targeting the region of ORF2 that amplified a 250 bp fragment selected using Primer3 software.3030 Untergasser A, Nijveen H, Rao X, Bisseling T, Geurts R, Leunissen JaM. Primer3 Plus, an enhanced web interface to Primer3. Nucleic Acids Res. 2007;35:W71-W74. In addition, the 16S rRNA gene from Escherichia coli was amplified using the primer pair FC27 and R530 as an endogenous internal control in each fecal sample evaluated for the specific presence of MAstV5.3131 Gontang EA, Fenical W, Jensen PR. Phylogenetic diversity of Gram-positive bacteria cultured from marine sediments. Appl Environ Microbiol. 2007;73(10):3272-3282. For partial genome amplification, sets of 12 pairs of sequencing primers were selected to amplify overlapping fragments of ORF1 (ORF1a and ORF1b) and capsid protein (ORF2) segment representing a consensus sequence of approximately 5000 nucleotides. The primer sequences are shown in Table 1.
Nested RT-PCR for MAstV5 detection
The cDNA was synthesized using SuperScript® III Reverse Transcriptase Kit (Life Technologies, USA) using the reverse primers in a total volume of 20 µL, following the manufacturer's instructions. The cDNA amplification was conducted in a final volume of 25 µL containing 1× PCR buffer, 1.5 mM of MgCl2, 0.2 mM of dNTP mix, 0.2 µM of each primer and 1 unit of Platinum® Taq DNA Polymerase (Life Technologies, USA). The first round of RT-PCR screening was carried out with an initial incubation at 94 °C for 3 min, 30 cycles of amplification consisting of denaturation at 94 °C for 1 min, annealing at 50 °C for 1 min, and extension at 72 °C for 1 min. The second round was performed in a final volume of 25 µL that contained 2 µL of the first reaction product and the thermocycler conditions were the same as those used for the first round.
The MAstV5-specific RT-PCR with specific and internal control primers was performed as a multiplex protocol. Cycling conditions were an initial cycle at 94 °C for 5 min, 25 cycles of denaturation at 94 °C for 30 s, annealing at 58 °C for 30 s and polymerization at 72 °C for 1 min, which was followed by a final extension cycle at 72 °C for 7 min. To confirm the specific amplification of MAstV5, RT-PCR products were submitted to purification using the NucleoSpin Extract II Kit (Macherey-Nagel, Germany) and sequenced. Both DNA strands were sequenced with an ABI PRISM 3100 Genetic Analyzer using a BigDye Terminator v.3.1 cycle Sequencing Kit (Applied Biosystems, USA).
Detection of other enteric viruses
All positive MAstV5 samples were also screened for other common enteric viruses through the amplification of cDNA/DNA. The primer pairs used for the detection of canine distemper virus (CDV), carnivore protoparvovirus 1 (canine parvovirus 2, CPV2), canine coronavirus (CCoV), canine rotavirus (CRV) and, canine adenovirus 1 (CAdV1) and CAdV2 are shown in Table 1. The cDNA/DNA amplification of the target sequences was conducted in a total volume of 25 µL containing 1× PCR buffer, 1.5 mM of MgCl2, 0.2 mM of dNTP mix, 0.2 µM of each primer pair and 1 unit of Taq DNA Polymerase (Ludwig Biotecnologia, Alvorada, RS, Brazil).
Genome amplification
Four MAstV5-positive samples were selected, taking into account their different geographical origins. ORF1a, ORF1b and ORF2 sequences were amplified using a nested touchdown RT-PCR method. The first round of amplification was conducted in a final volume of 25 µL. The cycling conditions included an initial denaturation at 95 °C for 5 min, 20 cycles of 30 s for denaturation at 95 °C, outer primer pair annealing for 30 s at 55-45 °C per the touchdown method, and 7 min of extension at 72 °C, with a final 7 min extension at 72 °C. The same final volume was used in the second round, which contained 2 µL of the amplification product of the first round. The second round cycling conditions were an initial denaturation at 95 °C for 5 min, 30 cycles of 30 s of denaturation at 95 °C, inner primer pair annealing for 30 s at 55-45 °C per the touchdown method, and 1 min of extension at 72 °C, with a 7 min final extension at 72 °C.
