Abstracts
We investigate the local and regional determinants of galling insect richness on the dioecious shrub Baccharis concinna Barroso (Asteraceae) in the southeastern portion of the Espinhaço Mountains in Brazil. The total number of galling species and the richness of galling Cecidomyiidae (Diptera) were influenced by plant gender. Male plants supported a higher richness of galling insects than female plants, supporting the hypothesis of sex-biased herbivory at the community level. The total number of galling species and the richness of galling cecidomyiids showed a peak at intermediate altitudes. The richness of all galling species and galling cecidomyiids were strongly influenced by habitat. Plants in xeric habitats supported more galling species than plants in mesic habitats, corroborating the hypothesis of hygrothermal/nutritional stress. Our results indicate that local factors may be the most important factors structuring the galling insect community on B. concinna.
Altitudinal gradient; Baccharis concinna; diversity; ecological community; herbivore
Foram investigados os determinantes locais e regionais da riqueza de insetos indutores de galhas sobre o arbusto dióico Baccharis concinna Barroso (Asteraceae) na porção sudeste da Cadeia do Espinhaço, MG. O número total de espécies de insetos indutores de galhas e a riqueza de Cecidomyiidae (Diptera) foram influenciados pelo sexo da planta. Plantas masculinas apresentaram maior riqueza de insetos indutores de galhas do que plantas femininas, corroborando a hipótese da herbivoria mediada pelo sexo na comunidade. O número total de espécies de insetos galhadores e a riqueza de Cecidomyiídeos mostraram um pico em altitudes intermediárias. A riqueza de todas as espécies de insetos galhadores e de cecidomiideos foi fortemente influenciada pelo habitat. Plantas no habitat xérico apresentaram mais espécies de insetos galhadores do que plantas no habitat mésico, corroborando a hipótese do estresse higrotérmico/nutricional. Os resultados indicam que os fatores locais podem ser importantes estruturadores da comundade de insetos galhadores em B. concinna.
Baccharis concinna; comunidade ecológica; diversidade; gradiente altitudinal; herbívoro
ECOLOGY, BEHAVIOR AND BIONOMICS
Convergence in the variation of local and regional galling species richness
Convergência na variação local e regional da riqueza de espécies de insetos galhadores
Marco A.A. CarneiroI, II; G.W. FernandesIII; O.F.F. De SouzaII
ILab. Padrões de Distribuição Animal, Depto. Ciências Biológicas ICEB, Univ. Federal de Ouro Preto, 35400 000, Ouro Preto, MG
IIDepto. Biologia Animal, Univ. Federal de Viçosa, 35571 000, Viçosa, MG, og.souza@mail.ufv.br
IIIEcologia Evolutiva de Herbívoros Tropicais/DBG, C. postal 486, ICB/ Univ. Federal de Minas Gerais, UFMG, 31270 901 Belo Horizonte, MG, gwilson@icb.ufmg.br
ABSTRACT
We investigate the local and regional determinants of galling insect richness on the dioecious shrub Baccharis concinna Barroso (Asteraceae) in the southeastern portion of the Espinhaço Mountains in Brazil. The total number of galling species and the richness of galling Cecidomyiidae (Diptera) were influenced by plant gender. Male plants supported a higher richness of galling insects than female plants, supporting the hypothesis of sex-biased herbivory at the community level. The total number of galling species and the richness of galling cecidomyiids showed a peak at intermediate altitudes. The richness of all galling species and galling cecidomyiids were strongly influenced by habitat. Plants in xeric habitats supported more galling species than plants in mesic habitats, corroborating the hypothesis of hygrothermal/nutritional stress. Our results indicate that local factors may be the most important factors structuring the galling insect community on B. concinna.
