Abstract
The objective of this work was to investigate the patterns of diversity and abundance of ciliates protozoa community in three tropical urban streams and verify which species can be considered bioindicators of water quality. In each stream, three regions (headwater, middle and mouth) were sampled in two years, in the rainy and dry seasons. The abiotic factors (dissolved oxygen, pH, temperature, turbidity, width, depth, flow and conductivity) and biotic factors (total coliforms, bacterioplankton, chlorophyll and flagellates) were analyzed using appropriate methods and ciliates were identified and counted through specialized literature. We identified 84 species distributed into 24 orders. Peniculida was the most representative order followed by Prorodontida. The RDA scores showed a spatial difference between streams. On the other hand, regarding the temporal variation, there was no separation of the sampled data. The Indval analysis showed ten species indicators, such as Coleps hirtus, Euplotes moebiusi and Tintinidium pusillum. The Cluster analysis per stream versus period showed a greater distinction of the streams in the dry season. On the other hand, a low similarity was observed between sections within the same stream. In summary, the results indicated that the ciliates can be used as important tools as bioindicators in lotic environments affected by different degrees of pollution, due to the fact that they have short life cycle, allowing the detection of impacts on a small time scale, as well as by having direct answers to environmental changes and high sensitivity to contaminants.
Keywords:
protists; urban stream; diversity; abundance; trophic
Resumo
O objetivo deste trabalho foi investigar os padrões de diversidade e abundância da comunidade de protozoários ciliados em três córregos urbanos tropicais, bem como verificar quais espécies podem ser consideradas bioindicadoras da qualidade da água. Em cada córrego foram amostradas três regiões (cabeceira, intermediária e foz) em dois anos, nas estações chuvosa e seca. Os fatores abióticos (oxigênio dissolvido, pH, temperatura, turbidez, largura, profundidade, fluxo e condutividade) e fatores bióticos (coliformes totais, bacterioplâncton, clorofila e flagelados) foram analisados por metodologia específica e os ciliados foram contados e identificados por meio de literatura especializada. Foram identificadas 84 espécies distribuídas em 24 ordens. Peniculida foi a ordem mais representativa, seguida por Prorodontida. Os escores da RDA mostraram uma diferença espacial entre os córregos. Por outro lado, em relação à variação temporal, não houve separação entre os dados amostrados. A análise Indval mostrou dez espécies indicadoras, como Coleps hirtus, Euplotes moebiusi e Tintinidium pusillum. A análise de Cluster realizada por riacho versus período evidenciou uma maior distinção dos córregos no período seco. Por outro lado, foi observada uma baixa similaridade entre os locais dentro do mesmo córrego. Em síntese, os resultados indicaram que os ciliados podem ser utilizados como ferramentas importantes como bioindicadores em ambientes lóticos afetados por diferentes graus de poluição, devido ao fato de apresentarem um curto ciclo de vida, permitindo a detecção de impactos em uma pequena escala de tempo, bem como por apresentarem respostas diretas às mudanças ambientais e alta sensibilidade a contaminantes.
Palavras-chave:
protistas; córregos urbanos; diversidade; abundância; trofia
1 Introduction
The demand for freshwater has grown ominously worldwide, but the degradation of its quality has further reduced its availability (Kuhl et al., 2010). Rural and urban watersheds suffer from undue human activities, which alter the characteristics, the balance and the dynamics of natural resources making it difficult the supply of good quality water. Along with these changes, the increase in discharge of pollutant loads into water systems and urbanization usually results in the interruption or limitation of the use of these resources given the impacts on the quality of these ecosystems (Madoni, 2005MADONI, P., 2005. Ciliated protozoan communities and saprobic evaluation of water quality in the hilly zone of some tributaries of the Po River (northern Italy). Hydrobiologia, vol. 541, no. 1, pp. 55-69. http://dx.doi.org/10.1007/s10750-004-4667-8.
http://dx.doi.org/10.1007/s10750-004-466...
).
Urban streams are usually subjected to degradation from high population density, with increased waterproofing, such as sidewalks, asphalt in street, avenues and roads. To monitor these impacts, both physical and chemical factors as the biotic variables have been used considering that they respond to environmental changes and the effects of pollution (Walsh et al., 2005WALSH, C.J., ROY, A.H., FEMINELLA, J.W., COTTINGHAM, P.D., GROFFMAN, P.M. and MORGAN II, R.P., 2005. The urban stream syndrome: Current knowledge and the search for a cure. Journal of the North American Benthological Society, vol. 24, no. 3, pp. 706-723. http://dx.doi.org/10.1899/0887-3593(2005)0240706:TUSSCK2.0.CO;2.
http://dx.doi.org/10.1899/0887-3593(2005...
). Thus, the integrated assessment of these factors allows us to evaluate the replenishment ability and sustainable use of resources, providing a systematic and integrated perception of environmental reality.
Ecological studies involving small and medium-sized streams including the fauna composition have increased in recent year. Such studies have focused on the diverse effects of human activities on these environments (Kühl et al., 2010KÜHL, A.M., ROCHA, C.L.M.S.C., ESPÍNDOLA, E.L.G. and LANSAC-TOHA, F.A., 2010. Rural and urban streams: anthropogenic influences and impacts on water and sediment quality. International Review of Hydrobiology, vol. 95, no. 3, pp. 260-272. http://dx.doi.org/10.1002/iroh.200911190.
http://dx.doi.org/10.1002/iroh.200911190...
; Camargo and Velho, 2011CAMARGO, J.C. and VELHO, L.F.M., 2011. Longitudinal variation of attributes from flagellate protozoan community in tropical streams. Acta Scientiarum Biological Sciences, vol. 33, no. 2, pp. 161-169.; Cunico et al., 2012CUNICO, A.M., FERREIRA, F.A., AGOSTINHO, A.A., BEAUMORD, A. and FERNANDES, A.C.R., 2012. The effects of local and regional environmental factors on the structure of fish assemblages in the Pirapó Basin, Southern Brazil. Landscape and Urban Planning, vol. 105, no. 3, pp. 336-344.).
Recent studies describe the effects of urbanization on streams, including hydrographic changes, rise in concentrations of nutrients and contaminants and changes in morphology and stability of the channel (Walsh et al., 2005WALSH, C.J., ROY, A.H., FEMINELLA, J.W., COTTINGHAM, P.D., GROFFMAN, P.M. and MORGAN II, R.P., 2005. The urban stream syndrome: Current knowledge and the search for a cure. Journal of the North American Benthological Society, vol. 24, no. 3, pp. 706-723. http://dx.doi.org/10.1899/0887-3593(2005)0240706:TUSSCK2.0.CO;2.
http://dx.doi.org/10.1899/0887-3593(2005...
; Cunico et al., 2012CUNICO, A.M., FERREIRA, F.A., AGOSTINHO, A.A., BEAUMORD, A. and FERNANDES, A.C.R., 2012. The effects of local and regional environmental factors on the structure of fish assemblages in the Pirapó Basin, Southern Brazil. Landscape and Urban Planning, vol. 105, no. 3, pp. 336-344.).
For a better understanding of a river water quality, data based on physical (temperature, turbidity, width, depth and flow), chemical (conductivity, pH, dissolved oxygen) and biological (algae, bacteria, fecal coliform and others) characteristics should be considered (Bere and Tundisi, 2010BERE, T. and TUNDISI, J.G., 2010. Biological monitoring of lotic ecosystems: the role of diatoms. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 70, no. 3, pp. 493-502. http://dx.doi.org/10.1590/S1519-69842010005000009. PMid:20737117.
http://dx.doi.org/10.1590/S1519-69842010...
