Abstract
The female of Navicordulia aemulatrix Pinto & Lamas is described and illustrated for the first time based on a single specimen from the same locality of the type series (state of Santa Catarina, [municipality of São Bento do Sul, 26°14'58"S, 49°22'59"W, railroad station] Rio Vermelho, 29.I.1952, in MZSP). In addition, further morphological notes for the male are provided based on three specimens collected at the type locality and at a new locality in the state of Santa Catarina (Timbó municipality). The pronotal process present in N. aemulatrix is re-evaluated and considered non-homologous to that found in Neocordulia setifera (Hagen in Selys) as previously suggested.
Anisoptera; Corduliinae; dragonfly; morphology; taxonomy
SYSTEMATICS, MORPHOLOGY AND PHYSIOLOGY
Description of the female of Navicordulia aemulatrix Pinto & Lamas and additional notes on the male (Odonata: Corduliidae)
AP PintoI, II; CJE LamasI
IMuseu de Zoologia, Univ de São Paulo, São Paulo, SP, Brazil
IIPPG em Ciências Biológicas (Zoologia), Instituto de Biociências, Univ de São Paulo, SP, Brazil; Collaborating researcher at the Lab de Biologia e Sistemática de Odonata, Depto de Entomologia, Museu Nacional, Univ Federal do Rio de Janeiro, Rio de Janeiro, RJ, Brazil
Correspondence Correspondence: Ângelo P Pinto Museu de Zoologia, Univ de São Paulo Av. Nazaré 481, Ipiranga 04263-000, São Paulo, SP, Brasil odonata_angelo@hotmail.com
ABSTRACT
The female of Navicordulia aemulatrix Pinto & Lamas is described and illustrated for the first time based on a single specimen from the same locality of the type series (state of Santa Catarina, [municipality of São Bento do Sul, 26°14'58"S, 49°22'59"W, railroad station] Rio Vermelho, 29.I.1952, in MZSP). In addition, further morphological notes for the male are provided based on three specimens collected at the type locality and at a new locality in the state of Santa Catarina (Timbó municipality). The pronotal process present in N. aemulatrix is re-evaluated and considered non-homologous to that found in Neocordulia setifera (Hagen in Selys) as previously suggested.
Keywords: Anisoptera, Corduliinae, dragonfly, morphology, taxonomy
Introduction
South American representatives of Corduliidae, commonly known as emerald dragonflies, due to their secretive habits are rare in collections, and general aspects on their biology, ecology and behavior are virtually unknown (Geijskes 1970, Machado & Costa 1995, Garrison et al 2006, Pinto & Carvalho 2010). Previous phylogenetic analyses suggested that the group comprises at least three distinct lineages, i.e. Corduliinae sensu stricto, GSI-group (Gomphomacromiinae, Synthemistinae and Idionychinae), and Macromiidae, thus Corduliidae, in its most traditional sense, is considered paraphyletic in relation to Libellulidae (Ware et al 2007, 2009, Bybee et al 2008). In South America, there are nine genera and 44 species of corduliids recognized, with 11 species in Navicordulia Machado & Costa, making it the second largest genus of the continent (Pinto & Lamas 2010).
Species of Navicordulia can be distinguished from all other corduliids occurring in the New World by the presence of an accentuated excavation distal to Hw anal triangle, by a pilose complex on male sternite S7, and by female subgenital plate boat-shaped, projected posteriorly beyond S10, with a supralaminar process projected posteriorly beyond apex of cerci (Machado & Costa 1995, Garrison et al 2006). Furthermore, the presence of a cylindrical tergal process in the prothorax of two species, Navicordulia aemulatrix Pinto & Lamas and Navicordulia errans (Calvert), character apparently shared only with Neocordulia setifera (Hagen in Selys), was also reported by Pinto & Lamas (2010).
Navicordulia aemulatrix was recently described based on three males. The specimens were collected in the locality of a deactivated railway station named Rio Vermelho in the municipality of São Bento do Sul, state of Santa Catarina, southern Brazil (Pinto & Lamas 2010). All of them are from the collection of Richard von Diringshofen, an amateur German-Brazilian entomologist, whose collection was inherited by the Museu de Zoologia da Universidade de São Paulo in the year of 1987 after his death (cf. Costa et al 2000). This large material, which lacks any sort of systematic organization, is still being incorporated into the main collection, so progress on its curatorial work has been very slow. As a result, some months after the manuscript describing N. aemulatrix was accepted for publication, we located four additional specimens, including a female, previously unknown. These specimens are described here, complementing the original description.