Key words: Altitudinal gradient, Baccharis concinna, diversity, ecological community, herbivore
RESUMO
Foram investigados os determinantes locais e regionais da riqueza de insetos indutores de galhas sobre o arbusto dióico Baccharis concinna Barroso (Asteraceae) na porção sudeste da Cadeia do Espinhaço, MG. O número total de espécies de insetos indutores de galhas e a riqueza de Cecidomyiidae (Diptera) foram influenciados pelo sexo da planta. Plantas masculinas apresentaram maior riqueza de insetos indutores de galhas do que plantas femininas, corroborando a hipótese da herbivoria mediada pelo sexo na comunidade. O número total de espécies de insetos galhadores e a riqueza de Cecidomyiídeos mostraram um pico em altitudes intermediárias. A riqueza de todas as espécies de insetos galhadores e de cecidomiideos foi fortemente influenciada pelo habitat. Plantas no habitat xérico apresentaram mais espécies de insetos galhadores do que plantas no habitat mésico, corroborando a hipótese do estresse higrotérmico/nutricional. Os resultados indicam que os fatores locais podem ser importantes estruturadores da comundade de insetos galhadores em B. concinna.
Palavras-chave:Baccharis concinna, comunidade ecológica, diversidade, gradiente altitudinal, herbívoro
How many species exist on earth? For arthropods alone, estimates vary from 10 to 30 million species (Gaston 1991). Such a discrepancy denotes the amount of uncertainty still involved in the problem. Patterns in the distribution and abundance of organisms are easily recognizable and more informative than the number of species. In this sense, ecology should profit best from identifying repeated patterns and formulating broad generalizations (MacArthur 1972; Lawton 1996).
At the global scale, the clearest pattern of diversity is the decrease of species number with increasing latitude (Pianka 1966, Price 1991, Rohde 1992, Willig et al. 2003). Several hypotheses have been proposed to explain such a gradient, but they are circular or weakly supported by empirical data (Rhode 1992, Willig et al. 2003). As far as it is known, galling insects are one of the exceptions to this pattern, presenting a peak of diversity at intermediate latitudes (Price et al. 1998). At the regional scale, such differences between gallers and other species are remarkably parallel: insect species diversity is, again, negatively correlated with altitude (Fernandes & Price 1988, 1991; McCoy 1990 and papers cited in; Fernandes & Lara 1993; Skuhravá 1994; Skuhravá & Skuhravy 1994), whereas in some cases galling insect diversity peaks at intermediate altitudes (Waring & Price 1990).
Such a convergence of latitudinal and altitudinal patterns in species diversity of gallers has led authors to speculate about the mechanisms involved (Fernandes et al. 2005). Fernandes & Price (1991) observed that the negative relationship between altitude at regional scale and galling species richness was heavily dependent on xeric habitat than mesci habitat, suggesting that altitude is a spurious correlate, and environmental harshness is the key factor determining galling species richness. Such a conclusion is supported by the latitudinal pattern, which galling species richness is higher at intermediate latitudes on warm habitats and sclerophyllous vegetation under water and nutrient stress (e.g., Cerrado or 'Brazilian savannas', Chaparral, and other Mediterranean-type vegetation, Price et al. 1998). At the local scale, species richness does not correlate either with host plant architecture (Fernandes & Price 1988) and is only weakly correlated with plant species richness (Fernandes & Price 1988, Blanche & Westoby 1995, but see Wright & Samways 1996).
The pattern of higher richness of galinng species in xeric habitat could arise from a few coupled mechanisms. Firstly, the gall itself may be an effective protection against the direct action of climate (Price et al. 1987, Stone & Schönrogge 2003). Secondly, given that plant nutrients become toxic to gallers at high levels, gallers would succeed better in harsh environments where plants tend to have low nutrient status (Fernandes & Price 1991). Galling species are able to circumvent the excess of chemical defense, typical of stressed plants (Müller et al. 1987, Stone et at. 2002), by inducing a phenol-free plant tissue when producing their galls (see Stone & Schönrogge 2003). Thirdly, the differential selective pressures inflicted by natural enemies and plant resistance between xeric and mesic habitats on gallmakers (Fernandes & Price 1988, 1992; Fernandes 1990, 1998).
