Abstracts
The taxonomy/systematics of the Erythrinidae fish is still imprecise, with several doubts on their relationships. Karyotypes and chromosomal characteristics of some species of the Hoplias lacerdae group (Erythrinidae), from different Brazilian hydrographic basins and pisciculture stations, were analyzed in the present study, using conventional Giemsa staining, C-banding, silver staining, Mithramycin and Distamycin/DAPI fluorochromes, and fluorescent in situ hybridization (FISH). A diploid chromosome number of 2n = 50 and karyotypes composed of meta- and submetacentric chromosomes without sex-related differences were found. Only one active NOR (Nucleolar Organizer Region) site was found, which was identified by silver staining (Ag-NOR) and FISH, located on the chromosome pair 11, although additional 45S rDNA sites were also mapped on other chromosome pairs only by FISH. The Ag-NOR of the chromosome pair 11 was found to be GC-rich, appearing positive after Mithramycin staining. Mithramycin-positive/DAPI-negative sites were also observed in the centromeric/pericentomeric regions of the chromosome pairs 4, 6, 15, and 19, which have also affinity to silver nitrate. However, these four sites were not detected by FISH with the rDNA probe, indicating to be only argentophilic GC-rich heterochromatic regions. Chromosome data show that the karyotype evolution in Hoplias lacerdae group is relatively conserved and follows a particular pathway concerning the other Erythrinidae fishes, such as Hoplias malabaricus, Hoplerythrinus unitaeniatus, and Erythrinus erythrinus, in which polytypic karyotypes are found. Thus, the H. lacerdae group shows chromosome features that are not closely related to those of the congeneric H. malabaricus group. These finds, together with genetic and morphologic data, are important tools to be considered in a major revision of the Erythrinidae family, as well as for conservation programs.
erythrinid; chromosome features; karyotypic evolution; cytotaxonomy
A taxonomia dos Erythrinidae encontra-se ainda por ser totalmente esclarecida, existindo várias dúvidas sobre suas relações filogenéticas. No presente estudo, foram analisados os cariótipos e características cromossômicas de algumas espécies do grupo Hoplias lacerdae (Erythrinidae), de diferentes bacias hidrográficas do Brasil e estações de piscicultura, empregando-se a coloração Giemsa convencional, bandamento C, coloração com nitrato de prata, fluorocromos Mitramicina e Distamicina/DAPI, e hibridação fluorescente in situ (FISH). O número diplóide de 2n = 50 cromossomos foi constante, sendo os cariótipos formados por cromossomos de dois braços, meta- e submetacêntricos, sem diferenciação relacionada ao sexo. Foi identificado apenas um sítio ativo de região organizadora de nucléolo (NOR), presente no par cromossômico no. 11, identificado tanto pela coloração com nitrato de prata (Ag-NOR) como por FISH com sonda de DNAr 45S, embora alguns sítios adicionais de DNAr tenham sido identificados em outros pares de cromossomos, apenas por FISH. O sítio de Ag-NOR no cromossomo 11 mostrou-se rico em pares de bases GC, sendo Mitramicina-positivo. Sítios Mitramicina-positivos/DAPI-negativos foram igualmente observados nas regiões centroméricas/pericentroméricas dos pares cromossômicos 4, 6, 15 e 19, os quais mostraram também afinidade pelo nitrato de prata. Contudo, estes quatro sítios não foram evidenciados por FISH com sonda de DNAr 45S, indicando serem somente regiões de heterocromatina GC-rica, com afinidade pela prata. Os dados cromossômicos mostram que a evolução cariotípica no grupo Hoplias lacerdae é relativamente conservada e segue um comportamento particular em relação aos outros eritrinídeos, como Hoplias malabaricus, Hoplerythrinus unitaeniatus e Erythrinus erythrinus, nos quais foram encontrados cariótipos politípicos. Assim sendo, o grupo H. lacerdae mostra características cromossômicas discordantes daquelas evidenciadas pelo grupo congenérico H. malabaricus. Estes dados, juntamente com os outros dados genéticos e morfológicos, constituem ferramentas importantes a serem consideradas em uma revisão ampla da família Erythrinidae, assim como para programas de conservação.
