Abstracts
In this study, habitat use of the armored catfishes Hypostomus ancistroides and Hypostomus sp. was investigated in 21 streams from the Upper Rio Paraná system in São Paulo State, Brazil, a pasture dominated region. The abundance of Hypostomus sp. was positively correlated with the amount of riffles and riparian vegetation condition, whereas H. ancistroides abundance was negatively correlated with riparian vegetation condition. Thirteen ecomorphological attributes analyzed by principal component analysis showed that when compared to H. ancistroides, Hypostomus sp. presented a wider mouth and more flattened body, suggesting an adapted body to faster waters. The most abundant populations of Hypostomus sp. were predominantly associated with good riffle availability and good riparian quality, suggesting that populational features of this species may be incorporated in the stream biotic integrity assessment in that region.
armored catfishes; upper Rio Paraná; ecomorphology; diet; habitat partitioning
Neste estudo investigamos o uso do hábitat dos cascudos Hypostomus ancistroides e Hypostomus sp. em 21 riachos localizados em uma paisagem dominada por pastagens no sistema do Alto Rio Paraná no Estado de São Paulo, Brasil. A abundância de Hypostomus sp. foi positivamente correlacionada com a extensão de corredeiras e condição da vegetação ripária enquanto que a abundância de H. ancistroides foi negativamente correlacionada com a condição da vegetação ripária. Treze atributos ecomorfológicos, processados em uma análise de componentes principais, mostraram que quando comparada com H. ancistroides, Hypostomus sp. possui abertura bucal mais ampla e corpo mais achatado, sugerindo uma forma de corpo adaptada a ambientes correntosos. Populações mais abundantes de Hypostomus sp. estiveram predominantemente associadas a riachos com boa disponibilidade de corredeiras e boa qualidade ripária, sugerindo que atributos populacionais dessa espécie possam ser posteriormente incorporados à avaliação da integridade biótica dos riachos de nossa região.
cascudos; Alto Rio Paraná; ecomorfologia; dieta; partilha de hábitat
ARTICLES
Habitat use by two species of Hypostomus (Pisces, Loricariidae) in southeastern brazilian streams
Lilian Casatti; Fabíola C. Rocha; Danusa C. Pereira
UNESP - Universidade Estadual Paulista, Laboratório de Ictiologia, Departamento de Zoologia e Botânica, Rua Cristóvão Colombo, 2265, 15054-000 São José do Rio Preto, SP, Brazil
ABSTRACT
In this study, habitat use of the armored catfishes Hypostomus ancistroides and Hypostomus sp. was investigated in 21 streams from the Upper Rio Paraná system in São Paulo State, Brazil, a pasture dominated region. The abundance of Hypostomus sp. was positively correlated with the amount of riffles and riparian vegetation condition, whereas H. ancistroides abundance was negatively correlated with riparian vegetation condition. Thirteen ecomorphological attributes analyzed by principal component analysis showed that when compared to H. ancistroides, Hypostomus sp. presented a wider mouth and more flattened body, suggesting an adapted body to faster waters. The most abundant populations of Hypostomus sp. were predominantly associated with good riffle availability and good riparian quality, suggesting that populational features of this species may be incorporated in the stream biotic integrity assessment in that region.
Key words: armored catfishes, upper Rio Paraná, ecomorphology, diet, habitat partitioning
RESUMO
Neste estudo investigamos o uso do hábitat dos cascudos Hypostomus ancistroides e Hypostomus sp. em 21 riachos localizados em uma paisagem dominada por pastagens no sistema do Alto Rio Paraná no Estado de São Paulo, Brasil. A abundância de Hypostomus sp. foi positivamente correlacionada com a extensão de corredeiras e condição da vegetação ripária enquanto que a abundância de H. ancistroides foi negativamente correlacionada com a condição da vegetação ripária. Treze atributos ecomorfológicos, processados em uma análise de componentes principais, mostraram que quando comparada com H. ancistroides, Hypostomus sp. possui abertura bucal mais ampla e corpo mais achatado, sugerindo uma forma de corpo adaptada a ambientes correntosos. Populações mais abundantes de Hypostomus sp. estiveram predominantemente associadas a riachos com boa disponibilidade de corredeiras e boa qualidade ripária, sugerindo que atributos populacionais dessa espécie possam ser posteriormente incorporados à avaliação da integridade biótica dos riachos de nossa região.
