Abstract
The fidelity of the genomes is defended by mechanism known as Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) systems. Three Type II CRISPR systems (CRISPR1- cas, CRISPR2 and CRISPR3-cas) have been identified in enterococci isolates from clinical and environmental samples. The aim of this study was to observe the distribution of CRISPR1-cas, CRISPR2 and CRISPR3-cas in non-clinical strains of Enterococcus faecalis and Enterococcus faecium isolates from food and fecal samples, including wild marine animals. The presence of CRISPRs was evaluated by PCR in 120 enterococci strains, 67 E. faecalis and 53 E. faecium. It is the first report of the presence of the CRISPRs system in E. faecalis and E. faecium strains isolated from wild marine animal fecal samples. The results showed that in non-clinical strains, the CRISPRs were more frequently detected in E. faecalis than in E. faecium. And the frequencies of CRISPR1-cas and CRISPR2 were higher (60%) in E. faecalis strains isolated from animal feces, compared to food samples. Both strains showed low frequencies of CRISPR3-cas (8.95% and 1.88%). In conclusion, the differences in the habitats of enterococcal species may be related with the results observe in distribution of CRISPRs systems.
Keywords:
Enterococcus faecalis; Enterococcus faecium; CRISPRs; food samples; fecal samples; wild marine animals
Resumo
A fidelidade dos genomas é defendida por mecanismos conhecidos como sistemas de repetições palindrômicas curtas agrupadas e regularmente interespaçadas (CRISPRs). Três tipos de sistemas CRISPR II (CRISPR1-cas, CRISPR2 e CRISPR3-cas) têm sido identificados em cepas de enterococos isolados de amostras clínicas e ambientais. O objetivo deste estudo foi observar a distribuição dos CRISPR1-cas, CRISPR2 e CRISPR3-cas em cepas não-clínicas de Enterococcus faecalis e Enterococcus faecium isoladas de amostras alimentícias e fecais, incluindo animais marinhos selvagens. A presenca dos CRISPRs foi determinada por PCR em 120 cepas de enterococos, sendo 67 E. faecalis e 53 E. faecium. É o primeiro relato da presença do sistema CRISPRs nas estirpes E. faecalis e E. faecium isoladas de amostras fecais de animais marinhos selvagens. Os resultados mostraram que em cepas não-clínicas, os CRISPRs foram mais frequentemente detectados em E. faecalis do que em E. faecium. E as frequências de CRISPR1-cas e CRISPR2 foram maiores (60%) em cepas de E. faecalis isoladas de fezes de animais, quando comparadas à amostras de alimentos. Ambas as cepas apresentaram baixas freqüências de CRISPR3-cas (8,95% e 1,88%). Em conclusão, as diferenças nos habitats das espécies de enterococos podem estar relacionadas com os resultados observados na distribuição dos sistemas CRISPRs.
Palavras-chave:
Enterococcus faecalis; Enterococcus faecium; CRISPRs; amostras alimentares; amostras fecais; animais marinhos selvagens
1. Introduction
Enterococci are important Gram-positive bacteria recognized as part of gut microbiota in humans and animals, and they are widely distributed in soil, water, plants and foods ( Cassenego et al., 2013 CASSENEGO, A.P.V., ELLWANGERI, J., D’AZEVEDO, P., FRAZZON, J. and FRAZZON, A.P.G., 2013. Virulence and biofilm formation by Enterococcus faecalis isolates from cloacal swabs of broilers infected with Eimeria spp. Pesquisa Veterinária Brasileira, vol. 33, no. 12, pp. 1433-1440. http://dx.doi.org/10.1590/S0100-736X2013001200007.
http://dx.doi.org/10.1590/S0100-736X201...
; Lebreton et al., 2014 LEBRETON, F., WILLEMS, R.J.L. and GILMORE, M.S., 2014. Enterococcus diversity, origins in nature, and gut colonization. In: M.S. Gilmore, D.B. Clewell, Y. Ike and N. Shankar, eds. Enterococci: from commensals to leading causes of drug resistant infection . Boston: Massachusetts Eye and Ear, pp. 1-59. PMid:24649510. ; Pieniz et al., 2015 PIENIZ, S., ANDREAZZA, R., OKEKE, B.C., CAMARGO, F.A. and BRANDELLI, A., 2015. Antimicrobial and antioxidant activities of Enterococcus species isolated from meat and dairy products. Brazilian Journal of Biology = Revista Brasileira de Biologia , vol. 75, no. 4, pp. 923-931. http://dx.doi.org/10.1590/1519-6984.02814. PMid:26675908.
http://dx.doi.org/10.1590/1519-6984.028...
; Santestevan et al., 2015 SANTESTEVAN, N.A., ZVOBODA, D.A., PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., MOURA, T.M., MEDEIROS, A.W., AMORIN, D.B., TAVARES, M., D’AZEVEDO, P.A., FRANCO, A.C., FRAZZON, J. and FRAZZON, A.P., 2015. Antimicrobial Resistance and Virulence Factor Gene Profiles of Enterococcus Spp. Isolates From Wild Arctocephalus Australis (South American Fur Seal) and Arctocephalus Tropicalis (Subantarctic Fur Seal). World Journal of Microbiology & Biotechnology , vol. 31, no. 12, pp. 1935-1946. http://dx.doi.org/10.1007/s11274-015-1938-7. PMid:26347323.
http://dx.doi.org/10.1007/s11274-015-19...
; Prichula et al., 2016 PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., TOLFO, N.C., SANTESTEVAN, N.A., MEDEIROS, A.W., TAVARES, M., FRAZZON, J., D’AZEVEDO, P.A. and FRAZZON, A.P., 2016. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Marine Pollution Bulletin , vol. 105, no. 1, pp. 51-57. http://dx.doi.org/10.1016/j.marpolbul.2016.02.071. PMid:26952995.
http://dx.doi.org/10.1016/j.marpolbul.2...
; Medeiros et al., 2017 MEDEIROS, A.W., AMORIM, D.B., TAVARES, M., MOURA, T.M., FRANCO, A.C., D’AZEVEDO, P.A., FRAZZON, J. and FRAZZON, A.P., 2017. Enterococcus species diversity in fecal samples of wild marine species as determined by Real-Time PCR. Canadian Journal of Microbiology, vol. 63, no. 2, pp. 129-136. http://dx.doi.org/10.1139/cjm-2016-0427. PMid:27991828.
http://dx.doi.org/10.1139/cjm-2016-0427...
). Their ubiquity reflects its ability to survive in a variety of environmental stressors ( Byappanahalli et al., 2012 BYAPPANAHALLI, M.N., NEVERS, M.B., KORAJKIC, A., STALEY, Z.R. and HARWOOD, V.J., 2012. Enterococci in the environment. Microbiology and Molecular Biology Reviews, vol. 76, no. 4, pp. 685-706. http://dx.doi.org/10.1128/MMBR.00023-12. PMid:23204362.
http://dx.doi.org/10.1128/MMBR.00023-12...
). Another important characteristic is their intrinsic resistance to some antimicrobials agents commonly prescribed to treat Gram-positive cocci such as cephalosporin, lincomycin, and low levels of penicillin and aminoglycosides. Furthermore, they are also able to acquire a variety of resistance genes via transposons or plasmids. Epidemiological data suggest that enterococci are important reservoirs for the spread of antibiotic resistance to different species of bacteria ( Lebreton et al., 2014 LEBRETON, F., WILLEMS, R.J.L. and GILMORE, M.S., 2014. Enterococcus diversity, origins in nature, and gut colonization. In: M.S. Gilmore, D.B. Clewell, Y. Ike and N. Shankar, eds. Enterococci: from commensals to leading causes of drug resistant infection . Boston: Massachusetts Eye and Ear, pp. 1-59. PMid:24649510. ).
