Abstracts
The zooplankton assemblages in Chilean Patagonian lakes are characterised mainly by their low biodiversity and high predominance of calanoids copepods, a pattern that has been studied for large and deep lakes between 38-51° S, and shallow ponds at 51° S. The aim of the present study was analyse the zooplankton assemblages in different water bodies located in coastal zones, middle valleys and mountain zones between 37-39° S. For this purpose, the following variables were considered: maximum depth, latitude, altitude, chlorophyll-a and species number, and to these variables, a Principal Component Analysis (PCA) was applied. A co-occurrence null model analysis was also applied for determining the existence of a random process in crustacean species associations. The results denoted low species richness, and different species associations for studied sites, and the null model analysis revealed the absence of a random process as regulator of species associations. Furthermore, a low species/genera ratio was identified, which denotes low productivity of the studied sites. These results were supported by a PCA analysis which denoted that the main determinant factors are chlorophyll concentration and species number that are directly associated. The obtained results are in agreement with descriptions in the literature for species diversity for lakes of Chilean Patagonia that describes oligotrophy as the main regulator of zooplankton assemblages. Other ecological and limnological topics are discussed in the present study.
lakes; oligotrophy; copepods; cladocerans
As comunidades zooplantônicas em lagos chilenos são caracterizadas, principalmente, por sua baixa biodiversidade e alta predominância de copépodos calanoides, um padrão que tem sido estudado em lagos grandes e profundos entre 38-51° S, e lagoas rasas em 51° S. O objetivo do presente estudo foi analisar as comunidades zooplantônicas em diferentes corpos de água localizados em zonas costeiras, vales intermediários e regiões de montanhas entre 37-39° S. Para este propósito, as seguintes variáveis foram consideradas: profundidade máxima, latitude, altitude, clorofila "a" e número de espécies, e a estas variáveis, foi aplicada a Análise de Componentes Principais (PCA). Um modelo nulo de coocorrência também foi aplicado para determinar a existência de um processo aleatório na associação de espécies de crustáceos. Os resultados denotam baixa diversidade de espécies e diferentes associações de espécies nos diferentes locais estudados, e o modelo nulo de análises revelou a ausência de um processo aleatório como regulador de associação de espécies. Além disso, a baixa relação espécie/gênero foi identificada, o que denota baixa produtividade dos locais estudados. Estes resultados foram apoiados pelas análises de PCA que mostraram que os fatores determinantes principais como a concentração de clorofila e número de espécies estão diretamente associados. Os resultados obtidos concordam com as descrições na literatura sobre a diversidade de espécies para lagos da Patagônia Chilena, que descreve a oligotrofia como principal regulador de comunidades zooplanctônicas. Outros tópicos ecológicos e limnológicos foram discutidos no presente estudo.
lagos; oligotrofia; copépodos; cladóceros
ECOLOGY
Microcrustacean assemblages composition and environmental variables in lakes and ponds of the Andean region South of Chile (37-39° S)
Composição de comunidades de microcustáceos e variáveis ambientais em lagos e lagoas da região andina sul do Chile (37-39° S)
De los Ríos-Escalante, P.* * e-mail: patorios@msn.com, prios@uct.cl ; Hauenstein, E.; Romero-Mieres, M.