Material and Methods
Specimens examined are deposited in the Museu de Zoologia da Universidade de São Paulo - MZSP.
The terminology used here, is the same adopted by Pinto & Lamas (2010). Measurements (in mm) were made with the aid of a stereomicroscope equipped with a camera lucida. Specimens were photographed with a Leica MZ16 stereomicroscope equipped with a Leica DFC420 camera, and source images combined using Auto-montage© software by The Synoptic Group. The following abbreviations were used along the text: Ax = antenodal crossveins; Fw = forewing; Hw = hindwing; LC = lateral abdominal carina; Px = postnodal crossveins; pt = pterostigma; S1-10 = abdominal segments; TC = transverse abdominal carina.
Geographic coordinates not provided in the collection labels were acquired from a digital database (IBGE 2007).
Navicordulia aemulatrixPinto & Lamas(Figs 1a-f, 2)
Material examined
Total 5 ♂ and 1 ♀. Holotype ♂ and 1 ♂ paratype. BRAZIL. State of Santa Catarina, [São Bento do Sul municipality, 26°14'58"S, 49°22'59"W, 858 m a.s.l.], [railway station] Rio Vermelho, II.1952, [Richard von] Dirings[hofen] leg.; additional material with same data as the types, but 1 ♂ and 1 ♀, 29.I.1952; same data, but 1 ♂, 10.II.1952; 1 ♂ [apparently under pre-reproductive period], Timbó municipality [26°49'22.80"S, 49°16'19.20"W, 69 m a.s.l.], 1954. All in MZSP.
Description of female
Head (Fig 1a). Face in anterior view four banded, with alternating dark and pale transverse stripes; covered with dark brown setae, except for yellow setae on labium and free margins of postclypeus. Labium light orange. Labrum orange, with a pair of small dark spots on each side and an irregular brown line on the clypeolabral suture. Clypeus brownish-orange; central portion of postclypeus irregularly spotted with orange. Frons with deep furrow; antefrons orange without metallic reflections, darkening to brownish-orange laterally; postfrons brownish-orange with faint metallic bluish-green reflections above, lateral parts brownish-orange. Vertex brown, with indistinct metallic reflections, dorsal area with a suboval orange spot. Antenna with scapus and pedicel dark brown to black, flagellum brown lighter distally. Occipital triangle brown; rear of head orange with irregular dark brown areas.
Thorax (Fig 1a-b). Pronotum with anterior margin of anterior lobe pale yellow; rest of prothorax color varying from brownish-orange to greenish-brown; suture between middle and posterior lobes with a small cylindrical yellowish-orange process directed dorsally (Fig 1b), similar to that of holotype. Synthorax brown with metallic green reflections; ventral surface, antealar sinus, interalar sclerite and metapostepimeron brown without metallic reflections; entire surface covered with yellow hair-like setae, except antealar sinus with short brown setae. Legs dark brown to black; coxa, postero-ventral surface of prothoracic trochanter, and femur lighter; tarsal claws brown.
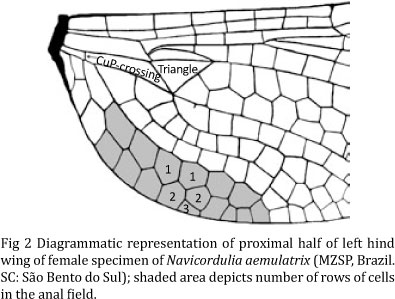
Wings (Figs 1a, 2). Membrane with ca. proximal 0.66 (up to third Px) hyaline, distal 0.33 tinged with an oblique large brown spot covering total width of the wing, center of some cells paler; spot faints distally and becomes hyaline at apex (Fig 1a); veins dark-brown to black, costal triangle pale-brown; pt brown, paler than adjacent veins, slightly trapezoidal, distal side most oblique; membranula brown with a small white spot on extreme antero-distal base. Venation as follows: 8-9 Ax in Fw, 6 Ax in Hw; 5 Px in Fw, 7 Px in Hw; 4 postsubnodals in Fw, 4-5 in Hw; bridge crossvein arising on RP1-2, before subnodus in Fw and under subnodus in Hw, left Hw with an additional bridge crossvein anterior to oblique vein; arc between Ax1-2, but closer to Ax2; not stalked, origin located ca. of basal 0.30 in all wings; RP3-4 not undulate; Rspl distinct, with one cell row of six cells in Fw, 6-7 in Hw; discoidal triangles and supratriangles not crossed in all wings; subtriangles with one crossvein in both Fw; Hw triangle base arising distinctly proximal to arc; space between CuP-crossing and proximal side of discoidal triangle not crossed in all wings (Fig 2); Fw discoidal field convergent, with two rows of cells over its entire length; Hw discoidal field divergent, with four rows of two cells, three of three cells, two of four cells and eight cells in the wing margin; Mspl indistinct in Fw (weakly defined with 3-4 cells), undefined in Hw; anal loop (Fig 2) elongated with distinct midrib, reaching distally near the RP-midfork level; total of cells in anal loop 15 (proximal row seven cells, distal eight); apex of anal loop slightly dilated with two cells, quadrangular; three paranals in Hw; space between anal loop and posterior border of wing with two cell rows and one row with three cells to triangle level (Fig 2); Fw PsA ends near the proximal angle of triangle.
