Abstract
The usage of aquatic plants represents an alternative in the treatment of residues originating from swine. In these systems, one of the N removal methods is the ammonium (NH4 +) uptake and volatilization of ammonia (NH3). In this way, the objective of this work was to evaluate the volatilization rates of NH3 in waste treatment systems swine fluids (SSF) with aquatic macrophytes, as well as the concentration of NH 4+ present in the swine fluids. The experiment was carried out at Campus II/UNOESTE. The treatment systems were composed of 16 boxes of PVC and characterized as: T1 = Control sample 50% of SSF/50% of water; T2 = 50% SSF/50% water + Eichhornia crassipes (Mart.) Solms; T3 = 50% SSF/50% water + Pistia stratiotes L.; T4 = 50% SSF/50% water + Salvinia auriculata Aubl. The design was randomized blocks, with 4 treatments and 4 replicates. The hydrogen potential (pH) and the NH4 + content of the effluent were analyzed weekly, and the volatilization of NH 3 by means of collectors installed in each treatment unit. The presence of aquatic macrophytes promoted the reduction of NH4+ concentration and of the pH values of swine fluids, and this resulted in the reduction of NH3 volatilization rates to the environment, with emphasis on the system with Eichhornia crassipes (Mart.) Solms, which presented the lowest rate of volatilization.
Keywords:
greenhouse gases; phytoremediation; wastewater; environmental management
Resumo
A utilização de plantas aquáticas representa uma alternativa no tratamento de resíduos oriundos da suinocultura. Nestes sistemas, uma das formas de remoção de nitrogênio (N) é a absorção de amônio (NH4+) pelas plantas, entretanto, também ocorre a volatilização de amônia (NH3). Dessa forma, o objetivo do trabalho foi avaliar as taxas de volatilização de NH3 em sistemas de tratamentos de dejetos líquidos de suínos (DLS) com macrófitas aquáticas, bem como a concentração de NH4+ presente nos dejetos. O experimento foi realizado em área de ambiente aberto no Campus II/UNOESTE. Os sistemas de tratamento foram constituídos de 16 caixas de PVC e caracterizados como: T1 = Testemunha 50% de DLS/50% de água; T2 = 50% de DLS/50% de água + Eichhornia crassipes (Mart.) Solms; T3 = 50% de DLS/50% de água + Pistia stratiotes L.; T4 = 50% de DLS/50% de água + Salvinia auriculata Aubl. O delineamento adotado foi em blocos casualizados, com 4 tratamentos e 4 repetições. Foram analisados o potencial hidrogeniônico (pH) e o teor de NH4 + do efluente semanalmente, e a volatilização de NH3 por meio de coletores instalados em cada unidade de tratamento. A presença das macrófitas aquáticas proporcionou a redução da concentração de NH4+ e dos valores de pH dos dejetos líquidos de suínos, e isto resultou na redução das taxas de volatilização de NH 3 ao meio ambiente, com destaque ao sistema com Eichhornia crassipes (Mart.) Solms, que apresentou a menor taxa de volatilização.
Palavras-chave:
gases do efeito estufa; fitorremediação; efluentes; manejo ambiental
1. Introduction
Pig farming represents a sector of great economic and social importance for Brazil ( Gonzatto et al., 2013 GONZATTO, R., MIOLA, E.C.C., DONEDA, A., PUJOL, S.B., AITA, C. and GIACOMINI, S.J., 2013. Volatilização de amônia e emissão de óxido nitroso após aplicação de dejetos líquidos de suínos em solo cultivado com milho. Ciência Rural, vol. 43, no. 9, pp. 1590-1596. http://dx.doi.org/10.1590/S0103-84782013000900009.
http://dx.doi.org/10.1590/S0103-8478201...
), responsible for increasing exports of meat and its industrialized products, and also for the generation of jobs. Brazilian pig farming occupies a prominent position in the world scenario, where Brazil is the fourth largest producer and exporter of pork ( ABCS, 2014 ASSOCIAÇÃO BRASILEIRA DOS CRIADORES DE SUÍNOS – ABCS, 2014. Produção de suínos: teoria e prática . 1st ed. Brasília: ABCS. 905 p. ).
However, pig farming is characterized as an activity with great potential for pollution, due to the effluent generation normally in the liquid form, with high load of organic and nutrient matter (mainly nitrogen and phosphorus, and also, potassium, calcium, sodium, magnesium, manganese, iron, zinc and copper) ( Steinmetz et al., 2009 STEINMETZ, R.L.R., KUNZ, A., DRESSLER, V.L., FLORES, É.M.M. and MARTINS, A.F., 2009. Study of metal distribution in raw end screened swine manure. Clean Soil Air Water , vol. 37, no. 3, pp. 239-244. http://dx.doi.org/10.1002/clen.200800156.
http://dx.doi.org/10.1002/clen.20080015...
). The pollutant load of liquid pig slurry may adversely affect environments as the water bodies, promoting the growth of microorganisms and the occurrence of eutrophication ( Meade et al., 2011 MEADE, G., PIERCE, J.V., O’DOHERTY, C., MUELLER, G., LANIGAN, G. and MC CABE, T., 2011. Ammonia and nitrous oxide emissions following land application of high and low nitrogen pig manures to winter wheat at three growth stages. Agriculture, Ecosystems & Environment , vol. 140, no. 1-2, pp. 208-217. http://dx.doi.org/10.1016/j.agee.2010.12.007.
http://dx.doi.org/10.1016/j.agee.2010.1...
).
Excess nutrients, applied to the soil at rates higher than those for capture, may get in surface and groundwater due to the flow and leaching ( Stone et al., 1998 STONE, K.C., HUNT, P.G., HUMENIK, F.J. and JOHNSON, M.H., 1998. Impact of swine waste application on ground and stream water quality in an Eastern Coastal Plain watershed. American Society of Agricultural and Biological Engineer, vol. 41, no. 6, pp. 1665-1670. http://dx.doi.org/10.13031/2013.17342.
http://dx.doi.org/10.13031/2013.17342 ...
). To reduce the nutrient load into the environment, alternative or additional ways of treating wastewater should be implemented. An option for the additional treating is phytoremediation, that is, the use of plants and associated microorganisms as an instrument for containment, isolation, removal or reduction of contaminant concentrations in solid, liquid or gaseous media ( EPA, 2000 UNITED STATES ENVIRONMENTAL PROTECTION AGENCY – EPA, 2000 [viewed 03 November 2017]. Introduction to phytoremediation. Cincinnati, Ohio: EPA. EPA/600/R-99/107. Available from: http://nepis.epa.gov
http://nepis.epa.gov ...
), at safe levels compatible with the protection of human health, as well as preventing the spread of harmful substances to the environment ( Andrade et al., 2007 ANDRADE, J.C.M., TAVARES, S.R.L. and MAHLER, C.F., 2007. Fitorremediação: o uso de plantas na melhoria da qualidade ambiental. São Paulo: Oficina de Textos. 176 p. ).
The systems made with aquatic macrophytes are used to reduce the nutrient concentration of pig slurry, minimize the impacts on aquatic ecosystems ( Poach et al., 2003 POACH, M.E., HUNT, P.G., VANOTTI, M.B., STONE, K.C., MATHENY, T.A., JOHNSON, M.H. and SADLER, E.J., 2003. Improved nitrogen by constructed wetlands receiving partially nitrified swine manure. Ecological Engineering, vol. 20, no. 2, pp. 183-197. http://dx.doi.org/10.1016/S0925-8574(03)00024-7.
http://dx.doi.org/10.1016/S0925-8574(03...