Nevertheless, most of the above patterns have been observed by gathering data from several environments and host plant species. Those analyses are, therefore, obscured by a variety of correlates, which may confound the results. Here we investigate local determinants of galling insect richness, in a unique system nearly void of confounding variables. We use the system composed by the dioecious shrub Baccharis concinna Barroso (Asteraceae) and its associated galling insect community, in a local altitudinal gradient within the same latitude. Using such a system, effects of latitude, plant species richness, and plant architecture need not be considered, reducing the number of confounding variables. Moreover, because we use altitudinal data on a local scale, regional contributions to altitudinal patterns need not to be considered. Our aim is to check the validity of some patterns already reported for galling species, namely, that altitude, plant gender and plant habitat influence the richness of this group of organisms.
Material and Methods
Plant and Galling Insects.Baccharis spp. (Asteraceae) are native to the New World and present the highest diversity of galling insects in the Neotropics (Fernandes et al. 1996). B. concinna is dioecious, and endemic to the Espinhaço range, Minas Gerais southeastern Brazil (Barroso 1976). This plant hosts nine species of galls induced by Cecidomyiidae (Diptera), two by Lepidoptera, two by Curculionidae (Coleoptera), one by Psyllidae (Homoptera), one induced by a fungus, and another by a parasitic plant (Loranthaceae) (Fernandes et al. 1996). The present study considered only galls induced by insects.
Study Sites. Collections were taken at Serra do Cipó, in the southern portion of the Espinhaço range, (19°15' e 19°30' S e 43°30' e 43°55' W) at 900, 1000, 1100, and 1300 m above sea level. One site was studied at each altitude. Habitats were typically xeric, with rocky soils, dominated by cerrado and rupestrian field vegetation (see Eiten 1978, Giulietti et al. 1987). At 1300 m additional collections were taken in a mesic habitat, in a site bordering a stream. At all sites, B. concinna occurred in patches. Since no obvious barrier was observed, so as to impair interpopulational exchanges, patterns and processes are considered "local", as opposed to "regional".
Sampling and Analyses. Fifty plants of similar size, belonging to each sex, were randomly chosen in each of the four xeric sites along the altitudinal gradient, except at 900 m where we only found 30 males and 20 females. The mesic site at 1300 m held 38 female and 37 male plants. A total of 425 plants were sampled. Gall richness was counted directly on the plants, in the rainy season of 1993. The terms "diversity" and "richness" are used here interchangeably to refer to the number of species. Gall richness refers to (i) the total number of galling insect species or (ii) the number of Cecidomyiidae species, each of these being used as y-variables in separate analyses. They are refered to below as "total richness" and "richness of Cecidomyiidae", respectively.
Statistical analyses were conducted in two stages. Firstly, data from xeric sites were used in a multiple regression to check for the effects of plant gender and local altitude on the number of galling species per plant. In the second stage, data from xeric and mesic sites were used in an analysis of variance to check for the effects of plant habitat and plant gender on the number of galling species per plant. All analyses were followed by inspection of residuals.
Multiple regressions followed the backward stepwise procedure, in which significance is assessed by removing terms from an initial (full) model and observing the F statistic associated to such a change. Terms making up the full model included: plant gender, local altitude, local altitude squared, and their respective interactions. The minimum adequate model achieved by deletion of insignificant terms was used to plot graphs of the relationships studied.
Data used in ANOVAs refer to gallers counted on plants inhabiting xeric and mesic sites at the same altitude (1300 m). Because galls were counted on female as well as on male plants, plant gender has also been included in these analyses. Thus, factors for the ANOVAs included plant habitat, plant gender, and their interactions.
After performing analyses on the total number of galling insect species, similar statistical procedures were applied to the number of galling Cecidomyiidae species per plant as a separate y variate. The reasoning behind such analyses is that Cecidomyiidae are the most common galling insect taxon (Gagné 1989). Therefore, most patterns might depend heavily on the responses of this one taxon (see Fernandes et al. 1994).