Sequencing and phylogenetic inferences
The RT-PCR products generated with the sets of sequencing primers were purified using the NucleoSpin Extract II Kit (Macherey-Nagel, Germany). Both DNA strands were sequenced with an ABI PRISM 3100 Genetic Analyzer using a BigDye Terminator v.3.1 cycle Sequencing Kit (Applied Biosystems, USA). Overlapping fragments were aligned and assembled using SeqMan software from the DNASTAR package (DNASTAR, USA).3232 Burland TG. DNASTAR's Lasergene sequence analysis software. Methods Mol Biol. 2000;132:71-91. [The open reading frames were identified using the NCBI ORF Finder software (http://www.ncbi.nlm.nih.gov/gorf/gorf.html).]
Sequence alignment was performed using the CLUSTAL W. For the phylogenetic inferences of the MAstV capsid protein region, the MAstV5 sequences that were submitted to genome amplification and 12 MAstV5 representative strains were included. MEGA6 software2929 Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. 2013;30:2725-2729. was used for phylogeny inference calculated using "find best DNA/protein model" tool from MEGA6. The Kimura 2-parameter substitution model was selected for the MAstV5 ORF2 nucleotide inference, and the LG substitution model (frequencies +F) was used for the amino acid inference. The substitution-rate variation among sites was modeled with a gamma distribution (shape parameter = 5). Statistical support was provided by 1000 non-parametric bootstrap analyses. A nucleotide distance matrix was calculated using an alignment with ORF2 and partial genome sequences (Table 2). The Mamastrovirus genotypes were distinguished based on the amino acid sequence of the full length ORF2, where the genetic distances (p-dist) 0.378-0.750 and 0.006-0.312 between and within groups, respectively, were used.55 Bosch A, Guix S, Krishna NK, et al. Family Astroviridae. In: King AMQ, Lefkowitz E, Adams MJ, Carstens EB, eds. Virus Taxonomy: Classification and Nomenclature of Viruses (Ninth Report of the International Committee on the Taxonomy of Viruses). 9th ed. 2011. New York. All of the sequence alignments used to construct the phylogenetic trees are available in Figshare (http://figshare.com/) with the DOI number https://doi.org/10.6084/m9.figshare.4596325. The nucleotide sequences obtained in this study were deposited in GenBank under accession numbers KR349488-KR349491.
Comparison of identity percentage between nucleotide sequence of the partial genomes and amino acid sequence identity percentage of open reading frame 2 (ORF2) from the sequenced MAstV5 compared with sequences available in GenBank.
Results
Detection of MAstV5 in fecal samples
The RT-PCR protocol using the screening primers for MAstV5 was positive in 22% (64/269) of the samples, and the protocol using the specific primers for MAstV5 identified 12% (32/269) of the samples tested. The sample was considered MAstV5 positive if the results of at least one of the two RT-PCR protocols were positive, which resulted in 26% (71/269) of the fecal samples being detected as positive. In addition, PCR products generated from both protocols were submitted to DNA sequencing to confirm the results (data not shown). The RT-PCR internal control from the 16S rRNA gene of E. coli was positive in all 269 of the samples tested.
Information about clinical signs of enteric disease was available for 49 of the 71 positive samples. Considering only these, in all of the MAstV5-positive samples derived from dogs with clinical signs suggestive of gastroenteritis, other enteric viruses were simultaneously detected. Detailed results for the supposed association with clinical signs and the detection of other enteric viruses are shown in Fig. 1 and Table 3, respectively.
Presumptive association between the MAstV5-positive samples (multiple or single infection) and the presence of clinical signs in the sampled dogs.
Animals were ranked in age category as puppies, adults and unknown age. Adults revealed a higher frequency of MAstV5-positive RT-PCR results than puppies (puppies: 44/166, 26.5%; adults: 6/16, 37.5%; unknown age: 21/91, 23.1%). No significant difference between dogs age and MAstV5 positive samples was observed in the different ranked ages (P = 0.56).
