Abstracts
The infection, known as yellow spot disease, produced by metacercariae of Clinostomum spp. was studied in fishes of the Taquari river, located in Jataizinho, Paraná State, Brazil. A total of 1,582 specimens, belonging to 36 species, were collected between March 1999 and April 2001. Yellow spot disease was observed only in Gymnotus carapo Linnaeus, 1814 (Gymnotiformes, Gymnotidae) and Cichlasoma paranaense Kullander, 1983 (Perciformes, Cichlidae). This parasitism was generated by metacercariae of Clinostomum complanatum (Rudolphi, 1814) (Digenea, Clinostomidae). Among the 88 specimens of G. carapo examined, 7 (prevalence = 8%) had cysts of the parasite. Four of them were captured in July 1999 and three in October 1999. In the other months, no specimen found was infected. Using relative condition factor (Kn) analysis, it was determined that both infected and non-infected specimens had a total weight equal to the theoretically expected value for each total length (Kn = 1.0). Among 56 individuals of Cichlasoma paranaense, 6 (prevalence = 10.7%) had between 1 and 27 metacercariae of C. complanatum (mean intensity of infection = 9.3 ± 9.6). In March 1999 and April of both 2000 and 2001, the specimens examined were not infected. The infected fish had a total weight higher than the expected value (Kn > 1.0), while the non-infected fish had a weight equal to the expected value (Kn = 1.0).
fish; parasitism; metacercariae; Clinostomum complanatum; Taquari river
Estudou-se a infecção por metacercárias de Clinostomum spp., conhecida como doença das manchas amarelas, em peixes do rio Taquari, localizado no município de Jataizinho, Paraná, Brasil. Um total de 1.582 espécimes pertencentes a 36 espécies foram capturados entre março de 1999 e abril de 2001. As manchas amarelas foram observadas apenas em Gymnotus carapo Linnaeus, 1814 (Gymnotiformes, Gymnotidae) e Cichlasoma paranaense Kullander, 1983 (Perciformes, Cichlidae). Esse parasitismo foi produzido por larvas de Clinostomum complanatum (Rudolphi, 1814) (Digenea, Clinostomidae), na fase de metacercária. Dentre os 88 espécimes de G. carapo examinados, 7 (prevalência = 8%) tinham cistos do parasita. Destes, 4 foram capturados em julho de 1999, quando foi observada a mais alta intensidade de infecção, e 3 em outubro de 1999. Nos outros meses, nenhum espécime encontrava-se infectado. Analisando o fator de condição relativo (Kn) foi possível determinar que tanto os exemplares parasitados como os não parasitados estavam com o peso igual ao teoricamente esperado para cada comprimento (Kn = 1,0). Entre os 56 indivíduos de Cichlasoma paranaense, 6 (prevalência = 10,7%) apresentavam entre 1 e 27 metacercárias de C. complanatum (intensidade média de infecção = 9,3 ± 9,6). A mais alta intensidade de infecção foi registrada em janeiro de 2000. Em março de 1999 e em abril de 2000 e de 2001, nenhum dos espécimes examinados estava infectado. Os peixes parasitados encontravam-se com o peso total acima do esperado (Kn > 1,0), enquanto os não parasitados apresentaram peso total igual ao esperado (Kn = 1,0).