eritrinídeos; características cromossômicas; evolução cariotípica; citotaxonomia
Evolutionary cytogenetics of the Hoplias lacerdae, Miranda Ribeiro, 1908 group. A particular pathway concerning the other Erythrinidae fish
Citogenética evolutiva do grupo Hoplias lacerdae. Um comportamento particular em relação aos outros peixes eritrinídeos
Morelli, S.I; Vicari, MR.II; Bertollo, LAC.III,* * e-mail: bertollo@power.ufscar.br
ILaboratório de Citogenética e Mutagênese, Instituto de Genética e Bioquímica, Universidade Federal de Uberlândia, Av. Pará, 1720, Campus Umuarama, CEP 38400-902, Uberlândia, MG, Brazil
IIDepartamento de Biologia Estrutural, Molecular e Genética, Universidade Estadual de Ponta Grossa, CEP 84030-900, Ponta Grossa, PR, Brazil
IIIDepartamento de Genética e Evolução, Universidade Federal de São Carlos UFSCar, CP 676, CEP 13565-905, São Carlos, SP, Brazil
ABSTRACT
The taxonomy/systematics of the Erythrinidae fish is still imprecise, with several doubts on their relationships. Karyotypes and chromosomal characteristics of some species of the Hoplias lacerdae group (Erythrinidae), from different Brazilian hydrographic basins and pisciculture stations, were analyzed in the present study, using conventional Giemsa staining, C-banding, silver staining, Mithramycin and Distamycin/DAPI fluorochromes, and fluorescent in situ hybridization (FISH). A diploid chromosome number of 2n = 50 and karyotypes composed of meta- and submetacentric chromosomes without sex-related differences were found. Only one active NOR (Nucleolar Organizer Region) site was found, which was identified by silver staining (Ag-NOR) and FISH, located on the chromosome pair 11, although additional 45S rDNA sites were also mapped on other chromosome pairs only by FISH. The Ag-NOR of the chromosome pair 11 was found to be GC-rich, appearing positive after Mithramycin staining. Mithramycin-positive/DAPI-negative sites were also observed in the centromeric/pericentomeric regions of the chromosome pairs 4, 6, 15, and 19, which have also affinity to silver nitrate. However, these four sites were not detected by FISH with the rDNA probe, indicating to be only argentophilic GC-rich heterochromatic regions. Chromosome data show that the karyotype evolution in Hoplias lacerdae group is relatively conserved and follows a particular pathway concerning the other Erythrinidae fishes, such as Hoplias malabaricus, Hoplerythrinus unitaeniatus, and Erythrinus erythrinus, in which polytypic karyotypes are found. Thus, the H. lacerdae group shows chromosome features that are not closely related to those of the congeneric H. malabaricus group. These finds, together with genetic and morphologic data, are important tools to be considered in a major revision of the Erythrinidae family, as well as for conservation programs.
Keywords: erythrinid, chromosome features, karyotypic evolution, cytotaxonomy.
RESUMO
A taxonomia dos Erythrinidae encontra-se ainda por ser totalmente esclarecida, existindo várias dúvidas sobre suas relações filogenéticas. No presente estudo, foram analisados os cariótipos e características cromossômicas de algumas espécies do grupo Hoplias lacerdae (Erythrinidae), de diferentes bacias hidrográficas do Brasil e estações de piscicultura, empregando-se a coloração Giemsa convencional, bandamento C, coloração com nitrato de prata, fluorocromos Mitramicina e Distamicina/DAPI, e hibridação fluorescente in situ (FISH). O número diplóide de 2n = 50 cromossomos foi constante, sendo os cariótipos formados por cromossomos de dois braços, meta- e submetacêntricos, sem diferenciação relacionada ao sexo. Foi identificado apenas um sítio ativo de região organizadora de nucléolo (NOR), presente no par cromossômico no. 11, identificado tanto pela coloração com nitrato de prata (Ag-NOR) como por FISH com sonda de DNAr 45S, embora alguns sítios adicionais de DNAr tenham sido identificados em outros pares de cromossomos, apenas por FISH. O sítio de Ag-NOR no cromossomo 11 mostrou-se rico em pares de bases GC, sendo Mitramicina-positivo. Sítios Mitramicina-positivos/DAPI-negativos foram igualmente observados nas regiões centroméricas/pericentroméricas dos pares cromossômicos 4, 6, 15 e 19, os quais mostraram também afinidade pelo nitrato de prata. Contudo, estes quatro sítios não foram evidenciados por FISH com sonda de DNAr 45S, indicando serem somente regiões de heterocromatina GC-rica, com afinidade pela prata. Os dados cromossômicos mostram que a evolução cariotípica no grupo Hoplias lacerdae é relativamente conservada e segue um comportamento particular em relação aos outros eritrinídeos, como Hoplias malabaricus, Hoplerythrinus unitaeniatus e Erythrinus erythrinus, nos quais foram encontrados cariótipos politípicos. Assim sendo, o grupo H. lacerdae mostra características cromossômicas discordantes daquelas evidenciadas pelo grupo congenérico H. malabaricus. Estes dados, juntamente com os outros dados genéticos e morfológicos, constituem ferramentas importantes a serem consideradas em uma revisão ampla da família Erythrinidae, assim como para programas de conservação.