MAstV5 genome sequences and phylogenetic inferences
Four partial genomes were obtained, namely, MAstV5_Sara/13/BRA, MAstV5_237/13/BRA, MAstV5_GRAV/13/BRA and MAstV5_5617/12/BRA, which were 4980, 5011, 5039 and 5012 nt in length, respectively, excluding the poly(A) tail and the 5′ untranslated regions (UTRs). The three first partial genomes were collected from different cities of Rio Grande do Sul State (Porto Alegre, Viamão and Gravataí, respectively), and one was from Londrina city, Paraná State. All of the four nearly complete genomes contained a typical AstV organization in the three predicted ORFs - ORF1a, ORF1b and ORF2. Further genome sequence comparison revealed that the four partial genomes had greater identities (94-96%) with Chinese strains, considered as the Italy, Hungary and United Kingdom strains identity ranged from 76-82%, 75-95% and 86-96% when compared to the Brazilian strains identified here, respectively (Table 2). In addition, the amino acid sequences of the ORF2 of the HUN/2012/126 (GenBank accession number KX599352), GI.E/Dog/ITA/2010/Zoid (GenBank accession number JN193534) strains showed lower identities (76 and 77%, respectively) compared to the Brazilian strains (Table 2).
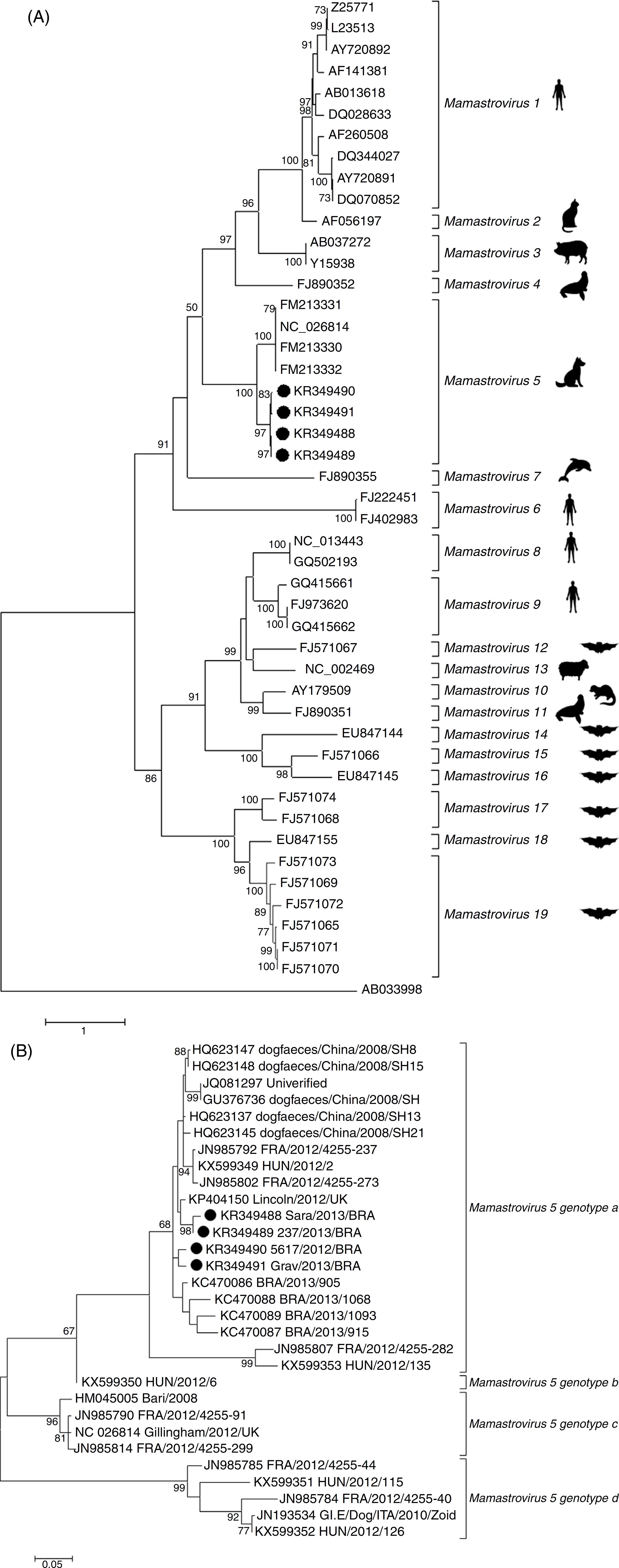
Phylogenetic inferences were also carried out with the partial and complete sequences of OFR2 at nucleotide and amino acid levels. The partial genome sequences of MAstV5 obtained in the present study and those available in GenBank, together with selected Mamastrovirus reference sequences from other species, generated two evolutionary trees (Fig. 2). Forty-three reference strains and the four sequences from this study corresponding to 19 Mamastrovirus species were delineated with high bootstrap support throughout the entire tree (Fig. 2A). In the MAstV5 clade, all of the present sequences clustered with "Gillingham/2012/UK" (GenBank accession number NC_026814), although with low amino acid identities of approximately 80% (Table 2). Consequently, these four new sequences grouped within Chinese sequences, suggesting a different genotype putatively named as MAsTV5a (Fig. 2B). Through a pairwise comparison of the ORF2 nucleotide sequence, a high degree of nucleotide identity, ranging from 95% to 99%, was detected among the Brazilian type strains of this study. The study sequences showed a closer relationship with the Chinese strains grouping in genotype a. More distant strains were observed among the HUN/2012/126 (GenBank accession number KX599352) Hungary strain composing the genogroup b, Bari/2008_ITA (GenBank accession number HM045005) and Gillingham/2012/UK (GenBank accession number NC_026814) strains from Italy and United Kingdom, respectively, grouping in the genotype c, and finally, forming the genotype d, HUN/2012/115 (GenBank accession number KX599351), HUN/2012/126 (GenBank accession number KX599352) strains from Hungary and GI.E/Dog/ITA/2010/Zoid (GenBank accession number JN193534) strain from Italy. This suggests a distinction of four sub-lineages among the MAstV5 species - MAstV5a to MAstV5d (Fig. 2B).
