Abstracts
Parasitoids are of great importance to forest ecosystems due to their ecological role in the regulation of the population of other insects. The species richness and abundance of parasitoids in the forest canopy and understory, both on the borders and in the interior of a tropical forest reserve in Central Amazonia were investigated. For a 12-month period, specimen collections were made every 15 days from suspended traps placed in the forest canopy and in the understory strata, both on the border and in the interior of forest areas. A total of 12,835 Hymenoptera parasitoids from 23 families were acquired. Braconidae, Diapriidae, Mymaridae, Eulophidae, and Scelionidae were the most represented in the area and strata samples. The results indicate that there were no significant differences in the species richness or abundance of Hymenoptera between the forest borders and the inner forest. The data does show that the presence of Hymenoptera is significantly greater in the understory in both the border and interior areas than in the canopy (vertical stratification). Aphelinidae and Ceraphronidae were significantly associated with the inner forest, while the other seven families with the border of the reserve. The abundance of Hymenoptera parasitoids presented seasonal variations during the year related to the rainy and dry seasons.
Hymenoptera; parasitoids; Amazonia; stratification; tropical forest
Os parasitoides são de grande importância nos ecossistemas florestais pelo seu papel ecológico na regulação da população de outros insetos. Foram investigadas a riqueza e a abundância de parasitoides no dossel e sub-bosque, tanto na borda como no interior de uma reserva florestal tropical na Amazônia Central. Durante 12 meses, foram realizadas coletas quinzenais por meio de armadilhas suspensas instaladas no dossel e subdossel da borda e interior da floresta. Foi contabilizado um total de 12.835 Hymenoptera parasitoides, distribuídos em 23 famílias, sendo Braconidae, Diapriidae, Mymaridae, Eulophidae e Scelionidae as famílias mais representativas nos locais e estratos amostrados. Os resultados indicaram não haver diferenças significativas na riqueza ou na abundância de Hymenoptera entre borda e interior da floresta. Os dados de riqueza e abundância de Hymenoptera são significativamente maiores no sub-bosque das duas áreas (interior e borda) em relação ao dossel (estratificação vertical). Aphelinidae e Ceraphronidae foram associadas significativamente ao interior da floresta; outras sete famílias foram relacionadas significativamente à borda da reserva. A abundância de Hymenoptera parasitoides apresentou efeito de sazonalidade durante o ano, relacionada ao período de seca e chuvas.
Hymenoptera; parasitoides; Amazonia; estratificação; floresta tropical
ECOLOGY
The spatial distribution of Hymenoptera parasitoids in a forest reserve in Central Amazonia, Manaus, AM, Brazil
Distribuição espacial de Hymenoptera parasitoides em uma reserva florestal na Amazônia Central, Manaus, AM, Brasil
Querino, RB.I, * * e-mail: ranyse@cpamn.embrapa.br ; Couceiro, SRM.II; Queiroz, LO.III; Penteado-Dias, AM.IV
IEmpresa Brasileira de Pesquisa Agropecuária - Embrapa Meio-Norte, Av. Duque de Caxias, 5650, CEP 64006-220, Teresina, PI, Brazil
IICoordenação de Pesquisas em Entomologia, Instituto Nacional de Pesquisas da Amazônia - INPA, Av. André Araújo, 2936, Aleixo, CEP 69011-970, Manaus, AM, Brazil
IIIRua Major Gabriel, 1104, Centro, CEP 69020-060, Manaus, AM, Brazil
IVDepartamento de Ecologia e Biologia Evolutiva, Universidade Federal de São Carlos - UFSCar, Rod. Washington Luíz, Km 235, Centro, CEP 13565-905, São Carlos, SP, Brazil
ABSTRACT
Parasitoids are of great importance to forest ecosystems due to their ecological role in the regulation of the population of other insects. The species richness and abundance of parasitoids in the forest canopy and understory, both on the borders and in the interior of a tropical forest reserve in Central Amazonia were investigated. For a 12-month period, specimen collections were made every 15 days from suspended traps placed in the forest canopy and in the understory strata, both on the border and in the interior of forest areas. A total of 12,835 Hymenoptera parasitoids from 23 families were acquired. Braconidae, Diapriidae, Mymaridae, Eulophidae, and Scelionidae were the most represented in the area and strata samples. The results indicate that there were no significant differences in the species richness or abundance of Hymenoptera between the forest borders and the inner forest. The data does show that the presence of Hymenoptera is significantly greater in the understory in both the border and interior areas than in the canopy (vertical stratification). Aphelinidae and Ceraphronidae were significantly associated with the inner forest, while the other seven families with the border of the reserve. The abundance of Hymenoptera parasitoids presented seasonal variations during the year related to the rainy and dry seasons.
