Abstracts
ABSTRACT Most information on catfishes of the genus Aspidoras has focused species kept in aquaria, in non-natural conditions. Biotic and abiotic parameters of environment inhabited by Aspidoras fuscoguttatus, associated ichthyofauna, as well as strategies involved in the successful occupation of different microhabitats in a first order stream, located in São José do Rio Preto, São Paulo State, were investigated. Data collection was monthly performed at three segments (headwaters, middle course, and mouth) from August 1999 to July 2000. The greatest values of water temperature, conductivity, turbidity, alkalinity, width, depth, and current velocity were obtained in the rainy season. Dissolved oxygen levels showed significant differences among three segments in all sampling months, except for September. Substrate composition was predominantly sandy, followed by silt and clay in middle course and mouth in rainy as much as in dry seasons. In headwaters a large increase of the sandy fraction occurred in the rainy season. In the marginal and aquatic vegetation 30 species of macrophytes and 5 of macroalgae were identified. The fauna included tadpoles of Anura and macroinvertebrates (Mollusca, Crustacea, Arachnida, and 8 orders of Insecta). A. fuscoguttatus was found throughout the entire stream in sympatry with 6 fish species. In middle course, cluster analysis revealed an association between A. fuscoguttatus immatures and low depth, suggesting a probable function of depth in that stage of ontogenetic development of catfishes. The results indicate that A. fuscoguttatus present plasticity in terms of microhabitat, including air breathing behavior in hypoxic conditions, previously unknown in genus Aspidoras.
ecology; stream; microhabitat; Aspidoras fuscoguttatus; Callichthyidae
A maioria das informações disponíveis sobre cascudos do gênero Aspidoras é proveniente de observações em aquário. No presente estudo, foram investigados aspectos da ecologia de Aspidoras fuscoguttatus em um riacho de primeira ordem na região de São José do Rio Preto, SP, incluindo caracterização do ambiente, levantamento da ictiofauna associada e análise de estratégias utilizadas na ocupação dos diferentes microhabitats do riacho. Os dados foram coletados mensalmente em três pontos (nascente, curso médio e desembocadura) no período de agosto de 1999 a julho de 2000. Os maiores valores de temperatura, condutividade elétrica, turbidez, alcalinidade, largura, profundidade e velocidade da água foram obtidos na estação chuvosa. As concentrações de oxigênio dissolvido apresentaram diferenças significativas entre os três pontos e em todos os meses amostrados, exceto setembro. Na composição do substrato, houve predomínio de areia, silte e argila no curso médio e na desembocadura, tanto na estação chuvosa como na seca. Na nascente ocorreu considerável aumento da fração arenosa no período chuvoso. Foram identificadas na vegetação marginal e aquática 30 espécies de macrófitas e 5 de algas. Quanto à fauna, foram encontrados girinos de Anura e invertebrados dos grupos Mollusca, Crustacea, Arachnida e 8 ordens de Insecta. A. fuscoguttatus foi encontrado em todo o riacho em simpatria com 6 espécies de peixes. No curso médio, a análise de agrupamentos revelou associação entre imaturos de A. fuscoguttatus e baixa profundidade, evidenciando provável função da profundidade nessa fase do desenvolvimento ontogenético. Os resultados indicam que essa espécie apresenta plasticidade em termos de microhabitat, incluindo comportamento de respiração aérea em condições de hipóxia, inédito no gênero Aspidoras.