Results
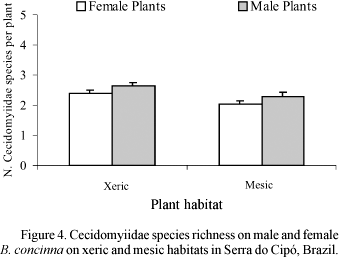
Plant Gender and Altitude. The total number of galling species was influenced by plant gender, with male plants supporting higher richness of gallers than female plants (Table 1, Fig. 1). Total richness peaked at intermediate altitudes (Fig. 1), independently of plant gender, as denoted by the absence of a significant interaction term in Table 1. The amount of variance in total richness explained by altitude alone was nearly ten times higher than the variance explained by plant gender (Table 1).
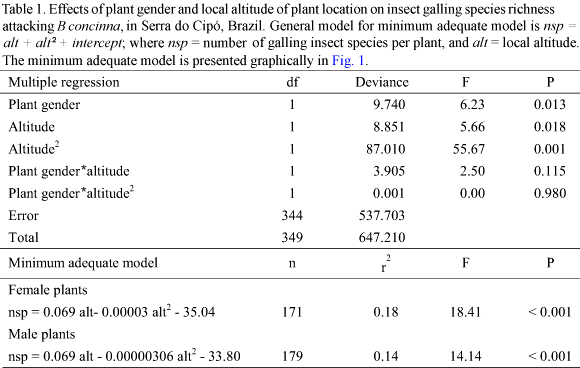
Similar patterns were found for the richness of Cecidomyiidae (Table 2, Fig. 2). The richness of Cecidomyiidae species was influenced by plant gender, with male plants supporting higher richness than female plants. A peak in richness was also observed at intermediate altitudes. Plant gender and altitude did not interact to influence Cecidomyiidae richness. The amount of variance explained by altitude was only 1.5 times higher than the variance explained by plant gender.
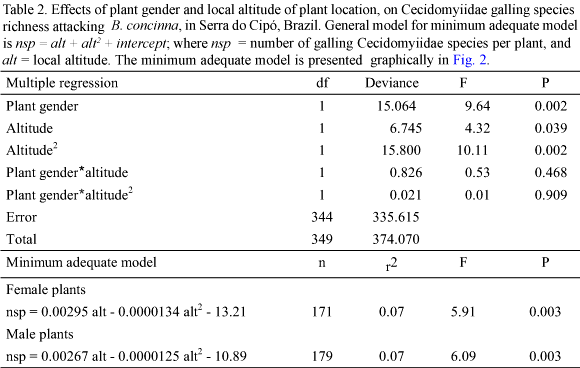
Plant Habitat. Galling species richness was strongly influenced by habitat (Table 3). Plants in xeric habitats supported more galling species than plants in mesic habitats (Fig. 3). In both habitat types, male and female plants did not differ in the richness of galling species (ANOVA F = 0.028; P = 0.867 xeric habitat; F = 2.916; P = 0.092 mesic habitat; Fig. 3).
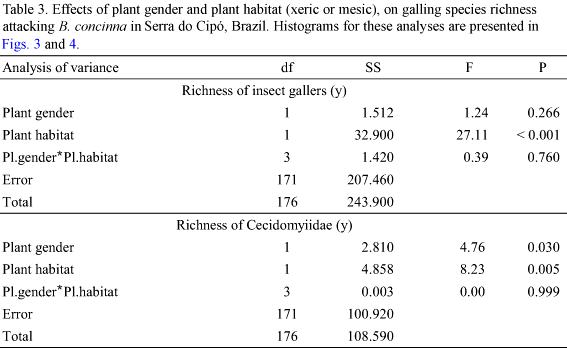
Richness of Cecidomyiidae was also affected by habitat (Table 3). Plants in xeric habitats supported more Cecidomyiidae species than plants in mesic habitats (Table 3, Fig. 4). Within both habitat types, male and female plants did not differ in the richness of galling Cecidomyiidae (ANOVA F = 2.323; P = 0.131 xeric habitat; F = 2.765; P = 0.121 mesic habitat; Fig. 3). However at 1300 m altitude, male plants supported more Cecidomyiidae species than female plants (Table 3, Fig. 4).