Facultad de Recursos Naturales, Escuela de Ciencias Ambientales, Universidad Católica de Temuco, Casilla 15-D, Temuco, Chile
ABSTRACT
The zooplankton assemblages in Chilean Patagonian lakes are characterised mainly by their low biodiversity and high predominance of calanoids copepods, a pattern that has been studied for large and deep lakes between 38-51° S, and shallow ponds at 51° S. The aim of the present study was analyse the zooplankton assemblages in different water bodies located in coastal zones, middle valleys and mountain zones between 37-39° S. For this purpose, the following variables were considered: maximum depth, latitude, altitude, chlorophyll-a and species number, and to these variables, a Principal Component Analysis (PCA) was applied. A co-occurrence null model analysis was also applied for determining the existence of a random process in crustacean species associations. The results denoted low species richness, and different species associations for studied sites, and the null model analysis revealed the absence of a random process as regulator of species associations. Furthermore, a low species/genera ratio was identified, which denotes low productivity of the studied sites. These results were supported by a PCA analysis which denoted that the main determinant factors are chlorophyll concentration and species number that are directly associated. The obtained results are in agreement with descriptions in the literature for species diversity for lakes of Chilean Patagonia that describes oligotrophy as the main regulator of zooplankton assemblages. Other ecological and limnological topics are discussed in the present study.
Keywords: lakes, oligotrophy, copepods, cladocerans.
RESUMO
As comunidades zooplantônicas em lagos chilenos são caracterizadas, principalmente, por sua baixa biodiversidade e alta predominância de copépodos calanoides, um padrão que tem sido estudado em lagos grandes e profundos entre 38-51° S, e lagoas rasas em 51° S. O objetivo do presente estudo foi analisar as comunidades zooplantônicas em diferentes corpos de água localizados em zonas costeiras, vales intermediários e regiões de montanhas entre 37-39° S. Para este propósito, as seguintes variáveis foram consideradas: profundidade máxima, latitude, altitude, clorofila "a" e número de espécies, e a estas variáveis, foi aplicada a Análise de Componentes Principais (PCA). Um modelo nulo de coocorrência também foi aplicado para determinar a existência de um processo aleatório na associação de espécies de crustáceos. Os resultados denotam baixa diversidade de espécies e diferentes associações de espécies nos diferentes locais estudados, e o modelo nulo de análises revelou a ausência de um processo aleatório como regulador de associação de espécies. Além disso, a baixa relação espécie/gênero foi identificada, o que denota baixa produtividade dos locais estudados. Estes resultados foram apoiados pelas análises de PCA que mostraram que os fatores determinantes principais como a concentração de clorofila e número de espécies estão diretamente associados. Os resultados obtidos concordam com as descrições na literatura sobre a diversidade de espécies para lagos da Patagônia Chilena, que descreve a oligotrofia como principal regulador de comunidades zooplanctônicas. Outros tópicos ecológicos e limnológicos foram discutidos no presente estudo.
Palavras-chave: lagos, oligotrofia, copépodos, cladóceros.
1. Introduction
The freshwater zooplankton assemblages of southern Chile are characterised by their low species richness and high predominance of calanoids copepods, due mainly to oligotrophy. Nevertheless, these studies are based mainly on descriptions for large and deep lakes located in Patagonia between 38-51° S (De los Ríos and Soto, 2006, 2007; Soto and Zúñiga, 1991). Northern Chilean Patagonia (Bio-Bio and Araucania regions 37-39° S) has numerous high mountains, middle valleys and coastal lakes (Steinhart et al., 2002; De los Ríos and Soto, 2007; De los Ríos et al., 2007). There are no studies on the zooplankton assemblages in these water bodies, although the studies on littoral crustaceans would indicate that these water bodies are oligotrophic and with a low number of species (De los Ríos et al., 2007; De los Ríos and Romero-Mieres, 2009).
The main point for studying communities are the null models, which propose that the community structures are random, or without regulator factors (Gotelli, 2000, 2001). The null models are more robust in comparison to other conventional methods (Gotelli, 2001). The aim of null models is based on neutral theory, which is the use of a random process for explaining ecological processes (Gotelli, 2000, 2001). The studies of these null models were carried out in terrestrial environments, and mainly the models based on species presence and absence (Tondoh, 2006; Tiho and Johens, 2007). The studies of null model species associations in Chilean lakes described for Southern Patagonian water bodies (44-51° S) described the presence of weak regulator factors, due to the presence of many repeated species in practically all sites (De los Ríos, 2008; De los Ríos et al., 2008a, b).