ecologia; riacho; microhabitat; Aspidoras fuscoguttatus; Callichthyidae
Ecology of a stream from upper Paraná River basin inhabited by Aspidoras fuscoguttatus Nijssen & Isbrüker, 1976 (Siluriformes, Callichthyidae)
Ecologia de um riacho da bacia do alto rio Paraná habitado por Aspidoras fuscoguttatus Nijssen & Isbrücker, 1976 (Siluriformes, Callichthyidae)
Araujo, R. B. deI; Garutti, V.I,II
ICentro de Aqüicultura da UNESP (CAUNESP), Jaboticabal, SP, Brazil
IIDepto. de Zoologia e Botânica, IBILCE/UNESP, São José do Rio Preto, SP, Brazil
Correspondence Correspondence to Renato Braz de Araujo Rua Joana Anderi Chalela, 335 CEP 15030-650, São José do Rio Preto, SP, Brazil e-mail: renatoaraujo@zipmail.com.br
ABSTRACT
Most information on catfishes of the genus Aspidoras has focused species kept in aquaria, in non-natural conditions. Biotic and abiotic parameters of environment inhabited by Aspidoras fuscoguttatus, associated ichthyofauna, as well as strategies involved in the successful occupation of different microhabitats in a first order stream, located in São José do Rio Preto, São Paulo State, were investigated. Data collection was monthly performed at three segments (headwaters, middle course, and mouth) from August 1999 to July 2000. The greatest values of water temperature, conductivity, turbidity, alkalinity, width, depth, and current velocity were obtained in the rainy season. Dissolved oxygen levels showed significant differences among three segments in all sampling months, except for September. Substrate composition was predominantly sandy, followed by silt and clay in middle course and mouth in rainy as much as in dry seasons. In headwaters a large increase of the sandy fraction occurred in the rainy season. In the marginal and aquatic vegetation 30 species of macrophytes and 5 of macroalgae were identified. The fauna included tadpoles of Anura and macroinvertebrates (Mollusca, Crustacea, Arachnida, and 8 orders of Insecta). A. fuscoguttatus was found throughout the entire stream in sympatry with 6 fish species. In middle course, cluster analysis revealed an association between A. fuscoguttatus immatures and low depth, suggesting a probable function of depth in that stage of ontogenetic development of catfishes. The results indicate that A. fuscoguttatus present plasticity in terms of microhabitat, including air breathing behavior in hypoxic conditions, previously unknown in genus Aspidoras.
Key words: ecology, stream, microhabitat, Aspidoras fuscoguttatus, Callichthyidae.
RESUMO
A maioria das informações disponíveis sobre cascudos do gênero Aspidoras é proveniente de observações em aquário. No presente estudo, foram investigados aspectos da ecologia de Aspidoras fuscoguttatus em um riacho de primeira ordem na região de São José do Rio Preto, SP, incluindo caracterização do ambiente, levantamento da ictiofauna associada e análise de estratégias utilizadas na ocupação dos diferentes microhabitats do riacho. Os dados foram coletados mensalmente em três pontos (nascente, curso médio e desembocadura) no período de agosto de 1999 a julho de 2000. Os maiores valores de temperatura, condutividade elétrica, turbidez, alcalinidade, largura, profundidade e velocidade da água foram obtidos na estação chuvosa. As concentrações de oxigênio dissolvido apresentaram diferenças significativas entre os três pontos e em todos os meses amostrados, exceto setembro. Na composição do substrato, houve predomínio de areia, silte e argila no curso médio e na desembocadura, tanto na estação chuvosa como na seca. Na nascente ocorreu considerável aumento da fração arenosa no período chuvoso. Foram identificadas na vegetação marginal e aquática 30 espécies de macrófitas e 5 de algas. Quanto à fauna, foram encontrados girinos de Anura e invertebrados dos grupos Mollusca, Crustacea, Arachnida e 8 ordens de Insecta. A. fuscoguttatus foi encontrado em todo o riacho em simpatria com 6 espécies de peixes. No curso médio, a análise de agrupamentos revelou associação entre imaturos de A. fuscoguttatus e baixa profundidade, evidenciando provável função da profundidade nessa fase do desenvolvimento ontogenético. Os resultados indicam que essa espécie apresenta plasticidade em termos de microhabitat, incluindo comportamento de respiração aérea em condições de hipóxia, inédito no gênero Aspidoras.
Palavras-chave: ecologia, riacho, microhabitat, Aspidoras fuscoguttatus, Callichthyidae.
INTRODUCTION
The Neotropical fish fauna contains the largest number of species in the world. However, knowledge about the ecology and systematics of its fish communities is relatively incomplete and geographically restricted (Böhlke et al., 1978; Lowe-McConnell, 1999). The main groups of Neotropical freshwater fishes include Characiformes, Siluriformes, Gymnotiformes, Cyprinodontiformes and Cichlidae (Britski et al., 1999). Siluriformes constitutes one of the dominant groups, with 15 families (Nelson, 1994). One of them, the Callichthyidae comprehends small armored South American catfishes characterized by the presence of two lateral and longitudinal series of body plates, three pairs of barbels and anterior mouth. This family has approximately 165 species, 8 genera and 2 subfamilies: Callichthyinae (Callichthys, Dianema, Hoplosternum, Lepthoplosternum and Megalechis) and Corydoradinae (Aspidoras, Corydoras and Brochis) (Reis, 1998).