Discussion
Patterns of galling insects diversity are currently well documented (e.g. Fernandes et al. 1994, Price et al. 1998), in spite of a poor understanding of the mechanisms generating them (see Fernandes 1990, 1998; Fernandes et al. 2005). Hypotheses so far proposed generally rely on local determinants, such as plant gender (Boecklen & Hoffman 1993), plant sclerophylly (Fernandes & Price 1988, 1991), plant water stress (Fernandes & Price 1988, Waring & Price 1990), and environmental-dependent mortality (Taper & Case 1987, Fernandes & Price 1992). A common difficulty faced by researchers in such area of knowledge is to distinguish the actual determinant of the patterns, among a variety of possible interdependent variables. This is a vital step when proposing hypotheses regarding underlying mechanisms for such patterns.
Our data present a unique situation that helps to clearly define the importance of some local determinants of galling species richness. Effects of latitude, plant architecture, plant richness, and regional geographical variation needed not be considered, since our system comprises by the entire galling insect community inhabiting a single plant species in closely neighboring sites. Therefore, by using a restricted set of variables, causal factors are more apparent, so that new light could be shed on previous hypotheses.
The first pattern we could clearly define is the effect of altitude on total number of galling species (Fig. 1). Our data revealed a peak of richness at intermediate altitudes (= 1100 m), independent of plant gender. A similar peak has been reported for Asphondylia spp. (Cecidomyiidae) on Larrea tridentata (DC) Coville by Waring & Price (1990), but with elevation studied over a regional scale, spanning 400 km, and the richest sites located around 700 m. These authors did not analyze explicitly the peak of richness. Rather, they analyzed the reasons why lower elevations would support fewer galling species. Because their lower elevation sites were located in a desert, they hypothesized that the actual causal factor was plant water stress. Whether or not our results reveal some effect of plant stress mediated by altitude, remains to be explicitly tested in future study. The most important novelty here is that the peak of richness at intermediate altitudes, already reported for cecidomyiid species in a single genus at a regional scale (Asphondylia spp, Waring & Price 1990), is also valid for the community of insect gallers on B. concinna at a local scale, and is independent of plant gender.
Our data also confirm that male plants support more galling species than female plants. Female plants have been shown to support lower herbivore abundance for a variety of organisms, from fungi to mammals (Herms & Mattson 1992, Boecklen & Hoffman 1993, and references therein). The simplest consequence of this would be that female plants, by supporting fewer galling individuals, would 'miss' rare species, thereby generating lower galler richness, as revealed by our data. An in depth analysis of galler abundance will be presented elsewhere.
Another pattern, which has been already reported for gallers on a variety of host plant species, is the increased richness in xeric environments (Fernandes & Price 1992). Our data, again, confirmed such a pattern, but more clearly than previous studies, since these data refer to a single plant species, in closely neighboring sites, at the local scale.
All but one of the above patterns were closely followed by the Cecidomyiidae alone: richness presented a peak at intermediate altitudes (Fig. 2), plant gender did not interact with altitude (Table 2), male plants presented higher galling species richness than female plants (Fig. 2), and xeric habitats present higher galling species richness than mesic habitats. However, Cecidomyiidae did not show any inter-sex bias in plant attack (Table 3, Fig. 4). Considering that Cecidomyiidae is the most common galling taxon (Gagné 1989, 1994), we tend to believe that this will be the general pattern for gallers elsewhere. In other words, plant habitat seems to be more important than plant gender in determining local galling richness.
In conclusion, local patterns observed here confirm general trends presented by galling species richness elsewhere, even when such trends are apparent at regional scales. This seems to suggest that whatever the mechanisms, underlying such patterns, the key factor may be operating at the local level. Also, a striking resemblance exists between patterns observed elsewhere for abundance of gallers and the patterns described here, despite the fact that our data refer only to species richness. When richness mirrors abundance, it means that losses in individuals represent proportional losses in species numbers. In other words, species rank abundances remain unchanged when population densities change, which suggests that community organization, should be more dependable on environmental conditions than on species interactions. We conclude that galling insect species on B. concinna seems to be regulated by local determinants unrelated to species interactions. Nevertheless studies should be extended to other populations along distribution at gradients of other suitable test this conclusion.