Palavras-chave: cascudos, Alto Rio Paraná, ecomorfologia, dieta, partilha de hábitat
Introduction
As part of aquatic ecosystems with well defined physical features, streams are useful models for developing ecological concepts related to distribution, abundance, and coexistence of organisms, as well as for evaluating the influence of physical disturbances affecting the biota (Esteves & Aranha 1999). In stream fish communities, resource partitioning is one of the most important ecological aspects, because it allows the comprehension of the inter-relations between species, showing the main resource dimensions in which species segregate (Ross 1986).
In small streams, many fishes present broad tolerance to habitat types and relative flexibility in their feeding behaviour, sharing many resources with other species (Lowe-McConnell 1987). Resource partitioning can also be suggested by ecomorphological models, which predict that one species is correspondent to its morphological mosaic (Miller 1984) and, therefore, species with similar morphological patterns could be able to use similar resources, maximizing resource partitioning to avoid competition.
With the rapid and widespread decline and the endangerment of drainages, there is an urgent need to define fish habitat requirements to elaborate adequate species management and future habitat restoration (Rosenfeld 2003), especially in those environments seriously threatened by anthropogenic pressures such as the northeastern portion of the São Paulo State in Brazil, where pasture represents more than 70% of the land use. In a recent survey on that area, we registered that the armored catfishes Hypostomus sp. and H. ancistroides (Fig. 1) show a non congruent pattern of distribution among streams, suggesting habitat partitioning. Aiming to investigate the habitat use for both species, we correlated their abundances with stream habitat features. Because habitat occupation is notably correlated to fish morphology and feeding, we also analyzed these two niche dimensions.
Material and methods
Study sites and sampling
The study area is located at the northwestern portion of São Paulo State, southeastern Brazil, and encompasses the Rio São José dos Dourados system, which covers 6,805 km2 (IPT 2000). Climate presents a wet season from October to March (January and February are the wettest months, with 53.7% of the annual rainfall), and a dry season from April to September (Barcha & Arid 1971). The maximum mean temperature is recorded in January (32°C) and the minimal in July (13°C) (IPT 2000). From the original vegetation in that river system (Semi-Deciduous forests and Savannah), only 3.3% remains, a percentage that is very altered, fragmented, and unconnected (SMA / IF 2005).
Sites (Table 1, Fig. 2) were randomly selected in a 1:50,000 map base, aiming to cover the entire extension of the São José dos Dourados system. Four habitat descriptors (amount of riffles, mean depth, large wood debris, and riparian vegetation condition, see Table 1) for each site were taken. The amount of riffles for each stream reach was visually estimated as (1) absent, (2) present (occurring in less than 50% of the stream reach), (3) extensive (occurring in more than 50% of the stream reach). Mean depth was evaluated by bathymetry in all mesohabitat. Large wood debris were estimated as (1) absent , (2) present (occurring only in isolated pools), (3) extensive (occurring in the majority of pools). Riparian vegetation condition was scored (from 0 to 20) according to previous published physical habitat protocols (Barbour et al. 1999, Roth et al. 2001), considering both width and integrity of riparian vegetation.
Fishes were collected during a dry season by electrofishing (modified from Mazzoni et al. 2000 and Castro et al. 2003), and fixed in 10% formalin solution. Later, they were preserved in a 70% EtOH solution and deposited in the collection of the "Departamento de Zoologia e Botânica, Universidade Estadual Paulista", São José do Rio Preto, São Paulo State, Brazil (DZSJRP 5844, 5853, 5865, 5879, 5888, 5911, 5932, 5948, 5949, 5967, 5979, 5997, 5998, 6007, 6007, 6016, 6036, 6037, 6048, 6055, 6071, 6083, 6105, 6106, 6118, 6136, 6174). Additional voucher specimens are deposited in the "Laboratório de Ictiologia de Ribeirão Preto, Universidade de São Paulo", Ribeirão Preto, São Paulo State, Brazil (LIRP 2716, 2672).
Analysis
Niche width for mesohabitat was calculated through the standardized Levin's measure (BA) (Krebs 1999), using proportional abundance for each species in each category of riffle availability (absent, present, extensive). To establish correlations between abiotic descriptors and species abundance in the sites where at least one species were collected (n=21), data set normality was initially tested and, afterwards, Spearman rank correlation coefficients were calculated (Zar 1999).