The Enterococcus genus comprises more than 50 species, and Enterococcus faecalis and Enterococcus faecium are the most frequent species isolated from humans, animals and food samples ( Lebreton et al., 2014 LEBRETON, F., WILLEMS, R.J.L. and GILMORE, M.S., 2014. Enterococcus diversity, origins in nature, and gut colonization. In: M.S. Gilmore, D.B. Clewell, Y. Ike and N. Shankar, eds. Enterococci: from commensals to leading causes of drug resistant infection . Boston: Massachusetts Eye and Ear, pp. 1-59. PMid:24649510. ). Moreover, they are the most prevalent species cultured from humans’ infected sites, including bacteremia, surgical site infections, and urinary tract infections.
The genomes of E. faecalis and E. faecium have suffered rearrangements over hundreds of millions of years in evolution, given them selective advantages to survive and disperse in the environment. As a result, the presence of the Enterococcus spp. has been investigated and monitored in a variety of habitats ( Van Tyne and Gilmore, 2014 VAN TYNE, D. and GILMORE, M.S., 2014. Friend turned foe: evolution of enterococcal virulence and antibiotic resistance. Annual Review of Microbiology, vol. 68, no. 1, pp. 337-356. http://dx.doi.org/10.1146/annurev-micro-091213-113003. PMid:25002090.
http://dx.doi.org/10.1146/annurev-micro...
).
To defend the genome against parasitic DNA invasion, and to maintain the fidelity of the genomes in stable ecosystems, there is a mechanism known as Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) systems ( Sorek et al. 2008 SOREK, R., KUNIN, V. and HUGENHOLTZ, P., 2008. CRISPR-- a widespread system that provides acquired resistance against phages in bacteria and archaea. Nature Reviews Microbiology , vol. 6, no. 3, pp. 181-186. http://dx.doi.org/10.1038/nrmicro1793. PMid:18157154.
http://dx.doi.org/10.1038/nrmicro1793 ...
; Palmer and Gilmore, 2010 PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
; Magadán et al., 2012 MAGADÁN, A.H., DUPUIS, M.È., VILLION, M. and MOINEAU, S., 2012. Cleavage of Phage DNA by the Streptococcus thermophilus CRISPR3-Cas System. PLoS One, vol. 7, no. 7, pp. e40913. http://dx.doi.org/10.1371/journal.pone.0040913. PMid:22911717.
http://dx.doi.org/10.1371/journal.pone....
). The CRISPR systems provides a type of defense in prokaryotes, leading to a resistance to plasmid uptake and phage infections, and a barrier to horizontal gene transfection ( Thomas and Nielsen, 2005 THOMAS, C.M. and NIELSEN, K.M., 2005. Mechanisms of, and barriers to, horizontal gene transfer between bacteria. Nature Reviews. Microbiology, vol. 3, no. 9, pp. 711-721. http://dx.doi.org/10.1038/nrmicro1234. PMid:16138099.
http://dx.doi.org/10.1038/nrmicro1234 ...
; Wiedenheft et al., 2012 WIEDENHEFT, B., STERNBERG, S.H. and DOUDNA, J.A., 2012. RNA-guided genetic silencing systems in bacteria and archaea. Nature, vol. 482, no. 7385, pp. 331-338. http://dx.doi.org/10.1038/nature10886. PMid:22337052.
http://dx.doi.org/10.1038/nature10886 ...
). This immunity depends on the presence of specific target-derived spacer sequences, the intervening repeat palindromes short and highly conserved, and nuclease activity encoded by the cas genes ( Haft et al., 2005 HAFT, D.H., SELENGUT, J., MONGODIN, E.F. and NELSON, K.E., 2005. A guild of 45 CRISPR-associated (Cas) protein families and multiple CRISPR/Cas subtypes exist in prokaryotic genomes. PLoS Computational Biology, vol. 1, no. 6, pp. e60. http://dx.doi.org/10.1371/journal.pcbi.0010060. PMid:16292354.
http://dx.doi.org/10.1371/journal.pcbi....
; Makarova et al., 2006 MAKAROVA, K., SLESAREV, A., WOLF, Y., SOROKIN, A., MIRKIN, B., KOONIN, E., PAVLOV, A., PAVLOVA, N., KARAMYCHEV, V., POLOUCHINE, N., SHAKHOVA, V., GRIGORIEV, I., LOU, Y., ROHKSAR, D., LUCAS, S., HUANG, K., GOODSTEIN, D.M., HAWKINS, T., PLENGVIDHYA, V., WELKER, D., HUGHES, J., GOH, Y., BENSON, A., BALDWIN, K., LEE, J.-H., DIAZ-MUNIZ, I., DOSTI, B., SMEIANOV, V., WECHTER, W., BARABOTE, R., LORCA, G., ALTERMANN, E., BARRANGOU, R., GANESAN, B., XIE, Y., RAWSTHORNE, H., TAMIR, D., PARKER, C., BREIDT, F., BROADBENT, J., HUTKINS, R., O’SULLIVAN, D., STEELE, J., UNLU, G., SAIER, M., KLAENHAMMER, T., RICHARDSON, P., KOZYAVKIN, S., WEIMER, B. and MILLS, D., 2006. Comparative genomics of the lactic acid bacteria. Proceedings of the National Academy of Sciences of the United States of America, vol. 103, no. 42, pp. 15611-15616. http://dx.doi.org/10.1073/pnas.0607117103. PMid:17030793.
http://dx.doi.org/10.1073/pnas.06071171...
; Sorek et al., 2008 SOREK, R., KUNIN, V. and HUGENHOLTZ, P., 2008. CRISPR-- a widespread system that provides acquired resistance against phages in bacteria and archaea. Nature Reviews Microbiology , vol. 6, no. 3, pp. 181-186. http://dx.doi.org/10.1038/nrmicro1793. PMid:18157154.
http://dx.doi.org/10.1038/nrmicro1793 ...
).
Type II CRISPR-Cas loci consists of a CRISPR array, the type-specific cas9 gene, and cas1 and cas2 genes. In enterococci strains, three Type II CRISPR-Cas loci have been identified, the CRISPR locus lacking cas genes, thus consisting solely of repeat-spacer arrays (orphan CRISPR2), and those carrying these cas genes (CRISPR1–Cas and CRISPR3–Cas) ( Makarova et al., 2011 MAKAROVA, K.S., HAFT, H.D., BARRANGOU, R., BROUNS, S.J., CHARPENTIER, E., HORVATH, P., MOINEAU, S., MOJICA, F.J., WOLF, Y.I., YAKUNIN, A.F., VAN DER OOST, J. and KOONIN, E.V., 2011. Evolution and classification of the CRISPR-Cas systems. Nature Reviews Microbiology , vol. 9, no. 6, pp. 467-477. http://dx.doi.org/10.1038/nrmicro2577. PMid:21552286.
http://dx.doi.org/10.1038/nrmicro2577 ...