).
The use of biological communities as assessment tools is increasingly becoming important, because they are good indicators of the influence of human expansion on the environment, and are important in assessing water quality (Munn et al., 2002MUNN, M.D., BLACK, R.W. and GRUBER, S.J., 2002. Response of benthic algae to environmental gradients in an agriculturally dominated landscape. Journal of the North American Benthological Society, vol. 21, no. 2, pp. 221-237. http://dx.doi.org/10.2307/1468411.
http://dx.doi.org/10.2307/1468411...
). Among the aquatic communities, protoplankton communities represent the first levels of trophic webs and essential in the transfer of matter and energy (Fenchel, 1987FENCHEL, T., 1987. Ecology of Protozoa: the Biology of free-living phagotrophic protists. Madison: Science Tech. Publishers, 197 p.).
According to some studies, ciliates are efficient quality bioindicators of freshwater environments and can be used in biomonitoring of streams, lakes and reservoirs under different levels of anthropogenic impact (Paiva and Silva-Neto, 2004PAIVA, T.S. and SILVA-NETO, I.D., 2004. Ciliate protists from Cabiúnas lagoon (restinga de Jurubatiba, Macaé, Rio de Janeiro) with emphasis on water quality indicator species and description of . Oxytricha marcili sp. nBrazilian Journal of Biology = Revista Brasileira de Biologia, vol. 64, no. 3, pp. 465-478. http://dx.doi.org/10.1590/S1519-69842004000300010. PMid:15622844.
http://dx.doi.org/10.1590/S1519-69842004...
; Madoni, 2005MADONI, P., 2005. Ciliated protozoan communities and saprobic evaluation of water quality in the hilly zone of some tributaries of the Po River (northern Italy). Hydrobiologia, vol. 541, no. 1, pp. 55-69. http://dx.doi.org/10.1007/s10750-004-4667-8.
http://dx.doi.org/10.1007/s10750-004-466...
; Dias et al., 2008DIAS, R.J.P., WIELOCH, A.H. and D’AGOSTO, M., 2008. The influence of environmental characteristics on the distribution of ciliates (Protozoa, Ciliophora) in an urban stream of southeast Brazil. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 68, no. 2, pp. 287-295. PMid:18660956.). In this sense, these organisms have favorable characteristics, being a large group found in all aquatic environments (Zingel, 2005ZINGEL, P., 2005. Vertical and seasonal dynamics of planktonic ciliates in a strongly stratified hypertrophic lake. Hydrobiologia, vol. 547, no. 1, pp. 163-174. http://dx.doi.org/10.1007/s10750-005-4157-7.
http://dx.doi.org/10.1007/s10750-005-415...
), with short life cycle and high reproduction rate, allowing the detection of environmental impacts in a short timescale (Groliére et al., 1990GROLIÈRE, C.A., CHAKLI, R., SPARAGANO, O. and PEPIN, D., 1990. Application de la colonisation d’un substratartificiel par les Ciliés à l’Étude de la qualité des eauxd’unerivière. European Journal of Protistology, vol. 25, no. 4, pp. 381-390. PMid:23196052.). Also, they are indicative of oxidizing or reducing conditions in the decomposition of organic matter, participating in phosphorus and nitrogen cycling, have high taxonomic density and are sensitive to different concentrations of pollutants, providing responses to different contamination levels (Paerl et al., 2003PAERL, H.W., DYBLE, J., MOISANDER, P.H., NOBLE, R.T., PIEHLER, M.F., PINCKNEY, J.L., STEPPE, T.F., TWOMEY, L. and VALDES, L.M., 2003. Microbial indicators of aquatic ecosystem change: current applications to eutrophication studies. Microbial Ecology, vol. 46, no. 3, pp. 233-246. http://dx.doi.org/10.1016/S0168-6496(03)00200-9. PMid:19719555.
http://dx.doi.org/10.1016/S0168-6496(03)...
).
Studies on the species diversity patterns of these protozoa are complicated by methodological problems especially in regard to the species identification (Auer and Arndt, 2001AUER, B. and ARNDT, H., 2001. Taxonomic composition and biomass of heterotrophic flagellates in relation to lake trophy and season. Freshwater Biology, vol. 46, no. 1, pp. 959-972. http://dx.doi.org/10.1046/j.1365-2427.2001.00730.x.
http://dx.doi.org/10.1046/j.1365-2427.20...
). Consequently, there are few studies on the longitudinal variation in species diversity and density of protozoa ciliates particularly in urban streams (Madoni and Bassanini, 1999MADONI, P. and BASSANINI, N., 1999. Longitudinal changes in the ciliated protozoa communities along a fluvial system polluted by organic matter. European Journal of Protistology, vol. 35, no. 4, pp. 391-402. http://dx.doi.org/10.1016/S0932-4739(99)80048-0.
http://dx.doi.org/10.1016/S0932-4739(99)...
; Dias et al., 2008DIAS, R.J.P., WIELOCH, A.H. and D’AGOSTO, M., 2008. The influence of environmental characteristics on the distribution of ciliates (Protozoa, Ciliophora) in an urban stream of southeast Brazil. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 68, no. 2, pp. 287-295. PMid:18660956.).
This study aims to investigate the patterns of diversity and abundance of the ciliate community and the factors involved in its structuring in three tropical urban streams, as well as to determine which species can be considered bioindicators of water quality.
2 Material and Methods
2.1 Study area and sampling design
The study area included three first-order urban streams, belonging to the upper Paraná River basin, sub-basins of the rivers São Francisco Verdadeiro and Piquiri (Figure 1). The São Francisco Verdadeiro River sub-basin drains about 10 municipalities in Western Paraná State, the municipalities of Cascavel and Toledo are the two largest urban centers in this region. The São Francisco Stream, in the municipality of Cascavel, is 2 km long, with banks occupied by residential areas and throughout its course, this stream receives drainage of surface waters from the surrounding neighborhoods. The Pinheirinho Stream, located in the municipality of Toledo, is affected by agricultural occupation in the surroundings, and is impacted by the residential areas, the introduction of species, pollutant input and other human activities. The Piquiri River basin crosses a region of intensive farming, and current urban development (Figure 1). The main use of this basin is the public water supply and a major problem is that only 10% of the people living in the cities located in the basin are served by sewage treatment (Gubiani et al., 2010GUBIANI, E.A., DAGA, V.S., FRANA, V.A., and GRAÇA, W.J., 2010. Fish, Toledo urban streams, São Francisco Verdadeiro River drainage, upper Paraná River basin, state of Paraná, Brazil. Check List, vol. 6, no. 1, pp. 45-48.).
Sampling sites in three streams located in the cities of Palotina, Toledo, and Cascavel, state of Paraná, southern Brazil.
The municipality of Palotina has about 30,000 inhabitants and various water bodies altered by urbanization, mainly related to lack of sanitation. An example of this is the Jequitibá Stream, which has approximately 3 km2 drainage area and entire length in the urban area of the municipality, with land use consisting of the urban and rural areas, with residential areas, farming and animal husbandry (Gubiani et al., 2010GUBIANI, E.A., DAGA, V.S., FRANA, V.A., and GRAÇA, W.J., 2010. Fish, Toledo urban streams, São Francisco Verdadeiro River drainage, upper Paraná River basin, state of Paraná, Brazil. Check List, vol. 6, no. 1, pp. 45-48.).
