Abstracts
Hydrochemical conditions in the Açu Lagoon are described using spatial and temporal variations of various limnological variables (water temperature, dissolved oxygen, electric conductivity, total alkalinity, carbon dioxide, dissolved and total nutrients (N, P and Si), and chlorophyll a). Collected data was used in order to understand the structure and functioning of an enclosed coastal lagoon strongly influenced by climatic conditions. Water samples were collected monthly (November 1999-December 2000) in five sampling stations established along the lagoon. A decreasing spatial gradient of electrical conductivity was observed beginning from a sand bar region between the lagoon and the sea in the direction of the sweet-water input area. The positive correlation observed between the pH and dissolved oxygen (DO) values, and the negative one observed between pH values and those of carbon dioxide (CO2), evidenced coupled biological processes, e.g., primary production and decomposition. Both spatial and temporal variation of dissolved nutrients showed fast increase and decrease in the beginning of summer, suggesting that nutrient input resulting from rainfall stimulates phytoplankton production, as reflected by chlorophyll a concentration increase.
coastal lagoon; hydrochemistry; dissolved nutrients
Condições hidroquímicas da lagoa do Açu são descritas usando a variação espacial e temporal de algumas variáveis limnológicas (temperatura da água, oxigênio dissolvido (OD), condutividade elétrica, alcalinidade total, CO2, nutrientes dissolvidos e totais (N, P, Si) e clorofila a para entender a estrutura e funcionamento de uma lagoa costeira isolada e fortemente influenciada pela sazonalidade. Amostras de água foram coletadas mensalmente (novembro 1999 a dezembro 2000) em cinco estações de amostragem estabelecido ao longo da lagoa. Um gradiente espacial decrescente de condutividade elétrica foi observado da região da barra de areia, que separa a lagoa do mar, em direção a região que recebe entrada de água doce. A correlação positiva entre os valores de pH e oxigênio dissolvido, e negativa observada destes com o CO2, evidencia o acoplamento de processos biológicos, como o de produção primaria e de decomposição. A variação espaço temporal de nutrientes dissolvidos mostrou um rápido incremento e redução da concentração de nutrientes dissolvidos no inicio do verão, sugerindo que a entrada de nutrientes pelas chuvas é um fator que estimula o crescimento fitoplanctônico, refletido pelo aumento da concentração de clorofila a.
lagoa costeira; hidroquímica; nutrientes dissolvidos
Seasonal hydrochemical variation in a tropical coastal lagoon (Açu Lagoon, Brazil)
Variação sazonal da hidroquímica em uma lagoa costeira tropical (Lagoa do Açu, Brasil)
Chagas, G. G.; Suzuki, M. S.
Laboratório de Ciências Ambientais, CBB, Universidade Estadual do Norte Fluminense, Av. Alberto Lamego, 2000, Pq. Califórnia, CEP 28013-600, Campos dos Goytacazes, RJ, Brazil
Correspondence to Correspondence to Gustavo Gomes Chagas Laboratório de Ciências Ambientais, CBB, Universidade Estadual do Norte Fluminense Av. Alberto Lamego, 2000, Pq. Califórnia CEP 28013-600, Campos dos Goytacazes, RJ, Brazil e-mail: ggchagas@yahoo.com.br
ABSTRACT
Hydrochemical conditions in the Açu Lagoon are described using spatial and temporal variations of various limnological variables (water temperature, dissolved oxygen, electric conductivity, total alkalinity, carbon dioxide, dissolved and total nutrients (N, P and Si), and chlorophyll a). Collected data was used in order to understand the structure and functioning of an enclosed coastal lagoon strongly influenced by climatic conditions. Water samples were collected monthly (November 1999-December 2000) in five sampling stations established along the lagoon. A decreasing spatial gradient of electrical conductivity was observed beginning from a sand bar region between the lagoon and the sea in the direction of the sweet-water input area. The positive correlation observed between the pH and dissolved oxygen (DO) values, and the negative one observed between pH values and those of carbon dioxide (CO2), evidenced coupled biological processes, e.g., primary production and decomposition. Both spatial and temporal variation of dissolved nutrients showed fast increase and decrease in the beginning of summer, suggesting that nutrient input resulting from rainfall stimulates phytoplankton production, as reflected by chlorophyll a concentration increase.