Palavras-chave: eritrinídeos, características cromossômicas, evolução cariotípica, citotaxonomia.
1. Introduction
Erythrinidae is a small freshwater fish family widely distributed throughout South America, with only three genera (Hoplias, Hoplerythrinus, and Erythrinus), with a low number of nominal species. However, the taxonomy/systematics of this family is not well understood. Hoplias malabaricus (Bloch, 1794), and H. lacerdae Miranda Ribeiro, 1908, for example, belong to two distinct species groups, the H. malabaricus and the H. lacerdae groups, respectively, in which several species do not present a definitive taxonomy (Oyakawa, 1990, 2003).
The taxonomic doubts are also supported by several cytogenetic data showing a great karyotypic diversity among different populations, indicating the occurrence of distinct hidden species. Indeed, H. malabaricus, Hoplerythrinus unitaeniatus (Agassiz, 1829), and Erythrinus erythrinus (Bloch and Schneider, 1801) have different karyotypes (cytotypes) easily characterized by the chromosome number and composition, as well as by the presence of distinct sex chromosome systems (Bertollo et al., 2000; Giuliano-Caetano et al. , 2001; Bertollo et al., 2004).
Concerning the H. lacerdae group, chromosome data are only available for specimens from two Brazilian populations. Although these two populations were widely isolated, one of them from the Southeastern region and the other one from the North region, they showed similar karyotypes with 2 n = 50 meta- and submetacentric chromosomes (Bertollo et al., 1978; Bertollo and Moreira-Filho, 1983). The present study deals with chromosome data from other distinct populations, using conventional analyses, fluorochrome staining, banding methods, and fluorescent in situ hybridization with an rDNA probe, in order to improve the cytogenetic data of the H. lacerdae group. The obtained data was also used to verify if their general karyotypic macrostructure remains stable or, on the contrary, if a karyotypic diversity is also found among populations, as occur in other erythrinids.
2. Material and Methods
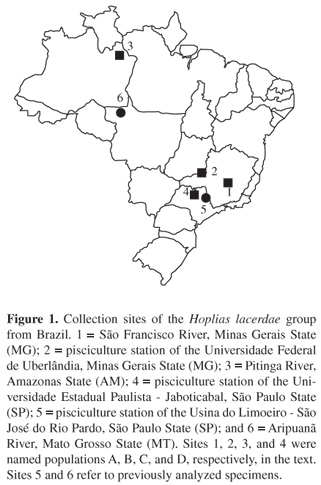
Specimens of the H. lacerdae group were collected in four distinct Brazilian localities (Figure 1): 1) São Francisco River Três Marias region, Minas Gerais State (14 males and 3 females), 2) pisciculture station of the Universidade Federal de Uberlândia Uberlândia, Minas Gerais State (11 males and 14 females), 3) Pitinga River left bank of the Amazon River in the Uatumã River drainage, Amazonas State (1 male and 1 female), and 4) pisciculture station of the Universidade Estadual Paulista UNESP Jaboticabal, São Paulo State (1 male). The specimens were deposited at the Laboratório de Citogenética, Departamento de Genética e Evolução, Universidade Federal de São Carlos, São Paulo State, Brazil. For convenience, these four populations were named A, B, C, and D, respectively. The sites 5 and 6 refer to specimens previously analyzed (Figure 1), and the respective populations were named E and F (Table 1).