), and provide an operationally passive form of wastewater treatment ( Hunt and Poach, 2001 HUNT, P.G. and POACH, M.E., 2001. State of the art for animal wastewater treatment in constructed wetlands. Water Science and Technology, vol. 44, no. 11-12, pp. 19-25. http://dx.doi.org/10.2166/wst.2001.0805. PMid:11804093.
http://dx.doi.org/10.2166/wst.2001.0805...
; Kadlec and Knight, 1996 KADLEC, R.H. and KNIGHT, R.L., 1996. Treatment wetlands. Boca Raton: Lewis Publishers. 893 p. ). Furthermore, they can effectively treat great amounts of animal excrements ( Knight et al., 2000 KNIGHT, R.L., PAYNE-JÚNIOR, V.W.E., BORER, R.E., CLARKE-JÚNIOR, R.A. and PRIES, J.H., 2000. Constructed wetlands for livestock wastewater management. Ecological Engineering, vol. 15, no. 1-2, pp. 41-55. http://dx.doi.org/10.1016/S0925-8574(99)00034-8.
http://dx.doi.org/10.1016/S0925-8574(99...
), especially in the nitrogen (N) removal ( Poach et al., 2003 POACH, M.E., HUNT, P.G., VANOTTI, M.B., STONE, K.C., MATHENY, T.A., JOHNSON, M.H. and SADLER, E.J., 2003. Improved nitrogen by constructed wetlands receiving partially nitrified swine manure. Ecological Engineering, vol. 20, no. 2, pp. 183-197. http://dx.doi.org/10.1016/S0925-8574(03)00024-7.
http://dx.doi.org/10.1016/S0925-8574(03...
).
According to Vesilind and Morgan (2011) VESILIND, P.A. and MORGAN, S.M., 2011. Introdução à engenharia ambiental. 2nd ed. São Paulo: Cengage Learning. 438 p. , the N is an important element in biological reactions, and may be connected to components that produce a lot of energy, such as amino acids and amines, also known as organic N. An intermediate element formed during the biological metabolism é the ammoniacal N. In accordance to Peng et al. (2005) PENG, J.F., WANG, B.Z. and WANG, L., 2005. Mult- stage ponds-wetlands ecosystem for effective wastewater treatment. Journal of Zhejiang University. Science, vol. 6B, no. 5, pp. 346-352. http://dx.doi.org/10.1631/jzus.2005.B0346.
http://dx.doi.org/10.1631/jzus.2005.B03...
, the organic and ammoniacal N are the main forms presented in wastewater, and are considered indicators of recent pollution. The organic N is converted to ammoniacal under anaerobic and aerobic conditions, with the reduction of ammoniacal nitrogen concentrations contributing to the reduction of total nitrogen, and the temperature and pH have an impact on the bioactivity and volatilization processes.
The wetlands, systems artificially designed for utilizing aquatic macrophytes, remove N through sedimentation, absorption, organic matter accumulation, microbial assimilation, nitrification/denitrification and volatilization of ammonia ( Brix, 1993 BRIX, H., 1993. Wastewater treatment in constructed wetlands: system design, removal processes, and treatment performance. In: G.A. MOSHIRI, ed. Constructed wetlands for water quality improvement. Boca Raton: CRC Press, pp. 9-22. ; Johnston, 1991 JOHNSTON, C.A., 1991. Sediment and nutrient retention by freshwater wetlands: effects on surface water quality. Critical Reviews in Environmental Control, vol. 21, no. 5-6, pp. 491-565. http://dx.doi.org/10.1080/10643389109388425.
http://dx.doi.org/10.1080/1064338910938...
; Poach et al., 2003 POACH, M.E., HUNT, P.G., VANOTTI, M.B., STONE, K.C., MATHENY, T.A., JOHNSON, M.H. and SADLER, E.J., 2003. Improved nitrogen by constructed wetlands receiving partially nitrified swine manure. Ecological Engineering, vol. 20, no. 2, pp. 183-197. http://dx.doi.org/10.1016/S0925-8574(03)00024-7.
http://dx.doi.org/10.1016/S0925-8574(03...
). Sooknah and Wilkie (2004) SOOKNAH, R.D. and WILKIE, A.C., 2004. Nutrient removal by floating aquatic macrophytes cultured in anaerobically disgested flushed dairy manure wastewater. Ecological Engineering , vol. 22, no. 1, pp. 27-42. http://dx.doi.org/10.1016/j.ecoleng.2004.01.004.
http://dx.doi.org/10.1016/j.ecoleng.200...
verify the reduction of N because of the direct absorption by the aquatic macrophytes to the nitrification carried out by the nitrifying bacteria and the volatilization of non-ionized ammonia (NH 3) which occurs predominantly in high pH environments. Ammonia emissions characterize a major threat to the environment, due to their implications, such as changes in the rainfall pH, contributions to the greenhouse effect, as well as effects on human and farmed animal health ( Felix and Cardoso, 2004 FELIX, E.P. and CARDOSO, A.A., 2004. Amônia (NH3) atmosférica: fontes, transformação, sorvedouros e métodos de análise. Quimica Nova, vol. 27, no. 1, pp. 123-130. http://dx.doi.org/10.1590/S0100-40422004000100022.
http://dx.doi.org/10.1590/S0100-4042200...
). Thus, monitoring these emissions is something fundamentally important for the control of atmospheric pollution.
Based on the previously said, this work aims at evaluating the losses of N to the atmosphere by volatilization of ammonia (NH3) in pig slurry treatment systems, with and without the presence of aquatic plants, as well as the concentration of ammonium (NH4 +) present in pig slurry, with the hypothesis that aquatic plants are able to reduce the concentration of NH4+ present in swine manures, as well as to reduce losses of N by NH3 volatilization.
2. Material and Methods
The experiment, carried out in May and June of 2016, was conducted in an open environment at Campus II/UNOESTE, whose geographic coordinates are: Latitude 22° 07’ S e Longitude 51° 09’ W, Presidente Prudente, São Paulo, Brazil. Located in a defined climate region, according to the climatic classification of Köppen, as Aw – tropical wet with dry winter, and average annual temperature of 29.2 °C. The average annual rainfall is 1254.9 mm ( CEPAGRI, 2017 CENTRO DE PESQUISAS METEOROLÓGICAS E CLIMÁTICAS APLICADAS A AGRICULTURA – CEPAGRI, 2017 [viewed 24 April 2017]. Clima dos municípios paulistas [online]. Campinas: Unicamp. Available from: http://www.cpa.unicamp.br/outras-informacoes/clima_muni_467.html
http://www.cpa.unicamp.br/outras-inform...
).
The treatment systems were composed of 16 polyvinyl chloride (PVC) containers with dimensions of 76.5 cm in height, 101.5 cm in superior diameter and 73.0 cm in inferior diameter, making a volume of 0.32 m3, in which only 0.25 m3 are used per container, margin adopted to avoid overflow.
Pig slurry was used, coming from the zootechnical center of Campus II/UNOESTE, for the composition of the treatments, which are: T1 = Control sample – 50% pig slurry and 50% water; T2 = 50% pig slurry and 50% water + Eichhornia crassipes (Mart.) Solms ( Figure 1 ); T3 = 50% pig slurry and 50% water + Pistia stratiotes L. ( Figure 2 ); T4 = 50% pig slurry and 50% water + Salvinia auriculata Aubl. ( Figure 3 ), distributed in a randomized block design (RBD), in split plots, with four repetitions. The plots are represented by the treatments (with and without the presence of aquatic macrophytes) and the split plots, by the collection periods.