Keywords: Hymenoptera, parasitoids, Amazonia, stratification, tropical forest.
RESUMO
Os parasitoides são de grande importância nos ecossistemas florestais pelo seu papel ecológico na regulação da população de outros insetos. Foram investigadas a riqueza e a abundância de parasitoides no dossel e sub-bosque, tanto na borda como no interior de uma reserva florestal tropical na Amazônia Central. Durante 12 meses, foram realizadas coletas quinzenais por meio de armadilhas suspensas instaladas no dossel e subdossel da borda e interior da floresta. Foi contabilizado um total de 12.835 Hymenoptera parasitoides, distribuídos em 23 famílias, sendo Braconidae, Diapriidae, Mymaridae, Eulophidae e Scelionidae as famílias mais representativas nos locais e estratos amostrados. Os resultados indicaram não haver diferenças significativas na riqueza ou na abundância de Hymenoptera entre borda e interior da floresta. Os dados de riqueza e abundância de Hymenoptera são significativamente maiores no sub-bosque das duas áreas (interior e borda) em relação ao dossel (estratificação vertical). Aphelinidae e Ceraphronidae foram associadas significativamente ao interior da floresta; outras sete famílias foram relacionadas significativamente à borda da reserva. A abundância de Hymenoptera parasitoides apresentou efeito de sazonalidade durante o ano, relacionada ao período de seca e chuvas.
Palavras-chave: Hymenoptera, parasitoides, Amazonia, estratificação, floresta tropical.
1. Introduction
In tropical rain forests many species are susceptible to the process of extinction, principally those that occur in low densities and participate in narrow and complex interactions with other species (Myers, 1987).
Evidence indicates that insect species respond to environmental alterations in different ways and are affected by the alterations in the interactions among species. The cascading effects of these individual responses affect the structure and composition of communities (Menéndez, 2007). Many studies have shown the importance of tritrophic interactions when evaluating the effect of environmental changes (e.g. Bezemer et al., 1998; Coley, 1998; Johns and Hughes, 2002; Hamilton et al., 2004; Menéndez, 2007; Thuiller, 2007; Battisti, 2008).
The large number of parasitoid species, combined with their abilities to respond to their density-dependence on the population of their hosts, are essential to the maintenance of ecological balance and contribute to the diversity of other organisms (LaSalle and Gauld, 1993). As climate changes affect the hosts and the parasitoids differently, it is to be expected that the distribution of each species changes and communities are altered, including the adaptation of the parasitoids to new species of hosts (Hance et al., 2007).
Parasitoids can be used to evaluate the effects of forest fragmentation which affect them much more than it does their phytophagous hosts (Kruess and Tscahrntke, 1994, 2000). Besides this, hymenopterous parasitoids are good biological indicators. For example, Braconidae are not only a regulating agent of diverse groups of herbivore insects, but also indicate the presence or absence of herbivores (Matthews, 1974; LaSalle, 1993).
Despite their importance, parasitoids have been little studied in the Amazon region where knowledge of diversity and distribution is still incipient. Habitats such as tropical forest canopies can constitute a niche for many new species because these forest tops are considered the heart of biotic diversity (Erwin, 1997). He defined the tropical forest canopy as the "last biotic frontier", referring to the vast richness of organisms in this habitat; mainly arthropods (e.g. Basset et al., 2003).
