Abstracts
BACKGROUND: Healing is a complex process that involves cellular and biochemical events. Several medicines have been used in order to shorten healing time and avoid aesthetic damage. OBJECTIVE: to verify the topical effect of ascorbic acid for the healing of rats' skin wounds through the number of macrophages, new vessels and fibroblast verifications in the experimental period; and analyse the thickness and the collagen fibre organization in the injured tissue. METHODS: Male Rattus norvegicus weighing 270 ± 30 g were used. After thionembutal anesthesia, 15 mm transversal incisions were made in the animals' cervical backs. They were divided into two groups: Control Group (CG, n = 12) - skin wound cleaned with water and soap daily; Treated Group (TG, n = 12) - skin wound cleaned daily and treated with ascorbic acid cream (10%). Samples of skin were collected on the 3rd, 7th and 14th days. The sections were stained with hematoxylin-eosin and picrosirius red for morphologic analysis. The images were obtained and analysed by a Digital Analyser System. RESULTS: The ascorbic acid acted on every stage of the healing process. It reduced the number of macrophages, increased the proliferation of fibroblasts and new vessels, and stimulated the synthesis of thicker and more organized collagen fibres in the wounds when compared to CG. CONCLUSION: Ascorbic acid was shown to have anti-inflammatory and healing effects, guaranteeing a suiTable environment and conditions for faster skin repair.
wound healing; ascorbic acid; collagen; fibroblasts; skin
FUNDAMENTOS: A cicatrização é um processo complexo que envolve eventos celulares e bioquímicos. Vários medicamentos têm sido empregados na tentativa de abreviar a cicatrização e evitar danos estéticos. OBJETIVO: verificar o efeito tópico do ácido ascórbico no processo de cicatrização de feridas cutâneas de ratos através da verificação do número de macrófagos, neovasos e fibroblastos presentes no período experimental; e analisar a espessura e a organização das fibras colágenas no tecido lesado. MÉTODOS: Foram utilizados Rattus norvegicus, machos, pesando 270 ± 30 g. Foi realizada incisão transversal na pele da região dorso-cervical de 15 mm de comprimento, após anestesia com Thionembutal. Os animais foram divididos em 2 grupos: grupo controle (GC, n = 12), feridas higienizadas diariamente com água e sabão; grupo tratado (GT, n = 12), feridas higienizadas e tratadas com creme de ácido ascórbico (10%). Os fragmentos para análise histológica foram coletados no 3º, 7º e 14º dia, e as lâminas coradas com hematoxilina-eosina e picrosírius red para análise morfológica. As imagens foram capturadas e analisadas por um sistema digitalizador. RESULTADOS: O ácido ascórbico atuou em todas as etapas da cicatrização, diminuindo o número de macrófagos, aumentando a proliferação dos fibroblastos e neovasos, e favorecendo a deposição de fibras colágenas mais espessas e organizadas nas feridas. CONCLUSÃO: O ácido ascórbico mostrou ter efeito antiinflamatório e cicatrizante, promovendo ambiente e condições favoráveis para a reparação tecidual, o que abreviou o tempo da cicatrização.
cicatrização de feridas; ácido ascórbico; colágeno; fibroblastos; pele
BIOLOGY
Ascorbic acid for the healing of skin wounds in rats
Ácido ascórbico na cicatrização de feridas cutâneas em ratos
Lima, CC.I; Pereira, APC.I; Silva, JRF.I; Oliveira, LS.I; Resck, MCC.I, II; Grechi, CO.III; Bernardes, MTCP.IV; Olímpio, FMP.IV; Santos, AMM.V; Incerpi, EK.VI; Garcia, JADI, * * e-mail: jadgarcia@uol.com.br
INúcleo de Pesquisa Farmacologia e Cirurgia Experimental, Universidade José do Rosário Vellano - UNIFENAS, Rod. MG 179, Km 0, CP 23, CEP 37130-000, Alfenas, MG, Brazil
IIDepartamento de Medicina Veterinária, Universidade José do Rosário Vellano - UNIFENAS, Rod. MG 179, Km 0, CP 23, CEP 37130-000, Alfenas, MG, Brazil
IIIEscola Estadual Judithy Vianna, Rua Gabriel M. Silva, 711, Alfenas, MG, Brazil
IVDepartamento de Farmácia, Universidade José do Rosário Vellano - UNIFENAS, Rod. MG 179, Km 0, CP 23, CEP 37130-000, Alfenas, MG, Brazil
VDepartamento de Biomedicina, Universidade José do Rosário Vellano - UNIFENAS, Rod. MG 179, Km 0, CP 23, CEP 37130-000, Alfenas, MG, Brazil
VIDepartamento de Farmacologia, Universidade de Três Corações - UNINCOR, Três Corações, MG, Brazil
ABSTRACT
BACKGROUND: Healing is a complex process that involves cellular and biochemical events. Several medicines have been used in order to shorten healing time and avoid aesthetic damage.