The aim of the present study is to compare the zooplankton species associations in different kinds of freshwater bodies some of these located in protected areas of Araucania and the Biobio region, Chile, with different kind of landscapes in their surroundings. For this purpose, the null models methods were used based on the presence and absence of species in order to determine the presence or absence of random causes as regulator factors (for example, low productivity and/or biogeographical process) of species associations and these results will be compared using multivariate analysis with the aim of determining if both analyses are in accordance in their results regarding the presence or absence of regulator factors.
2. Material and Methods
The sites visited were (Table 1): 1) Conguillio National Park that is characterised by the presence of Araucaria araucana (Molina), K.Koch, which coexists with Nothofagus forests, such as N. antarctica (G.Forst.) Oerst., N. pumilio (Poepp. and Endl.) Krasser and N. dombeyi (Mirb.) Oerst. (Marticorena and Rodríguez, 1995, 2003); 2) Huerquehue National Park, which is characterised by the presence of 240 plant species, A. araucana, N. dombeyi, N. pumilio and Saxe-gothaea conspicua Lindl. (Marticorena and Rodríguez, 1995, 2003; De los Ríos et al., 2007); 3) Galletue and Icalma lakes are located in the western zone of the Andes mountains, and both have in their surroundings forests of A. araucana, N. dombeyi and N. pumilio, and in ecotones it is possible find shrubs such as Berberis trigona Kunze ex P. et E., Baccharis magellanica (Lam.) Pers., Chusquea culeou Desv., Quinchamalium chilense Mol. and Festuca spp. (Hoffmann, 1997); and 4) Lanalhue and Lleulleu: both lakes are located in the Nahuelbuta mountains, that currently has intensive silviculture activities (Figueroa et al., 2008).
The studied sites were visited between September 2006 to January 2007, a period that corresponds to the season with maximum zooplankton abundances (Soto and Zúñiga, 1991), in different kinds of water bodies between 37-39° S (Table 1). The zooplankton samples were collected using vertical hauls of 20 m, using an Apstein net of 80 µm mesh size, collected specimens were fixed in ethanol and identified with the descriptions of Araya and Zúñiga (1985) and Bayly (1992), with the aim of obtaining species occurrence for collected samples.
The obtained data was analyzed in two steps. In the first step, the data was managed in a presence/absence matrix, to apply a null model for species occurrence with the objective of determining the potential effects of random process as regulator factors of species associations (Tondoh, 2007; Tiho and Johens, 2006) and to this end, it was applied using the following simulations: fixed-fixed; fixed-equiprobable and fixed-proportional (Tondoh, 2006; Tiho and Johens, 2007). This analysis was applied using the software Ecosim (Tondoh, 2006; Tiho and Johens, 2007; Gotelli and Entsminger, 2007).
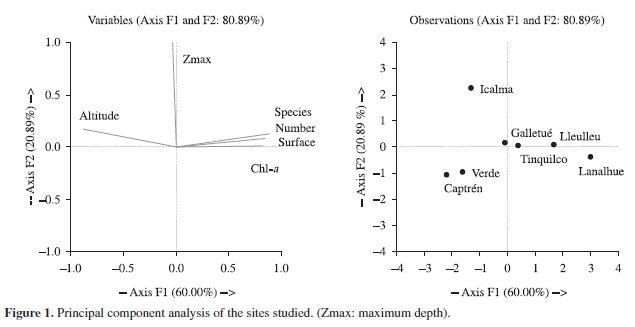
In a second step, samples were collected to determine chlorophyll-a concentration by acetone pigment extraction (Wetzel and Likens, 1991). Chlorophyll-a was considered because it is the best indicator of trophic status (Soto, 2002) and zooplankton assemblages (De los Ríos and Soto, 2006; Soto and De los Ríos, 2006) for Chilean lakes. This is because the trophic status in Chilean lakes is regulated by mixing depth instead of nutrients concentrations (Soto, 2002). In order to determine the principal components as determinants for grouping variables (surface, altitude, maximum depth, chlorophyll concentration and species number) for the sites studied, a principal component analysis (PCA) was applied which included the studied variables for all sites The aim of this analysis is to compare the obtained results with the null model analysis and determine the presence and absence of randomness in species associations.