Keywords: coastal lagoon, hydrochemistry, dissolved nutrients.
RESUMO
Condições hidroquímicas da lagoa do Açu são descritas usando a variação espacial e temporal de algumas variáveis limnológicas (temperatura da água, oxigênio dissolvido (OD), condutividade elétrica, alcalinidade total, CO2, nutrientes dissolvidos e totais (N, P, Si) e clorofila a para entender a estrutura e funcionamento de uma lagoa costeira isolada e fortemente influenciada pela sazonalidade. Amostras de água foram coletadas mensalmente (novembro 1999 a dezembro 2000) em cinco estações de amostragem estabelecido ao longo da lagoa. Um gradiente espacial decrescente de condutividade elétrica foi observado da região da barra de areia, que separa a lagoa do mar, em direção a região que recebe entrada de água doce. A correlação positiva entre os valores de pH e oxigênio dissolvido, e negativa observada destes com o CO2, evidencia o acoplamento de processos biológicos, como o de produção primaria e de decomposição. A variação espaço temporal de nutrientes dissolvidos mostrou um rápido incremento e redução da concentração de nutrientes dissolvidos no inicio do verão, sugerindo que a entrada de nutrientes pelas chuvas é um fator que estimula o crescimento fitoplanctônico, refletido pelo aumento da concentração de clorofila a.
Palavras-chave: lagoa costeira, hidroquímica, nutrientes dissolvidos.
INTRODUCTION
Coastal lagoons are highly productive aquatic ecosystems (Knoppers, 1994) due to intrinsic characteristics such as: shallow water; continuous dissolved and particulate matter input; particularly from high lands; and those caused by internal dynamics generated by external energy.
Lagoon characteristics and their intrinsic hydrochemical dynamics are directly related to the relative influence of continental and marine waters received, which generate a biogeochemical gradient from inner lagoon areas toward the sea (Castel et al., 1996; Suzuki et al., 2002). The generally lowland lagoon locations promote organic and inorganic materials accumulation, especially in coastal lagoons, which results in elevated degrees of natural eutrophication. Shallow waters and constant wind facilitate extensive modification of materials and complete water-column circulation, thus increasing the interaction between pelagic and benthic compartments (Nixon, 1981). Because it is productive (due to light penetration), the benthic compartment largely controls the functioning of these systems. In addition, sediment resuspension and interstitial water advection contribute to regenerating nutrients accumulated in the bottom (Nixon, 1982).
Lagoons are currently considered one of the environments most affected by anthropic activities such as domestic, industrial, and agricultural waste dumping; fish, crab, and shrimp catching and farming; and dredging (Esteves, 1998). Some of these activities can accelerate the natural eutrophication process, causing irreversible deterioration of these as yet not totally understood environments. Studying the structure and functioning of coastal lagoons can supply information useful in sustainably managing these sensitive ecosystems, among which are those of the northern area of Rio de Janeiro State. In this region, the lagoons have an important social and economic function because of fishery (mainly fish and crustaceans), which sustains many local families. They also serve as frequently visited leisure areas.
The aim of this study is to identify spatial and temporal variations of some limnological characteristics in the Açu Lagoon, to establish seasonal influence on the dynamics of these variables, and to determine the lagoons ecological heterogeneity.
Study area
In northern Rio de Janeiro State, the coastal lagoons whose origins are related to the formation process of Paraiba do Sul river delta are based geologically on quaternary fluvial marine sand deposits (Lamego, 1955). At present, these lagoons are isolated from the sea by a narrow (around 50 m wide) sandbar and a floodgate that sometimes is opened, especially in the rainy season (November to March).