Eichhornia crassipes (Mart.) Solms ( Lutzenberger, 1985 LUTZENBERGER, J., 1985. Ecologia: do jardim do poder. Porto Alegre: L&PM Editores, 102 p. ).
Pistia stratiotes L. ( Pott and Pott, 2000 POTT, V.J. and POTT, A., 2000. Plantas aquáticas do Pantanal. Brasília: Embrapa; Corumbá: Centro de Pesquisa Agropecuária do Pantanal. 404 p. ).
Salvinia auriculata Aubl. ( Gomes, 2011 GOMES, M.A.C., 2011. Efeito da salinidade sobre a biomassa, morfologia e fisiologia de Salvinia auriculata Aubl. Campos dos Goytacazes: Universidade Estadual do Norte Fluminense Darcy Ribeiro, 90 p. Dissertação de Mestrado em Biociências e Biotecnologia. ).
The Eichhornia crassipes (Mart.) Solms is characterized by being a free floating aquatic plant, native of South America, belonging to the monocotyledonous class, Pontederidaceae family, Pontederiales order ( Esteves, 1998 ESTEVES, F.A., 1998. Fundamentos de limnologia. 2nd ed. Rio de Janeiro: Interciência. 602 p. ), and by being an angiosperm with perennial life cycle ( Bortolotto and Guarim Neto, 2005 BORTOLOTTO, I.M. and GUARIM NETO, G., 2005. O uso do camalote, Eichhornia crassipes (Mart.) Solms, Pontederiaceae, para confecção de artesanato no Distrito de Albuquerque, Corumbá, MS, Brasil. Acta Botanica Brasílica , vol. 19, no. 2, pp. 331-337. http://dx.doi.org/10.1590/S0102-33062005000200016.
http://dx.doi.org/10.1590/S0102-3306200...
). Exotic in origin, it is distributed in all continents, in tropical semitropical latitudes and in some temperate countries ( Metcalf and Tchobanoglous, 1991 METCALF, E. and TCHOBANOGLOUS, G., 1991. Wastewater engineering: treatment, disposal and reuse. 3rd ed. New York: MacGraw Hill. ) and, in the absence of nutritional limitations, it rapidly develops in hot climate regions ( Andrade et al., 2007 ANDRADE, J.C.M., TAVARES, S.R.L. and MAHLER, C.F., 2007. Fitorremediação: o uso de plantas na melhoria da qualidade ambiental. São Paulo: Oficina de Textos. 176 p. ).
The Pistia stratiotes L. is commonly known as water lettuce, belonging to the Araceae family ( Kissmann and Groth, 1997 KISSMANN, K.G. and GROTH, D., 1997. Plantas infestantes e nocivas. 2nd ed. São Paulo: BASF Brasileira. ), extensively distributed throughout the world. Its origin, which is attributed to Africa or South America ( Lorenzi, 1982 LORENZI, H. 1982. Plantas daninhas do Brasil: terrestres, aquáticas, parasitas, tóxicas e medicinais. São Paulo: Nova Odessa. 425 p. ; Cardoso, et al., 2005 CARDOSO, L.R., MARTINS, D., MORI, E.S. and TERRA, M.A., 2005. Variabilidade genética entre populações de Pistia Stratiotes. Planta Daninha, vol. 23, no. 2, pp. 181-185. http://dx.doi.org/10.1590/S0100-83582005000200003.
http://dx.doi.org/10.1590/S0100-8358200...
), has not yet been fully defined. According to Pott and Pott (2000) POTT, V.J. and POTT, A., 2000. Plantas aquáticas do Pantanal. Brasília: Embrapa; Corumbá: Centro de Pesquisa Agropecuária do Pantanal. 404 p. , P. stratiotes is considered a cosmopolitan tropical and subtropical species, being widely distributed throughout Brazil, occurring both in natural ecosystems and in aquatic environments impacted by anthropic activities ( Henry-Silva and Camargo, 2000a HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2000a. Composição química de quatro espécies de macrófitas aquáticas e possibilidade de uso de suas biomassas. Naturalia, vol. 26, no. 8, pp. 111-125. ).
Belonging to the family Salviniaceae, the Salvinia auriculata Aubl. is a free floating aquatic plant, of annual or perennial occurrence ( Oliveira, 1981 OLIVEIRA, P., 1981. Plantas de aquário. Lisboa: Editorial Presença. 238 p. ). It presents a wide native distribution in the neotropics, extending from Mexico and the Galapagos Islands through Central America and the Antilles and most of South America to the south of Brazil ( Sculthorpe, 1967 SCULTHORPE, C.D., 1967. The biology of aquatic vascular plants. London: Edward Arnold Ltd. 610 p. ).
The aquatic macrophytes used were collected in lentic lakes from lands located in the West Paulista region, São Paulo, selected as young plants, with an established root system and aerial part of similar appearance. They were initially submitted to the environmental adaptation in boxes containing only water, at the experiment site, for a week. After this period, the treatment was initialized, and the amount of plants inserted in each experimental unit was determined to maintain an occupation of approximately 80% of the experimental units, as described by Henry-Silva and Camargo (2008) HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2008. Tratamento de efluentes de carcinicultura por macrófitas aquáticas flutuantes. Revista Brasileira de Zootecnia , vol. 37, no. 2, pp. 181-188. http://dx.doi.org/10.1590/S1516-35982008000200002.
http://dx.doi.org/10.1590/S1516-3598200...
.
During the experimental period, 16 samples of the effluent to analyse the pH and ammoniacal nitrogen in the ionized form (NH4+) were weekly collected, at the periods of 0 (day in which the experiment was implanted), 7, 14, 21 and 30 days. The pH measurements were obtained using the Micronal bench digital pH meter. The determination of the NH4+ content was obtained by the Kjeldahl method, consisted of three stages: sample digestion, distillation with the Kjeldahl nitrogen distiller and titration with sulfuric acid, through the method presented by Malavolta et al. (1997) MALAVOLTA, E., VITTI, G.C. and OLIVEIRA, S.A., 1997. Avaliação do estado nutricional das plantas: princípios e aplicações . 2nd ed. Piracicaba: Potafós. 319 p. . The method is based on the decomposition of organic matter by digestion of the sample with concentrated sulfuric acid at 350 °C, in the presence of catalyst salts that accelerate the oxidation of organic matter. The digestion will be terminated after obtaining a colorless or slightly greenish liquid. The nitrogen present in the resulting acid solution is determined by steam distillation, collected by the boric acid 2% (m/v) solution and indicators, followed by titration with sulfuric acid (0.02 N).
For the quantification of NH3 volatilization a PVC collector base ( Figure 4 ), with 15.0 cm in diameter and 14.0 cm in height, was placed in each treatment box, fixed to a 4.0 cm thick Styrofoam board, with a center opening beneath the surface of the effluent. Each base was protected at the top with a polyethylene plate with 20.0 cm in diameter, and a 2.0 cm opening between the plate and the collector base was kept, for establishing a steam pressure deficit and, thus, the NH3 volatilization could happen. Inside the PVC base a screen with an effluent height of 4.0 cm was place, fixed to a glass petri dish which housed in its interior a sponge with 2.0 cm thick and 7.0 cm width.
NH3 collector scheme (adapted from Ros et al., 2005 ROS, C.O., AITA, C. and GIACOMINI, S.J., 2005. Volatilização de amônia com aplicação de uréia na superfície do solo, no sistema plantio direto. Ciência Rural, vol. 35, no. 4, pp. 799-805. http://dx.doi.org/10.1590/S0103-84782005000400008.
http://dx.doi.org/10.1590/S0103-8478200... ).