peixes; parasitismo; metacercária; Clinostomum complanatum; rio Taquari
Parasitism of Cichlasoma paranaense Kullander, 1983 and Gymnotus carapo Linnaeus, 1814 by Clinostomum complanatum (Rudolphi, 1814) Metacercariae in the Taquari river
Parasitismo de Cichlasoma paranaense Kullander, 1983 e Gymnotus carapo Linnaeus, 1814 do rio Taquari por metacercárias de Clinostomum complanatum (Rudolphi, 1814)
Silva-Souza, A. T.I; Ludwig, G.I, II
IUniversidade Estadual de Londrina, Departamento de Biologia Animal e Vegetal, CCB, C.P. 6001, CEP 860510-990, Londrina, PR, Brazil
IIIC/CPG-UEL
Correspondence Correspondence to Ângela Teresa Silva e Souza Universidade Estadual de Londrina Departamento de Biologia Animal e Vegetal, CCB, C.P. 6001 CEP 86051-990, Londrina, Paraná, Brazil e-mail: ateresa@uel.br
ABSTRACT
The infection, known as yellow spot disease, produced by metacercariae of Clinostomum spp. was studied in fishes of the Taquari river, located in Jataizinho, Paraná State, Brazil. A total of 1,582 specimens, belonging to 36 species, were collected between March 1999 and April 2001. Yellow spot disease was observed only in Gymnotus carapo Linnaeus, 1814 (Gymnotiformes, Gymnotidae) and Cichlasoma paranaense Kullander, 1983 (Perciformes, Cichlidae). This parasitism was generated by metacercariae of Clinostomum complanatum (Rudolphi, 1814) (Digenea, Clinostomidae). Among the 88 specimens of G. carapo examined, 7 (prevalence = 8%) had cysts of the parasite. Four of them were captured in July 1999 and three in October 1999. In the other months, no specimen found was infected. Using relative condition factor (Kn) analysis, it was determined that both infected and non-infected specimens had a total weight equal to the theoretically expected value for each total length (Kn = 1.0). Among 56 individuals of Cichlasoma paranaense, 6 (prevalence = 10.7%) had between 1 and 27 metacercariae of C. complanatum (mean intensity of infection = 9.3 ± 9.6). In March 1999 and April of both 2000 and 2001, the specimens examined were not infected. The infected fish had a total weight higher than the expected value (Kn > 1.0), while the non-infected fish had a weight equal to the expected value (Kn = 1.0).
Key words: fish, parasitism, metacercariae, Clinostomum complanatum, Taquari river.
RESUMO
Estudou-se a infecção por metacercárias de Clinostomum spp., conhecida como doença das manchas amarelas, em peixes do rio Taquari, localizado no município de Jataizinho, Paraná, Brasil. Um total de 1.582 espécimes pertencentes a 36 espécies foram capturados entre março de 1999 e abril de 2001. As manchas amarelas foram observadas apenas em Gymnotus carapo Linnaeus, 1814 (Gymnotiformes, Gymnotidae) e Cichlasoma paranaense Kullander, 1983 (Perciformes, Cichlidae). Esse parasitismo foi produzido por larvas de Clinostomum complanatum (Rudolphi, 1814) (Digenea, Clinostomidae), na fase de metacercária. Dentre os 88 espécimes de G. carapo examinados, 7 (prevalência = 8%) tinham cistos do parasita. Destes, 4 foram capturados em julho de 1999, quando foi observada a mais alta intensidade de infecção, e 3 em outubro de 1999. Nos outros meses, nenhum espécime encontrava-se infectado. Analisando o fator de condição relativo (Kn) foi possível determinar que tanto os exemplares parasitados como os não parasitados estavam com o peso igual ao teoricamente esperado para cada comprimento (Kn = 1,0). Entre os 56 indivíduos de Cichlasoma paranaense, 6 (prevalência = 10,7%) apresentavam entre 1 e 27 metacercárias de C. complanatum (intensidade média de infecção = 9,3 ± 9,6). A mais alta intensidade de infecção foi registrada em janeiro de 2000. Em março de 1999 e em abril de 2000 e de 2001, nenhum dos espécimes examinados estava infectado. Os peixes parasitados encontravam-se com o peso total acima do esperado (Kn > 1,0), enquanto os não parasitados apresentaram peso total igual ao esperado (Kn = 1,0).
Palavras-chave: peixes, parasitismo, metacercária, Clinostomum complanatum, rio Taquari.
INTRODUCTION
Clinostomidae are digenetic trematodes that cause the parasitism known globally as yellow spot disease. The majority of digenetic trematodes do not pose a serious threat to fish health. However, the presence of parasite cysts makes the fish unattractive visually to consumers. According to Thatcher (1991), species of two genera of Clinostomidae, i.e., Clinostomum and Odheriotrema, cause yellow spot disease in Brazilian fishes.
Clinostomum complanatum (Rudolphi, 1814) is one of the most frequent species of Clinostomidae. The larvae do not seem to have high host specificity and their metacercariae have been found in several species of fish (Dias, 2002). The parasite cysts, which frequently have a yellow color, are surrounded by a thick capsule from the fish tegument.