Located in a sub-humid-dry region, the Açu Lagoon (Fig. 1), lies from within the city boundaries of Campos dos Goytacazes and São João da Barra. It presents a narrow, elongated shape, and runs parallel to the coast. Lagoon proximity to the sea, mainly on the northern portion, promotes a salinity gradient whose values decrease toward the southern portion, where the lagoon receives freshwater inputs especially from Quitinguta and São Bento canals and groundwater. The Açu lagoon watershed is covered by pastures, subsistance agriculture, and restinga vegetation. Due to the lagoons high salinity (about 30 us), mangrove species, especially of Avicenia sp, Laguncularia sp, and Rhizophora mangle, grow on its margins. Seasonal growth of benthonic macroalgae, e.g., Ulva sp, Cladophora sp, and Hypnea sp, near the sand bar that separates the lagoon from the sea. The inner area of the lagoon is also colonized by emergent aquatic macrophytes, especially Typha domingensis (Taboa).
METHODS
From November 1999 to December 2000, water samplings were carried out monthly in five stations along the Açu Lagoon (Fig. 1). The sampling stations had been chosen according to the morphology and visible biological characteristics of each, including varying degrees of settling macroalgae and aquatic macrophytes.
At each station, water samples were collected directly, using plastic bottles, from both surface (0.05 m below the water-column top) and 0.1 m above the bottom. Bottom water deeper than 0.5 m was collected with a Van Dorn bottle. Variables such as dissolved oxygen (DO), water temperature, electrical conductivity, and pH were measured at the water column itself using portable equipment (a YSI model 63 multiparameter and a YSI model 55 oximeter, with temperature compensation). All water samples collected were transported to the laboratory and processed immediately. Total alkalinity was measured following Grans procedures (Gran, 1952), and data were used to compute percentage saturation of dissolved inorganic carbon (CO2) (Carmouze, 1994). Subsamples were filtered in duplicate through Whatman GF/F membranes kept at - 20 °C and used to determine chlorophyll a by Nusch (1980) extraction method. The filtered samples were stored in plastic bottles, kept at - 5 °C, and used to identify dissolved inorganic N, P, and Si concentrations. Inorganic phophorus corrected for arsenate/silicate interference was determined using the molybdate blue methods of Strickland & Parson (1972). Reactive silicate was verified by the molybdenum blue method (Strickland & Parson, 1972). Nitrogen as ammonium was determined by indophenol blue methods of Grasshoff et al. (1983). Nitrite was determined by sulphanilamide; nitrate was verified by reduction to nitrite in a Cd-Cu column, using automatic flow injection analyzer (ASIA Ismatec System) methods. The DIN (dissolved inorganic nitrogen) was determined by adding the ammonium, nitrite, and nitrate. Total phophorus was determined by acid digestion. This required persulphate and spectophotometric measurements of inorganic phosphate forms. Total nitrogen was established using basic persulphate digestion, in the same way as that for nitrate.
Statistical analyses were done through the non-parametric Mann-Whitney and Spearman correlation (p < 0.05), using STATISTICA 5.0.
RESULTS
Shallow water (< 1.5 m) and local wind action prevented water column stratification. Consequently, no significant chemical variation was observed between surface and bottom waters (Mann-Whitney U-test: p > 0.05, n = 12 x 13). The results and discussion then were focused on the average value between surface and bottom water.
During the sampling period, the water temperature data strongly evidenced seasonality in the Açu lagoon area, with higher temperatures in summer (c. 30 °C, November-March) and a decline between fall and winter, (average temperature: 23 °C) caused by air temperature variation (Fig. 2A). The water temperature data from November 1999 indicated the arrival of a cold front in the area, which is not usual in the spring.