Chromosome preparations were obtained from the anterior kidney cells after in vivo treatment with colchicine (Bertollo et al., 1978). Some specimens were previously treated with yeast suspension (Lee and Elder, 1980), or cobalt chloride solution (Cucchi and Baruffaldi, 1990) for mitotic stimulation. In addition to conventional Giemsa staining, the chromosomes were analyzed after Mithramycin (Schmid, 1980) and Distamycin/DAPI (Schweizer, 1980) staining, in order to detect GC- and AT-rich sites on the chromosomes, respectively. Silver nitrate staining (Howell and Black, 1980), and C-banding (Sumner, 1972), were also used to analyze the active nucleolar organizer regions (Ag-NORs) and the C-positive heterochromatin, respectively. The rDNA sites were mapped on the chromosomes using the fluorescent in situ hybridization (FISH) with an rDNA probe, according to Pinkel et al. (1986). The probe consisted of a mixture of two recombinant plasmids (HM123 and HM456) containing rDNA fragments of Xenopus laevis (Meunier-Rotival et al., 1979) that comprise the totality of the 45S rDNA gene.
The diploid chromosome number was found for females and males; at least 30 metaphases per each specimen were analyzed. The classification of the chromosomes followed Levan et al. (1964).
3. Results
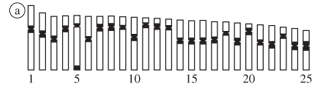
The specimens from the four populations possessed 2n = 50 chromosomes and the karyotypes were composed of meta- and submetacentric chromosomes with a very similar structure. The first chromosome pair is characterized by a larger size, while the remaining pairs present a gradual decrease in the size (Figure 2). Some minor interpopulational differences were observed only in a few chromosome pairs (Figure 2 - idiograms), related to the position of the centromere.
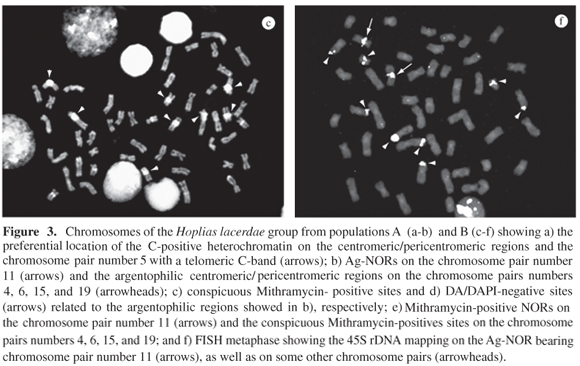
C-bands were located in the centromeric/pericentromeric region of all the chromosome pairs and several of them were very conspicuous. An exception was the specimens from population C which did not show satisfactory results for C-banding. In addition, chromosome number 5 showed a telomeric C-band on the long arm (Figures 3a, 4a).
One Ag-NOR site was identified close to the centromere, on the long arms of the chromosome pair number 11, in the four populations (Figures 3b, 4b), which is Mithramycin-positive (Figure 3e). However, the centromeric/pericentromeric sites of the chromosome pairs 4, 6, 15, and 19 were also highlighted after silver staining (Figures 3b, 4b), showing a conspicuously bright fluorescence after Mythramycin staining (Figures 3c, 3e, 4c) and negative fluorescence after Distamycin/DAPI staining (Figures 3d, 4d). Fluorescent in situ hybridization mapped the 45S rDNA on the Ag-NOR of the 11th chromosome pair, as well as in some other chromosome pairs, but not on the centromeric/pericentromeric regions of the chromosomes 4, 6, 15, and 19 (Figure 3f).
No karyotypic differentiation was observed between the males and females from the populations A, B, and C (Figure 2). In addition, the male karyotype from population D did not suggest the occurrence of chromosome heterogamety.