In Callichthyinae some biological aspects have been investigated as feeding (Saul, 1974) in Callichthys callichthys (Linnaeus, 1758); reproduction (Ramnarine, 1995), behavior (Andrade & Abe, 1997) and ontogenetical development (Machado-Allison, 1986) in Hoplosternum littorale (Hancock, 1828), and ecology (Mol, 1996) in H. thoracatum (Valenciennes, 1840). In Corydoradinae few studies were made regarding spatial occupation, feeding and reproductive period (Aranha et al., 1993) in Corydoras aeneus (Gill, 1858) and Corydoras gr. carlae Isbrücker, 1983, and populational dynamics (Bervian & Fontoura, 1994) and reproductive behavior (Pruzsinszky & Ladich, 1998) in C. paleatus (Jenyns, 1842).
Most information available on catfishes of the genus Aspidoras are restricted to the species kept in aquaria, in non-natural conditions (Burgess, 1989). According to Britto (2000), almost all 17 Aspidoras species occur in limited areas of some higher Brazilian river drainages, suggesting strong endemism (Nijssen & Isbrücker, 1976). Ecological studies on this genus, in natural environment, are scarce in Brazil. The only contribution dealing with Aspidoras fuscoguttatus Nijssen & Isbrücker, 1976 is that of Garutti (1988), analysing the longitudinal distribution of fish fauna in a stream from northwestern region of São Paulo State. Furthermore, there are still relatively few papers containing an ecological characterization of headwaters and streams from plateau areas inhabited primarily by small size fish species as A. fuscoguttatus, including main aquatic biotic and abiotic parameters.
Keeping this in mind, the aims of this work were: 1) to describe the aquatic environment occupied by A. fuscoguttatus (Fig. 1) focusing its main biotic and abiotic characteristics; 2) to make a survey of the fish species that occur in that environment; and 3) to investigate the possible strategies involved in the successful occupation by A. fuscoguttatus of different microhabitats in a small stream.
MATERIAL AND METHODS
Study area
The field work was carried out in a nameless stream situated in Engenheiro Schmidt, a district of São José do Rio Preto, São Paulo State, 20o52'06''S-49o16'40''W. It is a first order stream according to Strahler (1957), typical of plateau region, draining into a larger stream (Cedro Stream), which in turn is a tributary of Preto River, draining into the Turvo River. The Turvo River basin is part of the large Grande River drainage basin, which belongs to the Paraná River basin.
The regional topography is gentle and the relief is undulated and relatively uniform. Total slope is relatively small, the highest altitude reaching 550 m and the lowest 350 m. The soils are sandy and result from rock decomposition of Bauru Formation, with predominance of the sandy portion and a lower contribution of the finer (silt-clay) portion (Arid et al., 1970).
The original vegetation in the region of São José do Rio Preto is almost entirely restricted to the semi-deciduous marginal forests along the rivers or stream courses. A recent survey made by the Secretaria de Agricultura, São Paulo State, showed the following proportions of the land use in that region: 63% pastures, 6.4% annual cultured areas, 20% permanent cultured areas, 8% forests, and 2.6% unused or reforested areas.
The regional climate is the warm-moist tropical type (Arid et al., 1970). The mean annual temperatures are always higher than 25ºC, whereas the mean of the coolest months (June and July) is about 20ºC, and of the warmest months (January and February) is about 30ºC. Annual rainfall range from 1100 to 1250 mm, with a high concentration (85%) from October to March, the rainy season, and a low concentration (15%) from April to September, the dry season (Arid & Barcha, 1973).
Sample collection
All material was monthly sampled at three stream segments (headwaters, middle course, and mouth) from August 1999 to July 2000, except for January. Sampling was performed in the morning, between 7:10 and 11:40 am, in order to avoid diel variation of water physical and chemical parameters. Preliminary observations and collecting were undertaken from February to July 1999. Temperature was measured directly in the water at a depth of 10 cm using a mercury thermometer (10ºC to 110ºC). Specific conductivity was determined with a Crison field conductivimeter, model 524. Water samples were collected and transported to the laboratory for measurement of pH, turbidity, alkalinity, and dissolved oxygen.
Measurements of pH were determined with a Mettler Toledo digital pH-meter, model MP 220. Dissolved oxygen (mg L1) was determined by Winkler method (Golterman et al., 1978). Alkalinity (mg L1) was analysed by potentiometric titration according to Golterman et al. (1978). Turbidity (FTU) was measured with a Micronal turbidimeter, model B 250. All laboratory analyses for chemical and physical variables were performed on the same collection day with only one measurement, except for dissolved oxygen which was measured in three repetitions. Rainfall data were obtained from Escritório de Desenvolvimento Rural, Coordenadoria de Assistência Técnica Integral, São Paulo State, situated in Cedral, a town at a distance of 4 km from the sampling site.
