Abstracts
The present study analyses the glans penis and baculum morphology of three Brazilian tuco-tucos, Ctenomys torquatus Lichtenstein, 1830, Ctenomys minutus Nehring, 1887 and Ctenomys flamarioni Travi, 1981, in order to identify possible variations and understand some more about this taxonomically complex group. We used fixed penis from 15 previously listed adult specimens. For a more detailed baculum analysis, the penis underwent dissection and diaphanisation, whereas to analyse the glans penis surface we used Scanning Electron Microscopy (SEM). Results showed striking differences in baculum morphology among the three species. While C. minutus have a particular V-shaped proximal baculum tip, C. flamarioni baculum is thin throughout the shaft with rounded proximal and distal tips. Ctenomys torquatus have a shorter and larger baculum, similar to what has previously been described for the species. Glans penis surface microstructure analyses also revealed inter-specific differences, with penial spines varying in shape, size and, especially density. Although C. torquatus has a relatively small penis, it has the largest penial spine density, which suggests a more complex penial ornamentation in this species.
Ctenomys; Glans penis; morphology; Baculum; scanning electron microscopy
O presente estudo analisa a morfologia do glans penis e do baculum de três tuco-tucos do Brazil - Ctenomys torquatus Lichtenstein, 1830, Ctenomys minutus Nehring, 1887, e Ctenomys flamarioni Travi, 1981 - com a finalidade de identificar possíveis variações e elucidar mais sobre grupo taxonômico complexo. Foram usados pênis fixados de 15 indivíduos adultos das espécies listadas anteriormente. Para uma análise mais detalhada do baculum, o pênis foi submetido a dissecções e diafanização, enquanto que, para a análise da superfície do glans penis, foi usada a técnica de microscopia eletrônica de varredura (MEV). Os resultados exibiram significativas diferenças na morfologia do baculum entre as três espécies. Enquanto C. minutus apresenta um baculum com uma ponta em forma de V característica, o baculum de C. flamarioni é fino ao longo do osso, com as pontas proximal e distal arredondadas. Ctenomys torquatus apresenta um baculum mais curto e largo, similar ao descrito anteriormente para a espécie. Análises da microestrutura na superfície do glans penis revelaram diferenças interespecíficas, com os espinhos penianos variando em forma, tamanho e, especialmente, densidade. Apesar de C. torquatus ter um pênis relativamente pequeno, apresenta a maior densidade de espinhos penianos, o que sugere uma complexa ornamentação peniana nesta espécie.
Ctenomys; Glans penis; morfologia; Baculum; microscopia eletrônica de varredura
BIOLOGY
Penial morphology in three species of Brazilian Tuco-tucos, Ctenomys torquatus, C. minutus, and C. flamarioni (Rodentia: Ctenomyidae)
Morfologia peniana de três espécies de tuco-tuco do Brasil Ctenomys torquatus, C. minutus e C. flamarioni (Rodentia: Ctenomyidae)
Rocha-Barbosa, O.I,* * e-mail: or-barbosa@hotmail.com ; Bernardo, JSL.I,II; Loguercio, MFC.I; Freitas, TRO.III; Santos-Mallet, JR.IV; Bidau, CJ.II
ILaboratório de Zoologia de Vertebrados (Tetrapoda) LAZOVERTE, Departamento de Zoologia, Instituto de Biologia Roberto Alcântara Gomes, Universidade do Estado do Rio de Janeiro UERJ, Rio de Janeiro, RJ, Brazil
IILaboratório de Biologia e Parasitologia de Mamíferos Silvestres Reservatórios, Instituto Oswaldo Cruz FIOCRUZ, Pavilhão Arthur Neiva, sala 14, Rio de Janeiro, RJ, Brazil
IIIDepartamento de Genética, Instituto de Biociências, Universidade Federal do Rio Grande do Sul UFRGS, Porto Alegre, RS, Brazil
IVLaboratório de Transmissores de Leishmanioses, Setor de Entomologia Médica e Forense SEMEF, Instituto Oswaldo Cruz FIOCRUZ, Pavilhão Carlos Chagas, 5º andar, sala 16, Rio de Janeiro, RJ, Brazil
ABSTRACT
The present study analyses the glans penis and baculum morphology of three Brazilian tuco-tucos, Ctenomys torquatus Lichtenstein, 1830, Ctenomys minutus Nehring, 1887 and Ctenomys flamarioni Travi, 1981, in order to identify possible variations and understand some more about this taxonomically complex group. We used fixed penis from 15 previously listed adult specimens. For a more detailed baculum analysis, the penis underwent dissection and diaphanisation, whereas to analyse the glans penis surface we used Scanning Electron Microscopy (SEM). Results showed striking differences in baculum morphology among the three species. While C. minutus have a particular V-shaped proximal baculum tip, C. flamarioni baculum is thin throughout the shaft with rounded proximal and distal tips. Ctenomys torquatus have a shorter and larger baculum, similar to what has previously been described for the species. Glans penis surface microstructure analyses also revealed inter-specific differences, with penial spines varying in shape, size and, especially density. Although C. torquatus has a relatively small penis, it has the largest penial spine density, which suggests a more complex penial ornamentation in this species.