4. Discussion
All analyzed specimens have 2n = 50 meta- and submetacentric chromosomes, showing the same macro karyotype structure. Some minor differences were observed in the centromere position of the chromosome pair number 20 in the populations A and C, and in the chromosome pair number 23 in the populations B and D. However, it is difficult to conclude if these features indicate real differentiations among the populations or if they would be resultant of technical problems, such as the correct pairing in the karyotypes or the degree of the chromosomal condensation. Similar karyotypes were also previously observed in the specimens from Usina do Limoeiro pisciculture station, São José do Rio Pardo, São Paulo State (Bertollo et al., 1978) and from the Aripuanã River - Aripuanã, Mato Grosso State (Bertollo and Moreira-Filho, 1983). This karyotype stability is also reinforced by the similar pattern of the heterochromatin distribution, which is shared by the specimens of the distinct populations.
It is generally accepted that the nuclear organizer regions are GC-rich regions in lower vertebrates (Schmid and Guttenbach, 1988), i.e., they are Mithramycin- or Chromomycin A3-positive sites, which is also true for specimens of the H. lacerdae group. Fluorescent in situ hybridization, carried out in the population B, mapped the 45S rRNA genes in the NOR site on the chromosome pair 11, in addition to some sites in other chromosomes. These data indicate the occurrence of multiple NORs, although only that on the 11th pair was silver stained, i.e., an active NOR, which was also observed in the other analyzed specimens. Thus, all the additional 45S rDNA sites found in the population B appear to be under genetic inactivation. Until now, there is no available information on ribosomal gene mapping by FISH in the other populations.
On the other hand, the four argentophilic regions in the chromosome pairs 4, 6, 15, and 19 were Mithramycin-positive, a fact that, at first, could also indicate the occurrence of nucleolar organizer regions. However, ribosomal cistrons were not detected in these four chromosomes after FISH. Similar cases in which GC-rich sites were not correlated with NORs have also been observed in few other fish, such as in species of the genus Schizodon (Martins and Galetti Jr., 1998), Liposarcus and Leporinus (Artoni et al., 1999), Eigenmannia (Almeida-Toledo et al., 2000), and some Loricariidae (Kavalco et al., 2004). According to Sumner (1990), chromosome staining with silver nitrate may also highlight argentophilic heterochromatic sites, which can explain the present results.
The comparison between male and female karyotypes from populations A-C, as well as the analysis of the male karyotype from population D, indicate the absence of heteromorphic sex chromosomes. Bertollo et al. (1978), suggested an XX/XY sex chromosome system for Hoplias cf. lacerdae from Usina do Limoeiro pisciculture station, based on the comparative analysis between male and female karyotypes. Considering that it was the first study in this fish group using only conventional Giemsa stained chromosomes, the reevaluation of the proposed sex chromosome system would be necessary using more informative chromosomal analyses for its confirmation. Unfortunately, specimens from that locality are not more available hindering this confirmation. Thus, at this time, the Hoplias lacerdae group emerges as an Erythrinidae taxon where a heteromorphic sex chromosome system seems not to occur.
Despite the reduced number of species, several questions about the taxonomic state of the H. lacerdae group, as well as the whole Erythrinidae family, are still to be solved. Oyakawa (1990, 2003) recognizes H. lacerdae and H. brasiliensis (Agassiz, 1829) as valid species in the lacerdae group, which are found in the Ribeira de Iguape River (São Paulo and Paraná States), and in coastal basins (Bahia State), respectively. In addition, other species not yet formally described occur in several other South American hydrographic systems, such as Amazon basin, upper Paraná and upper São Francisco basins, Uruguay basin, and South Brazil. Strictly, none of the specimens from natural populations with available chromosomal data (Figure 1) must correspond to H. lacerdae and H. brasiliensis. Indeed, besides the fish from pisciculture stations (samples 2, 4, and 5) which have unknown origin, those from natural environments (samples 1, 3, and 6) are not representative specimens from the hydrographic basins where H. lacerdae and H. brasiliensis occur. Thus, it is very likely that they correspond to undescribed species from the Eastern and Northern Brazilian regions (São Francisco River, and Aripuanã and Pitinga Rivers, respectively).