Salinity, measured as electric conductivity, was varied substantially in time and space due to shallowness, macroclimatic conditions, and inputs of sea and fresh water. The spatial variation of conductivity values along the lagoon showed a clear chemical gradient going from less brackish water in the southern area (distal of the sand bar separating the lagoon from the sea; c. 26 mS.cm-1) to more brackish near the sand bar (average value: 45 mS.cm-1 in Station 5) due to the direct influence of the sea through the artificial sand bar openings, seawater percolation, and marine spray. Moreover, the highest electrical conductivity values found in Station 4 (generally 3 mS.cm-1 higher than in Station 5) suggested continuous sea-water infiltration through the sandland separating the lagoon from the sea. Decrease of conductivity values along the lagoon were noted during the first summer (Dec. 1999), when the rainfall levels rose. After December 1999, with reduced rainfall, electric conductivity values increased (Fig. 2B), showing that the evaporation rate (1844 mm.year-1, according to Meteorological Station of UFRRJ) exceeded freshwater input by rain (767 mm.y-1), groundwater, and the Quitinguta and São Bento canals. A rainy season characterized by low rainfall volume was registered between 2000 and 2001; increased conductivity resulted, with values reaching 63.7 mS.cm-1 (hypersalinity) on December 2000 at Station 4.
Both in space and time, pH and DO values showed a positive and significant correlation, while the correlation of pH with CDO was negative (Spearman test, r = 0.768 for pH and O2; pH and CO2 r = - 831, respectively; p < 0.0001; n = 62), which suggested that biological processes such as primary production and mineralization (decomposition/respiration) have a significant effect on these variations, particularly in stations 1, 3, and 5. The highest DO and pH values and the smallest CO2 values were observed at Station 5 (average values: 115% sat., 8.1, and 181% sat., respectively), probably due to development of benthonic macroalgae stands, which contribute significantly to regional primary production (Fig. 2D, 2E, 2F).
Reflecting both sea and fresh water influences, the total alkalinity values of Açu Lagoon presented minimum values of around 2 mEq.L-1 in the area closest to the sand bar, and values of around 4 mEq.L-1 in Station 1. From June to December 2000 this variable showed a certain homogeneity: all stations presented values of around 2 mEq.L-1 (Fig. 2C), probably due to reduced allochthonous organic matter (OM) inputs.
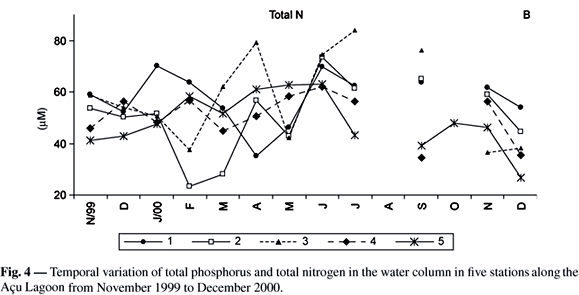
The temporal variation of concentrations of reactive silicate (Si-SiO4-3), dissolved inorganic nitrogen (N-NH4+ + N-NO2- + N-NO3-) and inorganic phosphorus (P-PO4-3) is shown in Fig. 3.
The reactive silicate showed highest concentrations in stations 1, 2, and 3 (around 200 µM) in December 1999, and lowest concentrations in stations 4 and 5 (around 35 µM). During the December 1999 rainy season, there was a higher input of continental nutrients into the lagoon through the canal, wetlands, and groundwater supplying the ecosystem with freshwater, thus establishing continental influence on dynamics of Si-SiO4-3 temporal variations (Fig. 3A). In addition, the data on this nutrient evidence the occurrence of an atypical summer between 2000 and 2001, when rainfall rates were lower than normal.
In general, dissolved inorganic nitrogen (DIN) presented a reduction from the area closest to the continent in the direction of the sand bar. The highest DIN value was 39 µM (Station 1, September 2000) and the smallest value was 1.6 µM (Station 5, February 2000; Fig. 3B). Inorganic phosphorus values were around 0.5 µM in all stations, except Station 5, which showed values above 0.9 µM throughout the sampling period (Fig. 3C).
Total nitrogen and total phosphorus values showed high values with no temporal and spacial patterns during the sampling period. The average total phosphorus value was 1.8 µM and the average total nitrogen value was 58 µM (Fig. 4A, 4B, respectively).