Sutton et al. (1983) observed vertical stratification in the tropical forests of Panama, Papua-New Guinea and Brunei with a marked preference for certain groups of insects including Agaonidae (Hymenoptera) in canopy areas. Stork (1991) observed that Hymenoptera was the group with the greatest richness of species in the tree tops of the forests of Borneo. In the Amazon forest, Adis et al. (1998) observed that Formicidae (Hymenoptera) and Diptera were dominant in the tops of Goupia glabra (Celastraceae), where the biotic interaction between dipteran gall formersand parasitoids was verified. The relatively rare species of Euglossinae (Hymenoptera) was found only in the canopy between 12 to 15 m high (Oliveira, 2001).
The aims of this study were to investigate the species richness and abundance of Hymenoptera parasitoids, taking into account their seasonal distribution in the understory and canopy, as well as in the inner forest and on the forest border of an Amazon forest reserve.
2. Materials and Methods
2.1. Study area
The study was conducted in the reserve at km 41 (2° 26' 56.26" S; 59° 46' 12.68" W) of the Dynamic Biology of Fragmented Forests Project (PDBFF) of INPA/Smithsonian Institute, in an area of approximately 2470 acres, located in the north of Manaus Municipality, Amazonas, Brazil. The vegetation in this area is composed of typical terra-firme forest (Gascon and Bierregarrd Junior, 2001).
2.2. Collection method
Suspended traps were used (adapted from Rafael and Gorayeb, 1982) with a yellow lower septum (Figure 1) and an 80% alcohol/glycerin solution (8:1) in the collecting tray. The traps were placed on 6 trails (AB, R, J, C, EE, LL), 700 m equidistant. On each trail traps were located, a set on the border and a set in the inner forest (a set being one in the canopy and one in the understory), a total of 24 traps (Figure 2) covering the total extension of the reserve. The traps placed in the inner forest were 500 m from the border. In the canopy, the traps were installed in the tree tops at an average height of 22 m. The understory traps were installed on trees at a height of approximately 1.8 m. Every 15 days, between May 2004, and April 2005, the captured insects were removed from the traps and transferred to flasks containing 70% alcohol.
2.3. Sweeping and identification
Sweeping of the material was done using a stereoscopic microscope, separating the Hymenoptera parasitoids from the other insects collected. The parasitoids were identified at the family level and thus the abundance of the groups collected was determined. The identification of groups was based on Gibson et al. (1997), Goulet and Huber (1993) and Hanson and Gould (1995). In this study, Vespoidea with parasitoid habits was not included. The material collected was deposited at the Invertebrates Collection of the Instituto Nacional de Pesquisas da Amazônia (INPA) and in the Taxonomic Collection, Department of Ecology and Evolutionary Biology, of the Universidade Federal de São Carlos (DCBU).
2.4. Analysis of the data
Comparisons of the richness and abundance of Hymenoptera parasitoids between the canopy and understory, as well as between the inner forest and the forest border were carried out using variance analysis (ANOVA). Nonmetric multidimensional scaling (NMDS) was used to compare the composition of Hymenoptera parasitoids between the canopy and the understory, and between the border and interior areas of the reserve. Species Indicators Analysis (Dufrêne and Legendre, 1997) with the Monte Carlo Test of 999 randomisations, a posteriori, were used to verify the association of the Hymenoptera families in determined vertical and horizontal strata.
3. Results and Discussion
About 12,835 specimens from 23 families of Hymenoptera parasitoids were collected (Table 1). The most representative in abundance on the border and in the interior areas, as well as in the canopies and understories in the reserve, were Broconidae, Diapriidae, Mymaridae, Eulophidae and Scelionidae.
There was not a significant difference in the species richness of Hymenoptera parasitoids on the border or in the interior of the reserve (Figure 3) when the vertical substrata of the canopy and understory are considered separately (F (1.10) = 0.082, p = 0.780; F (1.10) =0.652, p = 0.438, respectively). The abundance of parasitoids (Figure 4) was similar between the border and the interior of the forest, as well as between the canopy and the understory (F (1.10 = 1.191, p = 0.301; F (1.10) = 0.950, p = 0.353, respectively).