Fishes were collected with sieve (0.3 x 0.3 mm), casting net (0.9 x 0.9 mm), hand net (0.5 x 0.5 mm) and fish line. Vascular plants (terrestrial and aquatic macrophytes), macroalgae and substrate were sampled manually and with the aid of a small spade.
The fishes were fixed in 10% formaldehyde solution and after 48 hours transferred to 70% ethanol solution for permanent storage. Algae were preserved in 4% buffered formaldehyde. In the laboratory, total length (TL) was taken with a calliper. Voucher specimens were deposited in the fish collection of Departamento de Zoologia e Botânica, UNESP, São José do Rio Preto (DZSJRP 4414 to 4434 and 4436 to 4457).
For the microhabitat characterization, width and depth (measured with tape-measure), current velocity (estimated by the method of dislocation of a floating object), marginal and aquatic vegetation, and associated fauna (tadpoles and macroinvertebrates) were recorded. Two substrate samples, collected in May 1999 (dry season) and February 2000 (rain season), were analysed according to particle size classes by Allan (1995).
Statistical analysis
Differences in means for values of dissolved oxygen among the stream segments (headwaters, middle course and mouth) were tested by one-way analysis of variance (ANOVA) with subsequent mean separation by Tukey's test with a 0.05 significance level (Sokal & Rohlf, 1981). The value relative to January 2000 (not sampled) was estimated by the mean between anterior and posterior months. The analyses were performed with the Minitab statistical software (1996). Due to constant occurrence of some variables (biotic and abiotic) only in middle course along the study period, associations among these variables and sampling months were assessed by cluster analysis using Jaccard's similarity coefficient and unweighted group average method (UPGMA) from the Fitopac 1 software (Shepherd, 1995).
RESULTS
Stream characterization
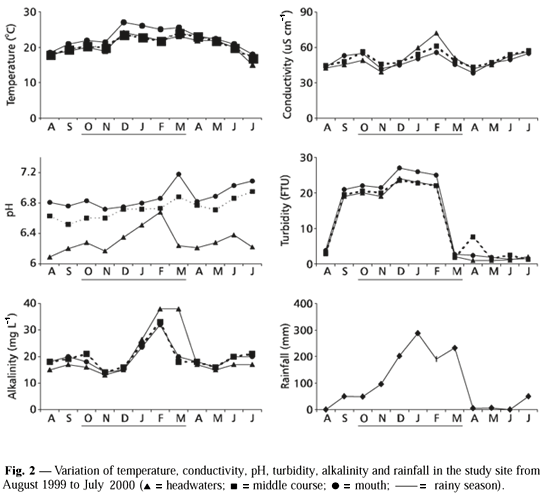
Seasonal fluctuations in temperature, conductivity, pH, turbidity, alkalinity and rainfall in three stream segments are given in Fig. 2. Temperature ranged from 15ºC in headwaters (winter) to 27ºC in mouth (summer). The lowest temperatures were recorded from July to August, while the highest values were observed from December to March. Conductivity was higher in February (rainy season) and lower in April (early dry season). pH showed a narrow variation around neutral in middle course and in mouth and lightly acid in headwaters. Turbidity demonstrated a remarkable seasonality with lower values during the dry period and higher in the rainy period. Alkalinity revealed maximum values in February and minimum from August to December and from April to July. Rainfall data presented high concentration (913.8 mm) from December to March (rainy season) and low (60.9 mm) from April to July (dry season).
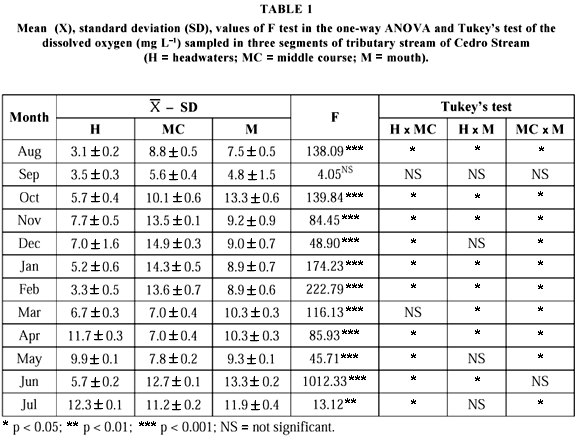
The lowest dissolved oxygen concentrations were observed from August to March in headwaters, while the highest were found from October to February in middle course. ANOVA indicated significant differences (p < 0.001) for oxygen among the three stream segments in all sampling months, except for September. Tukey's test revealed significant differences (p < 0.05) between headwaters and middle course throughout the study period, except for September and March. Between headwaters and mouth, there were no significant differences in September, December, May and July, while between middle course and mouth, the differences were not significant only in September and June (Table 1).