). Palmer and Gilmore (2010) PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
using comparative genomics found that pathogenic enterococci have multiple rearrangements that occurred during evolution and is followed by the loss of CRISPRs. In contrast, commensal strains have smaller genomes with fewer rearrangements, as demonstrated by whole-genome alignments. The same authors found an inverse relationship between the presence of a CRISPR-cas locus and acquired antibiotic resistance in E. faecalis, suggesting that antibiotic use inadvertently selects for enterococcal strains with compromised genome defense ( Palmer and Gilmore, 2010 PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
).
CRISPR1-Cas and CRISPR3-Cas loci occur with variable distribution in E. faecalis and they are absent in multi-resistant strains isolated from clinical samples ( Palmer and Gilmore, 2010 PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
). In addition, CRISPR2 locus is conserved across the species. Since the CRISPR system is reactive to the environment, it might play a critical role in the adaptation of the host to its surroundings and explains the persistence of particular bacterial strains in ecosystems where phages are present ( Horvath et al., 2008 HORVATH, P., ROMERO, D.A., COÛTÉ-MONVOISIN, A.C., RICHARDS, M., DEVEAU, H., MOINEAU, S., BOYAVAL, P., FREMAUX, C. and BARRANGOU, R., 2008. Diversity, activity, and evolution of CRISPR loci in Streptococcus thermophilus. Journal of Bacteriology, vol. 190, no. 4, pp. 1401-1412. http://dx.doi.org/10.1128/JB.01415-07. PMid:18065539.
http://dx.doi.org/10.1128/JB.01415-07 ...
).
The distribution of CRISPRs in clinical enterococci strains have been studied ( Palmer and Gilmore, 2010 PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
; Lindenstrauß et al., 2011 LINDENSTRAUß, A.G., PAVLOVIC, M., BRINGMANN, A., BEHR, J., EHRMANN, M.A. and VOGEL, R.F., 2011. Comparison of genotypic and phenotypic cluster analyses of virulence determinants and possible role of CRISPR elements towards their incidence in Enterococcus faecalis and Enterococcus faecium. Systematic and Applied Microbiology, vol. 34, no. 8, pp. 553-560. http://dx.doi.org/10.1016/j.syapm.2011.05.002. PMid:21943678.
http://dx.doi.org/10.1016/j.syapm.2011....
; Burley and Sedgley, 2012 BURLEY, K.M. and SEDGLEY, C.M., 2012. CRISPR-Cas, a prokaryotic adaptive immune system, in endodontic, oral, and multidrug-resistant hospital-acquired Enterococcus faecalis. Journal of Endodontics, vol. 38, no. 11, pp. 1511-1515. http://dx.doi.org/10.1016/j.joen.2012.07.004. PMid:23063226.
http://dx.doi.org/10.1016/j.joen.2012.0...
; Hullahalli et al., 2015 HULLAHALLI, K., RODRIGUES, M., SCHMIDT, B.D., LI, X., BHARDWAJ, P. and PALMER, K.L., 2015. Comparative analysis of the orphan CRISPR2 Locus in 242 Enterococcus faecalis strains. PLoS One, vol. 10, no. 9, pp. e0138890. http://dx.doi.org/10.1371/journal.pone.0138890. PMid:26398194.
http://dx.doi.org/10.1371/journal.pone....
), however, in non-clinical strains are still poorly studied ( Katyal et al., 2013 KATYAL, I., CHABAN, B., NG, B. and HILL, J.E., 2013. CRISPRs of Enterococcus faecalis and E. hirae isolates from pig feces have species-specific repeats but share some common spacer sequences. Microbial Ecology, vol. 66, no. 1, pp. 182-188. http://dx.doi.org/10.1007/s00248-013-0217-0. PMid:23535981.
http://dx.doi.org/10.1007/s00248-013-02...
; Lyons et al., 2015 LYONS, C., RAUSTAD, N., BUSTOS, M.A. and SHIARIS, M., 2015. Incidence of Type II CRISPR1-Cas systems in Enterococcus is species-dependent. PLoS One , vol. 10, no. 11, pp. e0143544. http://dx.doi.org/10.1371/journal.pone.0143544. PMid:26600384.
http://dx.doi.org/10.1371/journal.pone....
). Therefore, the goal of this study was to determine the distribution and frequency of CRISPR1–c as, orphan CRISPR2 and CRISPR3–cas in E. faecalis and E. faecium strains isolated from food and fecal samples, including wild marine animals.
2. Materials and Methods
2.1. Enterococcus strains
A total of 120 enterococci isolated from food and animal fecal samples were used in this study ( Table 1 ). Among these, 67 were identified as E. faecalis and 53 E. faecium. The enterococci were isolated from different foods samples (beetroot, potato, parsley, raw meat, buffalos milk and dairy products, such as mozzarella cheese), and animal feces (wild fur seals, chickens, wild Magellanic penguins and wild green turtles) in Southern Brazil, from 2009 to 2015. Strains were randomly selected from the culture collection of the Department of Microbiology, Universidade Federal do Rio Grande do Sul.
All strains were already identified as genus, species and virulence profile in previous studies ( Frazzon et al., 2009 FRAZZON, A.P.G., GAMA, B.A., HERMES, V., BIERHALS, C.G., PEREIRA, R.I., GUEDES, A.G., D’AZEVEDO, P.A. and FRAZZON, J., 2009. Prevalence of antimicrobial resistance and molecular characterization of tetracycline resistance mediated by tet(M) and tet (L) genes in Enterococcus spp. isolated from food in Southern Brazil. World Journal of Microbiology & Biotechnology, vol. 26, no. 2, pp. 365-370. http://dx.doi.org/10.1007/s11274-009-0160-x.
http://dx.doi.org/10.1007/s11274-009-01...
; Riboldi et al. 2009 RIBOLDI, G.P., FRAZZON, J., D’ AZEVEDO, P.A. and FRAZZON, A.P.G., 2009. Antimicrobial resistance profile of Enterococcus spp isolated from food in Southern Brazil. Brazilian Journal of Microbiology, vol. 40, no. 1, pp. 125-128. http://dx.doi.org/10.1590/S1517-83822009000100021. PMid:24031330.
http://dx.doi.org/10.1590/S1517-8382200...
; Cassenego et al., 2013 CASSENEGO, A.P.V., ELLWANGERI, J., D’AZEVEDO, P., FRAZZON, J. and FRAZZON, A.P.G., 2013. Virulence and biofilm formation by Enterococcus faecalis isolates from cloacal swabs of broilers infected with Eimeria spp. Pesquisa Veterinária Brasileira, vol. 33, no. 12, pp. 1433-1440. http://dx.doi.org/10.1590/S0100-736X2013001200007.
http://dx.doi.org/10.1590/S0100-736X201...
; Prichula et al., 2013 PRICHULA, J., ZVOBODA, D.A., PEREIRA, R.I., SANTESTEVAN, N.A., MEDEIROS, A.W., MOTTA, A.S., D’AZEVEDO, P.A., GIORDANI, A.R. and FRAZZON, A.P.G., 2013. Antimicrobial susceptibility profile and diversity of enterococci species isolated from raw milk of buffalo in South Brazil. Revista Brasileira de Ciência Veterinária, vol. 20, no. 2, pp. 104-109. http://dx.doi.org/10.4322/rbcv.2014.056.
http://dx.doi.org/10.4322/rbcv.2014.056...