The richness of species of Hymenoptera parasitoids (Figure 3) was significantly greater in the understory than in the canopy, both on the border (F (1.10) = 6.213, p = 0.032) and in the interior of the forest (F (1.10 = 10.714, p = 0.008). A similar pattern was observed for abundance (Figure 4) on the border (F (1.10 = 14.615, p = 0.003) and in the interior of the forest (F (1.10 = 35.732, p = 0001). These established facts are reflected in the composition in the sum of all parasitoids related to one vertical stratification (Figure 5). Eight families (Aphelinidae, Braconidae, Ceraphronidae, Chalcididae, Mymaridae, Scelionidae, Torymidae and Trichogrammatidae) were associated with the understory, while only one (Figitidae) was associated with the canopy (Table 2).
There was an association of seven families (Braconidae, Chalcididae, Figitidae, Mymaridae, Sceliondae, Torymidae and Trichogrammatidae) on the border of the reserve, while two families (Aphelinidae and Ceraphronidae) were associated with the interior (Table 2), since the richness and abundance of Hymenoptera were similar on the border and in the interior. These results indicate the less abundant Hymenoptera families are substituted in one or the other area (interior or border). The occurrence of Aphelinidae and Ceraphronidae tend to indicate a presence in the whole forest.
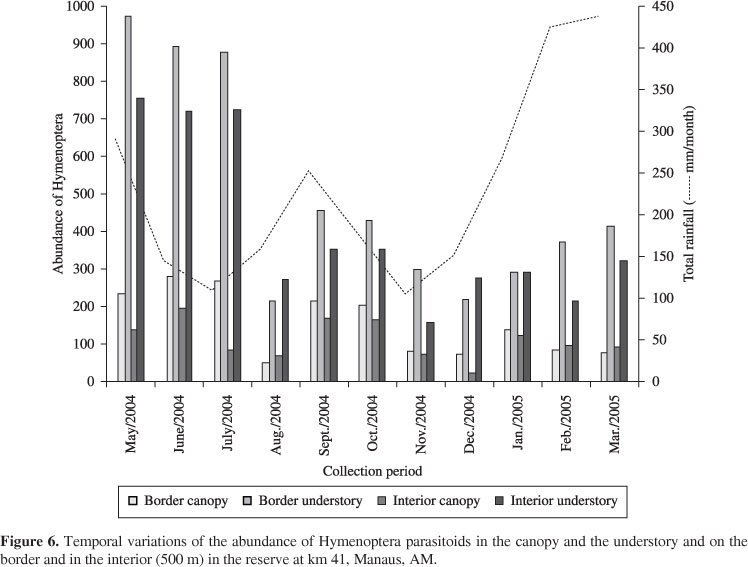
There was a tendency towards a reduction in the parasitoid population with the increase in rainfall which had a greater impact in the canopy (Figure 6).
The distribution of preferred hosts is one factor that could also explain the question about the lower number of parasitoids in the canopy. Parasitoids exploit an extensive range of hosts and respond to the density of, and fluctuations in them. Analysing other insects from the traps, it was verified that the understory had the greatest number of hosts.
No differences in the composition and richness of Hymenoptera in the interior and on the border were observed, probably because of the forest reserve´s proximity to an extensive area of continuous forest. Other studies have indicated differences in the composition and richness of Hymenoptera in the interior and on the border of forest fragments. Vasconcelos et al. (2001) studying a community of ants, verified that even though the effect on species richness in a fragment extension was not observed, evidence was found that this extension and its distance from the forest border affected the composition of the ant community in fragments of the Amazon region. Laranjeiro (2003) studied the insect fauna in a mosaic of eucalyptus plantations and nature conservation areas and verified that the border region between the eucalyptus growths and the forest presented the greatest number of Hymenoptera, mainly parasitoids. He also observed that the richness of the species decreased in the different ambiences in the following sequence: the border, the forest, and the eucalyptus stands. Martins (2001) found a small increase in species diversity of Drosophila Fallén (Diptera) on the border in relation to in the inner forest.