Abdomen (Fig 1a, c-f). Cylindrical, S1 to anterior 0.33 of S3 slightly swollen dorsally; regularly cylindrical posteriorly; S1-2 and S10 without LC. Ground color brown to black; latero-ventral surface of S1-2 and part of S3 orange-brown; dorsal of S2-3 and S4-10 with dull metallic green reflections; ventral carina of S3-8 yellowish-brown; ventro-tergal areas in all segments paler than dorsal, without distinct metallic reflections; sternites dark brown to black. Shape of S8-10 typical of females of the genus; supralaminar process dish-shaped, posterior margin covered by stiff brown setae (Fig 1c, f), width similar to that of subgenital plate in dorsal view; gonapophyses of S9 black, cylindrical, with their surfaces covered by micropunctures (Fig 1e); subgenital plate dark brown to black, projected posteriorly 0.6 mm beyond supralaminar process (Fig 1c-d, f). Epiproct (Fig 1c) dark brown to black; triangular, apex rounded; cerci black, cylindrical, obliquely directed slightly upwards in lateral view (Fig 1c).
Measurements (mm). Total length (incl. subgenital plate) 47.5; abdomen length (excluding subgenital plate) 35.5; maximum width of head 6.6; eyes seam length 0.9; length of Fw 31, Hw 30; width of wing (proximal to costal nodus) 8.5 in Fw, 11 in Hw; length of distance of base-nodus 18 in Fw, 14 in Hw; ratio between base-nodus distance / total length of wing 0.58 in Fw, 0.47 in Hw; pt length 2.7 in Fw, 2.6 in Hw; length of postnodal space (sensu May 1992) 0.49 in Fw, 0.43 in Hw; length of metathoracic femur 7.1; of metathoracic tibia 6.9; length of cercus in lateral view 0.95; of epiproct in lateral view 0.8; length of subgenital plate (vulvar lamina sensu Machado & Costa 1995) in lateral view 2.5; ratio between total length (from S8 posterior margin) of supralaminar process and total length of subgenital plate 0.8.
Remarks
The abdomen is crushed on S2-S4 (Fig 1a), and was broken in three pieces, which we joined using a water soluble glue. Afterwards it was transferred to a cellophane envelope.
Additional notes on the male
The three specimens are very similar to the type-series and only the differences are reported here.
Head. Two specimens with orange color of face fading, obscuring the transverse four-banded pattern observed in the type-series. Metallic reflections on postfrons greenish-blue. Vertex with metallic reflections ill-defined; two specimens with antero-dorsal area between barely developed tubercles regularly brown, without a suboval orange spot.
Thorax. Small cylindrical yellowish process on the suture between middle and posterior lobes of pronotum present in all specimens, but reduced as observed in the paratypes.
Wings. Pt pale brown and membranula grayish-brown (specimen from Timbó). Venation as follows: 4-5 postsubnodals in Fw and Hw, Rspl composed by 5-7 cells in Hw; one specimens from type locality with subtriangle in left Fw not crossed, with a vestige of crossvein, while right Fw has three cells; discoidal triangle in left Hw crossed with two cells (specimen from Timbó).
Abdomen. Ratio of carinated portion / total length of cercus 0.29-0.33, smaller than type-series; one specimen from type locality with ratio between cerci and epiproct total length in lateral view of 0.55, outside the range observed in the type series of 0.58-0.62.
Measurements. Length of Fw 32, Hw 31 (specimen from Timbó); pt length 2.2 in Fw in two specimens, 2.1 in Hw in one specimen from type locality; length of postnodal space (sensu May 1992) 0.47-0.49 in Fw, 0.41-0.45 in Hw; length of metathoracic femur 7.2-7.3; metathoracic tibia 7.0-7.1; maximum width of abdomen (between S7-8) in one specimen from type locality 3.2.