The infection of fish by Clinostomum complanatum metacercariae can unleash behavioral changes, disease, and death, on one hand, and economic loss on the other (Eiras, 1994; Vianna, 2001). Moreover, Clinostomum complanatum has zoonotic potential (Kamo et al., 1962), e.g., Clinostomidae specimens can infect the oral cavity of humans who eat raw parasitized fish (Chung et al., 1995; Kitagawa et al., 2003). According to Eiras (1994), this parasite can cause laryngopharyngitis and even death by asphyxia in humans. Kamo et al. (1962) reported the occurrence of anemia in a Thailandese male whose eyes were infected by Clinostomum sp. Tiewchaloern et al. (1999).
In Brazil, despite the large number of freshwater species of fish, only a few studies of parasitism by the larvae of Clinostomidae have been carried out. However, for several years fish species potentially infected by digenetic trematode larvae have been investigated in the Tibagi river and its tributaries. The aim of the present study was to evaluate yellow spot disease in fish from one of those tributaries, the Taquari river, and to determine the effect of parasitism on fish health.
MATERIAL AND METHODS
Fish were captured in the Taquari river, in the municipality of Jataizinho, Paraná State, Brazil, where samplings were made every three months between March 1999 and April 2000, and in November 2000 and March-April 2001. For each collection, a 20 m-long section was marked off by two nylon nets extending to the river bed and stretched from one bank of the river to the other. The specimens were captured in this closed space with fishing nets and bolters. All the collected fish were sacrificed by cerebral concussion, weighed (total weight in grams), measured (total and standard lengths in centimeters), identified, and fixed in 4% formalin. The specimens were then examined for parasites.
The cysts were carefully removed under a stereoscopic microscope and placed in Petri dishes with 0.65% physiological saline solution. The parasites were collected from the cysts, counted, and prepared routinely according to Amato et al. (1991).
Prevalence (P %), mean intensity of infection (MII), and mean abundance (MA) were calculated (Bush et al., 1997).
The total weight and the total length values of all the specimens examined were used to adjust the weight-length relationship curve. The a and b values were used first to calculate the theoretically expected weight (We) and then the relative condition factor (Kn = Wt/We) (LeCren, 1951; Eckmann, 1984). The mean Kn values of each infected fish group and non-parasitized group were compared to the standard Kn = 1.0 by Student's t-test. Statistical comparison of both the mean total length and mean total weight between the infected and non-infected fish groups was performed using Student's t-test. The data were considered significantly different when a bilateral probability of p < 0.05 was obtained.
RESULTS
A total of 1,582 individuals belonging to 36 fish species from the Taquari river were examined. Of these species, only Cichlasoma paranaense Kullander, 1983 (Perciformes, Cichlidae) and Gymnotus carapo Linnaeus, 1814 (Gymnotiformes, Gymnotidae) were found to have yellow spot disease. In both species, parasitism was the result of encystment of metacercariae of the digenetic trematode Clinostomum complanatum (Rudolphi, 1814) (Digenea, Clinostomidae).
In Cichlasoma paranaense the cysts, which were elongated and contained only one metacercaria, were located in the rays of the caudal, anal, dorsal, pectoral, and pelvic fins. In the case of Gymnotus carapo the majority of the cysts were rounded and had only one metacercaria, some of which were elongated and contained two metacercariae. The cysts were found in the head, throughout the ventral part of the body, in the operculum region, and in the base and rays of the anal fin.
Among the 56 individuals of C. paranaense captured in the Taquari river, six were infected (Prevalence = 10.7%) and had from one to 27 metacercariae of Clinostomum complanatum (MII = 9.33 ± 9.58). Despite the low prevalence, in most months at least one specimen was found with C. complanatum metacercariae. No infected individuals were discovered in collections made in March 1999, April 2000, and April 2001. The highest parasitic index was found in January 2000 (Table 1). Sampling at three-month intervals was inadequate to determine the infection period (this would require monthly evaluations). However, the parasitic index values observed suggest that infection occurred in summer.
Among the 88 specimens of Gymnotus carapo examined, only 7 were infected (8.0% prevalence). Four of them were captured in July and three in October 1999. In the other months, none of the individuals collected was infected (Table 2). The largest number of specimens were captured in July and October 1999, with July showing the higher value of infection intensity. Among those caught, four were infected, one so heavily that counting the number of metacercariae presented was not feasible. Therefore, determining the mean infection intensity and mean abundance was done by only taking into account the metacercariae in the other three fish (Table 2). The relative condition factor (Kn) was used to determine the probable health effects of parasitism on the specimens of Cichlasoma paranaense and Gymnotus carapo from the Taquari river.