Acknowledgements - We thank Rosaly Ale-Rocha and Beatriz Ronchi-Teles (INPA) for project support. To Malu C.B. Feitosa, Sérgio Geraldo M. da Costa, Eduardo T. Nascimento and Alison N. Gonçalves for help with the sweeping. Thanks to PDBFF for the financial support provided (no. 588 of the series technical). To CAPES for granting a Prodoc fellowship to the first author.
Received July 15, 2010
Accepted October 14, 2010
Distributed November 30, 2011
- ADIS, J., HARADA, AY., FONSECA, CRV., PAARMANN, W. and RAFAEL, JA., 1998. Arthropods obtained from the Amazonian tree species "Cupiuba" (Goupia glabra) by repeated canopy fogging with natural pyrethrum. Acta Amazônica, vol. 28, no. 3, p. 273-283.
- BASSET, Y., NOVATNY, V., MILLER, SE. and KITCHING, RL., 2003. Arthropods of tropical forest: special-temporal dynamics and resource use an the canopy Cambridge: Cambridge University Press. 474 p.
- BATTISTI, A., 2008. Forests and climate change - lessons from insects. Available from: <http://www.sisef.it/iforest/>
- BEZEMER, TM., JONES, TH. and KNIGHT, KJ., 1998. Long-term effects of elevated CO2 and temperature on populations of the peach potato aphid Myzus persicae and its parasitoid Aphidius matricariae Oecologia, vol. 116, p. 128-135. http://dx.doi.org/10.1007/s004420050571
- COLEY, PD., 1998. Possible effects of climate change on plant/herbivore interaction in moist tropical forest. Climate Change, vol. 39, p. 455-472. http://dx.doi.org/10.1023/A:1005307620024
- DUFRÊNE, M. and LEGENDRE, P., 1997. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecological Monographs, vol. 67, no. 3, p. 345-366.
- ERWIN, TL., 1997. A copa da floresta tropical: o coração da biodiversidade biótica. In WILSON, EO. and PETER, FM. (Eds.). Biodiversidade Rio de Janeiro: Nova Fronteira. p. 158-165.
- GASCON, C. and BIERREGAARD JUNIOR, RO., 2001. The Biological Dynamics of Forest Fragments Project - The study site, experimental design, and research activity. In BIERREGAARD JUNIOR, RO., GASCON, C., LOVEJOY, TE. and MESQUITA, R. (Eds.). Lesson from Amazonia: The ecology and conservation of a fragmented forest. Yale University Press. p. 31-45.
- GIBSON, GAP., HUBER, JT. and WOOLLEY, JB., 1997. Annotated keys to the Genera of Neartic Chalcidoidea (Hymenoptera) Ottawa: NRC Research Press. 794 p.
- GOULET, H. and HUBER, JT., 1993. Hymenoptera of the World: An identification guide to families. Ottawa: Agriculture Canada Publication. 668 p.
- HAMILTON, JG., ZANGERL, AR., BERENBAUM, MR., PIPPEN, J., ALDEA, M. and DE LUCIA, EH., 2004. Insect herbivory in an intact forest understory under experimental CO2 enrichment. Oecologia, vol. 138, p. 566-573.
- HANCE, T., BAAREN, J. van, VERNON, P. and BOIVIN, G., 2007. Impact of Extreme Temperatures on Parasitoids. Annual Review of Entomology, vol. 521, p. 07-26.
- HANSON, PE. and GAULD, ID., 1995. The Hymenoptera of Costa Rica Oxford: Oxford University Press. 893 p.
- JOHNS, CV. and HUGHES, L., 2002. Interactive effects of elevated CO2 and temperature on the leaf-miner Dialectia scalariella Zeller (Lepidoptera, Gracillariidae) in Paterson's Curse, Echium plantagineum (Boraginaceae). Global Change Biology, vol. 8, p. 142-152.