Stations 1 and 2 showed highest concentration of chlorophyll a (average ~ 6 and 7 µg.L-1). A spatial gradient was observed in Açu Lagoon, with higher values in the continental area close to the sand bar (Fig. 5A). The N:P inorganic ratio showed the same spatial behavior, with higher values in stations 1 and 2 (average: 72 and 24, respectively) than the other sampling stations (average: below 18). Station 5 showed very small values throughout the sampling period (average: 6) except for February 2000, when the N:P inorganic ratio was 82 (Fig. 5B).
DISCUSSION
In the area closer to the continent (stations 1 and 2), the freshwater input from the Quitinguta and São Bento canals probably carries a high amount of allochthonous dissolved (generously contributed to by well-developed stands of Typha domingensis) and particulate OM into the lagoon. The OM decomposition results in a darker-colored water and pH and DO reduction in the lagoons water column (Figs. 2D and 2E ). In Station 3, strong winds and the shallowness of the water column (< 0.5 m) combine to minimize organic material precipitation and accumulation; the sediment is, therefore, extremely sandy (average in lagoon = 33.5% DW OM, and in Station 3 = 1.4% DW OM; unpublished data). The low OM values of sediment in Station 3 is probably the result of organic matter mineralization in the water column, by which DO is consumed, thus producing CO2 and other acid compounds, in addition to releasing H+ ions into the water column. These effects are more evident in the rainy season (November-March) with the greater OM input from the drainage basin. In general, the sampling stations always presented DO subsaturation and CO2 supersaturation, specially in those closer to the continent in areas where the shores are colonized by mangrove species (stations 3 and 4) and Typha domingensis (stations 1 and 2). This vegetation possibly contributes the highest loads of organic material in the system, which in turn accounts for the elevated values of alkalinity caused by high anaerobic decomposition rates (sulphate reduction, ferrireduction, and denitrification) and the associated tendency to elevations in H+ consumption (Carmouze, 1994).
In the inner area, organic matter input and decomposition activity increased available DIN, especially in the N-NH4+ form. In the sand bar area, the development of macroalgae such as Ulva, which presents fast growth and high capacity to store nitrogen (Rosemberg & Ramus, 1982), may explain the low nitrogen concentration throughout the greater part of the year (March 2000-September 2000).
Although it coincided with the period of heaviest rainfall, and therefore of major nutrient discharges through runoff and input from the canals and groundwater, a reduction of inorganic nitrogen and phosphate concentration was observed in summer (December 1999-February 2000). This was probably because of the high assimilation rate associated with photosynthesis of the phytoplankton community, which developed intensely during this period (see chlorophyll a values in Figs. 4D, 3B, 3C).
High salinity near the sand bar promotes marine macroalgae genera such as Ulva, Cladophora, Gelidium, and Hypnea. These macroalgae compete for nutrients with other primary producers during the growth period (Smith & Horne, 1988; Fong et al., 1994). At the end of spring, when growth ceased and the seaweeds died, dissolved oxygen was consumed and pH and nutrient release into the water column decreased. The absence of macroalgae promoted the phytoplanktonic community, as is clear from increase in chlorophyll a and total phosphorus values, and decrease of those of inorganic phosphorus (Figs. 3, 4, and 5).
The region farthest from the sand bar (Station 1) differs from the other sampling areas by having the highest N:P inorganic ratio (Fig. 5A). Ammonification activity and the heavy input of organic material maintain a high concentration of dissolved inorganic nitrogen, whereas other processes, e.g., precipitation and phosphate immobilization in sediments related to the normally high dissolved iron concentration in this region account for these values being low throughout the year.