Acknowledgments
We thank ESA Marques and two anonymous reviewers for their comments on early drafts of this manuscript. Logistical support was provided by Parque Nacional da Serra do Cipó-Ibama/MG. Financial support was provided by CNPq (30 4851/2004-3), FAPEMIG (CBS 1950/95; CRA 583/03), and International Foundation for Science (H-2487/1). CNPq provided a scholarship to MAAC. This study was in partial fulfillment for a degree of master of sciences at Universidade Federal de Viçosa of MAAC.
Literature Cited
Received 28/X/03. Accepted 30/III/05.
- Barroso, G. 1976. Compositae-subtribo Baccharidinae Hoffmann estudo das espécies ocorrentes no Brasil. Rodriguésia 40: 7-273.
- Blanche, K.R. & M. Westoby. 1995. Gall-forming insect diversity is linked to soil fertility via host plant taxon. Ecology 76: 2334-2337.
- Boecklen, W.J. & M.T. Hoffman. 1993. Sex-biased herbivory in Ephedra trifurca: The importance of sex-by-environment interactions. Oecologia 96: 49-55.
- Eiten, G. 1978. Delimitation of the cerrado concept. Vegetatio 36: 169-178.
- Fernandes, G.W. 1990. Hypersensitivity: a neglected plant resistance mechanism against insect herbivores. Environ. Entomol. 19: 1173-1182.
- Fernandes, G.W. 1998. Hypersensitivity as a phenotypic basis of plant induced resistance against a galling insect (Diptera: Cecidomyiidae). Environ. Entomol. 27: 260-267.
- Fernandes, G.W. & A.C.F. Lara. 1993. Diversity of Indonesian gall-forming herbivores along altitudinal gradients. Biod. Lett. 1: 186-192.
- Fernandes G.W., A.C.F. Lara & P.W. Price. 1994. The geography of galling insects and the mechanisms resulting in patterns, p.42-48. In W.J. Mattson, Y. Baranchikov & P.W. Price (eds.), Gall-forming insects: Ecology, physiology, and evolution. North Carolina, United States Department of Agriculture, 222p.
- Fernandes, G.W., M.A.A. Carneiro, A.C.F. Lara, L.R. Allain, G.R. Julião, G.I. Andrade, T.R. Reis & I.M. Silva. 1996. Galling insects on neotropical species of Baccharis (Asteraceae). Trop. Zool. 9: 315-332.
- Fernandes, G.W. & P.W. Price. 1988. Biogeographical gradients in galling species richness: tests of hypotheses. Oecologia 76: 161-167.
- Fernandes G.W. & P.W. Price. 1991. Comparison of tropical and temperate galling species richness: The roles of environmental harshness and plant nutrient status. p.91-115. In P.W. Price, T.M. Lewinsohn, G.W. Fernandes & W.W. Benson (eds.), Plant-animal interactions: Evolutionary ecology in tropical and temperate regions. New York, John Wiley, 639p.
- Fernandes, G.W. & P.W. Price. 1992. The adaptive significance of insect gall distribution: Survivorship of species in xeric and mesic habitats. Oecologia 90: 14-20.
- Fernandes, G.W., S.J. Gonçalves-Alvim & M.A.A. Carneiro. 2005. Habitat-driven effects on the diversity of gall-inducing insects in the Brazilian cerrado, p.687-704. In A. Raman, C.W. Schaefer & T.M. Withers (eds.) Biology, ecology, and evolution of gall-inducing arthropods. New Hampshire, Science Publishers Inc., 774p.
- Gagné, R.J. 1989. The plant-feeding gall midges of North America. Ithaca, Cornell University Press, 368p.
- Gagné, R.J. 1994. The gall midges of the region neotropical. Ithaca, Cornell University Press, 352p.
- Gaston, K.J. 1991. The magnitude of global insect species richness. Conserv. Biol. 5: 283-296.
- Giiulietti, A.M., N.L. Menezes, J.R. Pirani, M. Meguro & M.G.L. Wanderley. 1987. Flora da Serra do Cipó: Caracterização e lista de espécies. Bolm. Bot. 9: 1-151.