Remarks
The specimen from Timbó represents a new locality record from Santa Catarina and was found fragmented in a big paper envelope with several specimens of Orthemis discolor (Burmeister) and some unidentified Macrothemis. The two specimens from the type locality were located in a drawer holding well-curated papered specimens belonging to many families and different localities. The specimens of N. aemulatrix were identified, likely by Richard von Diringshofen, with "124 Libell[en]"; similar handwriting label was found in the paratype formerly pinned (see Pinto & Lamas 2010). Studying Diringshofen's collection we realize that he maintained the finest preserved specimens of dragonflies in a distinct drawer with some kind of determination and chose one of them to be pinned. Curiously, Diringshofen was able to correctly associate these males to the same species, as we observed for several others specimens of distinct species designated with numbers or another sort of determination by him.
Discussion
Females of Navicordulia are poorly known. From the total of 11 species, just seven have the female described (including the probable Navicordulia nitens De Marmels; see below). Machado & Costa (1995) presented 41 female specimens in their synopsis, of which 82% belong to N. errans and N. leptostyla Machado & Costa, with 19 and 15 specimens each, respectively. Furthermore, Machado & Costa (1995: 197) included N. nitens in their females' key, based in the Venezuelan specimen illustrated by Rácenis (1970: 37, fig 7) as Paracordulia sericea (Selys), which was carefully studied by De Marmels (1983) under the provisional name "Paracordulia sp.1". However, as correctly highlighted by the same authors (Machado & Costa 1995: 188, 195), this specimen must be considered probably as N. nitens in agreement with De Marmels (1991:109) opinion: "...[Rácenis specimen] ist zweifellos eine weibliche Dorocordulia [Navicordulia]....könnte es sich um das Weibchen von D. nitens [N. nitens] oder einer ihr nahestehenden Art handeln". For this reason the actual identity of N. nitens female remains still uncertain and caution should be taken when identifying females when not associated with the corresponding males.
The female of N. aemulatrix keys out between Navicordulia longistyla Machado & Costa and Navicordulia mielkei Machado & Costa using the key provided by Machado & Costa (1995). A modification in couplet 6 will allow separating it from that of the two mentioned species.
6. Supralaminar process spoon-shaped with apex upturned and bearing a tuft of hair-like setae .................. N. longistyla
6'. Supralaminar process cup or dish-shaped with apex almost parallel to abdominal axis, if bearing a tuft of hair-like setae then not spoon-shaped ........................... 7
7. Supralaminar process shaped like a small cup and not provided with a tuft of hair-like setae; ratio between total length (from S8 posterior margin) of supralaminar process and total length of subgenital plate ca 0.66 ..................................................................................................... N. mielkei
7'. Supralaminar process dish-shaped and apex bearing a tuft of hair-like setae; ratio between total length (from S8 posterior margin) of supralaminar process and total length of subgenital plate 0.8 ..................... N. aemulatrix
The dish-like shape of the supralaminar process is undoubtedly similar to that of N. miersi Machado & Costa and N. mielkei (Machado & Costa 1995, figs 29-30). Navicordulia aemulatrix and N. mielkei can be distinguished by the characters in the key. Navicordulia miersi is known only from the female holotype from Joinville municipality, also in the state of Santa Catarina. This specimen is the only one of the genus with an additional crossvein to the CuP-crossing in Hw, and seems to be the unique distinction between these two species. The presence of an additional crossvein in the cubito-anal space may be a reliable parameter for specific distinction in Navicordulia, but the number of crossveins in this sector is particularly variable in other dragonfly species, such as in the Libellulidae genus Uracis Rambur (Costa & Santos 1997). Furthermore, it is relatively common to find additional crossveins in unexpected sectors of the wings, for example a pinned male specimen of Dasythemis mincki mincki Karsch in the Museu de Ciências e Tecnologia da PUCRS - MCTP has both Hw with one additional crossvein between CuP-crossing and proximal side of discoidal triangle, an uncommon feature among species of this genus. Probably, the number of cell rows in the anal field represents a more consistent character, since N. aemulatrix has two rows of cells and just one row with three cells (Fig 2), while N. miersi has three rows of cells for a distance of 3-4 cells. In addition, N. miersi has supralaminar process wider than subgenital plate in dorsal view (supralaminar process with same width of subgenital plate in N. aemulatrix) and ratio between total length of supralaminar process and total length of subgenital plate 0.66 (0.8 in N. aemulatrix). However, due the fact that both females are known from only one specimen, it is not possible to securely separate these two species without direct comparison.