The sponges, moistened with 30.0 mL of phosphoric acid (0.167 mol L-1) to capture the volatilized NH3 of the effluent, were replaced at the periods of 1, 4, 8, 12, 19 and 27 days after placing the collectors. In the collections, the sponges were gathered, stored in a plastic box with a lid and immediately taken to the laboratory of clinical analysis of vegetal tissues/UNOESTE to extract the ammonium phosphate solution ((NH4)3 PO4) formed from the chemical reaction between NH3 and phosphoric acid (H3PO4). Their washings were performed with 500.0 mL deionized water, in five continuous washings of 100.0 mL each. Finally, an aliquot of 20.0 mL of the solution was submitted to distillation with the Kjheldad distiller by the method described by Cantarella and Trivelin (2001) CANTARELLA, H. and TRIVELIN, P.C.O., 2001. Determinação de nitrogênio total em solos. In: B. RAIJ, J.C. ANDRADE, H. CANTARELLA and J.A. QUAGGIO, eds. Análise química para avaliação da fertilidade de solos tropicais . Campinas: Instituto Agronômico de Campinas, pp. 262-269. .
The efficiencies of the treatments in the reduction of the content of NH4+ were calculated according to Equation 1:
In which: E = Removal efficiency (%); Ci = Initial concentration (mg L-1); Cf = Final concentration (mg L-1).
The results were put under analysis of variance by the Test F and the comparison of the means by the Tukey Test, at the level of 5% probability, with software Assistat 7.7.
3. Results and Discussion
The values of Test F, obtained by the contrast of means between the treatments ( Table 1 ), show that there was a significant difference between the plant species used and the collection periods for the following variables: pH ammonium (NH4+) and ammonia (NH3).
Values of F for pH, NH4+ and the volatilization of NH3 calculated based on the Tukey Test, at the level of 5% probability, in the treatments with aquatic species, in collection periods.
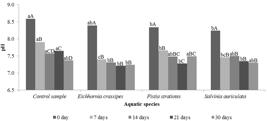
The pH values ( Figure 5 ) decreased, throughout the collection periods, in all treatments. It was possible to notice that in the period of 7 days, the Eichhornia crassipes (Mart.) Solms provided a significant reduction in the pH compared to the others, and according to Granato (1995) GRANATO, M., 1995. Utilização do aguapé no tratamento de efluentes com cianetos. Rio de Janeiro: CETEM/CNPq. 39 p. Série Tecnologia Ambiental, vol. 5. , Eichhornia crassipes (Mart.) Solms, in contact with alkaline solutions, has the ability of decreasing the pH of these solutions, due to the absorption of the potassium, calcium and magnesium nutrients, respectively, which are all present in the medium. According to Esteves (1998) ESTEVES, F.A., 1998. Fundamentos de limnologia. 2nd ed. Rio de Janeiro: Interciência. 602 p. , the organic matter decomposition process reduces the pH, since there is an increase in the carbon dioxide (CO2) concentration in the medium and, at the same time, the consumption of dissolved oxygen. Lin et al. (2005) LIN, Y.F., JING, S.R., LEE, D.Y., CHANG, Y.F., CHEN, Y.M. and SHIH, K.C., 2005. Performance of a constructed wetland treating intensive shrimp aquaculture wastewater under high hydraulic loading rate. Environmental Pollution, vol. 134, no. 3, pp. 411-421. PMid:15620586. also observed a reduction in the pH of the effluent from de Litopenaeus vannamei nurseries treated with constructed wetland. According to Shah et al. (2015) SHAH, M., HASHMI, H.N., GHUMMAN, A.R. and ZEESHAN, M., 2015. Performance assessment of aquatic macrophytes for treatment of municipal wastewater. Journal of the South African Institution of Civil Engineering, vol. 57, no. 3, pp. 18-25. http://dx.doi.org/10.17159/2309-8775/2015/V57N3A3.
http://dx.doi.org/10.17159/2309-8775/20...
, a pH of 6.0-9.0 is the most adequate for the aquatic macrophytes performance and, in accordance with CONAMA (2011) CONSELHO NACIONAL DO MEIO AMBIENTE – CONAMA, 2011. Resolução Conama nº 430 de 13 de maio de 2011. Dispõe sobre as condições e padrões de lançamento de efluentes, complementa e altera a Resolução n. 357, de 17 de março de 2005, do Conselho Nacional do Meio Ambiente - CONAMA
. Diário Oficial da República Federativa do Brasil, Brasília, DF, n. 92, 16 maio, p. 89. Resolution no. 430/2011, the pH values of the effluent from any polluting source, to be directly released in the receiving body, must be between 5.0 and 9.0. Thus, the results found in the treatments with plant species are in agreement with the ones required by the Brazilian legislation.
pH values obtained in the treatments with aquatic species, in five collection periods. Reference value according to CONAMA Resolution no 430/2011 = pH: 5-9. DMS for collection periods = 0.21; CV (%) = 1.47 classification on lowercase letters. DMS for aquatic species = 0.22; CV (%) = 1.49 classification on capital letters. Tukey Test was applied at the level of 5% probability.
For Henry-Silva and Camargo (2000b) HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2000b. Impacto do lançamento de efluentes urbanos sobre alguns ecossistemas aquáticos do município de Rio Claro (SP). Revista Ciências Biológicas e do Ambiente, vol. 2, pp. 317-330. , the highest pH found only in the effluent is probably related to the photosynthesis of the phytoplankton present in the site, which, by assimilating the CO 2 available in the water, increases the pH values of the medium.
Silva et al. (2014) SILVA, A.D.R., SANTOS, R.B., BRUNO, A.M.S.S., GENTELINI, A.L., SILVA, A.H.G. and SOARES, E.C., 2014. Eficiência do aguapé sobre variáveis limnológicas em canais de abastecimento utilizados no cultivo de tambaqui. Acta Amazonica , vol. 44, no. 2, pp. 255-262. http://dx.doi.org/10.1590/S0044-59672014000200011.
http://dx.doi.org/10.1590/S0044-5967201...
found out that the effluent from water supply channels with tambaqui farming treated with Eichhornia crassipes (Mart.) Solms showed lower pH values when compared to the treatments without plants. According to the authors, it happens because of the removal of bases that enable the growth of Eichhornia crassipes (Mart.) Solms. Gentelini et al. (2008) GENTELINI, A.L., GOMES, S.D., FEIDEN, A., ZENATTI, D., SAMPAIO, S.C. and COLDEBELLA, A., 2008. Produção de biomassa das macrófitas aquáticas Eichhornia crassipes (aguapé) e Egeria densa (egeria) em sistema de tratamento de efluente de piscicultura orgânica. Ciências Agrárias, vol. 29, no. 2, pp. 441-448. http://dx.doi.org/10.5433/1679-0359.2008v29n2p441.
http://dx.doi.org/10.5433/1679-0359.200...
observed that the organic pisciculture effluent, before passing through the system with aquatic macrophytes, was slightly alkaline, with a pH of 7.03, becoming somewhat acid after crossing the system. This same pattern of pH reduction, after passing through the treatment system, was observed by Henry-Silva and Camargo (2006) HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2006. Efficiency of aquatic macrophytes to treat Nile tilapia pond effluents. Scientia Agrícola, vol. 63, no. 5, pp. 433-438. http://dx.doi.org/10.1590/S0103-90162006000500003.
http://dx.doi.org/10.1590/S0103-9016200...
with three floating macrophytes, Eichhornia crassipes (Mart.) Solms, Pistia stratiotes L. and Salvinia molesta D. S. Mitch., in the treatment of pisciculture effluent.