; Santestevan et al., 2015 SANTESTEVAN, N.A., ZVOBODA, D.A., PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., MOURA, T.M., MEDEIROS, A.W., AMORIN, D.B., TAVARES, M., D’AZEVEDO, P.A., FRANCO, A.C., FRAZZON, J. and FRAZZON, A.P., 2015. Antimicrobial Resistance and Virulence Factor Gene Profiles of Enterococcus Spp. Isolates From Wild Arctocephalus Australis (South American Fur Seal) and Arctocephalus Tropicalis (Subantarctic Fur Seal). World Journal of Microbiology & Biotechnology , vol. 31, no. 12, pp. 1935-1946. http://dx.doi.org/10.1007/s11274-015-1938-7. PMid:26347323.
http://dx.doi.org/10.1007/s11274-015-19...
; Prichula et al., 2016 PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., TOLFO, N.C., SANTESTEVAN, N.A., MEDEIROS, A.W., TAVARES, M., FRAZZON, J., D’AZEVEDO, P.A. and FRAZZON, A.P., 2016. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Marine Pollution Bulletin , vol. 105, no. 1, pp. 51-57. http://dx.doi.org/10.1016/j.marpolbul.2016.02.071. PMid:26952995.
http://dx.doi.org/10.1016/j.marpolbul.2...
). The strains were preserved in 10% (w/v) skim milk (Difco, Sparks, MD, USA) solution supplemented with 10% (v/v) glycerol (LabSynth®) frozen at -20 °C.
2.2. DNA isolation
For DNA isolation, an aliquot of frozen bacterial cells was recovered in Brain Heart Infusion agar (BHA, Himedia®, India), and incubated at 35 °C for 24 h. Genomic DNA was extracted using the method described by Donato (2007) DONATO, S.T., 2007 [viewed 8 July 2017]. Comparison of conventional and semi-automatized methods to identify Enterococcus spp versus molecular biology in discrepant identifications [online]. Fortaleza: Universidade Federal de Fortaleza, 88 p. Dissertação de Mestrado em Medicina Clinica. Available from: http://www.repositorio.ufc.br/handle/riufc/1764
http://www.repositorio.ufc.br/handle/ri...
. All strains were confirmed to genus and species by polymerase chain reaction (PCR) assays according to the protocol established by Prichula et al. (2016) PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., TOLFO, N.C., SANTESTEVAN, N.A., MEDEIROS, A.W., TAVARES, M., FRAZZON, J., D’AZEVEDO, P.A. and FRAZZON, A.P., 2016. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Marine Pollution Bulletin , vol. 105, no. 1, pp. 51-57. http://dx.doi.org/10.1016/j.marpolbul.2016.02.071. PMid:26952995.
http://dx.doi.org/10.1016/j.marpolbul.2...
. The primers sequences and their amplification products are listed in Table 2 .
2.3. Detection of CRISPR-associated (cas) genes and orphan locus lacking cas genes
Primers for CRISPR amplification reported by Palmer and Gilmore (2010) PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
were used in PCR reaction. The primers and annealing temperatures used are listed in Table 2 . The PCR was performed in a total volume of 25 µL containing: 1x PCR buffer (10 mM Tris-HCl [pH 9.0], (Invitrogen, Carlsbad, CA, USA), 1.5 mM of MgCl2 (Invitrogen, Carlsbad, CA, USA), 200 μM of dNTPs (Ludwig Biotecnologia), 0.4 µM of each primer (Invitrogen, Carlsbad, CA, USA), 1 U of Taq polymerase (Invitrogen, Carlsbad, CA, USA), and 100 ng of genomic DNA. The reaction mixture was subjected to 30 cycles of denaturation at 94 °C for 1 min.; primer annealing for 1 min. (at appropriate temperature), and extension at 72 °C for 1 min., followed by a final extension at 72 °C for 10 min. The PCR products were analyzed on 1.2% agarose gels stained with SYBR safe (Invitrogen, Carlsbad, CA, USA) solution and visualized under UV light.
2.4. Sequencing of samples
In order to confirm the CRISPRs amplifications, PCR products amplified of CRISPR1–c as, orphan CRISPR2 and CRISPR3–cas genes were submitted to nucleotide sequence analysis. The DNA fragments were purified using illustra GFX™ PCR DNA and gel band purification kit (GE Healthcare-Buckinghamshire, United Kingdom - UK). Sequencing was carried out with the Big Dye Terminator Cycle Sequencing Ready Reaction (Applied Biosystems) in an ABI-PRISM 3100 Genetic Analyzer (ABI), according to the protocol of the manufacturer. The sequences obtained were compared with homologous nucleotide sequences deposited in GenBank).
3. Results
3.1. Detection of CRISPRs in enterococci isolates from food and animals fecal samples
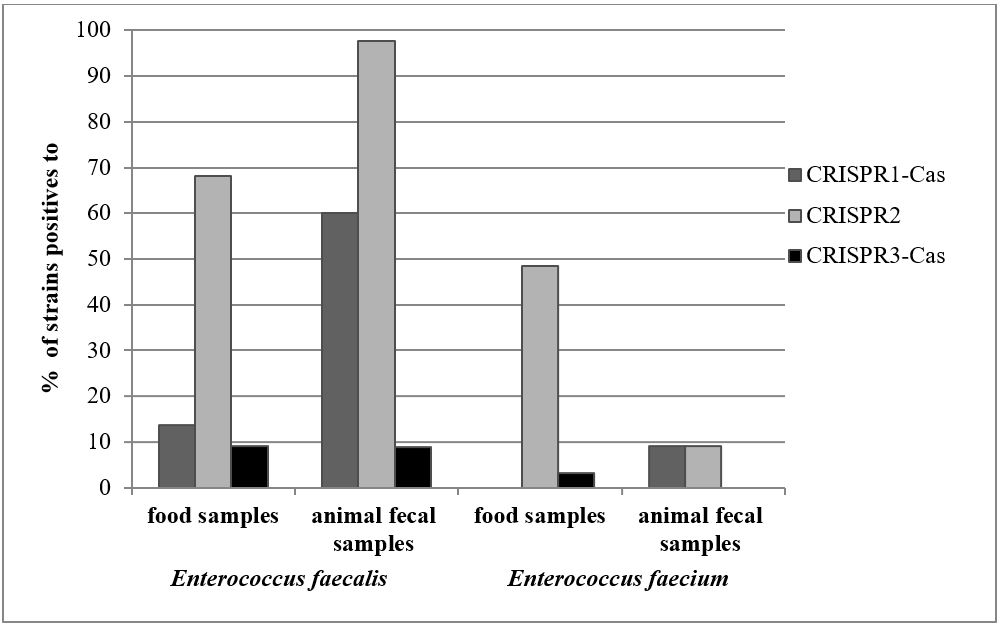
The distribution and the frequency of the Type II CRISPRs (CRISPR1-cas , CRISPR2, and CRISPR3-cas) in non-clinical strains of E. faecalis and E. faecium are present in Figure 1 . The CRISPRs were more frequently detected in E. faecalis than in E. faecium strains. In E. faecalis strains, the CRISPR1- cas and CRISPR2 were detected in 44.77% and 88.07%, respectively. On the other hand, in E. faecium the CRISPR1-cas were observed in 5.66% and CRISPR2 in 32.72% of the strains. Both strains showed low frequencies of CRISPR3- cas genes (8.95% and 1.88%).
Distribution of CRISPR systems in non-clinical Enterococcus faecalis and Enterococcus faecium isolated from food and animal fecal samples.