Our data demonstrate that the karyotype evolution in the H. lacerdae group is characterized by a macrostructural homogeneity, contrasting with other Erythrinidae species, such as H. malabaricus, Hoplerythrinus unitaeniatus, and Eryhtrinus erythrinus in which polytypic karyotypes are found. Indeed, many populations of the H. malabaricus group show distinct karyotypes (cytotypes) identified by their chromosome number and/or structure. The same is valid for the nominal species H. unitaeniatus and E. erythrinus from different Brazilian rivers. In addition, several populations of H. malabaricus and E. erythrinus also differ in the occurrence of distinct sex chromosome systems (XX/XY, X1X1X2X2/X1X2Y, and XX/XY1Y2), or in the absence of chromosome differentiation between sexes (Table 1). All these chromosomal features of H. malabaricus, H. unitaeniatus, and E. erythrinus are found among isolated populations in nature, or even among sympatric cytotypes without evidences of hybridization, showing that the chromosomal rearrangements are already fixed, suggesting the existence of several unnamed species (Bertollo et al., 2000; Diniz and Bertollo, 2003; Bertollo et al., 2004). Thus, it is remarkable that two very divergent karyotype pathways can be characterized within the genus Hoplias, i.e., H. malabaricus group, with a pronounced chromosome diversity and a smaller diploid number (2n = 39-42 chromosomes), and H. lacerdae group, with a relative chromosomal homogeneity and a higher diploid number (2 n = 50 chromosomes). In addition, in the H. lacerdae group only a single chromosome pair bearing active NORs (Ag-NORs) was found, in contrast to what occurs in the H. malabaricus group, where all the cytotypes showed multiple Ag-NORs (Bertollo, 1996), as well as in the other erythrinids (Table 1). Thus, H. malabaricus and H. lacerdae groups do not appear closely related if we consider their chromosomal features. As the taxonomy and systematics of the Erythrinidae fish are still imprecise showing several doubts about their relationships, the current data, together with other genetic and morphologic data, will be useful tools for the revision of this fish family, as well as for conservation programs.
Acknowledgments The authors thank Conselho Nacional Científico e Tecnológico (CNPq) for financial support, Dr. Yoshimi Sato (CODEVASF), and Dr. Jorge Porto (INPA) for providing specimens from the São Francisco River (MG) and Pitinga River (AM), respectively.
Received May 31, 2007
Accepted September 19, 2007
Distributed December 1, 2007
- ALMEIDA-TOLEDO, LF., FORESTI, F., DANIEL, MFZ. and TOLEDO-FILHO, SA., 2000. Sex chromosome evolution in fish: the formation of the neo-Y chromosome in Eigenmannia (Gymnotiformes). Chromosoma, vol. 109, p. 197-200.
- ARTONI, RF., MOLINA, WF., BERTOLLO, LAC. and GALETTI JR, PM., 1999. Heterochromatin analysis in the fish species Liporsarcus anisitsi (Siluriformes) and Leporinus elongatus (Characiformes). Genet. Mol. Biol., vol. 2, p. 39-44.
- BERTOLLO, LAC., 1996. The nucleolar organizer regions of Erythrinidae fish. An uncommon situation in the genus Hoplias Cytologia, vol. 61, p. 75-81.
- -, 2007. Chromosome evolution in the Neotropical Erythrinidae fish family: an overview. In PISANO, E., OZOUF-COSTAZ, C., FORESTI, F. and KAPOOR, BG. (eds.). Fish cytogenetics Oxford and IBH Publishing Co., New Delhi, p. 195-211.
- BERTOLLO, LAC., TAKAHASHI, CS. and MOREIRA-FILHO, O., 1978. Cytotaxonomic considerations on Hoplias lacerdae (Pisces, Erythrinidae). Brazil. J. Genet., vol. 1, p. 103-120.
- BERTOLLO, LAC. and MOREIRA-FILHO, O., 1983. Possível ocorrência de uma nova espécie do gênero Hoplias (Pisces, Erythrinidae): evidências morfológicas e citogenéticas. Cienc. Cult. (Suppl.), vol. 35, p. 685.
- BERTOLLO, LAC., BORN, GG., DERGAM, JA., FENOCCHIO, AS. and MOREIRA-FILHO, O., 2000. A biodiversity approach in the neotropical Erythrinidae fish, Hoplias malabaricus Karyotypic survey, geographic distribution of cytotypes and cytotaxonomic considerations. Chromosome Res., vol. 8, p. 603-613.