OBJECTIVE: to verify the topical effect of ascorbic acid for the healing of rats' skin wounds through the number of macrophages, new vessels and fibroblast verifications in the experimental period; and analyse the thickness and the collagen fibre organization in the injured tissue.
METHODS: Male Rattus norvegicus weighing 270 ± 30 g were used. After thionembutal anesthesia, 15 mm transversal incisions were made in the animals' cervical backs. They were divided into two groups: Control Group (CG, n = 12) - skin wound cleaned with water and soap daily; Treated Group (TG, n = 12) - skin wound cleaned daily and treated with ascorbic acid cream (10%). Samples of skin were collected on the 3rd, 7th and 14th days. The sections were stained with hematoxylin-eosin and picrosirius red for morphologic analysis. The images were obtained and analysed by a Digital Analyser System.
RESULTS: The ascorbic acid acted on every stage of the healing process. It reduced the number of macrophages, increased the proliferation of fibroblasts and new vessels, and stimulated the synthesis of thicker and more organized collagen fibres in the wounds when compared to CG.
CONCLUSION: Ascorbic acid was shown to have anti-inflammatory and healing effects, guaranteeing a suiTable environment and conditions for faster skin repair.
Keywords: wound healing, ascorbic acid, collagen, fibroblasts, skin.
RESUMO
FUNDAMENTOS: A cicatrização é um processo complexo que envolve eventos celulares e bioquímicos. Vários medicamentos têm sido empregados na tentativa de abreviar a cicatrização e evitar danos estéticos.
OBJETIVO: verificar o efeito tópico do ácido ascórbico no processo de cicatrização de feridas cutâneas de ratos através da verificação do número de macrófagos, neovasos e fibroblastos presentes no período experimental; e analisar a espessura e a organização das fibras colágenas no tecido lesado.
MÉTODOS: Foram utilizados Rattus norvegicus, machos, pesando 270 ± 30 g. Foi realizada incisão transversal na pele da região dorso-cervical de 15 mm de comprimento, após anestesia com Thionembutal. Os animais foram divididos em 2 grupos: grupo controle (GC, n = 12), feridas higienizadas diariamente com água e sabão; grupo tratado (GT, n = 12), feridas higienizadas e tratadas com creme de ácido ascórbico (10%). Os fragmentos para análise histológica foram coletados no 3º, 7º e 14º dia, e as lâminas coradas com hematoxilina-eosina e picrosírius red para análise morfológica. As imagens foram capturadas e analisadas por um sistema digitalizador.
RESULTADOS: O ácido ascórbico atuou em todas as etapas da cicatrização, diminuindo o número de macrófagos, aumentando a proliferação dos fibroblastos e neovasos, e favorecendo a deposição de fibras colágenas mais espessas e organizadas nas feridas.
CONCLUSÃO: O ácido ascórbico mostrou ter efeito antiinflamatório e cicatrizante, promovendo ambiente e condições favoráveis para a reparação tecidual, o que abreviou o tempo da cicatrização.
Palavras-chave: cicatrização de feridas, ácido ascórbico, colágeno, fibroblastos, pele.