Evolutionary relationship of MAstV5 with representative MAstV genera. The percentage of replicates in which the associated virus clustered together in the bootstrap test (1000 replicates) is shown next to the branches in each tree. The trees are drawn to scale; bars represent the number of substitutions per site. All positions except ambiguous positions were included. Bootstrap values <50 were excluded. GenBank accession numbers are shown on the tree. MAstV5 sequences obtained in the present study are indicated with a black dot (●). The Kimura 2-parameter substitution model was selected for the MAstV5 ORF2 nucleotide inference, and the LG substitution model (frequencies +F) was used for the amino acid inference. The substitution-rate variation among sites was modeled with a gamma distribution (shape parameter = 5). (A) Evolutionary tree based on the complete amino acid sequences of the ORF2 gene (capsid) of 47 nucleotide sequences of AstVs. (B) Evolutionary tree based on the partial nucleotide sequences of ORF2 from 30 sequences of MAstV5.
Discussion
Here, in a screening of dog fecal samples, 26% (71/269) of the dogs with and without diarrhea were MAstV5 positive, as determined using RT-PCR. Likewise, non-viral agents and factors such as bacteria, intestinal parasites, malnutrition and intoxications are able to promote enteric disease mainly in the young dog population. The search for other enteric viruses in the MAstV5-positive samples from dogs with gastroenteritis showed that the dogs were also infected with other known pathogens. Moreover, we found that single MAstV5 infection was associated only with the asymptomatic state, although there is a risk that the results will be biased, since the analyzes were conducted on the basis of convenience sampling and we can not exclude the possibility that the long term of viral shedding could be an explanation for the MAstV5-positive samples detected in asymptomatic dogs, based on previous study that demonstrated the comparison between virus load and clinical manifestation2626 Martella V, Moschidou P, Catella C, et al. Enteric disease in dogs naturally infected by a novel canine astrovirus. J Clin Microbiol. 2012;50(3):1066-1069. (Fig. 1 and Table 3). These findings were not unexpected, as mixed infections are common, but more studies will be necessary to real deduce the role of MAstV5 in the cases reported here.1515 Williams FP. Astrovirus-like, coronavirus-like, and parvovirus-like particles detected in the diarrheal stools of beagle pups. Arch Virol. 1980;66(3):215-226.