Keywords:Ctenomys, Glans penis, morphology, Baculum, scanning electron microscopy.
RESUMO
O presente estudo analisa a morfologia do glans penis e do baculum de três tuco-tucos do Brazil Ctenomys torquatus Lichtenstein, 1830, Ctenomys minutus Nehring, 1887, e Ctenomys flamarioni Travi, 1981 com a finalidade de identificar possíveis variações e elucidar mais sobre grupo taxonômico complexo. Foram usados pênis fixados de 15 indivíduos adultos das espécies listadas anteriormente. Para uma análise mais detalhada do baculum, o pênis foi submetido a dissecções e diafanização, enquanto que, para a análise da superfície do glans penis, foi usada a técnica de microscopia eletrônica de varredura (MEV). Os resultados exibiram significativas diferenças na morfologia do baculum entre as três espécies. Enquanto C. minutus apresenta um baculum com uma ponta em forma de V característica, o baculum de C. flamarioni é fino ao longo do osso, com as pontas proximal e distal arredondadas. Ctenomys torquatus apresenta um baculum mais curto e largo, similar ao descrito anteriormente para a espécie. Análises da microestrutura na superfície do glans penis revelaram diferenças interespecíficas, com os espinhos penianos variando em forma, tamanho e, especialmente, densidade. Apesar de C. torquatus ter um pênis relativamente pequeno, apresenta a maior densidade de espinhos penianos, o que sugere uma complexa ornamentação peniana nesta espécie.
Palavras-chave:Ctenomys, Glans penis, morfologia, Baculum, microscopia eletrônica de varredura.
1. Introduction
Structures such as penis and baculum are an important part of the mammalian reproductive system and they have been subject of several scientific studies (Patterson and Thaeler Junior, 1982; Bradley and Schmidly, 1987; Simson et al., 1995). Penis morphology has largely been used in taxonomical and systematic studies (Bradley and Schmidly, 1987; Bradley et al., 1989), as in other studies focused on better understanding copulation and sexual selection systems (Altuna and Lessa, 1985; Simson et al., 1995; Parag et al., 2006).
The baculum or os penis is a bone located within the male penis and it is the most diverse of bones (Romer, 1962; Patterson and Thaeler, 1982). It was first described by Thomas (1915) and it is found in the mammalian orders Rodentia, Chiroptera, Insectivora, Carnivora and Primates (Hamilton Junior, 1949). If the baculum has an adaptive function is still in debate, but three main hypotheses for its function have been proposed so far: firstly, to facilitate intromission through extra rigidity and support to overcome vaginal friction; secondly, to allow prolonged intromission to assist sperm transport; and last, to provide extra rigidity to stimulate female ovulation (Larivière and Ferguson, 2002).