- BERTOLLO, LAC., OLIVEIRA, C., MOLINA, WF., MARGARIDO, VP., FONTES, MS., PASTORI, MC., FALCÃO, JN. and FENOCCHIO, AS., 2004. Chromosome evolution in the erythrinid fish, Erythrinus erythrinus (Teleostei, Characiformes). Heredity, vol. 93, p. 228-233.
- CUCCHI, C. and BARUFFALDI, A., 1990. A new method for karyological studies in teleost fishes. J. Fish Biol, vol. 37, p. 71-75.
- DINIZ, D. and BERTOLLO, LAC., 2003. Karyotypic studies on Hoplerythrinus unitaeniatus (Pisces, Erythrinidae) populations. A biodiversity analysis. Caryologia, vol. 3, p. 303-313.
- GIULIANO-CAETANO, L., JORGE, LC., MOREIRA-FILHO, O. and BERTOLLO, LAC., 2001. Comparative cytogenetic studies on Hoplerythrinus unitaeniatus populations (Pisces, Erythrinidae). Cytologia, vol. 66, p. 39-43.
- HOWELL, WM. and BLACK, DA., 1980. Controlled silver-staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method. Experientia, vol. 36, p. 1014-1015.
- KAVALCO, KR., PAZZA, R., BERTOLLO, LAC. and MOREIRA-FILHO, O., 2004. Heterochromatin characterization of four species of the family Loricariidae (Siluriformes). Hereditas, vol. 141, p. 237-242.
- LEE, MR. and ELDER, FFB., 1980. Yeast stimulation of bone marrow mitosis for cytogenetic investigations. Cytogenet. Cell Genet, vol. 26, p. 36-40.
- LEVAN, A., FREDGA, K. and SANDBERG, AA., 1964. Nomenclature for centromeric position on chromosomes. Hereditas, vol. 52, p. 201-220.
- MARTINS, C. and GALETTI JR, PM., 1998. Karyotype similarity between two sympatric Schizodon fish species (Anostomidae, Characiformes) from the Paraguay river basin. Genet. Mol. Biol, vol. 21, p. 355-360.
- MEUNIER-ROTIVAL, M., CORTADAS, J., MACAYA, G. and BERNARDI, G., 1979. Isolation and organization of calf ribosomal DNA. Nucleic Acids Res., vol. 6, p. 2109-2123.
- OYAKAWA, OT., 1990. Revisão sistemática das espécies do gênero Hoplias grupo lacerdae da Amazônia Brasileira e região leste do Brasil. 114p. (Dissertação de Mestrado) - Universidade de São Paulo, São Paulo, SP.
- -, 2003. Family Erythrinidae. In REIS, RE., KULLANDER, SO. and FERRARIS JR, CJ. (eds.). Check list of the freshwater fishes of South and Central America EDIPUCRS, Porto Alegre, p. 238-240.
- PINKEL, D., STRAUME, T. and GRAY, JW., 1986. Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc. Natl. Acad. Sci, USA, vol. 83, p. 2934-2938.
- SCHMID, M., 1980. Chromosome banding in Amphibia. IV. Differentiation of GC- and AT-rich chromosome regions in Anura. Chromosoma, vol. 77, p. 83-103.
- SCHMID, M. and GUTTENBACH, M., 1988. Evolutionary diversity of reverse (R) fluorescent chromosome bands in vertebrates. Chromosoma, vol. 97, p. 101-114.
- SCHWEIZER, D., 1980. Simultaneous fluorescent staining of R bands and specific heterochromatic regions (DA/DAPI bands) in human chromosomes. Cytogenet. Cell Genet, vol. 27, p. 190-193.
- SUMNER, AT., 1972. A simple technique for demonstrating centromeric heterochromatin. Exp. Cell Res, vol. 75, p. 304-306
- -, 1990. Chromosome banding Unwin Hyman Ltd., London. 434p.
Publication Dates
-
Publication in this collection
11 Feb 2008 -
Date of issue
Dec 2007
History
-
Received
31 May 2007 -
Accepted
19 Sept 2007