In relation to the NH4+ concentration in the effluent, it should be noted that there was no significant interaction between the treatments (without and with the plants) and between the collection periods. However, after evaluating the means obtained in each treatment ( Figure 6 ), it was possible to notice that the Eichhornia crassipes (Mart.) Solms species statically differed from the others by presenting a lower NH4+ content in its effluent and, therefore, higher efficiency in the removal of this element (93.4%, according Table 2 ) in a 30 day detention time. Reidel et al. (2005) REIDEL, A., DAMASCENO, S., ZENATTI, D.C., SAMPAIO, S.C., FEIDEN, A. and QUEIROZ, M.M.F., 2005. Utilização de efluente de frigorífico, tratado com macrófita aquática, no cultivo de tilápia do Nilo. Revista Brasileira de Engenharia Agrícola e Ambiental, vol. 9, suppl, pp. 181-185. , when analysing the refrigerated effluent treatment system with Eichhornia crassipes (Mart.) Solms, found that the industrial effluent presented average concentration of 79.85 mg L-1 of NH4+, and the reductions of this element were 58.9% for a five day detention time, 86.1% for seven days and 97.7% for ten days, thus increasing the removal efficiency with an increased hydraulic detention time.
Available NH4+ concentration, in mg L-1, in the treatments with aquatic species. Reference value according to CONAMA Resolution no 430/2011 = NH4+: 20.0 mg L-1. DMS = 0.96; CV (%) = 10.47. The means followed by the same letter are not statistically different among themselves. Tukey Test was applied at the level of 5% probability.
Sezerino and Philippi (2000) SEZERINO, P.H. and PHILIPPI, L.S., 2000. Utilização de um sistema experimental por meio de “wetland” construído no tratamento de esgotos domésticos pós tanque séptico. In: Anais do Simpósio Luso-Brasileiro de Engenharia Sanitária e Ambiental; 2000; Porto Seguro. Rio de Janeiro: ABES, pp. 688-697. highlight that, in such treatment systems with plants, approximately 74.0% from the NH 4+ removal may be associated with the plant uptake. CONAMA Resolution no. 430/2011 puts a limit in the concentration of the NH4+ ion to values of 20 mg L-1 in effluents from any polluting source. Therefore, the values obtained in the treatments submitted to the aquatic plants ( Figure 6 ) are in accordance with the Brazilian legislation.
The NH3 volatilization (desorption to the atmosphere) is a physical process of disengaging this gas from the dissociation of the NH4+ ion in aquatic environments with high pH values ( Assunção, 2009 ASSUNÇÃO, F.A.L., 2009. Estudo da remoção de nitrogênio, com ênfase na volatilização de amônia, em lagoas de polimento de efluentes de reatores UASB tratando esgotos urbanos de Belo Horizonte/MG . Belo Horizonte: Universidade Federal de Minas Gerais, 89 p. Dissertação de Mestrado em Saneamento, Meio Ambiente e Recursos Hídricos. ). The NH3 volatilization was observed in the interaction between treatments with aquatic macrophytes and the evaluation periods ( Figure 7 ).
Available NH3 concentration, in mg L-1, in the treatments with aquatic species, in six collection periods. DMS for collection periods = 81.62; CV (%) = 30.33 classification on lowercase letters. DMS for aquatic species = 91.04; CV(%) = 31.25 classification on capital letters. Tukey Test was applied at the level of 5% probability.
According to Figure 7 , this volatilization was higher in the control sample treatment, stressing that the differentiation occurred, mainly, after the period of 12 days of collection. It is also stated that the systems containing aquatic species provided lower losses of NH3, with a noticeable decrease during the evaluation period. According to Körner and Vermaat (1998) KÖRNER, S. and VERMAAT, J.E., 1998. The relative importance of Lemna gibba , bacteria and algae for the nitrogen and phosphorus removal in duckweed – covered domestic wastewater. Water Research, vol. 32, no. 12, pp. 3651-366. http://dx.doi.org/10.1016/S0043-1354(98)00166-3.
http://dx.doi.org/10.1016/S0043-1354(98...
and Sooknah and Wilkie (2004) SOOKNAH, R.D. and WILKIE, A.C., 2004. Nutrient removal by floating aquatic macrophytes cultured in anaerobically disgested flushed dairy manure wastewater. Ecological Engineering , vol. 22, no. 1, pp. 27-42. http://dx.doi.org/10.1016/j.ecoleng.2004.01.004.
http://dx.doi.org/10.1016/j.ecoleng.200...
in treatment systems with aquatic macrophytes, the removal of N happens by the direct absorption of the plant, by the action of microorganisms fixed in the roots and by the NH3 volatilization itself.
It is noteworthy that at the initial collection period (1 to 4 days), the NH3 concentration was higher in the four treatments proposed. According to Assunção (2009) ASSUNÇÃO, F.A.L., 2009. Estudo da remoção de nitrogênio, com ênfase na volatilização de amônia, em lagoas de polimento de efluentes de reatores UASB tratando esgotos urbanos de Belo Horizonte/MG . Belo Horizonte: Universidade Federal de Minas Gerais, 89 p. Dissertação de Mestrado em Saneamento, Meio Ambiente e Recursos Hídricos. , when the pH is high, he balance between the free NH3 and NH4+ ion tends to shift toward the NH3 formation. For values above 9.26 there’s a predominance of NH3 (practically 100% at pH near 11.0) and for values close to neutrality (between 6.0 and 7.0), practically all NH3 is in the ionized form. For Kiehl (1985) KIEHL, E.J., 1985. Fertilizantes orgânicos. Piracicaba: Agronômica Ceres. 492 p. , the NH3 losses increase when the compound reaction is in the alkalinity zone and when the pH reaches values above 8.0 and 9.0, a large part of N turned into NH3, and may be lost in the atmosphere. The balance represented by the reaction NH4
+ ↔ NH3 + H+, at pH values around neutrality, is shifted to the left, and there is a predominance of NH3 only to pH values above 8.5 ( SCHMIDELL et al., 2007 SCHMIDELL, W., SOARES, H.M., ETCHEBEHERE, C., MENES, R.J., BERTOLA, N.C. and CONTRERAS, E.M., 2007. Tratamento biológico de águas residuárias . Florianópolis: Tribo da Ilha. ). Probably, due to the fact that the pH of the effluent was higher (above 8.0) in the first days (0 to 7 days) ( Figure 5 ), the release of NH3 was higher. Mkhabela et al. (2009) MKHABELA, M.S., GORDON, R., BURTON, D., SMITH, E. and MADANI, A., 2009. The impact of management practices and meteorological conditions on ammonia and nitrous oxide emissions following application of hog slurry to forage grass in Nova Scotia. Agriculture, Ecosystems & Environment, vol. 130, no. 1-2, pp. 41-49. http://dx.doi.org/10.1016/j.agee.2008.11.012.
http://dx.doi.org/10.1016/j.agee.2008.1...
stated that the initial high pH of the slurry is a factor that contributes to such emissions, and Meade et al. (2011) MEADE, G., PIERCE, J.V., O’DOHERTY, C., MUELLER, G., LANIGAN, G. and MC CABE, T., 2011. Ammonia and nitrous oxide emissions following land application of high and low nitrogen pig manures to winter wheat at three growth stages. Agriculture, Ecosystems & Environment , vol. 140, no. 1-2, pp. 208-217. http://dx.doi.org/10.1016/j.agee.2010.12.007.
http://dx.doi.org/10.1016/j.agee.2010.1...
observed that 95% of the NH3 emissions happened in the first 24 hours after applying the slurry.