Four samplings were conducted in two hydrological periods: rainy (April 2012 and 2013) and dry (September 2012 and February 2013) periods, to collect composite samples under the water surface, in three regions (headwaters, middle and mouth) of each stream.
2.2 Data collection/field sampling
For the rainfall of each sampling period, we considered the average of the five days prior to the date of each sampling. Data were provided by SIMEPAR - Paraná Meteorological Station, considering the months of April 2012 and 2013 as the rainy period and the months of September 2012 and February 2013 as the dry period.
For microbial communities (ciliates, heterotrophic flagellates and heterotrophic bacteria) were collected five liters of water for each sample, with polyethylene plastic bottles, stored in coolers, and transported to the laboratory for analysis. Of these 5 liters, 4 liters were separated for the analysis of ciliates community. In turn, for the bacterioplankton and heterotrophic flagellates were reserved approximately 100 mL of the total water collected from each sample, that were fixed with a solution composed of formaldehyde buffered with borate, alkaline Lugol´s solution and sodium thiosulfate (Sherr and Sherr, 1993SHERR, E.B. and SHERR, B.F., 1993. Preservation and storage of samples for enumeration of heterotrophic protists. In: P. KEMP, B. SHERR, E. SHERR and J. COLE, eds. Current methods in aquatic microbial ecology. New York: Lewis Publishers, pp. 207-212.).
For determination of the fecal coliforms, samples were collected in sterile glass flasks and kept under refrigeration. The Multiple Tube technique were used (MPN) for this determination (APHA, 2006AMERICAN PUBLIC HEALTH ASSOCIATION – APHA, 2006. Standard methods for the examination of water and wastewater. New York: APHA, 824 p.).
The electrical conductivity (μS/cm), dissolved oxygen (mg/L), pH, water temperature (°C), flow (m/s), depth (m) and width (m) were also measured with the help of portable devices and measuring tape. In situ, we collected 2 liters of water for analysis of chlorophyll-a (mg/L) and 10 mL of water for turbidity (NTU).
2.3 Laboratory analysis
2.3.1 Ciliates
Each ciliate sample were concentrated to 100 mL. Samples were examined in the laboratory and abundance ratios were determined within 5 h after collection. Estimates of ciliates species in each sample were made by direct observation in vivo under an optical microscope, at 100× and and 400× magnifications (Weisse, 1991WEISSE, T., 1991. The annual cycle of heterotrophic freshwater nanoflagellates: role of bottom-up versus top-down control. Journal of Plankton Research, vol. 13, no. 1, pp. 167-185. http://dx.doi.org/10.1093/plankt/13.1.167.
http://dx.doi.org/10.1093/plankt/13.1.16...
), and counting 10 slides of 100 µL per sample (Madoni and Bassanini, 1999MADONI, P. and BASSANINI, N., 1999. Longitudinal changes in the ciliated protozoa communities along a fluvial system polluted by organic matter. European Journal of Protistology, vol. 35, no. 4, pp. 391-402. http://dx.doi.org/10.1016/S0932-4739(99)80048-0.
http://dx.doi.org/10.1016/S0932-4739(99)...
; Madoni, 2005MADONI, P., 2005. Ciliated protozoan communities and saprobic evaluation of water quality in the hilly zone of some tributaries of the Po River (northern Italy). Hydrobiologia, vol. 541, no. 1, pp. 55-69. http://dx.doi.org/10.1007/s10750-004-4667-8.
http://dx.doi.org/10.1007/s10750-004-466...
). The total density was expressed as individuals/L. The identification was based on specialized literature (Foissner and Berger 1996FOISSNER, W. and BERGER, H., 1996. A user-friendly guide to the ciliates (Protozoa, Ciliophora) commonly used by hydrobiologists as bioindicators in rivers, lakes and waste waters, with notes on their ecology. Freshwater Biology, vol. 35, no. 1, pp. 375-482.; Foissner et al., 1999FOISSNER, W., BERGER, H. and SCHAUMBURG, J., 1999. Identification and ecology of limnetic plankton ciliates. Munich: Bavarian State Office for Water Management, 793 p.). The taxonomic classification was performed according to Lynn (2008)LYNN, D.H., 2008. The ciliated protozoa: characterization, classification, and guide to the literature. Dordrecht: Springer, 605 p..
2.3.2 Heterotrophic flagellates
Samples (100 mL) were fixed with a solution of formaldehyde, and alkaline lugol and thiosulfate (Sherr and Sherr, 1993SHERR, E.B. and SHERR, B.F., 1993. Preservation and storage of samples for enumeration of heterotrophic protists. In: P. KEMP, B. SHERR, E. SHERR and J. COLE, eds. Current methods in aquatic microbial ecology. New York: Lewis Publishers, pp. 207-212.). The density of heterotrophic flagellates was estimated by filtering subsamples of 10 mL through black Nucleopore Filters (0.8μm), stained with 1mL fluorochrome DAPI (4,6-diamidino-2-phenylindole) at 0.1%, for 15 minutes in the dark. Counting was performed in an epifluorescence microscope (Olympus BX51). To determine the flagellate density on each slide, we counted at least 50 fields under UV light, at 1000× magnification. The total density was expressed as individuals/L (Sherr and Sherr, 1993SHERR, E.B. and SHERR, B.F., 1993. Preservation and storage of samples for enumeration of heterotrophic protists. In: P. KEMP, B. SHERR, E. SHERR and J. COLE, eds. Current methods in aquatic microbial ecology. New York: Lewis Publishers, pp. 207-212.).
The differentiation of each fraction of flagellates was made simultaneously, using as a criterion the reddish color of autotrophs when subjected to blue light in contrast to the greenish color of heterotrophs, and only the data of heterotrophic flagellates were considered in this study.
2.3.3 Bacterioplankton
The process of fixation and estimation of density of the bacterioplankton were determined using the same protocol of filtration, mounting and storage of slides described for flagellates (Sherr and Sherr, 1993SHERR, E.B. and SHERR, B.F., 1993. Preservation and storage of samples for enumeration of heterotrophic protists. In: P. KEMP, B. SHERR, E. SHERR and J. COLE, eds. Current methods in aquatic microbial ecology. New York: Lewis Publishers, pp. 207-212.). However, subsamples of 1mL water were filtered through black Nucleopore/Watchman filter (0.2 μm) stained with 1 mL fluorochrome DAPI (4,6-diamidino-2-phenylindole) at 0.1%, for 5 minutes in the dark. Bacteria were quantified at 1000× magnification in an epifluorescence microscope (Sherr and Sherr, 1993SHERR, E.B. and SHERR, B.F., 1993. Preservation and storage of samples for enumeration of heterotrophic protists. In: P. KEMP, B. SHERR, E. SHERR and J. COLE, eds. Current methods in aquatic microbial ecology. New York: Lewis Publishers, pp. 207-212.). The total density was expressed in cells/mL.
2.3.4 Chlorophyll and turbidity
Concentrations of chlorophyll-a (µg/L) were determined at the Laboratory of Basic Limnology of Nupélia/UEM, according to Golterman et al. (1978)GOLTERMAN, H.L., CLYMO, R.S. and OHNSTAD, M.A.M., 1978. Methods for physical and chemical analysis of freshwaters. Oxford: Blackwell Scientific Publications..
The determination of turbidity was carried outat the LEPI - Laboratory of Fisheries and Ichthyology/UFPR - Campus Palotina, using a turbidimeter HACH 2100P (APHA, 2006AMERICAN PUBLIC HEALTH ASSOCIATION – APHA, 2006. Standard methods for the examination of water and wastewater. New York: APHA, 824 p.).