1. Introduction
The skin is the most frequently injured part of the body. When it is wounded, the connective tissue is exposed and a series of local cellular and biochemical events are triggered to restore tissue integrity. This sequence of events is called cicatrization and involves inflammation, proliferation and maturation (Irion, 2005; Micera, Vigneti et al., 2001; Contran, Kumar and Collins, 2001).
Several studies have shown the topical effect of medicines upon a wound, such as antibiotics (Raphael, 1965), association of an antibiotic with amino acids (Carvalho, Okamoto, 1977), antibacterial drugs (Carvalho, Oliveira, 1990), anti-inflammatories (Vasconcellos, Alberti et al., 2001; Chaves, Petroianu et al., 2004), 10% propolis (Magro-Filho, 1988) and insulin (Lima, Mahmoud et al., 2003) to shorten healing time and avoid infections and aesthetic damages.
Ascorbic acid (AA) or vitamin C (C6H8O6, ascorbate when in the ionised form) is a molecule used in the hydroxylation of various other cellular biochemical reactions. Its main function is the hydroxylation of collagen, a fibrillar protein that provides resistance to bones, teeth, tendons and blood vessel walls (Nelson and Cox, 2005). AA is essential to stimulate dermal fibroblasts (Azulay et al., 2003), and catecholamine biosynthesis. It is an antioxidant capable of neutralising the reactive oxygen species (Nelson and Cox, 2005; Naidu, 2003; Carr and Frei, 1999) and it is fundamental for the prevention and repair of blood vessel walls, and also for immunological defense (Azulay et al., 2003). Vitamin C is usually found in human skin, but it is rapidly lost in inflammatory processes. Topical vitamin C supplement for the skin, through lipid molecules, can be beneficial in dermatological treatment (Perricone, 1993). The topical application of vitamin C partially restores the anatomical structure of the epidermal-dermal junction in young skin and it increases the number of nutritive capillary loops in the papillary dermis (Kirsten et al., 2004). AA is highly labile, being easily oxidized by the enzyme dehydroascorbate reductase (Black and Hidiroglou, 1996).
Despite being frequently investigated, wound healing continues to challenge researchers. The aim of this study was to evaluate the topical effect of ascorbic acid on the healing process of rat skin wounds, mainly to quantify macrophages, newly formed blood vessels (neovessels) and fibroblasts, and analyse the thickness and organization of collagen fibres in the injured tissue.
2. Material and Methods
2.1. Animal protocol
White male wistar rats (Rattus norvegicus), three months old and weighing 270 ± 30 g, were obtained from the Universidade de Alfenas (UNIFENAS) Laboratory Animal Facility. The animals were intraperitoneally (IP) anesthetised with thionembutal (10 mg/kg). After manual depilation and antisepsis with povidone on the cervical back, a 15 mm long transversal incision was made with a scalpel (Schirato, Monteiro et al., 2006). The animals were randomly separated into two experimental groups: Control Group (CG, n = 12): wound cleaned with water and soap (neutral pH) once a day; Treated Group (TG, n = 12): wound cleaned with water and soap once a day and then treated with 1 g of a cream containing 10% ascorbic acid. The rats were all kept in cages and received ration and water ab libitum. On the 3rd, 7th and 14th days, four rats from each group were euthanised with an intraperitoneal overdose of thionembutal. The experimental protocol was approved by the UNIFENAS Committee of Ethics in Research under procedure no. 10A/2006.
2.2. Histological analysis
The material collected on the 3rd, 7th and 14th days as fixed in 10% formalin for 48 hours were embedded in paraffin. Five μm thick sections were placed on glass slides and histologically processed (Junqueira et al., 1979). Some of the sections were stained with hematoxylin-eosin for counting macrophages, neovessels, and fibroblasts; others were stained with picrosirius red to identify collagen fibres. The HE-stained sections were analysed through a common optical microscope, while those stained with picrosirius red were observed through a polarised light microscope. The histological images were captured by a digital camera through the "Software Image Pro-plus" with a 400× magnification. The "Software Fireworks" digitiser and image analyser was used to count macrophages, neovessels and fibroblasts (MEDEIROS et al., 2003). The thickness of the collagen fibres was measured by the "Image Tool" program. Eight fields of each histological section per animal were analysed, and the average number of macrophages, neovessels, fibroblasts, and the thickness of collagen fibres were calculated.