Among the rodents Octodontidea, the genus Ctenomys comprises fossorial herbivorous rodents (Nowak, 1999). This genus has a huge cariotipic diversity, including species varying from 2n = 10 to 2n = 70, which reflects in a complex evolutionary history, with still conflicting phylogenetic relations among species (Vitullo et al., 1988; Lessa and Cook, 1998; Contreras and Bidau, 1999; Mascheretti et al., 2000; Giménez et al., 2002; Gavas and Freitas, 2003, 2004; Bidau, 2006). Although, some evidence suggests that increase in penis ornamentation and decrease in baculum size are related to solitary behaviour (Parag et al., 2006), studies in Ctenomys penis indicate intra- and inter-specific variations such as in the intromitent sac and even in the baculum (Altuna and Lessa, 1985; Lessa and Cook, 1989; Contreras et al., 1993).
Herein, we aim to describe baculum and glans penis morphology in three species of Ctenomys genus found in the southern Brazilian territory: Ctenomys torquatus Lichtenstein, 1830, Ctenomys minutus Nehring, 1887 and Ctenomys flamarioni Travi, 1981, in order to elucidate some more about the morphology of these inconspicuous species.
2. Material and Methods
Overall, fifteen penises of adult specimens were examined. They were separated from the body at the base, and then preserved in 10% formalin and stored in the refrigerator. All specimens were collected in the state of Rio Grande do Sul and are deposited in the mammal collection of the Universidade Federal do Rio Grande do Sul UFRGS, Genetics Department. Numbers of collection and localities are as follows: C. minutus: TRO022 - Lagoa Emboaba, TRO391 - Capivari, TRO410, TRO416 and TRO418 - Lagoa dos Barros, TRO435 - Osório and TRO442 - Pitangueira; C. torquatus: TRO086 - Estação Ecológica do Taim, TRO373 - Barro Duro and SN - no data; C. flamarioni: TRO029 and TRO031 - Praia da Cidreira, TRO051 - Praia de Capão de novo, NC190 - no data and TRO049 - Lagoa dos Barros.
Four C. minutus specimens and two C. flamarioni were analysed using the diaphanisation technique. We used the Dingerkus and Uhler (1977) protocol with the following modifications: the enzimatic treatment consisted of immersing the material in a pancreatin solution, instead of in a trypsin solution; at the alcoholical series, the material was treated passing from a 30% glicerine solution to a 100% glicerine solution (abrupt osmotic shock).
One penis of each species was hydrated and then dissected ventrally to remove the baculum. Bacula were photographed using a Sony DSC S75 digital camera.
To study glans penis surface microstructure, two specimens of each species were used. The material was fixed in 2.5% glutaraldehyde in a sodium cacodylate buffer 0.1 M, pH 7.2 for 1 hour at room temperature. Soon after that, a cut was done on the glans penis basis to separate the glans from the penis body. Later, it was washed twice in buffer and post fixed in 1% osmium tetroxide in sodium cacodylate buffer 0.1 M, pH 7.2 for 1 hour at room temperature in the dark. The material was then dehydrated in an increasing series of acetone (7.5%, 15%, 30%, 50%, 70%, 90% and 100%) and subjected to the dry method by superdry CO2 critical point in Balzers devices. Then, the samples were put on metal brackets, using double-side tapes and were covered by gold in an evaporation system called sputtering, in which gold is removed from a massive electrode by ion bombardment in a high vacuum, using Balzers devices. Finally, the samples were observed in a scanning electron microscope Jeol JSM 6390LV (Akishima, Tokyo, Japan), at the Plataforma de Microspcopia Eletrônica do Instituto Oswaldo Cruz, at 15 Kv. All images were captured directly in a computer using the software Sem Aforce, which was also used to measure dorsal and ventral glans penis spines length.