Gonzatto et al. (2013) GONZATTO, R., MIOLA, E.C.C., DONEDA, A., PUJOL, S.B., AITA, C. and GIACOMINI, S.J., 2013. Volatilização de amônia e emissão de óxido nitroso após aplicação de dejetos líquidos de suínos em solo cultivado com milho. Ciência Rural, vol. 43, no. 9, pp. 1590-1596. http://dx.doi.org/10.1590/S0103-84782013000900009.
http://dx.doi.org/10.1590/S0103-8478201...
, when evaluating, under field conditions, the NH3 volatilization and N2 O emission after applying the pig slurry in maize, observed that approximately 80.0% of the N losses by volatilization happened in the first 22 hours after applying the slurry, quickly reducing with time. According to the authors, this kinetic observed in the NH3 emissions may be attributed to the high content of NH4+ of the pig slurry, which is one of the factors that influence the NH3 volatilization ( Sommer and Hutchings, 2001 SOMMER, S.G. and HUTCHINGS, N.J., 2001. Ammonia emission from field applied manure and its reduction: invited paper. European Journal of Agronomy, vol. 15, no. 1, pp. 1-15. http://dx.doi.org/10.1016/S1161-0301(01)00112-5.
http://dx.doi.org/10.1016/S1161-0301(01...
).
Harper et al. (2004) HARPER, L.A., SHARPE, R.R., PARKIN, T.B., VISSCHER, A., VAN CLEEMPUT, O. and BYERS, F.M., 2004. Nitrogen cycling through swine production systems: Ammonia, dinitrogen, and nitrous oxide emissions. Journal of Environmental Quality, vol. 33, no. 4, pp. 1189-1201. http://dx.doi.org/10.2134/jeq2004.1189. PMid:15254100.
http://dx.doi.org/10.2134/jeq2004.1189 ...
state that the main factors that can be correlated to the NH3 volatilization in pig ponds are the wind speed, temperature, NH4+ concentration and the effluent pH. Thus, it is observed that the NH4+ concentrations found in the pig slurry ( Figure 6 ) were higher in the beginning of the experiment; besides, in the aquatic macrophytes treatment, the NH3 volatilization may have been reduced from the 12 days of collection, compared to the control sample treatment ( Figure 7 ), due to the high absorption of NH4+ by the plants, as can be observed in Figure 6 . In the treatment with Eichhornia crassipes (Mart.) Solms, the NH 4+ concentrations in the effluent were lower and statistically differ from the others.
Zimmo et al. (2003) ZIMMO, O., VAN DER STEEN, N. and GIJZEN, H., 2003. Comparison of ammonia volatilization rates in algae and duckweed-based waste stabilization ponds treating domestic wastewater. Water Research, vol. 37, no. 19, pp. 4587-4594. http://dx.doi.org/10.1016/j.watres.2003.08.013. PMid:14568043.
http://dx.doi.org/10.1016/j.watres.2003...
verified that the NH3 volatilization rates in ponds with algae were higher than in ponds with aquatic plants (Lemna gibba). According to the authors, it can be explained by the lower NH3 values in ponds with plants due to the shadowing and to the lower pH values. In this case, the volatilization rate correlated to the free NH 3 concentration in the water of the pond.
When considering the percentages of reduction of the NH3 volatilization rates, it is possible to notice that the treatment systems with the species Eichhornia crassipes (Mart.) Solms and Salvinia auriculata Aubl. were more expressive, 77.8% and 76.2% in a 27 day retention time. This reduction in the NH3 volatilization shows the importance of the presence of aquatic plants in these treatment systems. Oron et al. (1988) ORON, G., DE-VEGT, A. and PORATH, D., 1988. Nitrogen removal and conversion by duckweed grown on wastewater. Water Research, vol. 22, no. 2, pp. 179-184. http://dx.doi.org/10.1016/0043-1354(88)90076-0.
http://dx.doi.org/10.1016/0043-1354(88)...
, when evaluating the treatment of domestic sewage under cultivation of floating aquatic plants of the family Lemnaceae, obtained NH3 removal efficiency of 90.0%, with a 10 day retention time and initial concentration of 520 mg L-1 of COD. Researches carried out in wetlands that treated pig slurry waters mention that the NH3 volatilization represented less than 20.0% of the N removed by the wetlands ( Poach et al., 2002 POACH, M.E., HUNT, P.G., SADLER, E.J., MATHENY, T.A., JOHNSON, M.H., STONE, K.C., HUMINEK, F.J. and RICE, J.M., 2002. Ammonia volatilization from constructed wetlands that treat swine wastewater. Transactions of the ASAE. American Society of Agricultural Engineers , vol. 45, no. 3, pp. 619-627. http://dx.doi.org/10.13031/2013.8825.
http://dx.doi.org/10.13031/2013.8825 ...
).
4. Conclusions
The presence of aquatic macrophytes promoted the reduction of NH4+ concentration and of the pH values of swine fluids, and this resulted in the reduction of NH 3 volatilization rates to the environment, with emphasis on the system with Eichhornia crassipes (Mart.) Solms, which presented the lowest rate of volatilization.
-
(With 7 figures)
References
- ANDRADE, J.C.M., TAVARES, S.R.L. and MAHLER, C.F., 2007. Fitorremediação: o uso de plantas na melhoria da qualidade ambiental São Paulo: Oficina de Textos. 176 p.
- ASSOCIAÇÃO BRASILEIRA DOS CRIADORES DE SUÍNOS – ABCS, 2014. Produção de suínos: teoria e prática . 1st ed. Brasília: ABCS. 905 p.
- ASSUNÇÃO, F.A.L., 2009. Estudo da remoção de nitrogênio, com ênfase na volatilização de amônia, em lagoas de polimento de efluentes de reatores UASB tratando esgotos urbanos de Belo Horizonte/MG . Belo Horizonte: Universidade Federal de Minas Gerais, 89 p. Dissertação de Mestrado em Saneamento, Meio Ambiente e Recursos Hídricos.
- BORTOLOTTO, I.M. and GUARIM NETO, G., 2005. O uso do camalote, Eichhornia crassipes (Mart.) Solms, Pontederiaceae, para confecção de artesanato no Distrito de Albuquerque, Corumbá, MS, Brasil. Acta Botanica Brasílica , vol. 19, no. 2, pp. 331-337. http://dx.doi.org/10.1590/S0102-33062005000200016.
» http://dx.doi.org/10.1590/S0102-33062005000200016 - BRIX, H., 1993. Wastewater treatment in constructed wetlands: system design, removal processes, and treatment performance. In: G.A. MOSHIRI, ed. Constructed wetlands for water quality improvement Boca Raton: CRC Press, pp. 9-22.
- CANTARELLA, H. and TRIVELIN, P.C.O., 2001. Determinação de nitrogênio total em solos. In: B. RAIJ, J.C. ANDRADE, H. CANTARELLA and J.A. QUAGGIO, eds. Análise química para avaliação da fertilidade de solos tropicais . Campinas: Instituto Agronômico de Campinas, pp. 262-269.
- CARDOSO, L.R., MARTINS, D., MORI, E.S. and TERRA, M.A., 2005. Variabilidade genética entre populações de Pistia Stratiotes. Planta Daninha, vol. 23, no. 2, pp. 181-185. http://dx.doi.org/10.1590/S0100-83582005000200003.