The experimental wounds were clinically and histologically evaluated during 14 days by only one observer.
2.3. Statistical analysis
The count values of macrophages, neovessels and fibroblasts were analysed and compared using the Student t-test, with 0.05 significance, and the results were expressed by the mean ± standard error of the mean (mean ± SEM).
3. Results
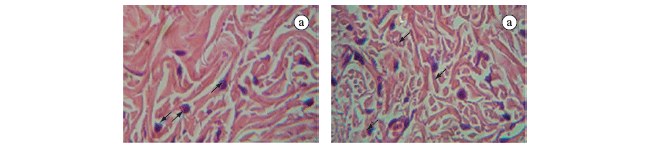
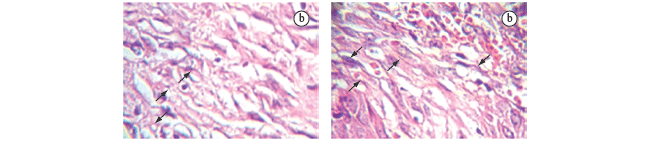
Less marked inflammatory signs and a more intense granulation tissue were evident in the treated group (TG) in relation to the control group (CG), but a serosanguineous secretion covered the wound bed in both groups (Figure 1a). Histologically, the TG wound showed a lower number of macrophages and higher number of neovessels when compared to the CG (Table 1). No significant differences were observed in either groups regarding the number of fibroblasts (Table 1 and Figure 1b). However, collagen fibres were thicker in the papillary layer of the TG (Table 1 and Figure 1c).
In relation to the CG, an easily removable scab on the TG rat wounds was macroscopically observed on the 7th day, probably due to the humidity provided by the ascorbic acid cream. No inflammatory signs were shown by the TG animals on the 7th day, while the CG animals maintained a marked inflammatory process. When compared to the CG, the TG histological sections of the wound showed a lower number of macrophages (Table 1 and Figure 2a), a higher number of neovessels (Table 1 and Figure 2b), a higher number of fibroblasts (Table 1 and Figure 2c) and thicker collagen fibers in the papillary layer (Table 1 and Figure 2d).
A complete contraction of the wound edges in the TG was clinically seen on day 8. The same occurred on day 12 in the CG.
The wounds were re-modelled in both groups on the 14th day, except for a cicatricial mark. Histologically, the number of macrophages in the TG is lower than in the CG (Table 1 and Figure 3a), and no significant difference occurred regarding the number of fibroblasts (Table 1 and Figure 3b). At this stage, collagen fibres were thicker (Table 1) and more organized (Figure 3c) in the TG than in the CG.
4. Discussion
With daily wound cleaning, none of the animals developed local infection. The presence of an easily removable scab in the TG wounds helped to handle the wounds during dressing without damaging the newly-formed granulation tissue.
The ascorbic acid took part in all the healing stages. The ascorbic acid cream (10%) had an anti-inflammatory and cicatrizing effect on skin wounds, which was characterised by a lower number of macrophages, a higher number of neovessels, a higher proliferation of fibroblasts and the production of thicker and more organized collagen fibres.
Skin wounds induce the release of several inflammatory mediators resulting from the aracdonic acid cascade, such as prostaglandins, leukotrienes and hydroxyeicosatetraenolic (Hete) acid (Trenam, Dabbagh et al., 1991). Such mediators attract neutrophils and macrophages to the wound and take part in all the clinical manifestations of the inflammatory process. These cells play an important role against bacterial contamination by means of phagocytosis, and production and release of proteinases and reactive oxygen species (ROS). The increased production of ROS is known as "respiratory burst", since the activation of neutrophils and macrophages use the NADPH oxidase cytochrome to reduce molecular oxygen into superoxide anions (Wientjes And Segal, 1995).