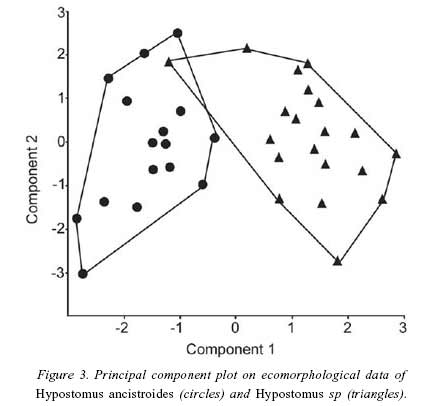
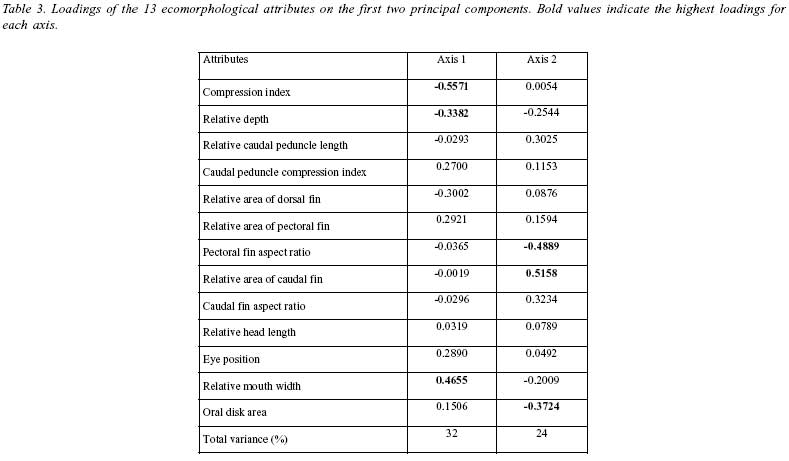
To investigate species segregation in the morphological space, 13 attributes were calculated for 18 individuals of each species belonging to similar size classes. Nine attributes were selected following previous ecomorphological studies (Mahon 1984, Watson & Balon 1984, Balon et al. 1986, Winemiller 1991, Beaumord & Petrere 1994): compression index, relative depth, relative caudal peduncle length, caudal peduncle compression index, relative area of dorsal fin, relative area of pectoral fin, pectoral fin aspect ratio, relative area of caudal fin, caudal fin aspect ratio, relative head length, eye position, and relative mouth width. An additional attribute, oral disc area, calculated by its projection on millimetric paper, was included herein, because this structure helps armored catfishes to remain attached to substrates (Buck & Sazima 1995, Casatti & Castro 1998), and larger areas are hypothesized to help the fish to hold position in fast flowing waters. Measurements were made with digital caliper (0.1 mm) and fin areas were calculated by their projection on millimetric paper (Beaumord & Petrere 1994). Multivariate analysis of morphological data was conducted by principal component analysis using a log-transformed data and a correlation matrix according to Valentin (1995).
Stomach contents were examined from eight adult individuals with similar size of each species collected in the only two streams (10 and 13) where such conditions were found. The anterior 1 cm of each digestive tube was removed, the contents were divided in two microscope slides which were directly examined in optical microscope. The identification was conducted with the help of specialized literature (Bicudo & Bicudo 1970) and revised by a specialist. For each item the frequency of occurrence (Bowen 1992) was calculated.
Results
Hypostomus ancistroides (n=168) was more abundant than Hypostomus sp. (n=89) and occurred in 20 localities whereas Hypostomus sp. was registered in seven streams (Table 1). Abundance of Hypostomus sp. was positively correlated to the amount of riffles and riparian vegetation condition, whereas H. ancistroides abundance was correlated with riparian vegetation condition (Table 2). Niche width was 0.15 for Hypostomus sp. and 0.92 for H. ancistroides, indicating a higher specialization in using riffle areas for the former species.
The first two axis of the PCA explained 56% of the total variance. PCA plot showed complete segregation between two species, notably in the first component (Fig. 3). When compared to H. ancistroides, Hypostomus sp. presents more flattened bodies and wider mouths (Table 3).
Cyanobacteria, chlorophytes, diatoms, and vegetal debris were the most frequent items (75% of occurrence) in the stomach contents (Table 4), but no significative differences in the diet composition were detected (U=28, p = 0.67).
Discussion
Several hypostomins are known for a closer association with fast flowing environments where they display a bottom-dwelling behavior, feeding on attached algae (Garavello & Garavello 2004). Although Hypostomus ancistroides is considered a typical stream species (Castro & Casatti 1997), the occupation of faster mesohabitats seems not a pattern for it in this study. In our samples, this species was more abundant in slower streams, in contrast to Hypostomus sp., more abundant in faster waters. Similar segregation between H. ancistroides and a non-determined Hypostomus was firstly mentioned by Uieda et al. (1997) in a stream from the Alto Rio Paraná system, where the authors observed H. ancistroides foraging in pools reaches and Hypostomus sp. in riffles. Concordantly, in a pristine first order stream H. ancistroides was abundant in the inferior reaches where pools predominated, in contrast to H. nigromaculatus, abundant in the upper reaches dominated by riffles (Casatti 2005).