3. Results
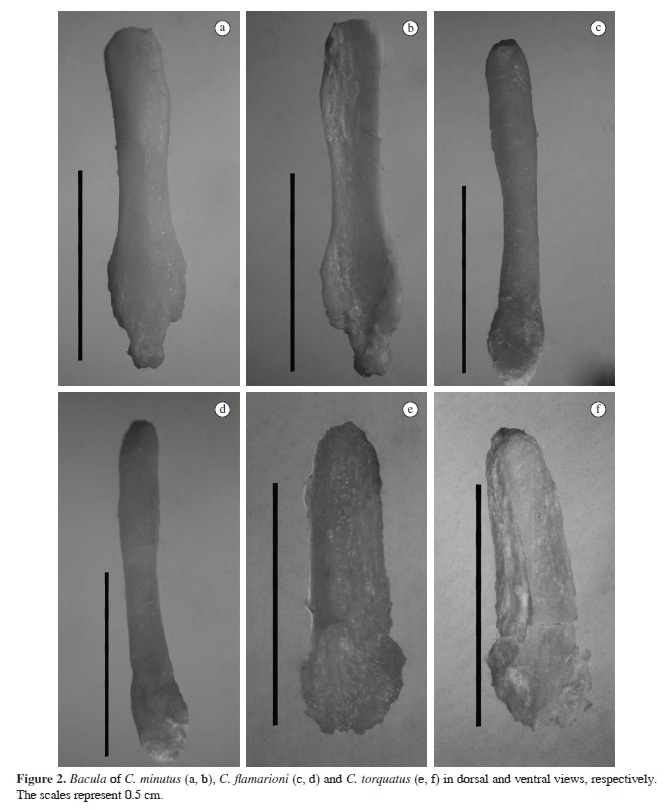
In all species, baculum was a thin and flat structure, located in the glans penis dorsal region as shown by diaphanisation and by dissections (Figures 1 and 2). But each species showed unique morphology.
The C. minutus baculum has a relatively "V-shaped" proximal tip. Its midlength is slightly narrower than both tips. The distal tip is round with a subtle concavity in the ventral surface. Length was 9.34 mm (Figure 2a, b). Meanwhile, C. flamarioni baculum is thin throughout the shaft. Both proximal and distal tips are round and its midlength is slightly constricted. It also has a slightly concave ventral surface. Length was 10.40 mm (Figure 2c, d). And C. torquatus baculum is relatively shorter and larger than in the other species. It has round proximal and distal tips. Its midlength is similar in width to both tips. There was no concavity in the ventral region. Length is 6.41 m (Figure 2e, f).
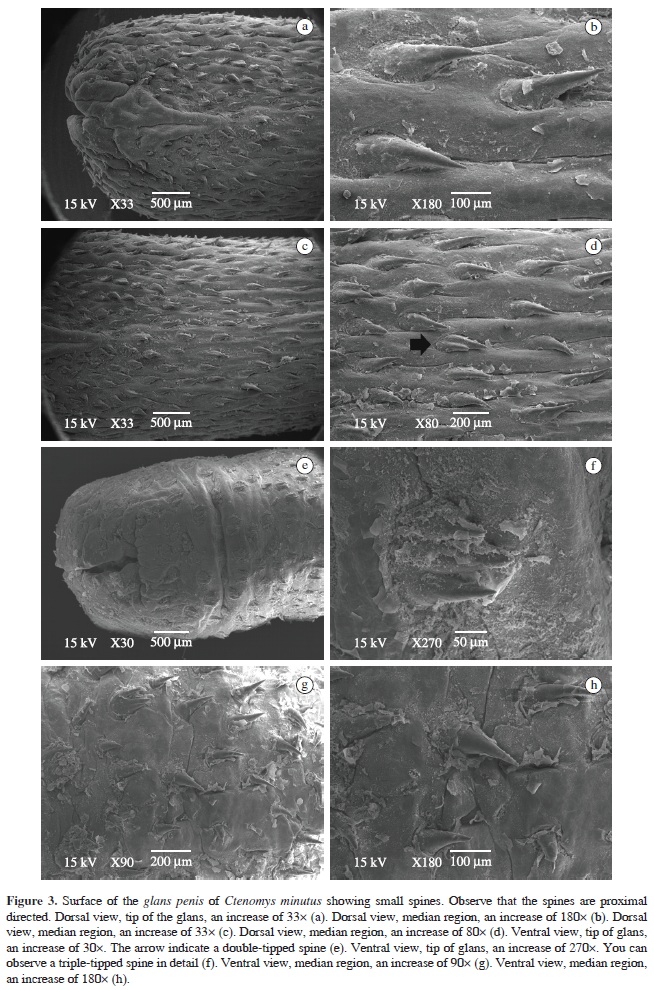
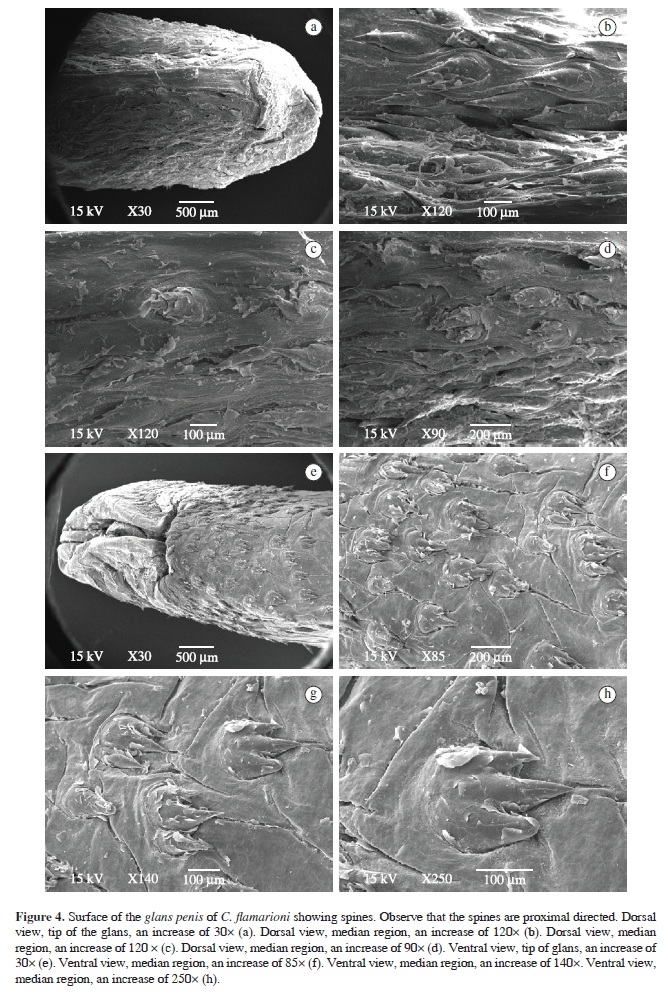
The glans penis surface microstructure analyses through Scanning Electron Microscopy (SEM) revealed inter-specific differences. Small spines varied in shape, size and density among the three species, as well as in ventral and dorsal regions. All specimens had proximal-directed spines, with their bases larger than their apices. In most cases, spines were sharp and internally curved.