2.3.5 Total coliforms
Thermotolerant coliforms were determined according to APHA (2006)AMERICAN PUBLIC HEALTH ASSOCIATION – APHA, 2006. Standard methods for the examination of water and wastewater. New York: APHA, 824 p.. The values of the analyzed characteristics were compared with the values recommended by CONAMA Resolution 357/2005 (Article 34) (Brasil, 2005BRASIL. Conselho Nacional de Meio Ambiente – CONAMA, 2005. Resolução n° 357 de 17 de março de 2005. Diário Oficial da República Federativa do Brasil, Brasília, 18 mar., pp. 58-63.), in order to check whether the parameters are within the acceptable standards by the aforementioned legislation.
2.4 Data analysis
2.4.1 Redundancy Analysis – RDA
We used Redundancy Analysis (RDA) (Legendre and Legendre, 1998LEGENDRE, P. and LEGENDRE, L., 1998. Numerical ecology. Amsterdam: Elsevier, 853 p.) to estimate the relative role of environmental predictors (water temperature, dissolved oxygen, pH, electrical conductivity, turbidity, width, depth, chlorophyll, total coliforms and flagellates) on the structure of the ciliate community.
This analysis was applied to investigate the influence of environmental variables, as well as bacteria, chlorophyll and flagellates on the ciliate community structure. To this end, we used a matrix with the density of the different species of ciliates and another matrix with the abiotic variables (water temperature, pH, electrical conductivity, dissolved oxygen) and biotic variables (chlorophyll, density of bacterioplankton, total coliforms and flagellate). Biotic and abiotic data were previously log transformed except for pH. This analysis was performed using the software R 3.0.1, and the package vegan.
2.4.2 Indicator species
We calculated for each stream and period a Indicator Value Analysis (INDVAL - Dufrêne and Legendre 1997DUFRÊNE, M. and LEGENDRE, P., 1997. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecological Monographs, vol. 67, no. 3, pp. 357-366.) to identify possible indicator species. This method determines the most characteristic organism for each environment and evaluates their specificity with a score ranging from 0 to 1, 1 being an organism present only in a given environment and totally absent from the others. This analysis was performed using the software PC-ORD (McCune and Mefford, 1999MCCUNE, B. and MEFFORD, M.J., 1999. PC-ORD multivariate analysis of Ecological data: Version 4. Gleneden Beach: MjM Software Design.).
2.4.3 Cluster Analysis
The general patterns of similarity between the streams and sampling sites within the streams were evidenced by a Cluster analysis using the software PCOrd 8.6. For this analysis, we considered the presence and absence of species of ciliates at each sampling site in different hydrological periods (rainy and dry). The analysis used as a similarity coefficient the Unweighted Pair Group Method Average (UPGMA) linkage method, in which the distance between two groups is the arithmetic average of the distance between all pairs of objects belonging to both groups (Anderberg, 2014ANDERBERG, M.R., 2014. Cluster Analysis for applications: probability and mathematical statistics: a series of monographs and textbooks. New York: Academic Press. 215 p.). In calculating the distance between the species (distance coefficient), we used a simple Euclidean distance for graphical representation through a dendrogram.
3 Results
3.1 Ciliate species composition
We identified 84 species distributed into 24 orders. Peniculida was the most representative order (12 species) followed by Prorodontida (8 species) (Table 1). The only taxa recorded in all samples of the three streams and in both hydrological periods was Vorticella aquadulcis complex (Order Sessilida).
Inventory of ciliated protozoa in three urbanized streams (Jequitibá, Pinheirinho and San Francisco) in each hydrological period where “x” indicates presence and “-” indicates absence of that species in that stream and period.
3.2 Influence of environmental variables on ciliate communities
The first and second axis of the RDA were significant (p<0.005) and were retained for interpretation (R2 adjusted= 0.40;p=0.006). The RDA analysis showed that the intervenient factors (biotic and abiotic variables) together with the most abundant ciliate species accounted for 40% of the data variability for the three streams in the rainy and dry periods (Figure 2).
Ordination diagram for the first two axes of Redundancy Analysis, with the scores of streams sampled according to the abiotic variables: Cond=electrical conductivity; Temp=temperature; Turb= turbidity; DO=dissolved oxygen; Chlor= chlorophyll a; Bac= bacterioplankton; Colif: coliforms and HF= heterotrophic flagellates. Streams: JeqR: Jequitibá Rainy; JeqD: Jequitibá Dry; PinR: Pinheirinho Rainy; PinD: Pinheirinho Dry; SFrR: San Francisco Rainy and SFrD: San Francisco Rainy.
RDA scores showed a spatial difference between streams (Figure 2). Thus, axis 1 separeted Jequitibá from São Francisco and Pinheirinho streams. As Jequitibá associated with most environmental variables, such as conductivity, dissolved oxygen, depth, pH, turbidity, temperature and flow, as well as flagellates heterotrophic density and chlorophyll concentration. While São Francisco and Pinheirinho streams were associated only with bacteria and total coliforms. Regarding the temporal variation, there was no separation of the sampled data.
Dissolved oxygen, conductivity and width along with chlorophyll and flagellates were positively associated to the distribution of the ciliates Caenomorpha sp., Dexiostoma campylum, Lembadion bullinum, L. lucens, Paramecium caudatum, Tetrahymena piryformis and Tintinnidium pusillum.
The abiotic variables flow, turbidity, temperature and pH influence the distribution of Coleps hirtus, Cyclidium glaucoma, Entamoeba histolytica, Halteria grandinella, Monodinium balbiani, Ophryoglena sp., Dexiostoma tetraedricum, Urocentrum turbo, Vorticella convallaria and V. campanula (Figure 2). Aspidica cicada, Cinetochilum margaritaceum, Glaucoma scintilans and Paramecium aurelia were influenced by the biological variables total coliforms and bacterioplankton (Figure 2).
3.3 Indicator species
The results of the Indicator Value Analysis (INDVAL) revealed the occurrence of 10 species with bioindicator potential regardless of the period, especially Coleps hirtus, Tintinidium pusilum and Euplotes moebiusi, which had the highest levels of significance (p <0.024, 0.019 and 0.018, respectively). The Jequitibá Stream had the largest number of bioindicator species, regardless of the hydrological period. On the other hand, the lowest number of bioindicator species was found in the Pinheirinho Stream (Table 2).
Value of indicator species of ciliates in the Jequitibá, Pinheirinho and San Francisco streams in the rainy and dry seasons, derived from a IndVal analysis, based on occurrence data and abundance of species, where p <0.05. The values in bold type indicate that the content was significant.
3.4 Cluster analysis
The Cluster analysis per stream versus period revealed a group formed by the streams Jequitibá in the dry period and São Francisco in both periods. The most distant group consisted of the streams Pinheirinho and Jequitibá in the dry period (Figure 3).
Dendogram Cluster analysis (UPGMA) showing the similarity between the streams and periods based on ciliates community. Jeq: Jequitibá; SFr: San Francisco; Pin: Pinheirinho; S: Dry; C: Rainy.
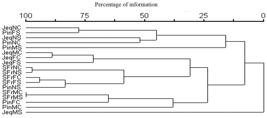
The Cluster analysis per stream section versus period evidenced a low similarity between sites within the same stream. The highest similarity occurred between JeqNC and PinFS, based on the species of ciliates. The most distant group was formed by PinMC and JeqMS (Figure 4).