16 Vieler E, Herbst W. Electron microscopic demonstration of viruses in feces of dogs with diarrhea. Tierarztl Prax. 1995;23(1):66-69.
17 Marshall JA, Healey DS, Studdert MJ, et al. Viruses and virus-like particles in the faeces of dogs with and without diarrhoea. Aust Vet J. 1984;61(2):33-38.
18 Toffan A, Jonassen CM, De Battisti C, et al. Genetic characterization of a new astrovirus detected in dogs suffering from diarrhoea. Vet Microbiol. 2009;139(1-2):147-152.
19 Zhu a L, Zhao W, Yin H, et al. Isolation and characterization of canine astrovirus in China. Arch Virol. 2011;156(9):1671-1675.
20 Castro TX, Cubel Garcia RCN, Costa EM, Leal RM, Xavier MDPT, Leite JPG. Molecular characterisation of calicivirus and astrovirus in puppies with enteritis. Vet Rec. 2013;172(21):557.
21 Grellet A, De Battisti C, Feugier A, et al. Prevalence and risk factors of astrovirus infection in puppies from French breeding kennels. Vet Microbiol. 2012;157(1-2):214-219.
22 Caddy SL, Goodfellow I. Complete genome sequence of canine astrovirus with molecular and epidemiological characterization of UK strains. Vet Microbiol. 2015;177(1-2):206-213.
23 Takano T, Takashina M, Doki T, Hohdatsu T. Detection of canine astrovirus in dogs with diarrhea in Japan. Arch Virol. 2015;160(6):1549-1553.-2424 Choi S, Lim S-I, Kim YK, Cho Y-Y, Song J-Y, An D-J. Phylogenetic analysis of astrovirus and kobuvirus in Korean dogs. J Vet Med Sci. 2014;76(8):1141-1145.
Several reports of MAstV5 suggest a clinical association of virus molecular detection and diseased dog clinical samples.1818 Toffan A, Jonassen CM, De Battisti C, et al. Genetic characterization of a new astrovirus detected in dogs suffering from diarrhoea. Vet Microbiol. 2009;139(1-2):147-152.,2020 Castro TX, Cubel Garcia RCN, Costa EM, Leal RM, Xavier MDPT, Leite JPG. Molecular characterisation of calicivirus and astrovirus in puppies with enteritis. Vet Rec. 2013;172(21):557.
21 Grellet A, De Battisti C, Feugier A, et al. Prevalence and risk factors of astrovirus infection in puppies from French breeding kennels. Vet Microbiol. 2012;157(1-2):214-219.
22 Caddy SL, Goodfellow I. Complete genome sequence of canine astrovirus with molecular and epidemiological characterization of UK strains. Vet Microbiol. 2015;177(1-2):206-213.-2323 Takano T, Takashina M, Doki T, Hohdatsu T. Detection of canine astrovirus in dogs with diarrhea in Japan. Arch Virol. 2015;160(6):1549-1553.,2626 Martella V, Moschidou P, Catella C, et al. Enteric disease in dogs naturally infected by a novel canine astrovirus. J Clin Microbiol. 2012;50(3):1066-1069.,3333 Martella V, Moschidou P, Lorusso E, et al. Detection and characterization of canine astroviruses. J Gen Virol. 2011;92(Pt 8):1880-1887. Furthermore, studies of the prevalence of MAstV5 in China showed that 12% (22/183) of the puppies displaying clinical signs of diarrhea were positive for MAstV5, as determined using RT-PCR, compared to none of 138 healthy dogs, although these studies did not look for other viruses that may be associated with diarrhea.1919 Zhu a L, Zhao W, Yin H, et al. Isolation and characterization of canine astrovirus in China. Arch Virol. 2011;156(9):1671-1675. In a study conducted in Italy, 24% of 110 stool samples collected from dogs with clinical signs tested positive for the presence of MAstV5 RNA, and 9% (10/110) of the samples showed an MAstV5-single infection, although other asymptomatic animals (9% of 75) were also positive for MAstV53333 Martella V, Moschidou P, Lorusso E, et al. Detection and characterization of canine astroviruses. J Gen Virol. 2011;92(Pt 8):1880-1887.. Therefore, the association with clinical signs and the shedding of the virus was described only in a case study of 2 animals, which is apparently an isolated case.2626 Martella V, Moschidou P, Catella C, et al. Enteric disease in dogs naturally infected by a novel canine astrovirus. J Clin Microbiol. 