In C. minutus, glans tips are recovered by spines, excluding the edge of the urethra (Figure 3a). In the dorsal surface, spines are very sharp; most of them with a single tip, but some double-tipped (Figure 3b-d). In the ventral surface, near the glans, spines had a flatter shape, very close to the surface and with two or three tips (Figure 3e‑f). In the median region (ventral view), near the glans base, spines had a sharper shape, like the dorsal spines (Figure 3g-h). Density was about 12 spines/mm2. Mean spine length was: 245.14 ± 49.81 µm (n = 41).
The spines observed in C. flamarioni specimens had a different shape. In the dorsal region, they have a sharp shape and sometimes a button shape, and a relatively small density (Figure 4a-d), while in the ventral region, spines had a round shape, flat and with two or three tips (Figure 4e-h). Density was about 9.5 spines/mm2. Mean length of the spines was 226.26 ± 32.42 µm (n = 41).
Ctenomys torquatus spines cover the entire glans tip, except at the urethra entry edges (Figure 5a). Dorsal surface has sharp spines, with convex shapes (Figure 5b‑d), whereas ventral surface has different shaped spines: shorter, rounded and concave (Figure 5e-h). Density was about 17.5 spines/mm2. Mean length of the spines was 189 ± 23.9 µm (n = 41).
4. Discussion
Our results confirmed the diverse penial morphology previously described in tuco-tucos by Lessa and Cook (1989) and Balbontin et al. (1996).