3. Results
The results denoted low species richness that varied from two (Verde lake, Table 1) and five species (Lanalhue lake, Table 1), and the presence of representative species for zooplankton assemblages in Chile, such as Tumeodiaptomus diabolicus (Dussart, 1979), Boeckella gracilipes (Daday, 1902), B. gracilis (Daday, 1902), D. Pulex (De Geer, 1877), and Ceriodaphnia dubia (Richard, 1894). It was denoted that T. diabolicus inhabits mainly in low altitude lakes, whereas B. gracilipes and B. gracilis inhabits high altitude water bodies, whereas D. pulex and C. dubia have a relatively wide distribution (Table 1).
The null models analysis denoted for all simulations the absence of a random process as regulators of species associations (Table 2). The correlation matrix revealed a significant inverse correlation between altitude and surface, and significant direct correlation between chlorophyll and number of species (Table 2). The PCA analysis revealed that the most important variables contribute with 60% to the first axis. These variables were surface, number of species, altitude, and chlorophyll-a concentration (Figure 1). For the second axis only the maximum depth (Zmax) contributed with the 20.89% (Figure 1). The PCA results revealed the existence of three main groups, first an oligotrophic lake with low species number (Figure 1), that can be separated into two small lakes (Verde and Captrén) and one large and deep lake (Icalma). Large oligo-mesotrophic lakes (Tinquilco and Galletué) can be denoted differently to the other sites, and finally the lakes Lanalhue and Lleulleu are notoriously different to the other lakes due to their high species number and chlrorophyll concentration (Figure 1).
4. Discussion
The genus Tumeodiaptomus and Boeckella are described mainly for Chilean oligotrophic lakes (Soto and Zúñiga, 1991). The presence of Tumeodiaptomus in lowland lakes and Boeckella in high mountain lakes (Table 1) agrees with the descriptions by Zúñiga (1975). Also, Boeckella is distributed mainly in high mountains water bodies in tropical and subtropical latitudes (Gaviria, 1989; Menu-Marque et al., 2000), and in this scenario the presence of B. gracilis agrees with biogeographical distribution for Andean water bodies (De los Ríos et al., 2008b). These results agree with observations for the spatial distribution of copepods in a Brazilian reservoir where the main regulator factors are geographical characteristics and phytoplankton (Velho et al., 2001).
Whereas a different situation occurs with D. pulex and C. dubia, that occurs in only in oligomesotrophic lakes (De los Ríos and Soto, 2007). These exposed antecedents would support the non random process for species associations in freshwater bodies (Table 2). The presence of regulator factors or absence of random in zooplankton species assemblages in according to null model agrees with descriptions for Patagonian water bodies, where the marked environmental heterogeneity is the main cause of the presence of regulator factors in zooplankton assemblages (De los Ríos, 2008). An opposite situation occurs in sites with environmental homogeneity or with many species repeated in many of studied sites (De los Ríos et al., 2008b; De los Ríos and Roa, 2010).
The result of PCA confirms the observations for Patagonian lakes that proposed the role of low food availability as a determinant of low species number (39-51° S; Soto and Zúñiga, 1991; De los Ríos and Soto, 2007). A marked situation was reported for other South American lakes and reservoirs with high food availability where it was possible to find a minimum of 10 species (Bonecker et al., 2001; Garrido, 2002; Sampaio et al., 2002; Santos-Wisniewski et al., 2002; de Azevedo and Bonecker, 2003; Bini et al., 2008) and high zooplankton biomass (Bonecker et al., 2007). These antecedents agree with descriptions for an African lake, where direct associations between food resources and species number and individual abundance were observed (Dejen et al., 2004).