In addition to inflammatory cells, other cell types, such as fibroblasts, can also produce ROS in response to pro-inflammatory cytokines (Meier, Radeke et al., 1989). The local ROS increase is important for the defense against microbial infection; however, the prolonged production of high levels of ROS can induce cell damages (Cerutti and Trump, 1991). ROS can also activate and maintain the inflammatory aracdonic acid cascade (Trenam, Dabbagh et al., 1991). Therefore, in the present study, the anti-inflammatory effect of AA observed in the TG rat wounds may be related to its antioxidant effects, given that AA takes part in the cell oxireduction processes, being responsible for transforming free radicals of oxygen into inert forms (Nelson and Cox, 2005; Naidu, 2003), and quenches signalling ROS induced by the cytokine-receptor interaction, preventing the activation of ROS-mediated responses, and also quenches ROS that results from signalling (Cárcamo et al., 2004).
The anti-inflammatory effect of AA on the TG animals did not delay tissue repair, but rather shortened the healing time in relation to the CG. The reduction of pH and oxygen tension in the skin wound induces angiogenesis (Diegelmann et al., 1981; Knighton et al., 1981), which was confirmed in the TG animals of our study. Therefore, topical application of AA on the wound keeps the integrity of blood vessel walls (Azulay et al., 2003), increases the number of neovessels and improves blood supply to the wound, which enhances the proliferation and viability of the cells involved in the healing process. The newly-formed blood vessels showed a normal anatomical structure in confocal microscopical examination and were apparently integrated in a healthy vascular architecture (Kirsten et al., 2004).
In the present study, we observed a higher number and a better arrangement of fibroblasts in the TG animals when compared to the CG, since the topical application of AA maintains an adequate skin concentration of this vitamin, which diffuses through the keratin layer and stimulates the proliferation of dermal fibroblasts. Such proliferation may occur by growth factor activation (Phillips et al., 1994).
In the presence of AA, fibroblasts have been shown to proliferate more rapidly, resulting in a gradual and dense secretion of collagen, which decreases after ten days (Chan, Lamande et al., 1990). During all our experiment, collagen fibres were thicker, more organized and parallel to one another in the TG wounds when compared to the CG. AA is a co-factor of lysil and prolyl hydroxylases, two essential enzymes in collagen biosynthesis (Pinnel, Murad and Darr, 1987). Lysil and prolyl hydroxylases are ferric enzymes (Boyera et al., 1998). AA as a co-factor and antioxidant prevents the oxidation of iron, thus protecting the enzymes against self-inactivation, besides stimulating the individual transcription of the genes (Junien et al., 1982). Ascorbic acid 2-phosphate (AA2P) increased the mitogenic stimulation of quiescent fibroblasts by serum factors and cell motility in the context of wound healing (Duart et al., 2008). Furthermore, AA2P-treated fibroblasts showed faster repair of oxidatively damaged DNA bases (Duart et al., 2008).
Skin wound repair is a complex process that requires local cellular and biochemical events which are activated by a series of mediators, such as cytokines, growth factors and enzymatic activity. In our study, it was shown that the topical application of AA influences cellular events, enhancing tissue repair and shortening healing time.
Acknowledgements - The authors are grateful to Dr. João Evangelista Fiorini (UNIFENAS), for permitting us to use his photomicroscope, and to Professor Vinicius Vieira Vignoli (UNIFENAS), for revising the text.
Received October 27, 2008 -
Accepted December 16, 2008 -
Distributed November 30, 2009 (With 3 figures)
- AZULAY, MM., MANDARIN de LACERDA, CA., PEREZ, MA., FILGUEIRA, AL. and CUZZI, T., 2003. Vitamina C. Anais Brasileiros de Dermatologia, vol. 78, no. 3, p. 265-274.
- BLACK, WD. and HIDIROGLOU, M., 1996. Pharmacokinetic study of ascorbic acid in sheep. Canadian Journal Veterinary Research, vol. 60, no. 3, p. 216-221.
- BOYERA, N., GALEY, I. and BERNARD, BA., 1998. Effect of vitamin c and its derivatives on collagen synthesis and cross-linking by normal human fibroblasts. International Journal of Cosmetic Science, vol. 20, no. 3, p. 151-158.