Inorganic nitrogen and phosphate concentrations in the water have often been used as nutrient limitation indications, with evidence suggesting that the optimum ratio may vary from 30:1 to 10:1, depending on the algae types present (Boynton et al., 1982; Atkinson & Smith, 1993). However, Fong et al. (1993) affirmed that other factors may limit phytoplankton growth. These authors further state that nutrient concentrations remain constant by virtue of a balance between the input and absorption pulses. Using the N:P ratio to evaluate nutrient limitation, only Station 1 in the Açu Lagoon presents phosphorus limitance, a point discussed below.
In general, the northern coastal lagoons of Rio de Janeiro State present low concentrations of dissolved nutrients because of fast cycling and assimilatory capacity of the biota (Suzuki et al.,1998; Machado & Knoppers, 1988). The low chlorophyll a values and relatively high availability of dissolved nutrients in the Açu Lagoon, when compared with those of other heavily anthropically- affected lagoons, e.g., the Grussaí and Imboassica lagoons (Table 1), suggest that the nutrient cycling process or nutrient input exceeds biota assimilation capacity. These facts corroborate the hypothesis that additional factors are limiting phytoplanktonic growth, although the N:P inorganic ratio indicated nutrient limitation only in Station 1. In stations 1 and 2 this factor is probably the reduced water clarity that restricts phytoplanktonic community growth. In stations 3 and 4, the high residence time of lagoon water (the last sand bar opening had occurred seven years before) favored the accumulation of organic compounds resulting from decomposing mangrove vegetation, which were proven to have inhibitory effects on freshwater phytoplankton communities and other microbial populations (Alongi, 1987; Lee et al., 1990; Serrano, 1992; Herrera-Silveira & Ramirez-Ramirez, 1996). In Station 5, high salinity favors macroalgae growth, which compared to the phytoplanktonic community shows higher competitive capacity. On the other hand, artificial eutrophication increase can result in phytoplanktonic community dominance, which alters internal nutrient and gases (CO2 and DO) dynamics as has been observed globally (Phlips et al., 2002; Menéndez et al., 2002; Nakata et al., 2000; Almunia et al., 1999; Castel et al., 1996; Sfriso & Pavoni, 1994).
CONCLUSION
The Açu Lagoon presents horizontal gradients under major or minor marine and continental influences, besides demonstrating seasonality patterns correlated with macroclimatic variables such as rainfall and evaporation, which determine the nutrient dynamics (input, availability, cycling).
The relation between spatial and temporal variations of salinity and inorganic nutrient concentrations showed a seasonal mineralization/input pattern of nutrients. The salinity affects organism distributions (macroalgae dominance in Station 5; high salinity and phytoplankton in the other stations). In the rainy period, macroalgae absence favored the phytoplanktonic community, thus changing the seasonal hydrochemistry dynamics in the Açu Lagoon.
Acknowledgements The authors wishes to thank Antônio Carlos de Oliveira Pessanha and André Taouil for invaluable help in the field and laboratory analyses, Eliane Brás for an earlier English revision, and the Universidade Estadual do Norte Fluminense (UENF) for making its facilities available. This research was funded by the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) This study is contribution number 007 from the graduate studies in Ecology and Natural Resources, Universidade Estadual do Norte Fluminense.
Received October 2, 2003 Accepted March 5, 2004 Distributed November 30, 2005
- ALMULIA, J., BASTERRETXEA, G., ARISTEGUI, J., & ULANOWICZ, R. G., 1999, Enthic-pelagic switching in coastal subtropical lagoon. Estuarine, Coastal and Shelf, Science, 52: 363-384.
- ALONGI, D. M., 1987, The influence of mangrove-derived tannins on intertidal meiobenthos in tropical estuaries. Oecologia, 71: 537-540.
- ATKINSON, M & SMITH, S., 1993, C:N:P ratios of bentic marine plants. Limnol. & Oceanogr., 28: 568-574.
- BOYNTON, W., KEMP, W., & KEEFE, C., 1982, Comparative analysis of nutrients and other factors influencing estuarine phytoplankton prodution, pp. 69-90. In: KENNEDY, V. (ed.), Estuarine Comparisons Academic Press, New York.