2012;50(3):1066-1069. A prevalence study in France found that 21% (66/316) of the puppies in 42% (14/33) of the breeding kennels surveyed were MAstV5 positive, as determined using RT-PCR.2121 Grellet A, De Battisti C, Feugier A, et al. Prevalence and risk factors of astrovirus infection in puppies from French breeding kennels. Vet Microbiol. 2012;157(1-2):214-219. In the same report, the authors observed that puppies that were less than 7 weeks old were especially susceptible to MAstV5 infection, although a direct association with clinical signs was not possible.2121 Grellet A, De Battisti C, Feugier A, et al. Prevalence and risk factors of astrovirus infection in puppies from French breeding kennels. Vet Microbiol. 2012;157(1-2):214-219. Lastly, recent studies found a MAstV5 prevalence of 6% in the United Kingdom and an infection rate of 33% in puppies under three months in Japan.2222 Caddy SL, Goodfellow I. Complete genome sequence of canine astrovirus with molecular and epidemiological characterization of UK strains. Vet Microbiol. 2015;177(1-2):206-213.,2323 Takano T, Takashina M, Doki T, Hohdatsu T. Detection of canine astrovirus in dogs with diarrhea in Japan. Arch Virol. 2015;160(6):1549-1553.
The partial genomic sequencing and characterization of selected samples revealed a remarkable genetic heterogeneity of MAstV5 of Brazilian origin. Because it was hypothesized that two strains of human AstV with less than 95% identity at the nucleotide level are serologically distinguishable,3434 Walter JE, Briggs J, Guerrero ML, et al. Molecular characterization of a novel recombinant strain of human astrovirus associated with gastroenteritis in children. Arch Virol. 2001;146(12):2357-2367. the lower identities (<85%) shown between the capsid gene sequences analyzed here may reflect the need for a novel species classification into four genotypes - MAstV5a to MAstV5d. Additionally, phylogenetic analysis indicated that the four MAstV5 strains reported here represent a lineage that is more closely related to the Chinese strains than to the others strains, based on the high sequence identity (97%) of ORF2, according to the species demarcation criteria established by the ICTV55 Bosch A, Guix S, Krishna NK, et al. Family Astroviridae. In: King AMQ, Lefkowitz E, Adams MJ, Carstens EB, eds. Virus Taxonomy: Classification and Nomenclature of Viruses (Ninth Report of the International Committee on the Taxonomy of Viruses). 9th ed. 2011. New York. (Table 2).
In summary, we found 26% MAstV5-positive fecal samples in dogs with or without gastroenteritis. Based on sequence analysis of the partial genome from four MAstV5-positive samples, we proposed a novel species classification into four genotypes - MAstV5a to MAstV5d. More studies are required to understand the biology and attempt the clinical and antigenic implications of astrovirus genotypes in dogs to elucidate the relative veterinary importance of different canine AstV types.
Acknowledgments
The authors would like to express their gratitude to the clinical practitioners who generously provided samples for analysis and to the graduate and post-graduate students of the Laboratório de Virologia for their excellent technical support in this work.
References
-
1Madeley CR, Cosgrove BP. Letter: 28 nm particles in faeces in infantile gastroenteritis. Lancet 1975;2(7932):451-452.
-
2De Benedictis P, Schultz-Cherry S, Burnham A, Cattoli G. Astrovirus infections in humans and animals - molecular biology, genetic diversity, and interspecies transmissions. Infect Genet Evol 2011;11(7):1529-1544.