The three species have single-tipped bacula differing from the trilobed ones described for Ctenomys rionegrensis Langguth and Abella, 1970 in Uruguay and Argentina (Reig et al., 1966; Altuna and Lessa, 1985). Although all bacula are located in the glans penis dorsal region as commonly found in others rodents (Simson et al., 1995), many striking differences are evident when comparing the three studied species.
The C. minutus paddle-shaped baculum has a similar morphology to the one described for the Uruguayan species Ctenomys pearsoni Lessa and Langguth, 1983 (Altuna and Lessa, 1985), even though the former has a more pronounced V-shaped proximal tip. We also observed a subtle concavity in the ventral region of the baculum as previously described by Contreras et al. (1993) to Octodontidae rodents.
Nevertheless, Reig et al. (1966) described a clearly different baculum for C. minutus from Santo Antonio da Patrulha, south Brazil. The Santo Antonio de Patrulha sample has a more rounded proximal tip and narrower midlength and distal apex, whereas our sample has a very pronounced paddle-shaped proximal tip and a slight constriction in the midlength. According to Freitas (2001), at this Brazilian region it is also found Ctenomys lami Freitas, 2001, a recently described species similar to C. minutus. Probably Reig et al. (1966) analysed C. lami baculum instead of C. minutus baculum, but further investigations are needed to corroborate this affirmation.
As expected C. torquatus baculum morphology was very similar to the paddle-shaped bacula described for C. torquatus from Médanos in Argentina (Reig et al., 1966). Both are very large throughout the shaft compared to the ones from the other Ctenomys species. Baculum from C. flamarioni is first described in the present study and it clearly differs from all previous morpho-types. It has both proximal and distal tips quite round and its midlength slightly constricted. Similarly to C. minutus, it is also subtly concave at its ventral region.
Bacula length also varied among the three species. While C. minutus and C. flamarioni had relatively similar lengths, C. torquatus had a much smaller baculum. Bacular length is usually considered to have allometric trends although it may vary independently from head and body length (Lessa and Cook, 1989). In contrast, in Ctenomys, the larger species (C. torquatus) has the smaller baculum. According to Long and Frank (1968), these morphological variations coupled with genetics may be closely related to reproductive isolation and speciation phenomena.
Our results revealed that in the three analysed species, glans penis surface is covered by small spines, whose shape and density seem to be species-specific. These small spines are very similar to penile spines found in others rodent groups (Bradley and Schmidly, 1987; Parag et al., 2006).
The penile spines of the three species analysed have larger bases than tips. Some of them are sharp and in all three species they are facing the penile base (proximal directed) as seen in other rodent families: Bathyergidae, which are also fossorial rodents like Ctenomys and Peromyscus from the Cricetidae family (Bradley and Schmidly, 1987; Bradley et al., 1989; Parag et al., 2006). Nevertheless, the number of spines per area varied in each species. Ctenomys torquatus despite having a relatively smaller penis than the others, showed the largest density, while C. flamarioni and C. minutus had nearly the same number of spines per area.
According to Parag et al. (2006), the complexity of rodent penis morphology, specifically the surface structures, is closely related to the level of sociability. The more social the animal is the less is the complexity of spines. That is, more solitary animals have a higher level of complexity of penile structures, as is the case of Ctenomys. According to Altuna and Lessa (1985), these microstructures would have greater adaptive value precisely in animals with a solitary lifestyle. Perhaps this suggests a more solitary social habit in C. torquatus than in C. minutus and C. flamarioni; nonetheless, we must take into account that interactions between social and reproductive tuco-tucos are much more diverse and complex than expected for a common solitary and territorial species (Zenuto et al., 2002).
Spines and other penis ornamentations can have various functions such as, for example, induce ovulation or act as encouragement to male acceptance through vaginal stimulation, or can even ensure genital lock, holding the walls of the vagina during intercourse (Altuna and Lessa, 1985). In species with high sperm competition, these spines may also have the function of removing the semen deposited in the vagina of the female from previous matings (Parag et al., 2006). Regardless of their possible role, the identification of these variations in glans penis surface and baculum morphology can contribute especially to enlighten our knowledge about the reproductive system of the complex Ctenomys group.