» http://dx.doi.org/10.1590/S0100-83582005000200003 - CENTRO DE PESQUISAS METEOROLÓGICAS E CLIMÁTICAS APLICADAS A AGRICULTURA – CEPAGRI, 2017 [viewed 24 April 2017]. Clima dos municípios paulistas [online]. Campinas: Unicamp. Available from: http://www.cpa.unicamp.br/outras-informacoes/clima_muni_467.html
» http://www.cpa.unicamp.br/outras-informacoes/clima_muni_467.html - CONSELHO NACIONAL DO MEIO AMBIENTE – CONAMA, 2011. Resolução Conama nº 430 de 13 de maio de 2011. Dispõe sobre as condições e padrões de lançamento de efluentes, complementa e altera a Resolução n. 357, de 17 de março de 2005, do Conselho Nacional do Meio Ambiente - CONAMA . Diário Oficial da República Federativa do Brasil, Brasília, DF, n. 92, 16 maio, p. 89.
- ESTEVES, F.A., 1998. Fundamentos de limnologia 2nd ed. Rio de Janeiro: Interciência. 602 p.
- FELIX, E.P. and CARDOSO, A.A., 2004. Amônia (NH3) atmosférica: fontes, transformação, sorvedouros e métodos de análise. Quimica Nova, vol. 27, no. 1, pp. 123-130. http://dx.doi.org/10.1590/S0100-40422004000100022.
» http://dx.doi.org/10.1590/S0100-40422004000100022 - GENTELINI, A.L., GOMES, S.D., FEIDEN, A., ZENATTI, D., SAMPAIO, S.C. and COLDEBELLA, A., 2008. Produção de biomassa das macrófitas aquáticas Eichhornia crassipes (aguapé) e Egeria densa (egeria) em sistema de tratamento de efluente de piscicultura orgânica. Ciências Agrárias, vol. 29, no. 2, pp. 441-448. http://dx.doi.org/10.5433/1679-0359.2008v29n2p441.
» http://dx.doi.org/10.5433/1679-0359.2008v29n2p441 - GOMES, M.A.C., 2011. Efeito da salinidade sobre a biomassa, morfologia e fisiologia de Salvinia auriculata Aubl Campos dos Goytacazes: Universidade Estadual do Norte Fluminense Darcy Ribeiro, 90 p. Dissertação de Mestrado em Biociências e Biotecnologia.
- GONZATTO, R., MIOLA, E.C.C., DONEDA, A., PUJOL, S.B., AITA, C. and GIACOMINI, S.J., 2013. Volatilização de amônia e emissão de óxido nitroso após aplicação de dejetos líquidos de suínos em solo cultivado com milho. Ciência Rural, vol. 43, no. 9, pp. 1590-1596. http://dx.doi.org/10.1590/S0103-84782013000900009.
» http://dx.doi.org/10.1590/S0103-84782013000900009 - GRANATO, M., 1995. Utilização do aguapé no tratamento de efluentes com cianetos Rio de Janeiro: CETEM/CNPq. 39 p. Série Tecnologia Ambiental, vol. 5.
- HARPER, L.A., SHARPE, R.R., PARKIN, T.B., VISSCHER, A., VAN CLEEMPUT, O. and BYERS, F.M., 2004. Nitrogen cycling through swine production systems: Ammonia, dinitrogen, and nitrous oxide emissions. Journal of Environmental Quality, vol. 33, no. 4, pp. 1189-1201. http://dx.doi.org/10.2134/jeq2004.1189. PMid:15254100.
» http://dx.doi.org/10.2134/jeq2004.1189 - HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2000a. Composição química de quatro espécies de macrófitas aquáticas e possibilidade de uso de suas biomassas. Naturalia, vol. 26, no. 8, pp. 111-125.
- HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2000b. Impacto do lançamento de efluentes urbanos sobre alguns ecossistemas aquáticos do município de Rio Claro (SP). Revista Ciências Biológicas e do Ambiente, vol. 2, pp. 317-330.
- HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2006. Efficiency of aquatic macrophytes to treat Nile tilapia pond effluents. Scientia Agrícola, vol. 63, no. 5, pp. 433-438. http://dx.doi.org/10.1590/S0103-90162006000500003.
» http://dx.doi.org/10.1590/S0103-90162006000500003 - HENRY-SILVA, G.G. and CAMARGO, A.F.M., 2008. Tratamento de efluentes de carcinicultura por macrófitas aquáticas flutuantes. Revista Brasileira de Zootecnia , vol. 37, no. 2, pp. 181-188. http://dx.doi.org/10.1590/S1516-35982008000200002.
» http://dx.doi.org/10.1590/S1516-35982008000200002 - HUNT, P.G. and POACH, M.E., 2001. State of the art for animal wastewater treatment in constructed wetlands. Water Science and Technology, vol. 44, no. 11-12, pp. 19-25. http://dx.doi.org/10.2166/wst.2001.0805. PMid:11804093.
» http://dx.doi.org/10.2166/wst.2001.0805 - JOHNSTON, C.A., 1991. Sediment and nutrient retention by freshwater wetlands: effects on surface water quality. Critical Reviews in Environmental Control, vol. 21, no. 5-6, pp. 491-565. http://dx.doi.org/10.1080/10643389109388425.
» http://dx.doi.org/10.1080/10643389109388425 - KADLEC, R.H. and KNIGHT, R.L., 1996. Treatment wetlands Boca Raton: Lewis Publishers. 893 p.
- KIEHL, E.J., 1985. Fertilizantes orgânicos Piracicaba: Agronômica Ceres. 492 p.
- KISSMANN, K.G. and GROTH, D., 1997. Plantas infestantes e nocivas 2nd ed. São Paulo: BASF Brasileira.
- KNIGHT, R.L., PAYNE-JÚNIOR, V.W.E., BORER, R.E., CLARKE-JÚNIOR, R.A. and PRIES, J.H., 2000. Constructed wetlands for livestock wastewater management. Ecological Engineering, vol. 15, no. 1-2, pp. 41-55. http://dx.doi.org/10.1016/S0925-8574(99)00034-8.
» http://dx.doi.org/10.1016/S0925-8574(99)00034-8 - KÖRNER, S. and VERMAAT, J.E., 1998. The relative importance of Lemna gibba , bacteria and algae for the nitrogen and phosphorus removal in duckweed – covered domestic wastewater. Water Research, vol. 32, no. 12, pp. 3651-366. http://dx.doi.org/10.1016/S0043-1354(98)00166-3.
» http://dx.doi.org/10.1016/S0043-1354(98)00166-3 - LIN, Y.F., JING, S.R., LEE, D.Y., CHANG, Y.F., CHEN, Y.M. and SHIH, K.C., 2005. Performance of a constructed wetland treating intensive shrimp aquaculture wastewater under high hydraulic loading rate. Environmental Pollution, vol. 134, no. 3, pp. 411-421. PMid:15620586.
- LORENZI, H. 1982. Plantas daninhas do Brasil: terrestres, aquáticas, parasitas, tóxicas e medicinais São Paulo: Nova Odessa. 425 p.
- LUTZENBERGER, J., 1985. Ecologia: do jardim do poder Porto Alegre: L&PM Editores, 102 p.
- MALAVOLTA, E., VITTI, G.C. and OLIVEIRA, S.A., 1997. Avaliação do estado nutricional das plantas: princípios e aplicações . 2nd ed. Piracicaba: Potafós. 319 p.
- MEADE, G., PIERCE, J.V., O’DOHERTY, C., MUELLER, G., LANIGAN, G. and MC CABE, T., 2011. Ammonia and nitrous oxide emissions following land application of high and low nitrogen pig manures to winter wheat at three growth stages. Agriculture, Ecosystems & Environment , vol. 140, no. 1-2, pp. 208-217. http://dx.doi.org/10.1016/j.agee.2010.12.007.
» http://dx.doi.org/10.1016/j.agee.2010.12.007 - METCALF, E. and TCHOBANOGLOUS, G., 1991. Wastewater engineering: treatment, disposal and reuse 3rd ed. New York: MacGraw Hill.