-
3Jiang B, Monroe SS, Koonin EV, Stine SE, Glass RI. RNA sequence of astrovirus: distinctive genomic organization and a putative retrovirus-like ribosomal frameshifting signal that directs the viral replicase synthesis. Proc Natl Acad Sci U S A 1993;90(22):10539-10543.
-
4Monroe SS, Jiang B, Stine SE, Koopmans M, Glass RI. Subgenomic RNA sequence of human astrovirus supports classification of Astroviridae as a new family of RNA viruses. J Virol 1993;67(6):3611-3614.
-
5Bosch A, Guix S, Krishna NK, et al. Family Astroviridae. In: King AMQ, Lefkowitz E, Adams MJ, Carstens EB, eds. Virus Taxonomy: Classification and Nomenclature of Viruses (Ninth Report of the International Committee on the Taxonomy of Viruses). 9th ed. 2011. New York.
-
6Finkbeiner SR, Li Y, Ruone S, et al. Identification of a novel astrovirus (astrovirus VA1) associated with an outbreak of acute gastroenteritis. J Virol 2009;83(20):10836-10839.
-
7Lewis DC, Lightfoot NF, Cubitt WD, Wilson SA. Outbreaks of astrovirus type 1 and rotavirus gastroenteritis in a geriatric in-patient population. J Hosp Infect 1989;14(1):9-14.
-
8Wunderli W, Meerbach A, Güngör T, et al. Astrovirus infection in hospitalized infants with severe combined immunodeficiency after allogeneic hematopoietic stem cell transplantation. PLoS ONE 2011;6(11):e27483.
-
9Gallimore CI, Taylor C, Gennery AR, et al. Use of a heminested reverse transcriptase PCR assay for detection of astrovirus in environmental swabs from an outbreak of gastroenteritis in a pediatric primary immunodeficiency unit. J Clin Microbiol 2005;43(8):3890-3894.
-
10Fu Y, Pan M, Wang X, et al. Complete sequence of a duck astrovirus associated with fatal hepatitis in ducklings. J Gen Virol 2009;90(5):1104-1108.
-
11Imada T, Yamaguchi S, Mase M, Tsukamoto K, Kubo M, Morooka A. Avian nephritis virus (ANV) as a new member of the family Astroviridae and construction of infectious ANV cDNA. J Virol 2000;74(18):8487-8493.
-
12Biđin M, Lojkić I, Tišljar M, Biđin Z, Majnarić D. Astroviruses associated with stunting and pre-hatching mortality in duck and goose embryos. Avian Pathol 2012;41(1):91-97.
-
13Blomström AL, Widén F, Hammer AS, Belák S, Berg M. Detection of a novel astrovirus in brain tissue of mink suffering from shaking mink syndrome by use of viral metagenomics. J Clin Microbiol 2010;48(12):4392-4396.
-
14Bouzalas IG, Wuthrich D, Walland J, et al. Neurotropic astrovirus in cattle with nonsuppurative encephalitis in Europe. J Clin Microbiol 2014;52(9):3318-3324.
-
15Williams FP. Astrovirus-like, coronavirus-like, and parvovirus-like particles detected in the diarrheal stools of beagle pups. Arch Virol 1980;66(3):215-226.
-
16Vieler E, Herbst W. Electron microscopic demonstration of viruses in feces of dogs with diarrhea. Tierarztl Prax 1995;23(1):66-69.
-
17Marshall JA, Healey DS, Studdert MJ, et al. Viruses and virus-like particles in the faeces of dogs with and without diarrhoea. Aust Vet J 1984;61(2):33-38.
-
18Toffan A, Jonassen CM, De Battisti C, et al. Genetic characterization of a new astrovirus detected in dogs suffering from diarrhoea. Vet Microbiol 2009;139(1-2):147-152.
-
19Zhu a L, Zhao W, Yin H, et al. Isolation and characterization of canine astrovirus in China. Arch Virol. 2011;156(9):1671-1675.
-
20Castro TX, Cubel Garcia RCN, Costa EM, Leal RM, Xavier MDPT, Leite JPG. Molecular characterisation of calicivirus and astrovirus in puppies with enteritis. Vet Rec 2013;172(21):557.