When cogenerics occur in the same stream, mesohabitats patches must shape the abundance of each species and such differential occupation can also be suggested by external morphology, suggesting that Hypostomus sp. has a body shape more adapted to fast flowing environments. In this species, a wider mouth can enhance grazing in faster waters and a more flattened body indicates a better capacity to maintain the position on the bottom (Mahon 1984).
Distinct mesohabitats dwelling could suggest distinct diet composition; we observed, however, high diet similarity between both species when collected in the same streams. This similarity indicates that periphyton is probably an abundant resource in the studied sites, and that partitioning, especially spatial, of feeding resources could be shaping the coexistence of both species. In Central America streams, Power (1984a) observed that periphyton grew faster in sunny pools than in dark pools, that the density of loricariids corresponded to algae productivity and a positive correlation between annual density of loricariids and canopy opening. None of the studied streams have dense canopies and periphyton must not be a limiting factor in these environments. Such availability of periphyton in streams was already suggested by Uieda et al. (1997) in southeastern Brazil.
Hypostomins are active mostly after sunset; during the day they remain under rocks or submerged logs (Weber 2003), but such a pattern is not a rule and it may vary according to body size (Power 1984b, Buck & Sazima 1995). For the species studied here, this pattern was not checked because the high water turbidity in the sampled streams restricted the use of underwater techniques for direct observation. Temporal segregation, however, was observed for another pair of Hypostomus species in a pristine stream, where H. nigromaculatus was active after the middle of the day up to the nightfall and H. ancistroides started foraging after nightfall, scraping periphyton off submerged large wood debris (Casatti 2002).
The main source of large wood debris for instream habitats is the riparian vegetation, influencing not only habitat features but also providing substrate for periphyton and aquatic invertebrates (Angermeier & Karr 1984). In the studied streams riparian vegetation do not play this role, because most localities are deforested and, even when riparian vegetation is present, its condition is poor (i.e., predominance of juvenile trees, bamboos, and arbustive vegetation). In pristine conditions, in contrast, woody fragments are abundant, being the preferable microhabitat, where H. ancistroides forages (Casatti 2002). In the absence of large wood debris, H. ancistroides - a more generalist species in respect to habitat occupation - forages in more diverse substrates, thus, probably representing a species which is less affected by structural habitat degradation, remarkably siltation enhancement that often is a consequence of riparian vegetation removal (Gregory et al. 1991, Rabeni & Smale 1995, Wichert & Rapport 1998). This assumption is reinforced by the negative correlation between riparian vegetation condition and H. ancistroides abundance. In contrast, expressive populations of Hypostomus sp. (15-20% of the ichthyocenosis abundance) were predominantly associated to streams with good riffle availability and good riparian quality, suggesting that populational features of this species may be incorporated in stream biotic integrity regional assessments.
Acknowledgements
We thank José Luiz Veronezi Jr., Daniel R. Lacerda, Renato M. Romero, Fernando R. Carvalho, Vanessa X. L. Andrade, Fábio F. S. Campos, Roselene S. C. Ferreira, Henrique F. Chaves, and Jane P. Serra for helping us during field work. Luis H. Zanini Branco assisted with algae determination. Ricardo M. C. Castro assisted with the figures 1A-B. Francisco Langeani assisted with fish identification and comments on the manuscript; Oscar A. Shibatta, anonymous reviewers, and Fernando B. Noll gave useful suggestions on various drafts of the manuscript; "Departamento de Zoologia e Botânica IBILCE-UNESP" for facilities use; and several landowners for work permissions. This study was made possible by funding from the BIOTA/FAPESP "O Instituto Virtual da Biodiversidade" (www.biota.org.br) from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP)" to the grant "Assessment of the biotic integrity in the northwestern streams of the São Paulo State using fish assemblages" (FAPESP grant 01/13340-7). The Instituto Brasileiro de Meio Ambiente e Recursos Naturais Renováveis (IBAMA) provided permission (authorization nº 01/2003) to collect and transport the fishes of this paper. LC and FCR receive grants from FAPESP (02/05996-2, 03/02753-4, 05/00929-3).
Date Received 05/04/2005
Revised 09/27/2005
Accepted 10/10/2005
ISSN 1676-0611
- ANGERMEIER, P.L. & KARR, J.R. 1984. Relationships between woody debris and fish habitat in a small warmwater stream. Trans. Am. Fish. Soc. 113:716-726.