The species reported denoted the occurrence of representative species of lakes between 38-41° S (De los Ríos and Soto, 2007). Also, another important factor is the effects of trophic webs in pelagic environments of lakes, in oligotrophy it predominate calanoids copepods and daphnids low abundance or absence and there is low species number in lakes with fishes (Soto and Zúñiga, 1991; De los Ríos and Soto, 2007). The studied sites revealed the presence of relatively high zooplankton species number with daphnids presence (Table 2), and a potential cause would be a cascade effect, due to the presence of salmonids that prey on zooplanktivorous fishes (Soto and Zúñiga, 1991). Similar descriptions about predator activities and its consequences in zooplankton, mainly in daphniids abundance was described by Gliwicz et al. (2001) and Gliwicz (2002), where alterations in reproductive activity in fish presence, interespecific competition in fish absence had been described.
The results of the present study denoted that the null model co-ocurrence species analysis denoted the presence of species associations and the principal component analysis revealed the role of chlorophyll concentration and geographical characteristics (specifically surface and maximum depth) as a regulator of species associations. The cause of the concordance of both analyses would be the environmental heterogeneity mainly in trophic status and geographical characteristics of studied sites.
Acknowledgements This study was supported by grant funding for development of Limnology by the Research Directorate of the Catholic University of Temuco (Projects DGI- 2005-04-01 and DGI-DCA-01). We also thank the staff of the Corporación Nacional Forestal CONAF-Chile, for access to the facilities of the National Parks Conguillio and Huerquehue.
Received October 5, 2009
Accepted February 3, 2010
Distributed May 31, 2011
- ARAYA, JM. and ZÚÑIGA, LR., 1985. Manual taxonómico del zooplancton lacustre de Chile. Boletín Limnológico, vol. 8, p. 1-169.
- BAYLY, IAE., 1992. Fusion of the genera Boeckella and Pseudoboeckella and a revision of their species from South America and Subantarctic islands. Revista Chilena de Historia Natural, vol. 65, no. 1, p. 17-63.
- BINI, LM., DA SILVA, LCF., VELHO, LFM., BONECKER, CC. and LANSAC-TOHA, FA., 2008. Zooplankton assemblage concordance patterns in Brazilian reservoirs. Hydrobiologia, vol. 598, p. 247-255. doi:10.1007/s10750-007-9157-3
- BONECKER, CC., LANSAC-TOHA, FA., VELHO, LFM. and ROSA, DC., 2001. The temporal distribution pattern in copepods in Corumbá Reservoir, State of Goias, Brazil. Hydrobiologia, vol. 453/454, p. 375-384.
- BONECKER, CC., NAGAE, CY., BLETLLER, MCM., VELHO, LFM. and LANDAC-TOHA, FA., 2007. Zooplankton biomass in tropical reservoirs in southern Brazil. Hydrobiologia, vol. 579, p. 115-123. doi:10.1007/s10750-006-0391-x
- DE AZEVEDO, F. and BONECKER, CC., 2003. Community structure of zooplanktonic assemblages in three lakes on the upper River Paraná flood plain, PR-MS, Brasil. Hydrobiologia, vol. 505, p. 147-158. doi:10.1023/B:HYDR.0000007303.78761.66
- DE LOS RÍOS, P. and ROA, G., 2010. Crustacean species assemblages in mountain shallow ponds: Parque Cañi (38° S Chile). Zoologia Curitiba, vol. 27, p. 81-86.
- DE LOS RÍOS, P. and ROMERO-MIERES, M., 2009. Littoral crustaceans in lakes of Conguillío National Park (38ş S), Araucania region, Chile. Crustaceana, vol. 82, no. 1, p. 117-119. doi:10.1163/156854008X363759
- DE LOS RÍOS, P., 2008. A null model for explain crustacean species associations in Central and Southern Patagonian inland waters. Anales del Instituto de la Patagonia, vol. 36, no. 1, p. 25-33.