The frequency of CRISPRs in E. faecalis and E. faecium strains and source of isolation were related. A higher frequency of CRISPR1-cas and CRISPR2 in non-clinical were observed in E. faecalis isolates from fecal samples, in contrast to food samples. In fecal strains, the CRISPR1-cas was observed in 60% of the E. faecalis strains, whereas in food strains in 13.66%. In relation to CRISPR2, it was positive in 97.11% and 68.18% of the E. faecalis isolated from fecal and food samples, respectively. Similar frequency of CRISPR3-cas gene was detected in both samples.
Identical proportions of CRISPR1-cas and CRISPR3-cas (3.22%) were observed in E. faecium isolated from food products, but a substantially high prevalence of CRISPR2 was observed in E. faecium (48.38%). In addition, equal frequency of CRISPR1-cas and CRISPR2 (9.095%) were observed in E. faecium isolated from animals fecal samples. The CRISPR3 -cas amplification was not detected in E. faecium strains.
3.2. Analysis of CRISPR sequences
The nucleotide BLAST of the PCR products from CRISPR1-cas, CRISPR2 and CRISPR3-cas genes showed that CRISPR1-cas gene had 99% identity with the DNA sequence of CRISPR1-cas of E. faecalis strain D32 isolated from pig feces in Denmark (GenBank: NC_018221), culture_collection E. faecalis strain OG1RF ATCC 47077 (GenBank: CP002621.1), and E. faecalis strain L12, isolated from suine in Brazil (GenBank: CP018102.1). The DNA sequence from CRISPR2 gene showed 91% of identity to CRISPR2 gene of E. faecalis strain L9, isolated from rectal swabs from suine in Brazil (GenBank: CP018004.1) and 88% to E. faecalis strain DENG1, isolated from sputum of human in China (GenBank: CP004081.1) and Enterococcus sp. strain 7L76 draft genome (GenBank: FP929058.1). The DNA sequence of CRISPR3-cas gene showed 98% of identity to the draft genome of Enterococcus sp. strain 7L76 (GenBank: FP929058.1).
4. Discussion
Differences in the distribution and the frequency of the Type II CRISPRs (CRISPR1- cas, CRISPR2, and CRISPR3-cas) in non-clinical E. faecalis and E. faecium were observed. These differences observed in CRISPRS distributions among enterococci species were reported before by Palmer and Gilmore (2010) PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
, Lindenstrauß et al. (2011) LINDENSTRAUß, A.G., PAVLOVIC, M., BRINGMANN, A., BEHR, J., EHRMANN, M.A. and VOGEL, R.F., 2011. Comparison of genotypic and phenotypic cluster analyses of virulence determinants and possible role of CRISPR elements towards their incidence in Enterococcus faecalis and Enterococcus faecium. Systematic and Applied Microbiology, vol. 34, no. 8, pp. 553-560. http://dx.doi.org/10.1016/j.syapm.2011.05.002. PMid:21943678.
http://dx.doi.org/10.1016/j.syapm.2011....
, Katyal et al. (2013) KATYAL, I., CHABAN, B., NG, B. and HILL, J.E., 2013. CRISPRs of Enterococcus faecalis and E. hirae isolates from pig feces have species-specific repeats but share some common spacer sequences. Microbial Ecology, vol. 66, no. 1, pp. 182-188. http://dx.doi.org/10.1007/s00248-013-0217-0. PMid:23535981.
http://dx.doi.org/10.1007/s00248-013-02...
, Hullahalli et al. (2015) HULLAHALLI, K., RODRIGUES, M., SCHMIDT, B.D., LI, X., BHARDWAJ, P. and PALMER, K.L., 2015. Comparative analysis of the orphan CRISPR2 Locus in 242 Enterococcus faecalis strains. PLoS One, vol. 10, no. 9, pp. e0138890. http://dx.doi.org/10.1371/journal.pone.0138890. PMid:26398194.
http://dx.doi.org/10.1371/journal.pone....
and Lyons et al. (2015) LYONS, C., RAUSTAD, N., BUSTOS, M.A. and SHIARIS, M., 2015. Incidence of Type II CRISPR1-Cas systems in Enterococcus is species-dependent. PLoS One , vol. 10, no. 11, pp. e0143544. http://dx.doi.org/10.1371/journal.pone.0143544. PMid:26600384.
http://dx.doi.org/10.1371/journal.pone....
. The incidence of CRISPR1-cas gene in Enterococcus species is recognized; sometimes cas sequences are distinctly different from each other, showing species-level evolution. CRISPR2 loci were detected in high frequency in all isolates evaluated, manly in animal fecal samples. Similar results were identified in other reports that evaluated E. faecalis strains ( Palmer and Gilmore, 2010 PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
; Hullahalli et al., 2015 HULLAHALLI, K., RODRIGUES, M., SCHMIDT, B.D., LI, X., BHARDWAJ, P. and PALMER, K.L., 2015. Comparative analysis of the orphan CRISPR2 Locus in 242 Enterococcus faecalis strains. PLoS One, vol. 10, no. 9, pp. e0138890. http://dx.doi.org/10.1371/journal.pone.0138890. PMid:26398194.
http://dx.doi.org/10.1371/journal.pone....
). Hullahalli et al. (2015) HULLAHALLI, K., RODRIGUES, M., SCHMIDT, B.D., LI, X., BHARDWAJ, P. and PALMER, K.L., 2015. Comparative analysis of the orphan CRISPR2 Locus in 242 Enterococcus faecalis strains. PLoS One, vol. 10, no. 9, pp. e0138890. http://dx.doi.org/10.1371/journal.pone.0138890. PMid:26398194.
http://dx.doi.org/10.1371/journal.pone....
showed that CRISPR2 locus is ubiquitous in E. faecalis. Until today, there are only a few reports evaluating the distribution of CRISPRs in non-clinical enterococci strains, isolated from food and animals fecal samples ( Katyal et al., 2013 KATYAL, I., CHABAN, B., NG, B. and HILL, J.E., 2013. CRISPRs of Enterococcus faecalis and E. hirae isolates from pig feces have species-specific repeats but share some common spacer sequences. Microbial Ecology, vol. 66, no. 1, pp. 182-188. http://dx.doi.org/10.1007/s00248-013-0217-0. PMid:23535981.
http://dx.doi.org/10.1007/s00248-013-02...
; Lindenstrauß et al., 2011 LINDENSTRAUß, A.G., PAVLOVIC, M., BRINGMANN, A., BEHR, J., EHRMANN, M.A. and VOGEL, R.F., 2011. Comparison of genotypic and phenotypic cluster analyses of virulence determinants and possible role of CRISPR elements towards their incidence in Enterococcus faecalis and Enterococcus faecium. Systematic and Applied Microbiology, vol. 34, no. 8, pp. 553-560. http://dx.doi.org/10.1016/j.syapm.2011.05.002. PMid:21943678.
http://dx.doi.org/10.1016/j.syapm.2011....
; Lyons et al., 2015 LYONS, C., RAUSTAD, N., BUSTOS, M.A. and SHIARIS, M., 2015. Incidence of Type II CRISPR1-Cas systems in Enterococcus is species-dependent. PLoS One , vol. 10, no. 11, pp. e0143544. http://dx.doi.org/10.1371/journal.pone.0143544. PMid:26600384.
http://dx.doi.org/10.1371/journal.pone....