- MKHABELA, M.S., GORDON, R., BURTON, D., SMITH, E. and MADANI, A., 2009. The impact of management practices and meteorological conditions on ammonia and nitrous oxide emissions following application of hog slurry to forage grass in Nova Scotia. Agriculture, Ecosystems & Environment, vol. 130, no. 1-2, pp. 41-49. http://dx.doi.org/10.1016/j.agee.2008.11.012.
» http://dx.doi.org/10.1016/j.agee.2008.11.012 - OLIVEIRA, P., 1981. Plantas de aquário Lisboa: Editorial Presença. 238 p.
- ORON, G., DE-VEGT, A. and PORATH, D., 1988. Nitrogen removal and conversion by duckweed grown on wastewater. Water Research, vol. 22, no. 2, pp. 179-184. http://dx.doi.org/10.1016/0043-1354(88)90076-0.
» http://dx.doi.org/10.1016/0043-1354(88)90076-0 - PENG, J.F., WANG, B.Z. and WANG, L., 2005. Mult- stage ponds-wetlands ecosystem for effective wastewater treatment. Journal of Zhejiang University. Science, vol. 6B, no. 5, pp. 346-352. http://dx.doi.org/10.1631/jzus.2005.B0346.
» http://dx.doi.org/10.1631/jzus.2005.B0346 - POACH, M.E., HUNT, P.G., SADLER, E.J., MATHENY, T.A., JOHNSON, M.H., STONE, K.C., HUMINEK, F.J. and RICE, J.M., 2002. Ammonia volatilization from constructed wetlands that treat swine wastewater. Transactions of the ASAE. American Society of Agricultural Engineers , vol. 45, no. 3, pp. 619-627. http://dx.doi.org/10.13031/2013.8825.
» http://dx.doi.org/10.13031/2013.8825 - POACH, M.E., HUNT, P.G., VANOTTI, M.B., STONE, K.C., MATHENY, T.A., JOHNSON, M.H. and SADLER, E.J., 2003. Improved nitrogen by constructed wetlands receiving partially nitrified swine manure. Ecological Engineering, vol. 20, no. 2, pp. 183-197. http://dx.doi.org/10.1016/S0925-8574(03)00024-7.
» http://dx.doi.org/10.1016/S0925-8574(03)00024-7 - POTT, V.J. and POTT, A., 2000. Plantas aquáticas do Pantanal Brasília: Embrapa; Corumbá: Centro de Pesquisa Agropecuária do Pantanal. 404 p.
- REIDEL, A., DAMASCENO, S., ZENATTI, D.C., SAMPAIO, S.C., FEIDEN, A. and QUEIROZ, M.M.F., 2005. Utilização de efluente de frigorífico, tratado com macrófita aquática, no cultivo de tilápia do Nilo. Revista Brasileira de Engenharia Agrícola e Ambiental, vol. 9, suppl, pp. 181-185.
- ROS, C.O., AITA, C. and GIACOMINI, S.J., 2005. Volatilização de amônia com aplicação de uréia na superfície do solo, no sistema plantio direto. Ciência Rural, vol. 35, no. 4, pp. 799-805. http://dx.doi.org/10.1590/S0103-84782005000400008.
» http://dx.doi.org/10.1590/S0103-84782005000400008 - SCHMIDELL, W., SOARES, H.M., ETCHEBEHERE, C., MENES, R.J., BERTOLA, N.C. and CONTRERAS, E.M., 2007. Tratamento biológico de águas residuárias . Florianópolis: Tribo da Ilha.
- SCULTHORPE, C.D., 1967. The biology of aquatic vascular plants London: Edward Arnold Ltd. 610 p.
- SEZERINO, P.H. and PHILIPPI, L.S., 2000. Utilização de um sistema experimental por meio de “wetland” construído no tratamento de esgotos domésticos pós tanque séptico. In: Anais do Simpósio Luso-Brasileiro de Engenharia Sanitária e Ambiental; 2000; Porto Seguro. Rio de Janeiro: ABES, pp. 688-697.
- SHAH, M., HASHMI, H.N., GHUMMAN, A.R. and ZEESHAN, M., 2015. Performance assessment of aquatic macrophytes for treatment of municipal wastewater. Journal of the South African Institution of Civil Engineering, vol. 57, no. 3, pp. 18-25. http://dx.doi.org/10.17159/2309-8775/2015/V57N3A3.
» http://dx.doi.org/10.17159/2309-8775/2015/V57N3A3 - SILVA, A.D.R., SANTOS, R.B., BRUNO, A.M.S.S., GENTELINI, A.L., SILVA, A.H.G. and SOARES, E.C., 2014. Eficiência do aguapé sobre variáveis limnológicas em canais de abastecimento utilizados no cultivo de tambaqui. Acta Amazonica , vol. 44, no. 2, pp. 255-262. http://dx.doi.org/10.1590/S0044-59672014000200011.
» http://dx.doi.org/10.1590/S0044-59672014000200011 - SOMMER, S.G. and HUTCHINGS, N.J., 2001. Ammonia emission from field applied manure and its reduction: invited paper. European Journal of Agronomy, vol. 15, no. 1, pp. 1-15. http://dx.doi.org/10.1016/S1161-0301(01)00112-5.
» http://dx.doi.org/10.1016/S1161-0301(01)00112-5 - SOOKNAH, R.D. and WILKIE, A.C., 2004. Nutrient removal by floating aquatic macrophytes cultured in anaerobically disgested flushed dairy manure wastewater. Ecological Engineering , vol. 22, no. 1, pp. 27-42. http://dx.doi.org/10.1016/j.ecoleng.2004.01.004.
» http://dx.doi.org/10.1016/j.ecoleng.2004.01.004 - STEINMETZ, R.L.R., KUNZ, A., DRESSLER, V.L., FLORES, É.M.M. and MARTINS, A.F., 2009. Study of metal distribution in raw end screened swine manure. Clean Soil Air Water , vol. 37, no. 3, pp. 239-244. http://dx.doi.org/10.1002/clen.200800156.
» http://dx.doi.org/10.1002/clen.200800156 - STONE, K.C., HUNT, P.G., HUMENIK, F.J. and JOHNSON, M.H., 1998. Impact of swine waste application on ground and stream water quality in an Eastern Coastal Plain watershed. American Society of Agricultural and Biological Engineer, vol. 41, no. 6, pp. 1665-1670. http://dx.doi.org/10.13031/2013.17342.
» http://dx.doi.org/10.13031/2013.17342 - UNITED STATES ENVIRONMENTAL PROTECTION AGENCY – EPA, 2000 [viewed 03 November 2017]. Introduction to phytoremediation. Cincinnati, Ohio: EPA. EPA/600/R-99/107. Available from: http://nepis.epa.gov
» http://nepis.epa.gov - VESILIND, P.A. and MORGAN, S.M., 2011. Introdução à engenharia ambiental 2nd ed. São Paulo: Cengage Learning. 438 p.
- ZIMMO, O., VAN DER STEEN, N. and GIJZEN, H., 2003. Comparison of ammonia volatilization rates in algae and duckweed-based waste stabilization ponds treating domestic wastewater. Water Research, vol. 37, no. 19, pp. 4587-4594. http://dx.doi.org/10.1016/j.watres.2003.08.013. PMid:14568043.
» http://dx.doi.org/10.1016/j.watres.2003.08.013
Publication Dates
-
Publication in this collection
13 Sept 2018 -
Date of issue
Jul-Sep 2019
History
-
Received
16 June 2017 -
Accepted
11 Jan 2018