- CÁRCAMO, JM., PEDRAZA, A., BÓRQUEZ-OJEDA, O., ZHANG, B., SANCHEZ, R. and GOLDE, DW., 2004. Vitamin C is a kinase inhibitor: dehydroascorbic acid inhibits iк bα kinase. Molecular and Cellular Biology, vol. 24, no. 15, p. 6645-52.
- CARR, A. and FREI, B., 1999. Does vitamin C act as a pro-oxidant under physiological condition? The Federation of American Societies for Experimental Biology Journal, vol. 13, no. 9, p. 1007-24.
- CARVALHO, ACP. and OKAMOTO, T., 1977. Cicatrização cutânea após aplicação tópica da associação antibióticos-aminoácidos: estudo clínico e histológico em ratos. ARS Cvrandi Odontologia, vol. 4, no. 3, p. 27-33.
- CARVALHO, PSP. and OLIVEIRA, GM., 1990. Cicatrização cutânea após aplicação tópica de nebacetin e gingilone em feridas infectadas: estudo clínico e histológico em ratos. Revista de Odontologia da UNESP, vol. 19, no. 1, p. 75-84.
- CHAN, D., LAMANDE, SR., COLE, WG. and BATEMAN, JF., 1990. Regulation of procollagen synthesis and processing during ascorbate-induced extracellular matrix accumulation in vitro. Biochemistry Journal, vol. 269, no. 1, p. 175-181.
- CHAVES, DN., PETROIANU, A., ALBERTI, LR. and PEREIRA, WA., 2004. Effects of Thalidomide, Cyclosporine and Diclofenac on Skin Allograft Survival in Rabbits. Transplantation proceedings, vol. 36, no. 4, p. 1018-1020.
- CERUTTI, PA. and TRUMP, BF., 1991. Inflammation and Oxidative Stress in Carcinogenesis. Cancer Cell, vol. 3, no. 5, p. 1-7.
- CONTRAN, RS., KUMAR, V. and COLLINS, T., 2001. ROBBINS: patologia estrutural e funcional. Rio de Janeiro: Guanabara Koogan. 1251 p.
- DIEGELMANN, RF., COHEN, IK. and KAPLAN, AM., 1981. The role of macrophages in wound repair: a review. Plastic and Reconstructive Surgery, vol. 68, no. 1, p. 107-113.
- DUARTE, TL., COOKE, MS. and JONES, GD., 2008. Gene expression profiling reveals new protective roles for vitamin C in human skin cells. Free Radical Biology & Medicine, vol. 46, no. 1, p.78-87.
- IRION, G., 2005. Feridas: novas abordagens, manejo clínico e atlas em cores. Rio de Janeiro: Guanabara Koogan. 329 p.
- JUNIEN, C., WEIL, D., MYERS, JC., CONG, N., CHU, ML., FOUBERT, C., GROSS, MS., PROCKOP, DJ., KAPLAN, JC. and RAMIREZ, F., 1982. Assignment of the human pro alpha 2(I) collagen structural gene (COLIA2) to chromosome 7 by molecular hybridization. American journal of human genetics, vol. 34, no. 3, p. 381-387.
- JUNQUEIRA, LC., BIGNOLAS, G. and BRENTANI, RR., 1979. Picrosirius red staining plus polarization microscopy, a specific method for collagen detection in tissue sections. The Histochemical Journal, vol. 11, no. 4, p. 447-455.
- KIRSTEN, S., SÖREN, J., URTE, K. and HORST, W., 2004. Topically applied vitamin C increases the density of dermal papillae in aged human skin. BMC Dermatology, vol. 4, p. 13.
- KNIGHTON, DR., SILVER, I. and HUNT, TK., 1981. Regulantion of wound-healing angiogenesis- effect of oxygen gradients and inspired oxygen. Surgery, vol. 90, no. 2, p. 262-270.