Dendogram Cluster analysis (UPGMA) showing the similarity between regions within each target stream and the hydrological period based on ciliates community. Jeq: Jequitibá; SFr: San Francisco; Pin: Pinheirinho; N: headwaters; M: middle; F: mouth; S: Dry; C: Rainy.
4 Discussion
The species richness recorded in this study (88 taxa) is within the range found in different lotic environments. Studies on species richness of ciliates in lotic environments have shown an approximate range from 20 to 200 species of these protozoa (Madoni, 2005MADONI, P., 2005. Ciliated protozoan communities and saprobic evaluation of water quality in the hilly zone of some tributaries of the Po River (northern Italy). Hydrobiologia, vol. 541, no. 1, pp. 55-69. http://dx.doi.org/10.1007/s10750-004-4667-8.
http://dx.doi.org/10.1007/s10750-004-466...
; Madoni and Braghiroli, 2007MADONI, P. and BRAGHIROLI, S., 2007. Changes in the ciliate assemblage along a fluvial system related to physical, chemical and geomorphological characteristics. European Journal of Protistology, vol. 43, no. 2, pp. 67-75. http://dx.doi.org/10.1016/j.ejop.2006.09.004. PMid:17222541.
http://dx.doi.org/10.1016/j.ejop.2006.09...
; Reiss and Schmid-Araya, 2008REISS, J. and SCHMID-ARAYA, J.M., 2008. Existing in plenty: abundance, biomass and diversity of ciliates and meiofauna in small streams. Freshwater Biology, vol. 53, no. 4, pp. 652-668. http://dx.doi.org/10.1111/j.1365-2427.2007.01907.x.
http://dx.doi.org/10.1111/j.1365-2427.20...
; Dias et al., 2008DIAS, R.J.P., WIELOCH, A.H. and D’AGOSTO, M., 2008. The influence of environmental characteristics on the distribution of ciliates (Protozoa, Ciliophora) in an urban stream of southeast Brazil. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 68, no. 2, pp. 287-295. PMid:18660956.; Bradley et al., 2010BRADLEY, M.W., ESTEBAN, G.F. and FINLAY, B.J., 2010. Ciliates in chalk stream habitats congregate in biodiversity hot spots. Microbiological Research, vol. 161, no. 7, pp. 619-625. PMid:20600862.; Pereira et al., 2014PEREIRA, P.S., FERNANDES, L.A.C., DIAS, R.J.P., SAMPAIO, M.S., SILVA-NETO, I.D., BAPTISTA, D.F. and OLIVEIRA, J.L.M., 2014. Ecological water quality assessment in the Guapiaçu-Macacu hydrographic complex (Rio de Janeiro, Brazil) using multiple indicators. Revista Ambiente & Água, vol. 9, no. 3, pp. 409-423.; Lobato Junior and Araújo, 2015LOBATO JÚNIOR, W.S. and ARAÚJO, M.F.F., 2015. Protozoários de vida livre (Ciliophora, Mastigophora e Sarcodina) em dois trechos de um ambiente lótico do nordeste do Brasil e seu potencial uso como bioindicadores. Ciência e Natura, vol. 37, no. 1, pp. 57-63.).
The most species-rich orders were Peniculida and Prorodontida, which include algivorous, bacterivorous, omnivorous and predator species. These orders are especially found in mesotrophic and eutrophic environments, conditions prevalent in the studied streams (Kim et al., 2003KIM, B.H., MOON, E.Y., HONG, S.S. and HAN, M.S., 2003. Seasonal variations of planktonic ciliates with reference to their prey in a shallow and eutrophic Korean stream. Journal of Freshwater Ecology, vol. 18, no. 4, pp. 577-584. http://dx.doi.org/10.1080/02705060.2003.9663998.
http://dx.doi.org/10.1080/02705060.2003....
; Gücker and Pusch, 2006GÜCKER, B. and PUSCH, M.T., 2006. Regulation of nutrient uptake in eutrophic lowland streams. Limnology and Oceanography, vol. 51, no. 3, pp. 1443-1453. http://dx.doi.org/10.4319/lo.2006.51.3.1443.
http://dx.doi.org/10.4319/lo.2006.51.3.1...
; Eckert and Carrick, 2014ECKERT, R.A. and CARRICK, H.J., 2014. Evidence for consumer regulation of biofilm–nutrient interactions among hard water streams (Pennsylvania, USA). Hydrobiologia, vol. 722, no. 1, pp. 183-198. http://dx.doi.org/10.1007/s10750-013-1699-y.
http://dx.doi.org/10.1007/s10750-013-169...
).
On the other hand, some orders, like Oligotrichida, showed low species richness in the studied streams, maybe because the species of this order are more adapted to oligotrophic environments (Beaver and Crisman, 1989BEAVER, J.R. and CRISMAN, T.L., 1989. Analysis of the community structure of planktonic ciliated Protozoa relative to trophic state in Florida lakes. Hydrobiologia, vol. 174, no. 3, pp. 177-184. http://dx.doi.org/10.1007/BF00008155.
http://dx.doi.org/10.1007/BF00008155...
), and to planktonic life, requiring long residence time of the water to develop their populations (Pauleto et al., 2009PAULETO, G.M., VELHO, L.F.M., BUOSI, P.R.B., BRÃO, A.F.S., LANSAC-TÔHA, F.A. and BONECKER, C.C., 2009. Spatial and temporal patterns of ciliate species composition (Protozoa: Ciliophora) in the plankton of the Upper Paraná River floodplain. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 69, no. 2, (Suppl.), pp. 517-527. http://dx.doi.org/10.1590/S1519-69842009000300007. PMid:19738959.
http://dx.doi.org/10.1590/S1519-69842009...
).
The presence of the Vorticella in all samplings and with high abundances has great representativeness in various works developed in rivers and streams (Madoni and Braghiroli, 2007MADONI, P. and BRAGHIROLI, S., 2007. Changes in the ciliate assemblage along a fluvial system related to physical, chemical and geomorphological characteristics. European Journal of Protistology, vol. 43, no. 2, pp. 67-75. http://dx.doi.org/10.1016/j.ejop.2006.09.004. PMid:17222541.
http://dx.doi.org/10.1016/j.ejop.2006.09...
; Andrushchyshyn et al., 2007ANDRUSHCHYSHYN, O.P., WILSON, P.K. and WILLIAMS, D.D., 2007. Ciliate communities in shallow groundwater: seasonal and spatial characteristics. Freshwater Biology, vol. 52, no. 9, pp. 1745-1761. http://dx.doi.org/10.1111/j.1365-2427.2007.01806.x.
http://dx.doi.org/10.1111/j.1365-2427.20...
).
The species of this genus have preferably periphytic habit (Foissner and Berger, 1996FOISSNER, W. and BERGER, H., 1996. A user-friendly guide to the ciliates (Protozoa, Ciliophora) commonly used by hydrobiologists as bioindicators in rivers, lakes and waste waters, with notes on their ecology. Freshwater Biology, vol. 35, no. 1, pp. 375-482.), having the ability to adhere to various substrates, making them more likely to resist mechanical action generated by the flow of lotic environments, therefore, considered as indicators of this type of environment (Kiss et al., 2009KISS, Á.K., ÁCS, É., KISS, K.T. and TÖRÖK, J.K., 2009. Structure and seasonal dynamics of the protozoan community (heterotrophic flagellates, ciliates, amoeboid protozoa) in the plankton of a large river (River Danube, Hungary). European Journal of Protistology, vol. 45, no. 2, pp. 121-138. http://dx.doi.org/10.1016/j.ejop.2008.08.002. PMid:19285382.
http://dx.doi.org/10.1016/j.ejop.2008.08...