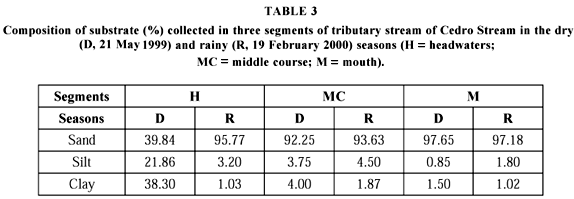
Environmental parameters for microhabitat characterization of each stream segment sampled are summarized in Table 2. Substrate composition in middle course and mouth during both seasons was predominantly sandy, followed by silt and clay, while in headwaters a considerable change in composition from dry to rainy season was verified (Table 3).
Marginal vegetation in headwaters and middle course showed open areas dominated by herbaceous plants as Poaceae, Cyperaceae, Zingiberaceae, Scrophulariaceae, Asteraceae, Polygonaceae, Lamiaceae, Begoniaceae, Leguminosae, Lithraceae and Commelinaceae, while near to mouth, shrubby-arboreous shaded by riparian canopy were noted as Melastomataceae, Boraginaceae, Hydrophyllaceae, Leguminosae and Myrtaceae. Aquatic vegetation in headwaters and middle course included filamentous green algae (Spyrogira sp. 1, Spyrogira sp. 2, Oedogonium sp., Microspora floccosa) and blue algae (Phormidium cf. retzii), macrophytes as Pontederiaceae (Heteranthera reniformis) and Onagraceae (Ludwigia aff. suffruticosa, L. caparosa, L. octovalvis, L. elegans). Adjacent vegetation throughout the entire stream was represented mainly by Poaceae.
Besides fishes, tadpoles of Hyla albopunctata and macroinvertebrates as Mollusca (Planorbidae), Crustacea (Macrobrachium sp.), Arachnida (Araneae) and 8 orders of Insecta including Odonata (larvae), Coleoptera (adult), Heteroptera (adult), Diptera (larvae), Trichoptera (larvae), Hymenoptera (adult), Lepidoptera (adult), and Phasmodea (adult) were also registered.
Ichthyofauna
A total of 661 fish specimens, belonging to 6 families, 7 genera and 7 species were collected at headwaters, middle course, and mouth of the tributary stream of Cedro Stream (Table 4) of which 651 (98.5%) were sampled with sieve and 10 (1.5%) with hand net, casting net and fish line.
Among the species reported, Aspidoras fuscoguttatus and Gymnotus carapo occurred in the three stream segments. The species with the greatest number of collected individuals were A. fuscoguttatus with 544 specimens (82.3%), P. reticulata with 54 (8.2%) and G. carapo with 42 (6.4%). The spatial distribution of the fish species showed a remarkable vertical stratification. Among the 7 species sampled, immatures and adults of A. fuscoguttatus were found in shallow (0.04 m) and deep (0.23 m) stretches, respectively. P. reticulata explored the water surface. G. carapo was captured in deep and shadow pools, among the submerged branches and roots of the marginal vegetation. H. ancistroides was found in the bottom of pools. S. marmoratus was captured in the macrophyte roots (Ludwigia caparosa). O. pintoi and A. altiparanae were encountered swimming actively at mid-water of slow flowing areas.
Cluster analysis of biotic and abiotic variables revealed four groupings in the middle course of stream (Fig. 3). Group 1 includes two variables (A. fuscoguttatus immatures and low depth) (87%) not associated only in October and co-occurring in seven months (August, September, March to July). Group 2 contains two aquatic organisms (Heteranthera reniformis and Planorbidae) (83%) co-existing in ten months (September, November to July). Group 3 presents three variables (tadpoles of H. albopunctata, A. fuscoguttatus and low depth) (71%) registered in five months (September, April to July). Group 4 comprehends four variables represented by Clorophyceae, H. albopunctata, A. fuscoguttatus and low depth (58%) found in only four common months (April to July).