). It is the first report of the presence of CRISPRs in E. faecalis and E. faecium isolates from fecal samples of wild marine animals. The high frequency of CRISPRs in E. faecalis strains isolated from animal fecal samples, especially in fecal samples of wild animal could be related with low frequency of antibiotic resistance genes in these strains ( Santestevan et al., 2015 SANTESTEVAN, N.A., ZVOBODA, D.A., PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., MOURA, T.M., MEDEIROS, A.W., AMORIN, D.B., TAVARES, M., D’AZEVEDO, P.A., FRANCO, A.C., FRAZZON, J. and FRAZZON, A.P., 2015. Antimicrobial Resistance and Virulence Factor Gene Profiles of Enterococcus Spp. Isolates From Wild Arctocephalus Australis (South American Fur Seal) and Arctocephalus Tropicalis (Subantarctic Fur Seal). World Journal of Microbiology & Biotechnology , vol. 31, no. 12, pp. 1935-1946. http://dx.doi.org/10.1007/s11274-015-1938-7. PMid:26347323.
http://dx.doi.org/10.1007/s11274-015-19...
; Prichula et al., 2016 PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., TOLFO, N.C., SANTESTEVAN, N.A., MEDEIROS, A.W., TAVARES, M., FRAZZON, J., D’AZEVEDO, P.A. and FRAZZON, A.P., 2016. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Marine Pollution Bulletin , vol. 105, no. 1, pp. 51-57. http://dx.doi.org/10.1016/j.marpolbul.2016.02.071. PMid:26952995.
http://dx.doi.org/10.1016/j.marpolbul.2...
). To fecal strains, the results are partial in agreement with Lyons et al. 2015 and Katyal et al. 2003 that reported CRISPRs in E. faecalis isolated from fecal specimens. Although Lyons et al. (2015) LYONS, C., RAUSTAD, N., BUSTOS, M.A. and SHIARIS, M., 2015. Incidence of Type II CRISPR1-Cas systems in Enterococcus is species-dependent. PLoS One , vol. 10, no. 11, pp. e0143544. http://dx.doi.org/10.1371/journal.pone.0143544. PMid:26600384.
http://dx.doi.org/10.1371/journal.pone....
described a high frequency of CRISPR1-cas in Enterococcus spp. isolates from animal fecal samples; CRISPR1-cas was not detected in E. faecalis strains. Instead, Katyal et al. (2013) KATYAL, I., CHABAN, B., NG, B. and HILL, J.E., 2013. CRISPRs of Enterococcus faecalis and E. hirae isolates from pig feces have species-specific repeats but share some common spacer sequences. Microbial Ecology, vol. 66, no. 1, pp. 182-188. http://dx.doi.org/10.1007/s00248-013-0217-0. PMid:23535981.
http://dx.doi.org/10.1007/s00248-013-02...
reported a similar high frequency of CRISPR1-cas (52%) in E. faecalis isolates from pig feces; they have not reported the presence of CRISPR3-cas . We observed for the first time the presence of this gene in animal fecal samples.
Our results demonstrated a low frequency of CRISPRs Type II in E. faecium strains isolated from animal and food samples. A similar result was observed to Palmer and Gilmore (2010) PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
in clinical E. faecium strains. There is only one study which tested CRISPRs in environmental E. faecium from strains, and the authors did not detect any CRISPRs ( Lindenstrauß et al., 2011 LINDENSTRAUß, A.G., PAVLOVIC, M., BRINGMANN, A., BEHR, J., EHRMANN, M.A. and VOGEL, R.F., 2011. Comparison of genotypic and phenotypic cluster analyses of virulence determinants and possible role of CRISPR elements towards their incidence in Enterococcus faecalis and Enterococcus faecium. Systematic and Applied Microbiology, vol. 34, no. 8, pp. 553-560. http://dx.doi.org/10.1016/j.syapm.2011.05.002. PMid:21943678.
http://dx.doi.org/10.1016/j.syapm.2011....
). So, our data is the first report of the presence of CRISPR1-cas and CRISPR2 in E. faecium isolated from animal feces.
For some enterococcal lineages, CRISPR system has been recognized as a prokaryotic self-defense that provides a type of acquired immunity. Palmer and Gilmore (2010) PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
http://dx.doi.org/10.1128/mBio.00227-10...
observed an inverse correlation between CRISPRs presence and antibiotic resistance. In this study, we also observed that strains which contain less antibiotic resistant genes tended to have more cas genes (data not show). In previous studies from our group evaluating the same strains, was observed in food strains a large number of antibiotic resistance genes (data not published), and in animals strains, mainly wild animals, a low frequency of antibiotic resistance genes ( Santestevan et al., 2015 SANTESTEVAN, N.A., ZVOBODA, D.A., PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., MOURA, T.M., MEDEIROS, A.W., AMORIN, D.B., TAVARES, M., D’AZEVEDO, P.A., FRANCO, A.C., FRAZZON, J. and FRAZZON, A.P., 2015. Antimicrobial Resistance and Virulence Factor Gene Profiles of Enterococcus Spp. Isolates From Wild Arctocephalus Australis (South American Fur Seal) and Arctocephalus Tropicalis (Subantarctic Fur Seal). World Journal of Microbiology & Biotechnology , vol. 31, no. 12, pp. 1935-1946. http://dx.doi.org/10.1007/s11274-015-1938-7. PMid:26347323.
http://dx.doi.org/10.1007/s11274-015-19...
; Prichula et al., 2016 PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., TOLFO, N.C., SANTESTEVAN, N.A., MEDEIROS, A.W., TAVARES, M., FRAZZON, J., D’AZEVEDO, P.A. and FRAZZON, A.P., 2016. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Marine Pollution Bulletin , vol. 105, no. 1, pp. 51-57. http://dx.doi.org/10.1016/j.marpolbul.2016.02.071. PMid:26952995.
http://dx.doi.org/10.1016/j.marpolbul.2...
).
In the conclusion, we detected different proportions and distributions of CRISPRs in E. faecalis and E. faecium isolates from food and fecal samples. The differences in the habitats of enterococcal species influenced this result. The CRISPRs genes were obtained more frequently between E. faecalis strains isolated from fecal samples of animals, highlighting to wild marine animals fecal strains. These strains isolated from wild animals are theoretically under an extremely small selective pressure when compared to strains isolated from food and clinical samples.
Acknowledgements
We thank the government agencies Conselho Nacional de Desenvolvimento Científico e Tecnológico do Brasil (CNPq - #444335/2014-5, #300912/2012-9, and #303251/2014-0) and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) of the Brazilian government. We are grateful to Grupo de Estudos de Mamíferos Aquáticos do Rio Grande do Sul and all staff from Coastal, Limnological and Marine Studies (CECLIMAR) da Universidade Federal do Rio Grande do Sul by help in the data collection.
-
(With 1 figure)
References
- BURLEY, K.M. and SEDGLEY, C.M., 2012. CRISPR-Cas, a prokaryotic adaptive immune system, in endodontic, oral, and multidrug-resistant hospital-acquired Enterococcus faecalis. Journal of Endodontics, vol. 38, no. 11, pp. 1511-1515. http://dx.doi.org/10.1016/j.joen.2012.07.004. PMid:23063226.
» http://dx.doi.org/10.1016/j.joen.2012.07.004 - BYAPPANAHALLI, M.N., NEVERS, M.B., KORAJKIC, A., STALEY, Z.R. and HARWOOD, V.J., 2012. Enterococci in the environment. Microbiology and Molecular Biology Reviews, vol. 76, no. 4, pp. 685-706. http://dx.doi.org/10.1128/MMBR.00023-12. PMid:23204362.