).
The high abundance of ciliates as Caenomorpha sp., D. campylum, L. bullinum, L. lucens, P. caudatum, T. piryformis and T. pusilum in environments with high concentrations of resources, such as chlorophyll and total coliforms, is probably related to their feeding habits, since these species primarily consume algae and bacteria (Zingel, 2005ZINGEL, P., 2005. Vertical and seasonal dynamics of planktonic ciliates in a strongly stratified hypertrophic lake. Hydrobiologia, vol. 547, no. 1, pp. 163-174. http://dx.doi.org/10.1007/s10750-005-4157-7.
http://dx.doi.org/10.1007/s10750-005-415...
).
Furthermore, the positive relationship between the bacterivorous species A. cicada, C. margaritaceum, G. scintilans and P. aurelia and total coliforms and bacterioplankton, suggests that bacterial growth has been sufficient to maintain a high abundance of species with such feeding habits (Madoni, 2005MADONI, P., 2005. Ciliated protozoan communities and saprobic evaluation of water quality in the hilly zone of some tributaries of the Po River (northern Italy). Hydrobiologia, vol. 541, no. 1, pp. 55-69. http://dx.doi.org/10.1007/s10750-004-4667-8.
http://dx.doi.org/10.1007/s10750-004-466...
).
The presence of some predatory ciliates, such as D. campylum, indicates the presence of flagellates in the sampled sites. According to Camargo and Velho (2011)CAMARGO, J.C. and VELHO, L.F.M., 2011. Longitudinal variation of attributes from flagellate protozoan community in tropical streams. Acta Scientiarum Biological Sciences, vol. 33, no. 2, pp. 161-169., the success of flagellate protozoa in streams can be attributed to human activities in the region, as some groups are considered indicators of impacted environments because of their preference for environments rich in organic matter (Reynolds and Descy, 1996REYNOLDS, C.S. and DESCY, J.P., 1996. The production, biomass and structure of phytoplankton in large rivers. Archiv für Hydrobiologie, vol. 113, no. 4, pp. 161-187.).
The variables flow, turbidity, temperature and pH influence the distribution of some species of ciliates, because, depending on the amount and origin of suspended material, they can be favorable to the occurrence of bacterivorous, algivorous and omnivorous ciliates, such as C. hirtus, C. glaucoma, E. histolytica, H. grandinella, M. balbiani, Ophryoglena. sp., D. tetraedricum, U. turbo, V. convallaria and V. campanula (Foissner and Berger, 1996FOISSNER, W. and BERGER, H., 1996. A user-friendly guide to the ciliates (Protozoa, Ciliophora) commonly used by hydrobiologists as bioindicators in rivers, lakes and waste waters, with notes on their ecology. Freshwater Biology, vol. 35, no. 1, pp. 375-482.). Another important factor that may have contributed to the record of these species in these sites is the frequent physical disturbance of the bed during floods and anthropogenic changes nearby (Matthaei and Townsend, 2000MATTHAEI, C.D. and TOWNSEND, C.R., 2000. Long- term effects of local disturbance history on mobile stream invertebrates. Oecologia, vol. 125, no. 1, pp. 119-126. http://dx.doi.org/10.1007/PL00008883.
http://dx.doi.org/10.1007/PL00008883...
), increasing or not the water flow in these environments, suggesting a continuous input of allochthonous organic matter into the investigated streams (Zingel, 2005ZINGEL, P., 2005. Vertical and seasonal dynamics of planktonic ciliates in a strongly stratified hypertrophic lake. Hydrobiologia, vol. 547, no. 1, pp. 163-174. http://dx.doi.org/10.1007/s10750-005-4157-7.
http://dx.doi.org/10.1007/s10750-005-415...
). It is noteworthy that higher temperatures favor the consumption of bacteria, since bacterivory is temperature dependent and increases proportionally to this (Sherr et al., 1988SHERR, B., SHERR, R. and RASSOULZADEGAN, F., 1988. Rates of Digeston of Bacteria by Marine Phagotrophic Protozoa: Temperatura Dependence. Applied and Environmental Microbiology, vol. 54, no. 5, pp. 1095-1988. PMid:16347623.). Regarding the influence of temperature and pH, Mansano et al. (2014)MANSANO, A.S., HISATUGO, K.F., HAYASHI, L.H. and REGALI-SELEGHIM, M.H., 2014. The importance of protozoan bacterivory in a subtropical environment (Lobo-Broa Reservoir, SP, Brazil). Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 74, no. 3, pp. 569-578. http://dx.doi.org/10.1590/bjb.2014.0081. PMid:25296204.
http://dx.doi.org/10.1590/bjb.2014.0081...
reported the highest densities of C. glaucoma and H. grandinella with higher values of pH and temperature.
None of the species indicated by the IndVal analysis is bioindicator of oligotrophic environments. This points out that the environments studied, although they are not under critical organic pollution, presented some species typical of beta-alpha-mesosaprobic, oligo-beta-saprobic and alpha-meso-saprobic environments (Foissner et al., 1995FOISSNER, W., BLATTERER, H., BERGER, H. and KOHMANN, F., 1995. Taxonomischeund okologische Revision der Ciliaten des Saprobien- systems – Band IV: Gymnostomatea, Loxodes, Suctoria. Informations Berichte des Bayerischen Landesamtes für Wasserwirtschaft, vol. 1, no. 1, pp. 1-540.).
Besides, there was a greater similarity in ciliate species composition between streams than between sections within each stream. This may be due to different characteristics in each section of the stream. Herein, the environments exhibited a low similarity to each other, with many rare species; only 26 species were recorded in more than two sampling sites, indicating a high turnover rate of species between sites.
This may be due to the conditions of each water body, as they are often disturbed by natural processes, such as changes in water flow and sediment input, and man-made activities, such as deforestation and removal of riparian vegetation, thus reducing the stability of the banks, increasing erosion and sedimentation processes, affecting in this way, the diversity of aquatic communities (Libório et al., 2013LIBÓRIO, R.A., SOUZA, A.L.T., FONSECA, D.G. and TANAKA, M.O., 2013. Influence of riparian vegetation and forest structure on the water quality of rural low-order streams in SE Brazil. Forest Ecology and Management, vol. 341, no. 1, pp. 12-18.).
Although some sampled sites have been characterized by a relative load of organic pollution and some species have been indicator of these conditions, the community structure did not show the pattern expected for impacted environments, that is, anthropogenic changes reduce the richness of taxa to few generalist and tolerant species, leading to greater homogenization of biological communities, thereby decreasing diversity in streams (Rahel, 2002RAHEL, F.J., 2002. Homogenization of fish faunas across the United States. Science, vol. 288, no. 1, pp. 854-856. PMid:10797007.).
In this sense, the ciliate community distribution was mainly influenced by biotic and abiotic variables and not by seasonal conditions. There is greater similarity in ciliate species composition between streams than between sections within each stream. In general, the sites studied presented species associated with a certain trophic state, nevertheless, the community showed high diversity, indicating that these environments are not under severe pollution.
The results indicated that the ciliates can be used as important tools as bioindicators in lotic environments affected by different degrees of pollution, due to the fact that they have short life cycle, allowing the detection of impacts on a small time scale, as well as by having direct answers to environmental changes and high sensitivity to contaminants.
-
(With 4 figures)
References
- AMERICAN PUBLIC HEALTH ASSOCIATION – APHA, 2006. Standard methods for the examination of water and wastewater. New York: APHA, 824 p.