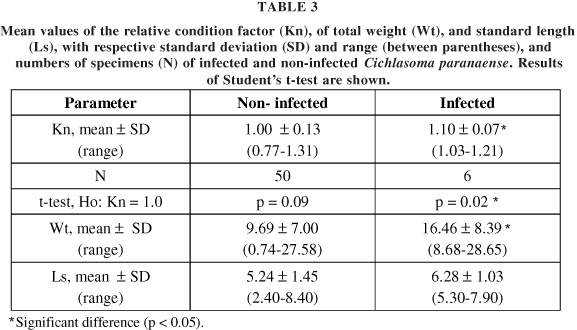
In Cichlasoma paranaense, the specimens' weight ranged between 0.74 and 28.65 g; standard length was between 2.4 and 8.4 cm. The values of a and b were 0.0555 and 3.0056, respectively. The statistical results of the relative condition factor, total weight, and total length of the specimens studied are summarized in Table 3.
Although the average standard length of the infected individuals did not differ significantly from that of the non-infected fish (p = 0.09), the mean value of the infected fishes' total weight was statistically higher (p = 0.03). The same was true of the relative condition factor (Kn), i.e., the infected fish had a mean Kn significantly higher than that of the non-infected fish (p = 0.04). Comparing the mean Kn with the standard value (1.0) showed that the infected fish had a greater weight than expected (Kn > 1.0), while the weight of the non-infected fish was equal to the expected theoretical value (Kn = 1.0).
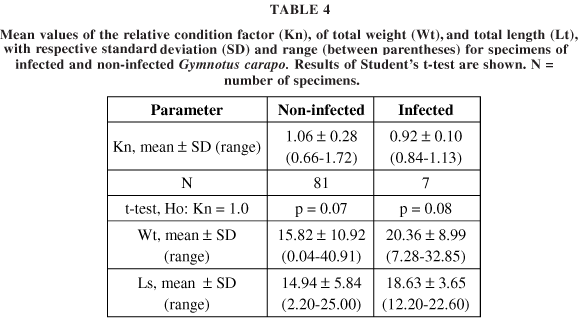
The specimens of Gymnotus carapo weighed from 0.04 to 40.91 g and measured length ranged between 2.2 and 25.0 cm. The values of a and b were 0.0059 and 2.795, respectively. The statistical results for the relative condition factor, total weight, and total length of the specimens studied are summarized in Table 4. The infected and non-infected individuals did not differ in relation to total length and total weight (p = 0.11 and p = 0.29, respectively). The same was found when comparing the values for the relative condition factor (Kn) between infected and non-infected fish (p = 0.21). The comparison of Kn values with the standard value Kn = 1.0 showed that in both parasitized and non-parasitized fish, the specimens had a total weight equal to the theoretically expected value (Kn = 1.0).
DISCUSSION
Metacercariae of Clinostomum complanatum have been found in many fish species, e.g., Lepomis macrochirus, L. gulosus, L. auritus, Pomoxis nigromaculatus, Micropterus salmoides (Hanzen & Esch, 1978), Trichogaster fasciatus (Siddiqui & Nizami, 1982), Aphanius dispar (Kalantan et al., 1987), Rhamdia guatemalensis (Pérez-Ponce de León et al., 1992), Aloophorus robustus (Guzmán-Cornejo & García-Pietro, 1999), Rhamdia cf. quelen (Vianna, 2001), Hoplias malabaricus, Loricaria sp., Hoplosternum littorale, Loricariichthys platymetopon, and Parauchenipterus galeatus (Dias et al., 2003). Metacercariae of Clinostomum complanatum have also been found in toads, frogs, salamanders, and tritons (Yamaguti, 1971; Muzzall, 1991; Goldberg et al., 1998), as well as in many fish species, thus demonstrating the diversity of hosts this parasite can exploit.
In Rhamdia guatemalensis, metacercariae of C. complanatum were found in the gills, operculum, and muscle (Pérez-Ponce de León et al., 1992). In Aphanius dispar, Kalantan et al. (1987) detected cysts mostly in the trunk region and, secondly, in that of the head. Rhamdia cf. quelen presented metacercariae in the head region and in the base of the fins, mainly the pectoral and caudal (Vianna, 2001). Dias (2002) observed experimentally induced infection by metacercariae in the operculum of Gymnotus carapo. Fish species from the Paraná river were shown to have cysts in the base of the fins, muscle, visceral cavity, operculum cavity, palate, and oral cavity (Dias, op. cit.).