DISCUSSION
During the study period, the stream occupied by A. fuscoguttatus showed a remarkable seasonality, with the highest values of water temperature, conductivity, turbidity, alkalinity, width, depth and current velocity obtained in the rainy season. Substrate composition presented a notable alteration in headwaters probably due to stream dumping provoked by the high amount of rain in February (190.9 mm). The influence of these factors in water quality associated with the changes in aquatic and marginal vegetation and fauna accentuates the importance of the microhabitats formed among the aquatic macrophytes after the rainfall. Considering the representatives of fauna hereby recorded, such microhabitats may be contributing as refuge, source of food, protection against predators and site of reproduction for aquatic organisms.
The highest percentage of fishes sampled with sieves was similar to that of other works. Pavanelli & Caramaschi (1997), studying the composition of the ichthyofauna of two streams (also in the Paraná basin), captured more specimens (85%) with sieves. Uieda (1984) investigating the seasonal, spatial and temporal distribution of fish fauna in a stream in the same basin also had more success with the use of sieves.
In the present research, the low depth, small size of specimens and abundant aquatic macrophytes, especially during the rainy season, probably contributed to achieve satisfactory results with sieves, because the efficiency of sampling methods depends on habitat features and biology of species (Uieda & Uieda, 2001). The Neotropical ichthyofauna presents the most diversified and richest freshwater ichthyofauna of the world (Böhlke et al., 1978). However, most studies focus mainly large rivers and more commercially important species, while the small size fishes (mostly less than 12 cm in standard length), usually inhabiting streams and shallow waters at the side of rivers, are restricted to few investigations (Lowe-McConnell, 1999).
Garutti (1988) studying the ichthyofauna of Barra Funda Stream, Preto River basin (approximately 14 km of length), recorded the same species found in the present investigation, including A. fuscoguttatus, found in this study in all sampling months and in the three stream segments. Garutti (op. cit.) also verified that the main characteristic in the longitudinal succession was the addition of species, progressing from the headwaters toward the mouth. Besides size differences, addition of species was also evidenced in this work, with 2 species occurring in headwaters (A. fuscoguttatus and G. carapo), 5 in middle course (A. fuscoguttatus, G. carapo, H. ancistroides, P. reticulata, S. marmoratus), and 6 in mouth (A. fuscoguttatus, G. carapo, H. ancistroides, P. reticulata, O. pintoi, A. altiparanae).
Cluster analysis showed an ecological group of A. fuscoguttatus immatures and low depth in the middle course indicating a probable function of this abiotic parameter in that stage of ontogenetic development. The association between the macrophyte Heteranthera reniformis (Pontederiaceae) and the mollusc Planorbidae also showed constancy along the study period. According to Correa-Soares (1985), H. reniformis constitutes source of food for Biomphalaria tenagophila (Planorbidae) during all months of the year.
The fish species composition encountered in the tributary stream of Cedro Stream indicates that in this stream some species are successful as A. fuscoguttatus, which demonstrated a plasticity in terms of microhabitat, pH and dissolved oxygen. In headwaters, specimens were captured in pools or among macrophytes roots (Brachiaria sp.) throughout sampling months; in middle course, immatures were collected in low depth and in the macrophyte Heteranthera reniformis, while adults were found in deep and shallow stretches, and in mouth, the catfishes were sampled in pools or riffles. Such plasticity can be reinforced by a preliminary sampling performed in February 1999, when it was observed an air breathing behavior for this species in headwaters with a dissolved oxygen concentration of 1.0 mg L1. Some representatives of the subfamily Callichthyinae exhibit air breathing as Hoplosternum thoracatum (Gee & Graham, 1978), H. littorale and Callichthys callichthys (Carter & Beadle, 1931), and Megalechis personata (Mol et al., 1999). In Corydoradinae there are evidences only for Brochis splendens (Gee & Graham, 1978) and Corydoras aeneus (Kramer & McClure, 1980). Therefore, based on indicators above mentioned, there is a hypothesis that must be tested aiming to know if A. fuscoguttatus is the first recorded species of the genus with air breathing as an adaptation to hypoxic waters.