-
21Grellet A, De Battisti C, Feugier A, et al. Prevalence and risk factors of astrovirus infection in puppies from French breeding kennels. Vet Microbiol 2012;157(1-2):214-219.
-
22Caddy SL, Goodfellow I. Complete genome sequence of canine astrovirus with molecular and epidemiological characterization of UK strains. Vet Microbiol 2015;177(1-2):206-213.
-
23Takano T, Takashina M, Doki T, Hohdatsu T. Detection of canine astrovirus in dogs with diarrhea in Japan. Arch Virol. 2015;160(6):1549-1553.
-
24Choi S, Lim S-I, Kim YK, Cho Y-Y, Song J-Y, An D-J. Phylogenetic analysis of astrovirus and kobuvirus in Korean dogs. J Vet Med Sci 2014;76(8):1141-1145.
-
25Mihalov-Kovács E, Martella V, Lanave G, et al. Genome analysis of canine astroviruses reveals genetic heterogeneity and suggests possible inter-species transmission. Virus Res 2016, http://dx.doi.org/10.1016/j.virusres.2016.12.005
» http://dx.doi.org/10.1016/j.virusres.2016.12.005 -
26Martella V, Moschidou P, Catella C, et al. Enteric disease in dogs naturally infected by a novel canine astrovirus. J Clin Microbiol 2012;50(3):1066-1069.
-
27Boom R, Sol CJ, Salimans MM, Jansen CL, Wertheim-van Dillen PM, van der Noordaa J. Rapid and simple method for purification of nucleic acids. J Clin Microbiol 1990;28(3):495-503.
-
28Chu DKW, Poon LLM, Guan Y, Peiris JSM. Novel astroviruses in insectivorous bats. J Virol 2008;82(18):9107-9114.
-
29Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. 2013;30:2725-2729.
-
30Untergasser A, Nijveen H, Rao X, Bisseling T, Geurts R, Leunissen JaM. Primer3 Plus, an enhanced web interface to Primer3. Nucleic Acids Res. 2007;35:W71-W74.
-
31Gontang EA, Fenical W, Jensen PR. Phylogenetic diversity of Gram-positive bacteria cultured from marine sediments. Appl Environ Microbiol 2007;73(10):3272-3282.
-
32Burland TG. DNASTAR's Lasergene sequence analysis software. Methods Mol Biol. 2000;132:71-91.
-
33Martella V, Moschidou P, Lorusso E, et al. Detection and characterization of canine astroviruses. J Gen Virol 2011;92(Pt 8):1880-1887.
-
34Walter JE, Briggs J, Guerrero ML, et al. Molecular characterization of a novel recombinant strain of human astrovirus associated with gastroenteritis in children. Arch Virol. 2001;146(12):2357-2367.
-
35Buonavoglia C, Martella V, Pratella A, et al. Evidence for evolution of canine parvovirus type 2 in Italy. J Gen Virol. 2001;82(12):3021-3025.
-
36Linné T. Differences in the E3 regions of the canine adenovirus type 1 and type 2. Virus Res 1992;23:119-133.
-
37Herrewegh AAPM, Smeenk I, Horzinek MC, Rottier PJM, De Groot RJ. Feline coronavirus type II strains 79-1683 and 79-1146 originate from a double recombination between feline coronavirus type I and canine coronavirus. J Virol. 1998;72:4508-4514.
-
38Gouvea V, Glass RI, Woods P, et al. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. J Clin Microbiol 1990;28:276-282.
-
39Castilho JG, Brandão PE, Carnieli P, et al. Molecular analysis of the N gene of canine distemper virus in dogs in Brazil. Arq Bras Med Vet Zootec. 2007;59:654-659.
-
40Frisk AL, König M, Moritz A, Baumgärtner W. Detection of canine distemper virus nucleoprotein RNA by reverse transcription-PCR using serum, whole blood, and cerebrospinal fluid from dogs with distemper. J Clin Microbiol 1999;37:3634-3643.
Edited by
Publication Dates
-
Publication in this collection
Jul-Sep 2018
History
-
Received
16 Mar 2017 -
Accepted
26 Sept 2017