The present study demonstrated that the infection site varied according to the host, as well as to the type of cysts. The specimens of Cichlasoma paranaense showed cysts exclusively in fin rays. But in Gymnotus carapo the cysts, some of which contained two metacercariae instead of one, were located in various body regions, as has been recorded for species of fish caught in other places. The prevalence of Clinostomidae species can vary according to environmental characteristics (Eiras, 1994). In studying parasitism of Rhamdia cf. quelen by C. complanatum metacercariae, Vianna (2001) found a higher prevalence of these parasites in spring (95.5%) and a lower incidence in summer (33.3%). Similar values of prevalence (65.6% and 65.4%, respectively) were found in autumn and winter.
In the Taquari river, C. paranaense had a low intensity of infection in most months, while higher parasitic index values were observed in the summer. G. carapo specimens were infected in both months in which the species was found; however, heavily parasitized individuals were collected in the winter. The very few specimens captured of either host species precludes conclusions about seasonal variation of C. complanatum infection in fish from the Taquari river.
Elliot & Russert (1949) (apud Vianna, 2001) detected no relation between infection by Clinostomum marginatum and the condition factor (K) of Perca flavescens. Silva-Souza (1998) compared values of the relative condition factor (Kn) of Plagioscion squamosissimus (Sciaenidae) infected by metacercariae of Diplostomum (Austrodiplostomum) compactum and found that infected fish had a total weight lower than that theoretically expected for each length, while the weight of non-infected specimens was higher than that expected. Ranzani-Paiva et al. (2000) showed that the parasites found in specimens of Schizodon borellii and Prochilodus lineatus of the Paraná river did not significantly affect host condition. In both species, the infected and non-infected individuals had a total weight near the theoretically expected value (Kn = 1.0).
In addition, the health of Cichlasoma paranaense and Gymnotus carapo from the Taquari river did not seem to be influenced by infection with metacercariae of Clinostomum complanatum. This is probably due to the low infection intensity in the hosts. However, the most heavily parasitized specimen of G. carapo was not the one with the lowest Kn value. Similar results were obtained by Vianna (2001) who compared the Kn values of specimens of Rhamdia cf. quelen, some of which were infected by C. complanatum, and found no significant differences.
This is the first report of Cichlasoma paranaense and Gymnotus carapo infection by matecercariae of Clinostomum complanatum in the natural environment.
Received October 24, 2003 Accepted December 15, 2003 Distributed August 31, 2005
- AMATO, J. F. R., BOEGER, W. A. & AMATO, S. B., 1991, Protocolos para laboratório-coleta e processamento de parasitas do pescado Imprensa Universitária, Universidade Federal do Rio de Janeiro, Rio de Janeiro, 81p.
- BUSH, A. O., LAFFERTY, K. D., LOTZ, J. M. & SHOSTAK, A. W., 1997, Parasitology meets ecology on its own terms: Margolis et al. revisited. Journal of Parasitology, 83(4): 575-583.
- CHUNG, D. I., MOON, H. H., CHOI, D. W. & LIM, D. K., 1995, The first case of Clinostomum complanatum (Trematoda: Clinostomidae) infection in Korea. Korean J. Parasitol., 33(3): 219-223.
- DIAS, M. L. G. G., 2002, Aspectos ecológicos e ciclo de vida de Clinostomum complanatum (Rud., 1814) (Trematoda: Clinostomidae), parasito de peixes e aves da planície de inundação do alto rio Paraná, Brasil Ph.D. Thesis (Ecology of continental aquatic environments Environmental Sciences), Universidade Estadual de Maringá, Maringá, PR.
- DIAS, M. L., EIRAS, J. C., MACHADO, M. H., SOUZA, G. T. & PAVANELLI, G. C., 2003, The life cycle of Clinostomum complanatum (Rudolphi, 1814) (Digenea, Clinostomidae) on the floodplain of the high Paraná river, Brazil. Parasitology Research, 89(6): 506-508.
- ECKMANN, R., 1984, Induced reproduction in Brycon cf. erythropterus Aquaculture, 38: 379-382.
- EIRAS, J. C., 1994, Elementos de ictioparasitologia Fundação Eng. António de Almeida, Porto, Portugal, 339p.