Pronounced differences among dissolved oxygen concentrations along the study period were found. The lowest oxygen levels were obtained in headwaters as verified by Necchi (1992) also in the region of São José do Rio Preto. Besides the characteristics inherent to each stream segment, the variation in the amount of oxygen in the three sampling sites suggests adaptative strategies that enable A. fuscoguttatus to inhabit the whole stream with its distinct microhabitats. Thus, considering that some Aspidoras species are very appreciated in aquaria as A. fuscoguttatus (pers. obs.), A. pauciradiatus and A. menezesi (Pinkerton, 1987), A. raimundi (Schindler & Staeck, 1996), ecological data obtained in natural environment, as did in this study, can contribute to the production of these ornamental fishes in artificial conditions, reducing the irregular and predatory extraction made in the Brazilian hydrographic basins.
Acknowledgments This research was supported by a CAPES personal grant to the first author. We are grateful to Antonio F. M. Camargo, Virgínia S. Uieda, Dorotéia R. S. Souza, and Francisco Langeani, for their criticism and valuable suggestions; to Neusa T. Ranga for vascular plants identification; to Orlando Necchi Júnior and Luiz H. Z. Branco for macroalgae identification; to Antonio J. Manzato for statistical orientation; to Andréia A. Rezende for help in the use of Fitopac software; to Nilson D. Menezes for granulometric analysis of substrate samples; to Henrique F. Chaves and Roselene S. C. Ferreira for help in field and laboratory work; to Patrícia R. Delboni for help in the preparation of figures, and to Carlos Daghlian for helping to improve the english text.
Received January 14, 2002
Accepted May 16, 2002
Distributed August 31, 2003
- ALLAN, J. D., 1995, Stream ecology: structure and function of running waters Chapman & Hall, London, 388p.
- ANDRADE, D. V. & ABE, A. S., 1997, Foam nest production in the armoured catfish. J. Fish Biol., 50: 665-667.
- ARANHA, J. M. R., CARAMASCHI, E. P. & CARAMASCHI, U., 1993, Ocupação espacial, alimentação e época reprodutiva de duas espécies de Corydoras Lacépede (Siluroidei, Callichthyidae) coexistentes no rio Alambari (Botucatu, São Paulo). Rev. Bras. Zool., 10: 453-466.
- ARID, F. M. & BARCHA, S. F., 1973, Água subterrânea na formação Bauru: região norte-ocidental do Estado de São Paulo. Bolm. Ciênc., 1: 70-101.
- ARID, F. M., CASTRO, P. R. M. & BARCHA, S. F., 1970, Estudos hidrogeológicos no município de São José do Rio Preto, SP. Bol. Soc. Brasil. Geol., 19: 43-69.
- BERVIAN, G. & FONTOURA, N. F., 1994, Dinâmica populacional da ictiofauna da Lagoa Fortaleza, Cidreira, Rio Grande do Sul. III. Corydoras paleatus (Jenyns, 1842) (Teleostei, Callichthyidae). Biociências, 2: 15-23.
- BÖHLKE, J. E., WEITZMAN, S. H. & MENEZES, N. A., 1978, Estado atual da sistemática dos peixes de água doce da América do Sul. Acta Amazonica, 8: 657-677.
- BRITSKI, H. A., SILIMON, K. Z. & LOPES, B. S., 1999, Peixes do Pantanal: manual de identificação Embrapa-SPI, Brasília/Embrapa-CPAP, Corumbá, 184p.
- BRITTO, M. R., 2000, Aspidoras depinnai (Siluriformes: Callichthyidae): a new species from northeastern Brazil. Copeia, 2000: 1048-1055.
- BURGESS, W. E., 1989, An atlas of freshwater and marine catfishes: a preliminary survey of the Siluriformes TFH Publications, Neptune City, 784p.
- CARTER, G. S. & BEADLE, L. C., 1931, The fauna of Swamps of the Paraguayan Chaco in relation to its environment. II. Respiratory adaptations in the fishes. J. Linn. Soc, 37: 327-368.
- CORREA-SOARES, B. E., 1985, Crescimento e fecundidade de Biomphalaria tenagophila (Mollusca, Planorbidae) submetida a dietas exclusivas de Heteranthera reniformis, Nasturtium officinale ou Lactuca savita. Dissertação de Mestrado, Instituto Oswaldo Cruz, Rio de Janeiro, RJ, 61p.
- GARUTTI, V., 1988, Distribuição longitudinal da ictiofauna em um córrego da região noroeste do Estado de São Paulo, bacia do rio Paraná. Rev. Brasil. Biol., 48: 747-759.
- GEE, J. H. & GRAHAM, J. B., 1978, Respiratory and hydrostatic functions of the intestine of catfishes Hoplosternum thoracatum and Brochis splendens (Callichthyidae). J. Exp. Biol, 74: 1-16.