- LIMA, MHM., MAHMOUD, JGE., GASPARETTI, AL., PARISI, MC., VELLOSO, LA. and SAAD, MJA., 2003. Efeito da insulina na cicatrização de feridas em ratos diabéticos e não diabéticos. In: Semana Científica: cuidando do trabalho de Enfermagem. Caderno de Resumos, vol. 1, p. 10.
- MAGRO-FILHO, O., 1988. Reparação de alvéolo dental e de ferida cutânea após irrigação com solução de própolis: estudo histológico em ratos. Araçatuba: Universidade Estadual de São Paulo. [Dissertação de Mestrado]
- MEDEIROS, AC., FILHO, AMD., NETO, TA., LIMA, FP., AZEVÊDO, IM. and ALVES, SG., 2003. O fator de crescimento de fibroblastos básico melhora a cicatrização de anastomoses duodenais em ratos. Acta Cirúrgica Brasileira, vol. 18, supl.1, p. 10-14.
- MEIER, B., RADEKE, HH., SELLE, S., YOUNES, M., SIES, H. RESCH K. and HABERMEHL, GG., 1989. Human fibroblasts release reactive oxygen species in response to interleukin-1 or tumour necrosis factor-α. Biochemical Journal , vol. 263, no. 2, p. 539-545.
- MICERA, A., VIGNETI, E., PICKHOLTZ, D., REICH, R., PAPPO, O., BONINI, S., MAQUART, FX., ALOE, L. and LEVI-SCHAFFER, F., 2001. Nerve growth factor display stimulatory effects on human skin and lung fibroblasts, demonstrating a direct role for this factor in tissue repair. Proceedings of the National Academy of Sciences of the United States of America, vol. 98, no. 11, p. 6162-6167.
- NAIDU, KA., 2003. Vitamin C in human health and disease is still a mystery? An overview. Nutrition Journal, vol. 2, no. 7, p. 1-10.
- NELSON, DL. and COX, MM., 2005. Lehninger Principles of Biochemistry 4 ed. New York: W. H. Freeman. 1119 p.
- PERRICONE, NV., 1993. The photoprotective and anti-inflammatory effects of topical ascorbyl palmitate. The Journal of Geritric Dermatology, vol. 1, no. 1, p. 5-10.
- PHILLIPS, CL., COMBS, SB. and PINNEL, SR., 1994. Effects of ascorbic acid on proliferation and collagen synthesis in relation to the donor age of human dermal fibroblasts. Journal of Investigative Dermatology, vol. 103, no. 2, p. 228-232.
- PINNEL, SR., MURAD, S. and DARR, D., 1987. Induction of collagen synthesis by ascorbic acid. a possible mechanism. Archives of Dermatology, vol. 23, no. 12, p. 1684-1686.
- RAPHAEL, A., 1965. Emprego tópico dos antibióticos em dermatologia. In LACAZ, CS. (Org.). Antibióticos São Paulo: Editorial Procienx. 459 p.
- SCHIRATO, GV., MONTEIRO, FMF., SILVA, FO., FILHO, JLL., LEÃO, AMA. and PORTO, ALF., 2006. O polissacarídeo do Anacardium Occidentale L. na fase inflamatória do processo cicatricial de lesões cutâneas. Ciência Rural, vol. 36, no. 1, p. 149-154.
- TRENAM, CW., DABBAGH, AJ., BLAKE, DR. and MORRIS, CJ., 1992. The role of iron in an acute model of skin inflammation induced by reactive oxygen species. British Journal of Dermatology, vol. 126, no. 3, p. 250-256.
- VASCONCELLOS, LS., ALBERTI, LR., PETROIANU, A. and TRIVELATO, LP., 2001. Efeito da hidrocortisona sobre a resistência cicatricial da pele em camundongos. Revista do Colégio Brasileiro de Cirurgiões, vol. 28, no. 4, p. 438-443.
- WIENTJES, FB. and SEGAL, AW., 1995. NADPH oxidase and the respiratory burst. Seminars in Cell Biology, vol. 6, no. 6, p. 357-365.
Publication Dates
-
Publication in this collection
04 Dec 2009 -
Date of issue
Nov 2009
History
-
Received
27 Oct 2008 -
Accepted
16 Dec 2008