- DE LOS RÍOS, P. and SOTO, D., 2006. Structure of the zooplanktonic crustaceous Chilean lacustre assamblages: role of the trophic status and protection resources. Crustaceana, vol. 79, no. 1, p. 23-32.
- -, 2007. Crustacean (Copepoda and Cladocera) zooplankton richness in Chilean Patagonian lakes. Crustaceana, vol. 80, no. 3, p. 285-296. doi:10.1163/156854007780162433
- DE LOS RÍOS, P., HAUENSTEIN, E., ACEVEDO, P. and JAQUE, X., 2007. Littoral crustaceans in mountain lakes of Huerquehue National Park (38° S, Araucania region, Chile). Crustaceana, vol. 80, no. 4, p. 401-410. doi:10.1163/156854007780440885
- DE LOS RÍOS, P., RIVERA, N. and GALINDO, M., 2008a. The use of null model to explain crustacean zooplankton associations of the Magellan region, Chile. Crustaceana, vol. 81, no. 10, p. 1219-1228. doi:10.1163/156854008X374540
- DE LOS RÍOS, P., ACEVEDO P., RIVERA, R. and ROA, G., 2008b. Comunidades de crustáceos litorales de humedales del norte de la Patagonia chilena (38° S): rol potencial de la exposición a la radiación ultravioleta. In FERNÁNDEZ, L. and VOLPEDO, A. (Ed). Efectos de los Cambios Globales sobre la Biodiversidad 209-218 p. CYTED Red 406RT0285.
- DEJEN, E., VIJVERBERG, J., NAGELKERKE, LAJ. and SIBBING, FA., 2004. Temporal and spatial distribution of microcrustacean zooplankton in relation to turbidity and other environmental factors in a large tropical lake (L. Tana, Ethiopia). Hydrobiologia, vol. 513, p. 39-49.
- FIGUEROA, R., CRUCES, L., ARANEDA, A. and CRUCES, F., 2008. el problema del luchecillo (Egeria densa), una maleza en sistemas lénticos del centro-sur de Chile. In Congreso Internacional en gestión sostenible del agua, 2008, Medellín, Colombia. p. 1-23.
- GARRIDO, GG., 2002. Zooplancton del embalse Yacyretá, Argentina-Paraguay. Revista de Ecología Latinoamericana, vol. 9, no. 1, p. 9-15.
- GAVIRIA, S., 1989. The calanoid fauna (Crustacea, Copepoda) of the Cordillera Oriental of the Colombian Andes. Hydrobiologia, vol. 178, p. 113-134.
- GLIWICZ, ZM., 2002. On the different nature of top-down and bottom-up effects in pelagic food webs. Freshwater Biology, vol. 47, no. 12, p. 2296-2312. doi:10.1046/j.1365-2427.2002.00990.x
- GLIWICZ, ZM., ORŁOWSKA, A. and ŚLUSARCZYK, M., 2001. Life history synchronization in a long-life-span single-cohort Daphnia population of an alpine lake free of fish. Oecologia, vol. 128, no. 3, p. 368-378. doi:10.1007/s004420100673
- GOTELLI, NJ., 2000. Null models of species co-occurrence patterns. Ecology, vol. 81, no. 9, p. 2606-2621. doi:10.1890/0012-9658(2000)081[2606:NMAOSC]2.0.CO;2
- -, 2001. Research frontiers in null model analysis. Global Ecology and Biogeography, vol. 10, p. 337-343. doi:10.1046/j.1466-822X.2001.00249.x
- GOTELLI, NJ. and ENTSMINGER, GL., 2007. EcoSim: Null models software for ecology. Version 7. Acquired Intelligence Inc. and Kesey-Bear. Jericho, VT 05465. Available from: <http://garyentsminger.com/ecosim.htm>
- HOFFMANN, A., 1997. Flora Silvestre de Chile: Zona Araucana. Chile: Ediciones Fundación Claudio Gay.