- LARANJEIRO, AJ., 2003. Estabilidade da entomofauna num mosaico de plantações de eucalipto e áreas naturais de conservação Piracicaba: Universidade de São Paulo. 142 p. Tese de Doutorado em Entomologia.
- KRUESS, A. and TSCHARNTKE, T., 1994. Habitat fragmentation, species loss and biological control. Science, vol. 264, p. 1581-1584. PMid:17769603. http://dx.doi.org/10.1126/science.264.5165.1581
- -, 2000. Species richness and parasitism in a fragmented landscape: experiments and field studies with insects on Vicia sepium Oecologia, vol. 122, p. 129-137. http://dx.doi.org/10.1007/PL00008829
- LASALLE, J., 1993. Parasitic Hymenoptera, Biological Control and Biodiversity. In LASALLE, J. and GAULD, ID. (Eds.). Hymenoptera and Biodiversity Wallingford: C.A.B. International. p. 197-215.
- MARTINS, MB., 2001. Drosophilid Fruit-Fly Guilds in Forest Fragments. In BIERREGAARD JUNIOR, RO., GASCON, C., LOVEJOY, TE. and MESQUITA, RCG. (Eds.). Lessons from Amazonia: the ecology and conservation of a fragmented forest. North Carolina: Yale University. p. 175-186.
- MATTHEWS, W., 1974. Biology of Braconidae. Annual Review of Entomology, vol. 19, p. 15-32. http://dx.doi.org/10.1146/annurev.en.19.010174.000311
- MENÉNDEZ, R., 2007. How are insects responding to global warming? Tijdschrift voor Entomologie, vol. 150, p. 355-365.
- MYERS, N., 1987. The extinction spasm impending: synergisms at work. Conservation Biology, vol. 1, no. 1, p. 14-21. http://dx.doi.org/10.1111/j.1523-1739.1987.tb00004.x
- OLIVEIRA, ML., 2001. Stingless Bees (Meliponini) and Orchid Bees (Euglossini) in Terra Firme Tropical Forest and Forest Fragments. In BIERREGAARD JUNIOR, RO., GASCON, C., LOVEJOY, TE. and MESQUITA, RCG. (Eds.). Lessons from Amazonia: the ecology and conservation of a fragmented forest. North Carolina: Yale University. p. 208-218.
- RAFAEL, JA. and GORAYEB, IS., 1982. Tabanidae (Diptera) da Amazônia, I - Uma nova armadilha suspensa e primeiros registros de copas de árvores. Acta Amazônica, vol. 12, no. 1, p. 232-236.
- STORK, NE., 1991. The composition of the arthropod fauna of Bornean lowland rain forest trees. Journal of Tropical Ecology, vol. 7, p. 161-180. http://dx.doi.org/10.1017/S0266467400005319
- SUTTON, SL., ASH, CP. and GRUNDY, A., 1983. The vertical distribution of flying insects in lowland rain-forest of Panama, Papua New Guinea and Brunei. Zoological Journal of the Linnean Society, vol. 78, p. 287-297. http://dx.doi.org/10.1111/j.1096-3642.1983.tb00868.x
- THUILLER, W., 2007. Climate change and the ecologist. Nature, vol. 448, no. 2, p. 550-552. PMid:17671497. http://dx.doi.org/10.1038/448550a
- VASCONCELOS, HL., CARVALHO, KS. and DELABIE, JHC., 2001. Landscape modifications and ant communities. In BIERREGAARD JUNIOR, RO., GASCON, C., LOVEJOY, TE. and MESQUITA, RCG. (Eds.). Lessons from Amazonia: the ecology and conservation of a fragmented forest. North Carolina: Yale University. p. 199-207.
Publication Dates
-
Publication in this collection
24 Nov 2011 -
Date of issue
Nov 2011
History
-
Received
15 July 2010 -
Accepted
14 Oct 2010