- CARMOUZE, J. P., 1994, Metabolismos dos ecossistemas aquáticos: fundamentos teóricos, métodos de estudo e análises químicas. 1st ed. São Paulo: Edgard Blucher/Fapesp. 254 p.
- CASTEL, J., CAUMETTE, P., & HERBERT, R., 1996, Eutrophication gradients in coastal lagoons as exemplified by the bassin dArcachon and the Etang du Prévost. Hydrobiologia, 329: ix - xxviii.
- ESTEVES, F. A, ISHII, I. H., & CAMARGO, A. F. M., 1984, Pesquisas limnológicas em 14 lagoas do estado do Rio de Janeiro, pp. 443-454. In: LACERDA, L. D., ARAÚJO, D. S. D., CERQUEIRA, R. & TURCQ, B. (org.) Restingas: origem, estrutura e processos. CEUFF, Niterói.
- ESTEVES, F. A. (Ed.), 1998, Ecologia das lagoas costeiras do Parque Nacional da Restinga de Jurubatiba e do Município de Macaé (RJ) NUPEM, Macaé, 442 p.
- FARJALLA, V. F., 1998, Nutrientes limitantes ao crescimento do Bacterioplâncton nas lagoas de Carapebus, Comprida, Cabiúnas e Iodada, pp. 147-158. In: ESTEVES, F. A. (Ed.), 1998, Ecologia das lagoas costeiras do Parque Nacional da Restinga de Jurubatiba e do Município de Macaé (RJ) NUPEM, Macaé, 442 p.
- FONG, P., ZEDLER, J. B. & DONOHOC, R. M., 1993, Nitrogen vs. phosphorus limitation of algal biomass in shallow lagoons. Limnol. Oceanogr., 38 (5): 906-92.
- FONG, P., ZEDLER, J. B. & DONOHOC, R. M., 1994, A simulation model of lagoon algae based on nitrogen competition and internal storage. Ecol.Monogr., 64: 224-247.
- GRAN, G., 1952, Determination of equivalent point in poletiometric titration. Analyst: 77: 661-671.
- GRASSHOFF, K. A, ERHHARDT, M. & KREMLING, K., 1983, Methods of seawater analysis, 2nd ed. Verlag Chemie, 419 pp.
- HERRERASILVERA, J. A & RAMÍREZ-RAMIREZ, J., 1996, Effects of natural phenolic material (tannin) on phytoplankton growth. Limnol. Oceanogr., 41 (3): 1108-1023.
- KNOPPERS, B., 1994, Aquatic primary production in coastal lagoon processes. In: KJERFVE, B. (ed). Coastal Lagoon Procesess, Elsevier Oceanography Series 60, Amsterdam, pp. 243-286.
- KOZLOWISHY-SUZUKI, B., 1998, Efeito do enriquecimento artificial e abertura de barra sobre a dinâmica das variáveis na lagoa de Imboassica, Macaé, RJ. Dissertação de mestrado, Rio de Janeiro, Universidade Federal do Rio de Janeiro, 94 p.
- LAMEGO, A. R., 1955, Geologia da quadrícula de Campos, São Tomé, Lagoa Feia e Xéxé. Bol. Div. Geol. Min.,154: 1-60.
- LEE, K. H., MORAN, M. A., BENNER, R. & HODSON, R. E., 1990, Influence of soluble components of red mangrove (Rhizophora mangle) leaves on microbial decomposition of structural (lignocellu-losic) leaf components in seawater. Mar. Sci. Bull., 42: 74-386.
- MACHADO, E. C. & KNOPPERS, B., 1988, Sediment oxygen consumption in an organic-rich subtropical lagoon, Brazil. The Science of Total Environment, 75: 341-349.
- MENÉNDEZ, M., HERNANDEZ, O., & COMIN, F. A., 2002, Spatial distribution and ecophysiological characteristics of macrophytes in a Mediterranean coastal lagoon. Estuarine, Coastal and Shelf Science,55: 403-413.