» http://dx.doi.org/10.1128/MMBR.00023-12 - CASSENEGO, A.P.V., ELLWANGERI, J., D’AZEVEDO, P., FRAZZON, J. and FRAZZON, A.P.G., 2013. Virulence and biofilm formation by Enterococcus faecalis isolates from cloacal swabs of broilers infected with Eimeria spp. Pesquisa Veterinária Brasileira, vol. 33, no. 12, pp. 1433-1440. http://dx.doi.org/10.1590/S0100-736X2013001200007.
» http://dx.doi.org/10.1590/S0100-736X2013001200007 - CHENG, S., MCCLESKEY, F.K., GRESS, M.J., PETROZIELLO, J.M., LIU, R., NAMDARI, H., BENINGA, K., SALMEN, A. and DELVECCHIO, V.G., 1997. A PCR assay for identification of Enterococcus faecium. Journal of Clinical Microbiology, vol. 35, no. 5, pp. 1248-1250. PMid:9114416.
- DEPARDIEU, F., PERICHON, B. and COURVALIN, P., 2004. Detection of the van alphabet and identification of enterococci and staphylococci at the species level by multiplex PCR. Journal of Clinical Microbiology, vol. 42, no. 12, pp. 5857-5860. http://dx.doi.org/10.1128/JCM.42.12.5857-5860.2004. PMid:15583325.
» http://dx.doi.org/10.1128/JCM.42.12.5857-5860.2004 - DONATO, S.T., 2007 [viewed 8 July 2017]. Comparison of conventional and semi-automatized methods to identify Enterococcus spp versus molecular biology in discrepant identifications [online]. Fortaleza: Universidade Federal de Fortaleza, 88 p. Dissertação de Mestrado em Medicina Clinica. Available from: http://www.repositorio.ufc.br/handle/riufc/1764
» http://www.repositorio.ufc.br/handle/riufc/1764 - FRAZZON, A.P.G., GAMA, B.A., HERMES, V., BIERHALS, C.G., PEREIRA, R.I., GUEDES, A.G., D’AZEVEDO, P.A. and FRAZZON, J., 2009. Prevalence of antimicrobial resistance and molecular characterization of tetracycline resistance mediated by tet(M) and tet (L) genes in Enterococcus spp. isolated from food in Southern Brazil. World Journal of Microbiology & Biotechnology, vol. 26, no. 2, pp. 365-370. http://dx.doi.org/10.1007/s11274-009-0160-x.
» http://dx.doi.org/10.1007/s11274-009-0160-x - HAFT, D.H., SELENGUT, J., MONGODIN, E.F. and NELSON, K.E., 2005. A guild of 45 CRISPR-associated (Cas) protein families and multiple CRISPR/Cas subtypes exist in prokaryotic genomes. PLoS Computational Biology, vol. 1, no. 6, pp. e60. http://dx.doi.org/10.1371/journal.pcbi.0010060. PMid:16292354.
» http://dx.doi.org/10.1371/journal.pcbi.0010060 - HORVATH, P., ROMERO, D.A., COÛTÉ-MONVOISIN, A.C., RICHARDS, M., DEVEAU, H., MOINEAU, S., BOYAVAL, P., FREMAUX, C. and BARRANGOU, R., 2008. Diversity, activity, and evolution of CRISPR loci in Streptococcus thermophilus. Journal of Bacteriology, vol. 190, no. 4, pp. 1401-1412. http://dx.doi.org/10.1128/JB.01415-07. PMid:18065539.
» http://dx.doi.org/10.1128/JB.01415-07 - HULLAHALLI, K., RODRIGUES, M., SCHMIDT, B.D., LI, X., BHARDWAJ, P. and PALMER, K.L., 2015. Comparative analysis of the orphan CRISPR2 Locus in 242 Enterococcus faecalis strains. PLoS One, vol. 10, no. 9, pp. e0138890. http://dx.doi.org/10.1371/journal.pone.0138890. PMid:26398194.
» http://dx.doi.org/10.1371/journal.pone.0138890 - KATYAL, I., CHABAN, B., NG, B. and HILL, J.E., 2013. CRISPRs of Enterococcus faecalis and E. hirae isolates from pig feces have species-specific repeats but share some common spacer sequences. Microbial Ecology, vol. 66, no. 1, pp. 182-188. http://dx.doi.org/10.1007/s00248-013-0217-0. PMid:23535981.
» http://dx.doi.org/10.1007/s00248-013-0217-0 - KE, D., PICARD, F.J., MARTINEAU, F., MENARD, C., ROY, P.H., OUELLETTE, M. and BERGERON, M.G., 1999. Development of a PCR assay for rapid detection of Enterococci. Journal of Clinical Microbiology, vol. 37, no. 11, pp. 3497-3503. PMid:10523541.
- LEBRETON, F., WILLEMS, R.J.L. and GILMORE, M.S., 2014. Enterococcus diversity, origins in nature, and gut colonization. In: M.S. Gilmore, D.B. Clewell, Y. Ike and N. Shankar, eds. Enterococci: from commensals to leading causes of drug resistant infection . Boston: Massachusetts Eye and Ear, pp. 1-59. PMid:24649510.
- LINDENSTRAUß, A.G., PAVLOVIC, M., BRINGMANN, A., BEHR, J., EHRMANN, M.A. and VOGEL, R.F., 2011. Comparison of genotypic and phenotypic cluster analyses of virulence determinants and possible role of CRISPR elements towards their incidence in Enterococcus faecalis and Enterococcus faecium. Systematic and Applied Microbiology, vol. 34, no. 8, pp. 553-560. http://dx.doi.org/10.1016/j.syapm.2011.05.002. PMid:21943678.
» http://dx.doi.org/10.1016/j.syapm.2011.05.002 - LYONS, C., RAUSTAD, N., BUSTOS, M.A. and SHIARIS, M., 2015. Incidence of Type II CRISPR1-Cas systems in Enterococcus is species-dependent. PLoS One , vol. 10, no. 11, pp. e0143544. http://dx.doi.org/10.1371/journal.pone.0143544. PMid:26600384.
» http://dx.doi.org/10.1371/journal.pone.0143544 - MAGADÁN, A.H., DUPUIS, M.È., VILLION, M. and MOINEAU, S., 2012. Cleavage of Phage DNA by the Streptococcus thermophilus CRISPR3-Cas System. PLoS One, vol. 7, no. 7, pp. e40913. http://dx.doi.org/10.1371/journal.pone.0040913. PMid:22911717.
» http://dx.doi.org/10.1371/journal.pone.0040913 - MAKAROVA, K., SLESAREV, A., WOLF, Y., SOROKIN, A., MIRKIN, B., KOONIN, E., PAVLOV, A., PAVLOVA, N., KARAMYCHEV, V., POLOUCHINE, N., SHAKHOVA, V., GRIGORIEV, I., LOU, Y., ROHKSAR, D., LUCAS, S., HUANG, K., GOODSTEIN, D.M., HAWKINS, T., PLENGVIDHYA, V., WELKER, D., HUGHES, J., GOH, Y., BENSON, A., BALDWIN, K., LEE, J.-H., DIAZ-MUNIZ, I., DOSTI, B., SMEIANOV, V., WECHTER, W., BARABOTE, R., LORCA, G., ALTERMANN, E., BARRANGOU, R., GANESAN, B., XIE, Y., RAWSTHORNE, H., TAMIR, D., PARKER, C., BREIDT, F., BROADBENT, J., HUTKINS, R., O’SULLIVAN, D., STEELE, J., UNLU, G., SAIER, M., KLAENHAMMER, T., RICHARDSON, P., KOZYAVKIN, S., WEIMER, B. and MILLS, D., 2006. Comparative genomics of the lactic acid bacteria. Proceedings of the National Academy of Sciences of the United States of America, vol. 103, no. 42, pp. 15611-15616. http://dx.doi.org/10.1073/pnas.0607117103. PMid:17030793.