- MARTICORENA, C. and RODRÍGUEZ, R., 1995. Flora de Chile. Pteridophyta - Gymnospermae, vol. 1, p. 1-351.
- -, 2003. Flora de Chile. Berberidaceae - Betulaceae, vol. 3, p. 1-93.
- MENU-MARQUE, S., MORRONE, JJ. and LOCASCIO DE MITROVICH, C., 2000. Distributional patterns of South American species of Boeckella (Copepoda, Centropagidae): a track analysis. Journal of Crustacean Biology, vol. 20, no. 2, p. 262-272. doi:10.1651/0278-0372(2000)020[0262:DPOTSA]2.0.CO;2
- SAMPAIO, EV., ROCHA, O., MATSUMURA-TUNDISI, C. and TUNDISI, JG., 2002. Composition and abundance of zooplankton in the limnetic zone of seven reservoirs of the Paranapanema River, Brazil. Brazilian Journal of Biology, vol. 63, no. 3, p. 525-545.
- SANTOS-WISNIESWKI, MJ., ROCHA, O., GUNTZEL, AM. and MATSUMURA-TUNDISI, C., 2002. Cladocera Chydoridae of high altitude water bodies (Serra da Mantiquera), in Brazil. Brazilian Journal of Biology, vol. 62, no. 4A, p. 681-687.
- SOTO, D., 2002. Oligotrophic patterns in southern Chile lakes: the relevance of nutrients and mixing depth. Revista Chilena de Historia Natural, vol. 75, p. 377-393.
- SOTO, D. and ZÚÑIGA, L., 1991. Zooplankton assemblages of Chilean temperate lakes: a comparison with North American counterparts. Revista Chilena de Historia Natural, vol. 64, no. 3, p. 569-581.
- SOTO, D. and DE LOS RÍOS, P., 2006. Trophic status and conductitivity as regulators of daphnids dominance and zooplankton assemblages in lakes and ponds of Torres del Paine National Park. Biologia, Bratislava, vol. 61, no. 5, p. 541-546. doi:10.2478/s11756-006-0088-7
- STEINHART, GS., LIKENS, GE. and SOTO, D., 2002. Physiological indicators of nutrient deficiency in phytoplankton in southern Chilean lakes. Hydrobiologia, vol. 489, p. 21-27. doi:10.1023/A:1023271331086
- TIHO, S. and JOHENS, J., 2007. Co-occurrence of earthworms in urban surroundings: a null models of community structure. European Journal of Soil Biology, vol. 43, no. 2, p. 84-90. doi:10.1016/j.ejsobi.2006.10.004
- TONDOH, JE., 2006. Seasonal changes in earthworm diversity and community structure in central Côte d'Ivoire. European Journal of Soil Biology, vol. 42, supl. 1, p. 334-340.
- VELHO, LFM., LANSAC-TOHA, FA., BONECKER, CC., BINI, LM. and ROSA, DC., 2001. The longitudinal distribution of copepods in Corumbá Reservoir, State of Goiás, Brazil. Hydrobiologia, vol. 453/454, p. 385-391.
- WETZEL, R. and LIKENS, G.L., 1991. Limnological analysis New York: Springer. 391 p.
- ZÚÑIGA, LR., 1975. Sobre Diaptomus diabolicus Brehm (Crustacea, Copepoda, Calanoidea). Noticiario Mensual del Museo Nacional de Historia Natural Santiago de Chile, vol. 228, p. 3-9.
Publication Dates
-
Publication in this collection
15 July 2011 -
Date of issue
May 2011
History
-
Received
05 Oct 2009 -
Accepted
03 Feb 2010