Acknowledgements The authors would like to especially thank Hugo Ricardo Secioso Santos and Dr. Catarina Macedo Lopes for technical support. The work was funded by: CNPq, FAPERJ, Prociência/UERJ, REUNI/CAPES.
Received January 31, 2012
Accepted May 30, 2012
Distributed February 28, 2013
- ALTUNA, CA. and LESSA EP., 1985. Penial Morphology in Uruguayan Species of Ctenomys (Rodentia: Octodontidae). Journal of Mammalogy, vol. 66, no. 3, p. 483-488. http://dx.doi.org/10.2307/1380923
- BALBONTIN, J., REIG, S. and MORENO, S., 1996. Evolutionary relationships of Ctenomys (Rodentia:Octodontidae) from Argentina, based on penis morphology. Acta Theriologica, vol. 41, p. 237-253.
- BIDAU, CJ., 2006. Família Ctenomyidae. In BÁRQUEZ, RJ., DÍAZ, MM. and OJEDA, RA. (Eds.). Mamíferos de Argentina Sistemática y distribución. SAREM. p. 212-231.
- BRADLEY, RD. and SCHMIDLY, JD., 1987. The glans penis and bacula in latin American taxa of the Peromyscus boylii group. Jounal of mammalogy, vol. 68, p. 595-616.
- BRADLEY, RD., SCHMIDLY, JD. and OWEN, RD.,1989. Variation in the glans penis and bacula among latin american populations of the Peromyscus boylii species complex. Journal of mammalogy, vol. 70, no. 4, p. 712-725. http://dx.doi.org/10.2307/1381706
- CONTRERAS, JR. and BIDAU, CJ., 1999. Líneas generales del panorama evolutivo de los roedores excavadores sudamericanos del género Ctenomys (Mammalia, Rodentia, Caviomorpha, Ctenomyidae). Ciencia Siglo, vol. 21, no. 1, p. 1-22.
- CONTRERAS, LC., TORRES-MURA, JC., SPOTORNO, AE. and CATZEFLIS, FM., 1993. Morphological variation of the glans penis of South American Octodontid and Abrocomid rodents. Journal of. Mammalogy, vol. 74, p. 926-935. http://dx.doi.org/10.2307/1382431
- DINGERKUS, G. and UHLER, LD., 1977. Enzyme clearing of alcian blue stained whole small vertebrates for demonstration of cartilage. Stain Technology, vol. 52, p. 229-232.
- FREITAS, TRO., 2001. Tuco-tucos (Rodentia-Octodontidae) in southern Brazil: Ctenomys lami spec. nov. separated from C. minutus Nehring 1887. Studies on Neotropical Fauna and Environment, vol. 36, p. 1-8. http://dx.doi.org/10.1076/snfe.36.1.1.8882
- GAVA, A. and FREITAS, TRO., 2003. Inter and Intra-Specific Hybridization in Tuco-Tucos (Ctenomys) from Brazilian Coastal Plains (Rodentia: Ctenomyidae) Inter and Intra-specific. Genética, vol. 119, p. 11-17. PMid:12903742. http://dx.doi.org/10.1023/A:1024465429856
- -, 2004. Microsatellite analyses of a hybrid zone between chromosomally divergent populations of Ctenomys minutus from southeastern Brazil (Rodentia-Ctenomyidae). Journal of Mammalogy, vol. 85, p. 1201-1206. http://dx.doi.org/10.1644/BEL-017.1
- GIMÉNEZ, MD., MIROL, CJ., BIDAU, CJ. and SEARLE, JB., 2002. Molecular analysis of populations of Ctenomys (Caviomorpha, Rodentia) with high karyotypic variability. Cytogenetic and Genome Research, vol. 96, p.130-136. PMid:12438789. http://dx.doi.org/10.1159/000063051
- HAMILTON JUNIOR, WJ., 1949.Tthe bacula of some North AmericanVespertilionid bats. Journal of Mammalogy, vol. 30, p. 97-102. PMid:18121317. http://dx.doi.org/10.2307/1375254
- LESSA, EP. and COOK, JA., 1989. Interespecific variation in penial characters in the genus Ctenomys (Rodentia: Octodontidae). Journal of Mammalogy, vol. 70, p. 856- 860. http://dx.doi.org/10.2307/1381728
- -, 1998. The molecular phylogenetics of tuco-tucos (genus Ctenomys, Rodentia: Octodontidae) suggests an early burst of speciation. Molecular Phylogenetics and Evolution, vol. 9, p. 88‑99. PMid:9479698. http://dx.doi.org/10.1006/mpev.1997.0445
- LARIVIÈRE, S. and FERGUSON, SH., 2002. On the evolution of the mammalian baculum: vaginal friction, prolonged intromission or induced ovulation? Mammal Review, vol. 32, p. 283-294. http://dx.doi.org/10.1046/j.1365-2907.2002.00112.x
- LONG, CA. and FRANK, T., 1969. Morphometric variation and function in the baculum, with comments on correlation of parts. Journal of Mammalogy, vol. 49, p. 32-43.