- GOLDBERG, S. R., BURSEY, C. R. & CHEAM, H., 1998, Helminths of two native frog species (Rana chiricahuensis, Rana yavapaiensis) and one introduced frog species (Rana catesbeiana) (Ranidae) from Arizona. J. Parasitol., 84(1): 175-177.
- GUZMÁN-CORNEJO, M. del C. & GARCÍA-PRIETO, L., 1999, Trematodiasis en algunos peces del lago del Cuitzeo, Michoacán, México. Rev. Biol. Trop., 47(3): 593-596.
- HANZEN, T. C. & ESCH, G. W., 1978, Observations on the ecology of Clinostomum marginatum in Largemouth bass (Micropterus salmoides). J. Fish Biol., 12: 411-420.
- KALANTAN, A. M., ARFIN, M. & NIZAMI, W. A., 1987, Seasonal incidence and pathogenicity of the metacercariae of Clinostomum complanatum in Aphanius dispar Jap. J. Parasitol., 36(1): 17-23.
- KAMO, H., OGINO, K. & HATSUSHIKA, A. R. A., 1962, Unique infection of man with Clinostomum sp., a small Trematoda causing acute laryngitis. Yonago Acta Medica, 6: 37-40.
- KITAGAWA, N., ODA, M., TOTOKI, T., WASHIZAKI, S., ODA, M. & KIFUNE, T., 2003, Lidocaine spray used to capture a live Clinostomum parasite causing laryngitis. Am. J. Otolaryngol., 24(5): 341-343.
- LeCREN, E. D., 1951, The length-weight relationship and seasonal cycle in gonad weight and condition in perch Perca fluviatilis Journal of Animal Ecology, 20: 201-219.
- MUZZALL, P. M., 1991, Helminth infracommunities of the Newt, Notophthalmus viridescens, from Turkey Marsh, Michigan. J. Parasiol., 77(1): 87-91.
- PÉREZ-PONCE DE LÉON, G., OSORIO-SARABIA, D. & GARCÍA-PRIETO, L., 1992, Helmintofauna del "Juile" Rhamdia guatemalensis (Pisces: Pimelodidae), del lago de Catemaco, Veracruz. Rev. Soc. Mex. Hist. Nat., 43: 25-31.
- RANZANI-PAIVA, M. J. T., SILVA-SOUZA, A. T., PAVANELLI, G. C. & TAKEMOTO, R. M., 2000, Hematological characteristics and relative condition factor (Kn) associated with parasitism in Schizodon borellii (Osteichthyes, Anostomidae) and Prochilodus lineatus (Osteichthyes, Prochilodontidae) from Paraná river, Porto Rico region, Paraná, Brazil. Acta Scientiarum, 22(2): 515-521.
- SIDDIQUI, A. & NIZAMI, W. A., 1982, Seasonal population dynamics of the metacercariae of Clinostomum complanatum (Trematoda: Digenea) in relation to sex of the host. Rev. Parasitol., 43(2): 275-279.
- SILVA-SOUZA, A. T., 1998, Estudo do parasitismo em Plagioscion squamosissimus (Heckel, 1840) (Perciformes, Sciaenidae) por Diplostomum (Austrodiplostomum) compactum (Lutz, 1928) (Trematoda, Digenea) no rio Tibagi, PR. Ph.D. Thesis, Universidade Federal de São Carlos, São Carlos, SP.
- THATCHER, V. E., 1991, Amazon fish parasites. Amazoniana, 11(3/4): 263-571.
- TIEWCHALOERN, S., UDOMKIJDECHA, S., SUVOUTTHO, S., CHUNCHAMSRI, K. & WAIKAGUL, J., 1999, Clinostomum trematode from human eye. Southeast Asian J. Trop. Med. Public Health, 30(2): 382-384.
- VIANNA, R. T., 2001, Clinostomum complanatum (Rudolphi, 1814) em Rhamdia cf. quelen (Quoy & Gaimard, 1824), no Arroio Sarandi, Rio Grande, RS. M.S. Thesis, Universidade Federal de Santa Maria, Santa Maria, RS.
- YAMAGUTI, S., 1971, Synopsis of digenetic trematodes of vertebrates Keigakur Publishing Co. Tokyo, Japan, 1072p.
Correspondence to
Publication Dates
-
Publication in this collection
13 Dec 2005 -
Date of issue
Aug 2005
History
-
Received
24 Oct 2003 -
Accepted
15 Dec 2003