The knowledge of females of Atlantic Forest species of Navicordulia is still far to be satisfactory. Considering the five species (N. aemulatrix, N. atlantica Machado & Costa, N. kiautai Machado & Costa, N. mielkei and N. miersi), three of them have the females described (N. miersi, N. mielkei, and now N. aemulatrix). The fact that N. miersi is known only by its female holotype turns the situation worse, since N. atlantica, described from only one male collected in the same locality (municipality of Joinville, state of Santa Catarina, Brazil), are badly preserved, with some parts lost, including the anal area of Hw (Machado & Costa 1995: 205), precluding the observation of the presence of an additional crossvein to CuP-crossing. Odonatologists have avoided describing new species based in female specimens, as females, in general, are less informative for species recognition. Indeed, in some cases, females are virtually undistinguishable, such as some species of the New World Calopterygidae (Garrison 2006).
Based in venational features and vesica spermalis morphology, Navicordulia most probably belongs to Corduliidae sensu stricto (cf. Ware et al 2007). Pfau (2005) considered p-ps sclerite, which is present in the vesica spermalis of Navicordulia, as characteristic of the primitive corduloid glans mechanism. This sclerite is reduced or completely absent in the GSI-group and Libellulidae species. The long subgenital plate found in all species of Navicordulia is uncommon in 'Corduliidae', as stated by Machado & Costa (1995). These authors discussed the probable relationship with other 'Corduliidae' taxa such as Gomphomacromia Brauer (Andean), Somatochlora Selys (Holarctic) and Synthemistinae genera (Australasian). However, only Somatochlora (Corduliidae s.s.) is likely to be closely related to Navicordulia. Furthermore, the subgenital plate of the Australasian genus Metaphya Laidlaw possess a great similarity to that of Navicordulia (cf. Campion 1921: 66, Laidlaw 1913: 66, plate IV, Theischinger & Hawking 2006: 242) and investigations are required to establish what is the phylogenetic relationship between these two genera.
In general Navicordulia can be considered highland forest-dependent dragonflies. The administrative center of Timbó is located in a valley at low altitude (69 m a.s.l.); however, based in the locality of the other specimens, we believe that it was probably collected in the adjacent mountains, with elevations little higher than 650 m a.s.l. The same interpretation could be applied to the known localities of N. mielkei and N. miersi, known from Joinville, whose administrative center is located at maximum 100 m a.s.l., but there are adjacent forested mountains with more than 900 m a.s.l.
The presence of a developed pronotal process in the female of N. aemulatrix (Fig 1b), similar to the one presented by its males (cf. Pinto & Lamas 2010), allowed us to reevaluate this structure. In the complete series of seven specimens only the holotype and the female have a fully developed process, and it is less conspicuous in the remaining specimens. Although Pinto & Lamas (2010) stated the possibility of N. setifera also presenting this structure, direct comparison revealed that it probably should not be considered homologous to the one of Navicordulia, which seems to be exclusive. In order to test this hypothesis, examination of a large sample of species and even of other 'corduliid' genera will be necessary.
Acknowledgments
We thank Sônia A. Casari (MZSP curator of Aquatic Insects collection), Dr. Alcimar L. Carvalho (MNRJ, Brazil) for allowing APP to use his laboratory equipment, personal odonatological library, and for reviewing the manuscript; Dr. Angelo B. M. Machado (UFMG, Brazil) for sending all the reprints of his Corduliidae papers and for sharing his knowledge on Odonata systematics. This study was partially supported by Brazilian government agencies, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, doctoral fellowship to the first author, CAPES PROAP-2011), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, fellowship to second author) and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ process numbers E-26/170.644/2004 and E-26/171.281/2006).
Received 17 May 2011 and accepted 30 July 2011
Edited by Roberto A Zucchi - ESALQ/USP
- Bybee SM, Ogden TH, Branham MA, Whiting MF (2008) Molecules, morphology and fossils: a comprehensive approach to odonate phylogeny and the evolution of the odonate wing. Cladistics 23: 1-38.
- Campion H (1921) Odonata collected in New Caledonia by the late Mr. Paul D. Monugue. Ann mag nat hist 8, series 9: 33-67, pls. VIII-IX.