- GOLTERMAN, H. L., CLYMO, R. S. & OHNSTAD, M. A. M., 1978, Methods for physical and chemical analysis of freshwater Blackwell Sci. Publ., London, 213p.
- KRAMER, D. A. & McCLURE, M., 1980, Aerial respiration in the catfish Corydoras aeneus (Callichthyidae). Can. J. Zool, 58: 1984-1991.
- LOWE-McCONNELL, R. H., 1999, Estudos ecológicos de comunidades de peixes tropicais EDUSP, São Paulo, 534p.
- MACHADO-ALLISON, A., 1986, Aspectos sobre la historia natural del ''curito'' Hoplosternum littorale (Hancock, 1828) (Siluriformes, Callichthyidae) en el bajo llano de Venezuela: desarrolo, alimentación y distribución espacial. Acta Cient. Venez., 37: 72-78.
- MINITAB, 1996, Minitab for windows, release 11 Minitab Inc., State College, PA, USA.
- MOL, J. H., 1996, Impact of predation on early stages of the armoured catfish Hoplosternum thoracatum (Siluriformes: Callichthyidae) and implications for the syntopic occurrence with other related catfishes in a neotropical multi-predator swamp. Oecology, 107: 395-410.
- MOL, J. H., ATSMA, W., FLIK, G. & BOUWMEESTER, H., 1999, Effect of low ambient mineral concentrations on the acumulation of calcium, magnesium and phosphorus by early life stages of the air-breathing armoured catfish Megalechis personata (Siluriformes, Callichthyidae). J. Exp. Biol, 202: 2121-2129.
- NECCHI, O. Jr., 1992, Macroalgae dynamics in a spring in São Paulo State, southeastern Brazil. Arch. Hydrobiol, 124: 489-499.
- NELSON, J. S., 1994, Fishes of the world John Wiley & Sons Inc., 3. ed., 600p.
- NIJSSEN, H. & ISBRÜCKER, I. J. H., 1976, The South American plated catfish genus Aspidoras R. von Ihering, 1907, with descriptions of nine new species from Brazil (Pisces, Siluriformes, Callichthyidae). Bijdr. Dierk, 46: 107-131.
- PAVANELLI, C. S. & CARAMASCHI, E. P., 1997, Composition of the ichthyofauna of two small tributaries of the Paraná river, Porto Rico, Paraná State, Brazil. Ichthyol. Explor Freshwaters, 8: 23-31.
- PINKERTON, A., 1987, Spawning and rearing Aspidoras Trop. Fish Hobb, 35: 34-41.
- PRUZSINSZKY, I. & LADICH, F., 1998, Sound production and reproductive behaviour of the armoured catfish Corydoras paleatus (Callichthyidae). Env. Biol. Fish, 53: 183-191.
- RAMNARINE, I. W., 1995, Induction of nest-building and spawing in Hoplosternum littorale. J. Fish Biol, 47: 555-557.
- REIS, R. E., 1998, Anatomy and phylogenetic analysis of the neotropical callichthyid catfishes (Ostariophysi, Siluriformes). Zool. J. Linn. Soc, 124: 105-168.
- SAUL, W. G., 1974, An ecological study of fishes at a site in upper Amazonian Ecuador. Proc. Acad. Nat. Sci. Philad., 127: 93-134.
- SHEPHERD, G. J., 1995, Programa FITOPAC 1 Universidade Estadual de Campinas, Campinas.
- SCHINDLER, I. & STAECK, W., 1996, Ein aquaritstisch neuer Panzerwels: Aspidoras raimundi (Steindachner, 1907). Datz, 49: 630-633.
- SOKAL, R. R. & ROHLF, F. J., 1981, BiometryW. H. Freeman, New York, 859p.
- STRAHLER, A. N., 1957, Quantitative analysis of watershed geomorphology. Trans. Am. Geophys. Un, 38: 913-920.
- UIEDA, V. S., 1984, Ocorrência e distribuição dos peixes em um riacho de água doce. Rev. Brasil. Biol, 44: 203-213.
- UIEDA, V. S. & UIEDA, W., 2001, Species composition and spatial distribution of a stream fish assemblage in the east coast of Brazil: composition of two field study methodologies. Braz. J. Biol, 61: 377-388.
Publication Dates
-
Publication in this collection
21 Jan 2004 -
Date of issue
Aug 2003
History
-
Accepted
16 May 2002 -
Received
14 Jan 2002