- ANDERBERG, M.R., 2014. Cluster Analysis for applications: probability and mathematical statistics: a series of monographs and textbooks. New York: Academic Press. 215 p.
- ANDRUSHCHYSHYN, O.P., WILSON, P.K. and WILLIAMS, D.D., 2007. Ciliate communities in shallow groundwater: seasonal and spatial characteristics. Freshwater Biology, vol. 52, no. 9, pp. 1745-1761. http://dx.doi.org/10.1111/j.1365-2427.2007.01806.x
» http://dx.doi.org/10.1111/j.1365-2427.2007.01806.x - AUER, B. and ARNDT, H., 2001. Taxonomic composition and biomass of heterotrophic flagellates in relation to lake trophy and season. Freshwater Biology, vol. 46, no. 1, pp. 959-972. http://dx.doi.org/10.1046/j.1365-2427.2001.00730.x
» http://dx.doi.org/10.1046/j.1365-2427.2001.00730.x - BEAVER, J.R. and CRISMAN, T.L., 1989. Analysis of the community structure of planktonic ciliated Protozoa relative to trophic state in Florida lakes. Hydrobiologia, vol. 174, no. 3, pp. 177-184. http://dx.doi.org/10.1007/BF00008155
» http://dx.doi.org/10.1007/BF00008155 - BERE, T. and TUNDISI, J.G., 2010. Biological monitoring of lotic ecosystems: the role of diatoms. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 70, no. 3, pp. 493-502. http://dx.doi.org/10.1590/S1519-69842010005000009 PMid:20737117.
» http://dx.doi.org/10.1590/S1519-69842010005000009 - BRADLEY, M.W., ESTEBAN, G.F. and FINLAY, B.J., 2010. Ciliates in chalk stream habitats congregate in biodiversity hot spots. Microbiological Research, vol. 161, no. 7, pp. 619-625. PMid:20600862.
- BRASIL. Conselho Nacional de Meio Ambiente – CONAMA, 2005. Resolução n° 357 de 17 de março de 2005. Diário Oficial da República Federativa do Brasil, Brasília, 18 mar., pp. 58-63.
- CAMARGO, J.C. and VELHO, L.F.M., 2011. Longitudinal variation of attributes from flagellate protozoan community in tropical streams. Acta Scientiarum Biological Sciences, vol. 33, no. 2, pp. 161-169.
- CUNICO, A.M., FERREIRA, F.A., AGOSTINHO, A.A., BEAUMORD, A. and FERNANDES, A.C.R., 2012. The effects of local and regional environmental factors on the structure of fish assemblages in the Pirapó Basin, Southern Brazil. Landscape and Urban Planning, vol. 105, no. 3, pp. 336-344.
- DIAS, R.J.P., WIELOCH, A.H. and D’AGOSTO, M., 2008. The influence of environmental characteristics on the distribution of ciliates (Protozoa, Ciliophora) in an urban stream of southeast Brazil. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 68, no. 2, pp. 287-295. PMid:18660956.
- DUFRÊNE, M. and LEGENDRE, P., 1997. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecological Monographs, vol. 67, no. 3, pp. 357-366.
- ECKERT, R.A. and CARRICK, H.J., 2014. Evidence for consumer regulation of biofilm–nutrient interactions among hard water streams (Pennsylvania, USA). Hydrobiologia, vol. 722, no. 1, pp. 183-198. http://dx.doi.org/10.1007/s10750-013-1699-y
» http://dx.doi.org/10.1007/s10750-013-1699-y - FENCHEL, T., 1987. Ecology of Protozoa: the Biology of free-living phagotrophic protists. Madison: Science Tech. Publishers, 197 p.
- FOISSNER, W. and BERGER, H., 1996. A user-friendly guide to the ciliates (Protozoa, Ciliophora) commonly used by hydrobiologists as bioindicators in rivers, lakes and waste waters, with notes on their ecology. Freshwater Biology, vol. 35, no. 1, pp. 375-482.
- FOISSNER, W., BERGER, H. and SCHAUMBURG, J., 1999. Identification and ecology of limnetic plankton ciliates. Munich: Bavarian State Office for Water Management, 793 p.
- FOISSNER, W., BLATTERER, H., BERGER, H. and KOHMANN, F., 1995. Taxonomischeund okologische Revision der Ciliaten des Saprobien- systems – Band IV: Gymnostomatea, Loxodes, Suctoria. Informations Berichte des Bayerischen Landesamtes für Wasserwirtschaft, vol. 1, no. 1, pp. 1-540.
- GOLTERMAN, H.L., CLYMO, R.S. and OHNSTAD, M.A.M., 1978. Methods for physical and chemical analysis of freshwaters. Oxford: Blackwell Scientific Publications.
- GROLIÈRE, C.A., CHAKLI, R., SPARAGANO, O. and PEPIN, D., 1990. Application de la colonisation d’un substratartificiel par les Ciliés à l’Étude de la qualité des eauxd’unerivière. European Journal of Protistology, vol. 25, no. 4, pp. 381-390. PMid:23196052.
- GUBIANI, E.A., DAGA, V.S., FRANA, V.A., and GRAÇA, W.J., 2010. Fish, Toledo urban streams, São Francisco Verdadeiro River drainage, upper Paraná River basin, state of Paraná, Brazil. Check List, vol. 6, no. 1, pp. 45-48.
- GÜCKER, B. and PUSCH, M.T., 2006. Regulation of nutrient uptake in eutrophic lowland streams. Limnology and Oceanography, vol. 51, no. 3, pp. 1443-1453. http://dx.doi.org/10.4319/lo.2006.51.3.1443
» http://dx.doi.org/10.4319/lo.2006.51.3.1443 - KIM, B.H., MOON, E.Y., HONG, S.S. and HAN, M.S., 2003. Seasonal variations of planktonic ciliates with reference to their prey in a shallow and eutrophic Korean stream. Journal of Freshwater Ecology, vol. 18, no. 4, pp. 577-584. http://dx.doi.org/10.1080/02705060.2003.9663998
» http://dx.doi.org/10.1080/02705060.2003.9663998 - KISS, Á.K., ÁCS, É., KISS, K.T. and TÖRÖK, J.K., 2009. Structure and seasonal dynamics of the protozoan community (heterotrophic flagellates, ciliates, amoeboid protozoa) in the plankton of a large river (River Danube, Hungary). European Journal of Protistology, vol. 45, no. 2, pp. 121-138. http://dx.doi.org/10.1016/j.ejop.2008.08.002 PMid:19285382.
» http://dx.doi.org/10.1016/j.ejop.2008.08.002 - KÜHL, A.M., ROCHA, C.L.M.S.C., ESPÍNDOLA, E.L.G. and LANSAC-TOHA, F.A., 2010. Rural and urban streams: anthropogenic influences and impacts on water and sediment quality. International Review of Hydrobiology, vol. 95, no. 3, pp. 260-272. http://dx.doi.org/10.1002/iroh.200911190
» http://dx.doi.org/10.1002/iroh.200911190 - LEGENDRE, P. and LEGENDRE, L., 1998. Numerical ecology. Amsterdam: Elsevier, 853 p.
- LIBÓRIO, R.A., SOUZA, A.L.T., FONSECA, D.G. and TANAKA, M.O., 2013. Influence of riparian vegetation and forest structure on the water quality of rural low-order streams in SE Brazil. Forest Ecology and Management, vol. 341, no. 1, pp. 12-18.