- Herms, D.A. & W.J. Mattson. 1992. The dilema of plants: To grow or defend. Quart. Rev. Biol. 67: 283-335.
- Lara, A.C.F. & G.W. Fernandes. 1996. The highest diversity of galling insects: Serra do Cipó, Brazil. Biod. Lett. 3: 111-114.
- Lawton, J. H. 1996. Patterns in ecology. Oikos 75: 145-147.
- MacArthur, R.H. 1972. Geographical ecology: Patterns in the distribution of species. New York, Harper & Row, 253p.
- McCoy, E.D. 1990. The distribution of insects along elevational gradients. Oikos 58: 313-322.
- Müller, R.N., P.J. Kalisz & T.W. Kimmerer. 1987. Intraspecific variation in production of astringent phenolics over a vegetation-resource availability gradient. Oecologia 72: 211-215.
- Pianka, E.R. 1966. Latitudinal gradients in species diversity: A review of concepts. Am. Nat. 100: 33-46.
- Price, P.W. 1991. Patterns in communities along latitudinal gradients, p.51-70. In P.W. Price; T.M. Lewinsohn, G.W. Fernandes & W.W. Benson (eds), Plant-animal interactions: Evolutionary ecology in tropical and temperate regions. New York, John Wiley, 639p.
- Price P.W., G.W. Fernandes, A.C.F. Lara, J. Brawn, D. Gerling, H. Barrios, M.G. Wright, S.P. Ribeiro & N. Rothcliff. 1998. Global patterns in local number of insect galling species. J. Biogeogr. 25: 581-591.
- Price, P.W., G.W. Fernandes & G.L. Waring. 1987. Hypotheses on the adaptive nature of galls. Environ. Entomol. 16: 15-24.
- Ricklefs, R. & D. Schluter. 1993. (eds.) Species diversity in ecological communities: Historical and geographical perspectives. Chicago, University of Chicago Press, 416p.
- Rohde, K. 1992. Latitudinal gradients in species diversity: the search for the primary cause. Oikos 65: 514-527.
- Taper, M.L. & T.J. Case. 1987. Interactions between oak tannins and parasite community structure: Unexpected benefits of tannins to cynipid gall-wasps. Oecologia 71: 254-261.
- Skuhravá, M. 1994. The zoogeography of gall midges (Diptera: Cecidomyiidae) of the Czech Republic I. Evaluation of faunistic researches in the 1855-1990 period. Soc. Zool. Bohemia 57: 211-293.
- Skuhravá, M. & V. Skuhravy. 1994. European gall midges (Diptera: Cecidomyiidae): Their host plants and environment, p.93-95. In W.J. Mattson, Y. Baranchikov & P.W. Price (eds.), Gall-forming insects: Ecology, physiology, and evolution. North Carolina, United States Department of Agriculture, 222p.
- Stone, G.N. & K. Schönrogge. 2003. The adaptive significance of insect gall morphology. TREE 18: 512-522.
- Stone, G.N., K. Schönrogge, R.J. Atkinson, D. Bellido & J. Pujade-Villar. 2002. The population biology of oak gall wasps (Hymenoptera: Cynipidae). Ann. Rev. Entomol 47: 633-668.
- Waring, G.L. & P.W. Price. 1990. Plant water stress and gall formation (Cecidomyiidae: Asphondylia spp.) on creosote bush. Ecol. Entomol 15: 87-95.
- Wheeler, Q.D. 1990. Insect diversity and cladistic constraints. Ann. Entomol. Soc. Am. 83: 1031-1047.
- Willig, M.R., D.M. Kaufman & R.D Stevens. 2003. Latitudinal gradients of Biodiversity: Pattern, process, scale and synthesis. Annu. Rev. Ecol. Evol. Syst. 34: 273-309.
- Wright, M.G. & M.J. Samways. 1996. Gall-insect species richness in African Fynbos and Karoo vegetation: The importance of plant species richness. Biod. Lett. 3: 151-155.
Publication Dates
-
Publication in this collection
26 Sept 2005 -
Date of issue
Aug 2005
History
-
Accepted
30 Mar 2005 -
Received
28 Oct 2003