- NAKATA, K, HORIGUCHI, F., & YAMAMURO, M., 2000, Model study of lakes Shiji and Nakaumi a coupled coastal lagoon system. J. Mar. Syst. 26 (2):145-169.
- NIXON, S.W., 1981, Remineralization and nutrient cycling in coastal marine ecosystems. In: NEILSON, B. J. & CRONIN, L. E. (ed.) Estuaries and nutrients Clifton, New Jersey, Humana Press, pp. 111-138.
- NIXON, S. W., 1982, Nutrient dynamics, primary production and fisheries yields of lagoons. Oceanologia Acta, 4: 357-371.
- NUSCH, E. A., 1980, Comparison of different metods for chlorophyll and phaeopigment determination. Arch. Hydrobiol., 14: 14-36.
- PEREIRA, E. A., 2000, Metabolismo total e pelágico e hidroquímica na lagoa de Grussaí- São João da Barra RJ (Dissertação de mestrado) - Campos, Universidade Estadual do Norte Fluminense, 78 p.
- PHLIPS, E. J., BADYLAK, S., & GROSSKOPF, T., 2002, Factors affecting the abundance of phytoplankton in a restricted subtropical lagoon, the Indian River Lagoon, Florida. Estuarine, Coastal and Shelf Science, 55: 385-402.
- ROLAND, F., 1998, Produção fitoplanctônica em diferentes classes de tamanho nas lagoas Imboassica e Cabiúnas, pp. 159-175. In: ESTEVES, F. A. Ecologia das lagoas costeiras do Parque Nacional da Restinga de Jurubatiba e do Município de Macaé (RJ). NUPEM, Macaé, 442 p.
- ROSEMBERG, C. & RAMUS, J., 1982, Ecological growth strategies in the seaweed Gracilaria foliifera (Rodophyceae) and Ulva sp (Chlorophyceae) soluble nitrogen and reverve carbohydrates. Mar. Biol., 66: 251-247.
- SERRANO, L., 1992, Efecto de los compuestos polifenólicos, taninos y ligninas sobre las comunidades planctónicas de las lagunas peridunares del Parque Nacional de Doñana. Ph.D. Dissertation. Fac. de Biología, Univ. de Sevilla, Spain, 126 pp.
- SFRISO, A. & PAVONI, B., 1994, Macroalgae and phytoplankton competition in the central Venice lagoon. Environ. Tech., 55: 1-14.
- SMITH, D. W. & HORNE, A. J., 1988, Experimental measurement of resource competition between planktonic microalgae and macroalgae (seaweeds) in mesocosms simulating the San Francisco Bay Estuary, California. Hydrobiologia, 159: 259-268.
- STRICKLAND, J. D. H. & PARSON, T. R., 1972, A practical handbook of seawater analysis, 2nd ed. Fish. Res. Bd. Can. Bull., Ottawa, Canada, 122 p.
- SUZUKI, M. S., 1997, Aberturas da barra da lagoa de Grussaí, São João da Barra, RJ: Aspectos hidroquímicos, dinâmica da comunidade fitoplanctônica e metabolismo. Tese de Doutorado. Campos, Universidade Estadual do Norte Fluminense, 125 p.
- SUZUKI, M. S., OVALLE, A. R. C & PERREIRA, E. A., 1998, Effects of sand bar openings on some limnological variables in a hypertrophic tropical coastal lagoon of Brazil. Hydrobiologia,368: 111-122.
- SUZUKI, M. S., FIGUEIREDO, R. O., CASTRO, S. C., SILVA, C. F., PERREIRA, E. A., SILVA, J. A. & ARAGON, G. T., 2002, Sandbar opening in a coastal lagoon ( Iquiparí), in the Northern region of Rio de Janeiro State: Hydrological and Hydrochemical changes. Braz. J. Biol.,62(1): 51-62.
Publication Dates
-
Publication in this collection
06 Mar 2006 -
Date of issue
Nov 2005
History
-
Accepted
05 Mar 2004 -
Received
02 Oct 2003