- LOBATO JÚNIOR, W.S. and ARAÚJO, M.F.F., 2015. Protozoários de vida livre (Ciliophora, Mastigophora e Sarcodina) em dois trechos de um ambiente lótico do nordeste do Brasil e seu potencial uso como bioindicadores. Ciência e Natura, vol. 37, no. 1, pp. 57-63.
- LYNN, D.H., 2008. The ciliated protozoa: characterization, classification, and guide to the literature. Dordrecht: Springer, 605 p.
- MADONI, P., 2005. Ciliated protozoan communities and saprobic evaluation of water quality in the hilly zone of some tributaries of the Po River (northern Italy). Hydrobiologia, vol. 541, no. 1, pp. 55-69. http://dx.doi.org/10.1007/s10750-004-4667-8
» http://dx.doi.org/10.1007/s10750-004-4667-8 - MADONI, P. and BASSANINI, N., 1999. Longitudinal changes in the ciliated protozoa communities along a fluvial system polluted by organic matter. European Journal of Protistology, vol. 35, no. 4, pp. 391-402. http://dx.doi.org/10.1016/S0932-4739(99)80048-0
» http://dx.doi.org/10.1016/S0932-4739(99)80048-0 - MADONI, P. and BRAGHIROLI, S., 2007. Changes in the ciliate assemblage along a fluvial system related to physical, chemical and geomorphological characteristics. European Journal of Protistology, vol. 43, no. 2, pp. 67-75. http://dx.doi.org/10.1016/j.ejop.2006.09.004 PMid:17222541.
» http://dx.doi.org/10.1016/j.ejop.2006.09.004 - MANSANO, A.S., HISATUGO, K.F., HAYASHI, L.H. and REGALI-SELEGHIM, M.H., 2014. The importance of protozoan bacterivory in a subtropical environment (Lobo-Broa Reservoir, SP, Brazil). Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 74, no. 3, pp. 569-578. http://dx.doi.org/10.1590/bjb.2014.0081 PMid:25296204.
» http://dx.doi.org/10.1590/bjb.2014.0081 - MATTHAEI, C.D. and TOWNSEND, C.R., 2000. Long- term effects of local disturbance history on mobile stream invertebrates. Oecologia, vol. 125, no. 1, pp. 119-126. http://dx.doi.org/10.1007/PL00008883
» http://dx.doi.org/10.1007/PL00008883 - MCCUNE, B. and MEFFORD, M.J., 1999. PC-ORD multivariate analysis of Ecological data: Version 4. Gleneden Beach: MjM Software Design.
- MUNN, M.D., BLACK, R.W. and GRUBER, S.J., 2002. Response of benthic algae to environmental gradients in an agriculturally dominated landscape. Journal of the North American Benthological Society, vol. 21, no. 2, pp. 221-237. http://dx.doi.org/10.2307/1468411
» http://dx.doi.org/10.2307/1468411 - PAERL, H.W., DYBLE, J., MOISANDER, P.H., NOBLE, R.T., PIEHLER, M.F., PINCKNEY, J.L., STEPPE, T.F., TWOMEY, L. and VALDES, L.M., 2003. Microbial indicators of aquatic ecosystem change: current applications to eutrophication studies. Microbial Ecology, vol. 46, no. 3, pp. 233-246. http://dx.doi.org/10.1016/S0168-6496(03)00200-9 PMid:19719555.
» http://dx.doi.org/10.1016/S0168-6496(03)00200-9 - PAIVA, T.S. and SILVA-NETO, I.D., 2004. Ciliate protists from Cabiúnas lagoon (restinga de Jurubatiba, Macaé, Rio de Janeiro) with emphasis on water quality indicator species and description of . Oxytricha marcili sp. nBrazilian Journal of Biology = Revista Brasileira de Biologia, vol. 64, no. 3, pp. 465-478. http://dx.doi.org/10.1590/S1519-69842004000300010 PMid:15622844.
» http://dx.doi.org/10.1590/S1519-69842004000300010 - PAULETO, G.M., VELHO, L.F.M., BUOSI, P.R.B., BRÃO, A.F.S., LANSAC-TÔHA, F.A. and BONECKER, C.C., 2009. Spatial and temporal patterns of ciliate species composition (Protozoa: Ciliophora) in the plankton of the Upper Paraná River floodplain. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 69, no. 2, (Suppl.), pp. 517-527. http://dx.doi.org/10.1590/S1519-69842009000300007 PMid:19738959.
» http://dx.doi.org/10.1590/S1519-69842009000300007 - PEREIRA, P.S., FERNANDES, L.A.C., DIAS, R.J.P., SAMPAIO, M.S., SILVA-NETO, I.D., BAPTISTA, D.F. and OLIVEIRA, J.L.M., 2014. Ecological water quality assessment in the Guapiaçu-Macacu hydrographic complex (Rio de Janeiro, Brazil) using multiple indicators. Revista Ambiente & Água, vol. 9, no. 3, pp. 409-423.
- RAHEL, F.J., 2002. Homogenization of fish faunas across the United States. Science, vol. 288, no. 1, pp. 854-856. PMid:10797007.
- REISS, J. and SCHMID-ARAYA, J.M., 2008. Existing in plenty: abundance, biomass and diversity of ciliates and meiofauna in small streams. Freshwater Biology, vol. 53, no. 4, pp. 652-668. http://dx.doi.org/10.1111/j.1365-2427.2007.01907.x
» http://dx.doi.org/10.1111/j.1365-2427.2007.01907.x - REYNOLDS, C.S. and DESCY, J.P., 1996. The production, biomass and structure of phytoplankton in large rivers. Archiv für Hydrobiologie, vol. 113, no. 4, pp. 161-187.
- SHERR, E.B. and SHERR, B.F., 1993. Preservation and storage of samples for enumeration of heterotrophic protists. In: P. KEMP, B. SHERR, E. SHERR and J. COLE, eds. Current methods in aquatic microbial ecology. New York: Lewis Publishers, pp. 207-212.
- SHERR, B., SHERR, R. and RASSOULZADEGAN, F., 1988. Rates of Digeston of Bacteria by Marine Phagotrophic Protozoa: Temperatura Dependence. Applied and Environmental Microbiology, vol. 54, no. 5, pp. 1095-1988. PMid:16347623.
- WALSH, C.J., ROY, A.H., FEMINELLA, J.W., COTTINGHAM, P.D., GROFFMAN, P.M. and MORGAN II, R.P., 2005. The urban stream syndrome: Current knowledge and the search for a cure. Journal of the North American Benthological Society, vol. 24, no. 3, pp. 706-723. http://dx.doi.org/10.1899/0887-3593(2005)0240706:TUSSCK2.0.CO;2
» http://dx.doi.org/10.1899/0887-3593(2005)0240706:TUSSCK2.0.CO;2 - WEISSE, T., 1991. The annual cycle of heterotrophic freshwater nanoflagellates: role of bottom-up versus top-down control. Journal of Plankton Research, vol. 13, no. 1, pp. 167-185. http://dx.doi.org/10.1093/plankt/13.1.167
» http://dx.doi.org/10.1093/plankt/13.1.167 - ZINGEL, P., 2005. Vertical and seasonal dynamics of planktonic ciliates in a strongly stratified hypertrophic lake. Hydrobiologia, vol. 547, no. 1, pp. 163-174. http://dx.doi.org/10.1007/s10750-005-4157-7
» http://dx.doi.org/10.1007/s10750-005-4157-7
Publication Dates
-
Publication in this collection
17 May 2016 -
Date of issue
Oct-Dec 2016
History
-
Received
01 June 2015 -
Accepted
17 Sept 2015