- MASCHERETTI, S., MIROL, PM., GIMÉNEZ, MD., BIDAU, CJ., CONTRERAS, JR. and SEARLE, JB., 2000. Phylogenetics of the speciose and chromosomally variable genus Ctenomys (Ctenomyidae, Octodontoidea), based on mitochondrial cytochrome b sequences. Biological Journal of the Linnean Society, vol. 70, p. 361-376. http://dx.doi.org/10.1111/j.1095-8312.2000.tb00215.x
- NOWAK, RM., 1999. Walker's mammals of the world 6nd ed. Baltimore, London, Johns Hopkins University Press. 1629 p.
- PARAG, A., BENNETT, NC., FAULKES, CG. and BATEMAN, PW., 2006. Penile morphology of African mole rats (Bathyergidae): structural modification in relation to mode of ovulation and degree of sociality. Journal of Zoology, vol. 270, p. 323-329. http://dx.doi.org/10.1111/j.1469-7998.2006.00141.x
- PATTERSON, BD. and THAELER JUNIOR, CS., 1982. The mammalian baculum: hypotheses on the nature of bacular variabillity. Journal of Mammalogy, vol. 63, p. 1-15. http://dx.doi.org/10.2307/1380665
- REIG ,OA., CONTRERAS, JR. and PIANTANIDA, MJ., 1966. Contribución a la elucidacion de la sistematica de las entidades del genero Ctenomys. I. Relaciones de parentesco entre ocho poblaciones de tuco-tucos inferidas del estudio de variables del fenotipo y su correlación con las caracteristicas del cariotipo. Contribución Científica, Serie Zoológica, vol. 2, p. 301-352.
- ROMER, AS. 1962. The vertebrate body 3rd ed. Philadelphia, Saunders. 627 p.
- SIMSON, S., FERRUCCI, L., KURTONUR, C., OZKAN, B. and FOLIPPUCCI, MG., 1995. Phalli and bacula of European dormice: description and comparisson. Hystrix, vol. 6, p. 231-244.
- THOMAS, O., 1915. The penis bone, or "baculum", as a guide to the classification of certain squirrels. Annals and Magazine of Natural History, vol. 15, p. 383-387. http://dx.doi.org/10.1080/00222931508693653
- VITULLO, AD., ROLDAN, ERS. and MERANI, MS., 1988. On the morphology of spermatozoa of tuco-tucos, Ctenomys (Rodentia, Ctenomyidae): new data and its implications for the evolution of the genus. Journal of Zoology, vol. 215, p. 675-683. http://dx.doi.org/10.1111/j.1469-7998.1988.tb02403.x
- ZENUTO, RR., VASSALLO, AI. and BUSCH, C., 2002. Comportamiento social y reproductivo del roedor subterráneo solitario Ctenomys talarum (Rodentia: Ctenomyidae) en condiciones de semicautiverio. Revista Chilena de Historia Natural, vol. 75, p. 165-177.
Publication Dates
-
Publication in this collection
18 Apr 2013 -
Date of issue
Feb 2013
History
-
Received
31 Jan 2012 -
Accepted
30 May 2012