» http://dx.doi.org/10.1073/pnas.0607117103 - MAKAROVA, K.S., HAFT, H.D., BARRANGOU, R., BROUNS, S.J., CHARPENTIER, E., HORVATH, P., MOINEAU, S., MOJICA, F.J., WOLF, Y.I., YAKUNIN, A.F., VAN DER OOST, J. and KOONIN, E.V., 2011. Evolution and classification of the CRISPR-Cas systems. Nature Reviews Microbiology , vol. 9, no. 6, pp. 467-477. http://dx.doi.org/10.1038/nrmicro2577. PMid:21552286.
» http://dx.doi.org/10.1038/nrmicro2577 - MEDEIROS, A.W., AMORIM, D.B., TAVARES, M., MOURA, T.M., FRANCO, A.C., D’AZEVEDO, P.A., FRAZZON, J. and FRAZZON, A.P., 2017. Enterococcus species diversity in fecal samples of wild marine species as determined by Real-Time PCR. Canadian Journal of Microbiology, vol. 63, no. 2, pp. 129-136. http://dx.doi.org/10.1139/cjm-2016-0427. PMid:27991828.
» http://dx.doi.org/10.1139/cjm-2016-0427 - PALMER, K.L. and GILMORE, M.S., 2010. Multidrug-resistant enterococci lack CRISPR-cas. mBio, vol. 1, no. 4, pp. e00227-10. http://dx.doi.org/10.1128/mBio.00227-10. PMid:21060735.
» http://dx.doi.org/10.1128/mBio.00227-10 - PIENIZ, S., ANDREAZZA, R., OKEKE, B.C., CAMARGO, F.A. and BRANDELLI, A., 2015. Antimicrobial and antioxidant activities of Enterococcus species isolated from meat and dairy products. Brazilian Journal of Biology = Revista Brasileira de Biologia , vol. 75, no. 4, pp. 923-931. http://dx.doi.org/10.1590/1519-6984.02814. PMid:26675908.
» http://dx.doi.org/10.1590/1519-6984.02814 - PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., TOLFO, N.C., SANTESTEVAN, N.A., MEDEIROS, A.W., TAVARES, M., FRAZZON, J., D’AZEVEDO, P.A. and FRAZZON, A.P., 2016. Resistance to antimicrobial agents among enterococci isolated from fecal samples of wild marine species in the southern coast of Brazil. Marine Pollution Bulletin , vol. 105, no. 1, pp. 51-57. http://dx.doi.org/10.1016/j.marpolbul.2016.02.071. PMid:26952995.
» http://dx.doi.org/10.1016/j.marpolbul.2016.02.071 - PRICHULA, J., ZVOBODA, D.A., PEREIRA, R.I., SANTESTEVAN, N.A., MEDEIROS, A.W., MOTTA, A.S., D’AZEVEDO, P.A., GIORDANI, A.R. and FRAZZON, A.P.G., 2013. Antimicrobial susceptibility profile and diversity of enterococci species isolated from raw milk of buffalo in South Brazil. Revista Brasileira de Ciência Veterinária, vol. 20, no. 2, pp. 104-109. http://dx.doi.org/10.4322/rbcv.2014.056.
» http://dx.doi.org/10.4322/rbcv.2014.056 - RIBOLDI, G.P., FRAZZON, J., D’ AZEVEDO, P.A. and FRAZZON, A.P.G., 2009. Antimicrobial resistance profile of Enterococcus spp isolated from food in Southern Brazil. Brazilian Journal of Microbiology, vol. 40, no. 1, pp. 125-128. http://dx.doi.org/10.1590/S1517-83822009000100021. PMid:24031330.
» http://dx.doi.org/10.1590/S1517-83822009000100021 - RIBOLDI, G.P., MATTOS, E.P., FRAZZON, A.P.G., D’AZEVEDO, P.A. and FRAZZON, J., 2008. Phenotypic and genotypic heterogeneity of Enterococcus species isolated from food in Southern Brazil. Journal of Basic Microbiology, vol. 48, no. 1, pp. 31-37. http://dx.doi.org/10.1002/jobm.200700226. PMid:18247393.
» http://dx.doi.org/10.1002/jobm.200700226 - SANTESTEVAN, N.A., ZVOBODA, D.A., PRICHULA, J., PEREIRA, R.I., WACHHOLZ, G.R., CARDOSO, L.A., MOURA, T.M., MEDEIROS, A.W., AMORIN, D.B., TAVARES, M., D’AZEVEDO, P.A., FRANCO, A.C., FRAZZON, J. and FRAZZON, A.P., 2015. Antimicrobial Resistance and Virulence Factor Gene Profiles of Enterococcus Spp. Isolates From Wild Arctocephalus Australis (South American Fur Seal) and Arctocephalus Tropicalis (Subantarctic Fur Seal). World Journal of Microbiology & Biotechnology , vol. 31, no. 12, pp. 1935-1946. http://dx.doi.org/10.1007/s11274-015-1938-7. PMid:26347323.
» http://dx.doi.org/10.1007/s11274-015-1938-7 - SOREK, R., KUNIN, V. and HUGENHOLTZ, P., 2008. CRISPR-- a widespread system that provides acquired resistance against phages in bacteria and archaea. Nature Reviews Microbiology , vol. 6, no. 3, pp. 181-186. http://dx.doi.org/10.1038/nrmicro1793. PMid:18157154.
» http://dx.doi.org/10.1038/nrmicro1793 - THOMAS, C.M. and NIELSEN, K.M., 2005. Mechanisms of, and barriers to, horizontal gene transfer between bacteria. Nature Reviews. Microbiology, vol. 3, no. 9, pp. 711-721. http://dx.doi.org/10.1038/nrmicro1234. PMid:16138099.
» http://dx.doi.org/10.1038/nrmicro1234 - VAN TYNE, D. and GILMORE, M.S., 2014. Friend turned foe: evolution of enterococcal virulence and antibiotic resistance. Annual Review of Microbiology, vol. 68, no. 1, pp. 337-356. http://dx.doi.org/10.1146/annurev-micro-091213-113003. PMid:25002090.
» http://dx.doi.org/10.1146/annurev-micro-091213-113003 - WIEDENHEFT, B., STERNBERG, S.H. and DOUDNA, J.A., 2012. RNA-guided genetic silencing systems in bacteria and archaea. Nature, vol. 482, no. 7385, pp. 331-338. http://dx.doi.org/10.1038/nature10886. PMid:22337052.
» http://dx.doi.org/10.1038/nature10886
Publication Dates
-
Publication in this collection
04 Oct 2018 -
Date of issue
Jul-Sep 2019
History
-
Received
08 July 2017 -
Accepted
16 Jan 2018