- Costa C, Ide S, Rosado Neto GH, Galileo MHM, Fonseca CRV, Valente RM, Monné MA (2000) Diagnóstico del conocimiento de las principales colecciones brasileñas de Coleoptera, p.115-136. In Martín-Piera F, Morrone JJ, Melic A (eds) Hacia un proyecto CYTED para el inventario y estimación de la diversidad entomológica en Iberoamérica: PrIBES-2000. vol. 1, Zaragoza, Monografías Tercer Milenio, 326p.
- Costa JM, Santos TC (1997) Intra- and interspecific variation in the genus Uracis Rambur, 1842, with a key to the known species (Anisoptera: Libellulidae). Odonatologica 26: 1-7.
- De Marmels J (1983) The Odonata of the region of mount Auyantepui and the Sierra de Lema, in Venezuelan Guyana. 3. Additions to the families Gomphidae, Aeshnidae and Corduliidae, with description of Progomphus racenisi spec. nov. Odonatologica 12: 1-13.
- De Marmels J (1991) Dorocordulia nitens sp. n., eine neue Smaragdlibelle aus Venezuela (Odonata: Corduliidae). Mitteilungen der Entomologischen Gesellschaft Basel 41: 106-111.
- Garrison RW (2006) A synopsis of the genera Mnesarete Cowley, Bryoplathanon gen.nov., and Ormenoplebia gen. nov. (Odonata: Calopterygidae). Contrib Sci 506: 1-84.
- Garrison RW, von Ellenrieder N, Louton JA (2006) Dragonfly genera of the New World: an illustrated and annotated key to the Anisoptera. Baltimore, The Johns Hopkins University Press, 368p.
- Geijskes DC (1970) Generic characters of the South American Corduliidae, with descriptions of the species found in the Guyanas. Uitg Natuurwet Studiekring Suriname Ned Antillen 60 [Studies on the Fauna of Suriname and other Guianas 44]: 1-42, pls. I-II.
- Instituto Brasileiro de Geografia e Estatística - IBGE (2007) Malha municipal digital do Brasil - 2005. Available at: http://www.ibge.gov.br/servidor_arquivos_geo/ (accessed in 10 October 2007).
- Laidlaw FF (1913) Contributions to a study of the Dragonfly fauna of Borneo - Part I. The Corduliinae: the genus Amphicnemis: the legion Protoneura Proc Zool Soc Lond: 63-80, pl. IV
- Machado ABM, Costa JM (1995) Navicordulia gen. nov. a new genus of Neotropical Corduliidae, with descriptions of seven new species (Anisoptera: Corduliidae). Odonatologica 24: 187-218.
- May M (1991) [distribution date 1992]. A review of the genus Neocordulia, with a description of Mesocordulia subgen. nov. and of Neocordulia griphus spec. nov. from Central America, and a note on Lauromacromia (Odonata: Corduliidae). Folia Entomol Mex 82: 17-67.
- Pfau HK (2005) Structure, function and evolution of the 'glans' of the anisopteran vesica spermalis (Odonata). Int J Odonatol 8: 259-310.
- Pinto AP, Carvalho AL (2010) A new species of Lauromacromia (Odonata: Corduliidae) from Southeastern Brazil, with a cladistic analysis of the genus and comments on Neotropical dragonfly biogeography. Zootaxa 2425: 45-68.
- Pinto AP, Lamas CJE (2010) Navicordulia aemulatrix sp. nov. (Odonata: Corduliidae) from northeastern Santa Catarina State, Brazil. Rev Bras Entomol 54: 608-617.
- Rácenis J (1970) Los odonatos de la región del Auyantepui y de La Sierra de Lema, en La Guayana Venezolana. 2. Las familias Gomphidae, Aeshnidae y Cordulildae. Acta Biol Venez 7: 23-29.
- Theischinger G, Hawking J (2006) The complete field guide to dragonflies of Australia. Collingwood, CSIRO Publishing, 374p.
- Ware JL, May M, Kjer K (2007) Phylogeny of the higher Libelluloidea (Anisoptera: Odonata): An exploration of the most speciose superfamily of dragonflies. Mol Phylogenet Evol 45: 289-310.
- Ware JL, Simaika JP, Samways MJ (2009) Biogeography and divergence time estimation of the relict Cape dragonfly genus Syncordulia: global significance and implications for conservation. Zootaxa 2216: 22-36.
Publication Dates
-
Publication in this collection
06 Feb 2012 -
Date of issue
Dec 2011
History
-
Received
17 May 2011 -
Accepted
30 July 2011