- BALON, E.K., CRAWFORD, S.S. & LELEK, A. 1986. Fish communities of the upper Danube River (Germany, Austria) prior to the new Rhein-Main-Donau connection. Env. Biol. Fishes 154:242-271.
- BARBOUR, M.T., GERRITSEN, J., SNYDER, B.D. & STRIBLING, J.B. 1999. Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish. Second edition. EPA 841-B-99-002. U. S. Environmental Protection Agency, Office of Water, Washington, D.C.
- BARCHA, S.F. & ARID, F.M. 1971. Estudos da evapotranspiração na região norte-ocidental do Estado de São Paulo. Rev. Ciên. Votuporanga 1:99-122.
- BEAUMORD, A.C. & PETRERE Jr., M. 1994. Fish communities of Manso River, Chapada dos Guimarães, MT, Brazil. Acta Bio. Venez. 152:21-35.
- BICUDO, C.E.M. & BICUDO, R.M.T. 1970. Algas de águas continentais brasileiras: chave ilustrada para identificação de gêneros. Fundação Brasileira para o Desenvolvimento do Ensino das Ciências, São Paulo.
- BOWEN, S.H. 1992. Quantitative description of the diet. In Fisheries techniques (L.A. Nielsen & D.L. Johnson, eds.). American Fisheries Society, Bethesda, p.325-336.
- BUCK, S. & SAZIMA, I. 1995. An assemblage of mailed catfishes (Loricariidae) in southeastern Brazil: distribution, activity, and feeding. Ichthyol. Explor. Freshwaters 6:325-332.
- CASATTI, L. 2002. Alimentação dos peixes em um riacho do Parque Estadual Morro do Diabo, bacia do Alto Rio Paraná, sudeste do Brasil. Biota Neotrop. 2(2) http://www.biotaneotropica.org.br/v2n2/pt/abstract?article+BN02502022002
- CASATTI, L. 2005. Fish assemblage structure in a first order stream, southeastern Brazil: longitudinal distribution, seasonality, and microhabitat diversity. Biota Neotrop. 5(1) http://www.biotaneotropica.org.br/v5n1/pt/abstract?article+BN02505012005
- CASATTI, L. & CASTRO, R.M.C. 1998. A fish community of the São Francisco River headwaters riffles, southeastern Brazil. Ichthyol. Explor. Freshwaters 9:229-242.
- CASTRO, R.M.C. & CASATTI, L. 1997. The fish fauna from a small forest stream of the upper Paraná River Basin, southeastern Brazil. Ichthyol. Explor. Freshwaters 7:337-352.
- CASTRO, R.M.C., CASATTI, L., SANTOS, H.F., FERREIRA, K.M., RIBEIRO, A.C., BENINE, R.C., DARDIS, G.Z.P., MELO, A.L.A., STOPIGLIA, R., ABREU, T.X., BOCKMANN, F.A., CARVALHO, M., GIBRAN, F.Z. & LIMA, F.C.T. 2003. Estrutura e composição da ictiofauna de riachos do Rio Paranapanema, sudeste do Brasil. Biota Neotrop. 3(1) http://www.biotaneotropica.org.br/v3n1/pt/abstract?article+BN01703012003
- ESTEVES, K.E. & ARANHA, J.M.R. 1999. Ecologia trófica de peixes de riachos. In Ecologia de Peixes de Riachos (E.P. Caramaschi, R. Mazzoni & P.R. Peres-Neto, eds.). Série Oecologia Brasiliensis, Programa de Pós-Graduação em Ecologia, Instituto de Biologia-UFRJ, Rio de Janeiro, p.157-182.
- GARAVELLO, J.C. & GARAVELLO, J.P. 2004. Spatial distribution and interaction of four species of the catfish genus Hypostomus Lacépède with bottom of Rio São Francisco, Canindé do São Francisco, Sergipe, Brazil (Pisces, Loricariidae, Hypostominae). Braz. J. Biol. 64B:591-598.
- GREGORY, S.V., SWANSON, F.J., MCKEE, W.A. & CUMMINS, K.W. 1991. An ecosystem perspective of riparian zones. BioScience 41:540-551.
- IPT (Instituto de Pesquisas Tecnológicas do Estado de São Paulo). 2000. Diagnóstico da situação atual dos recursos hídricos e estabelecimento de diretrizes técnicas para a elaboração do Plano da Bacia Hidrográfica do São José dos Dourados. Comitê da Bacia Hidrográfica do São José dos Dourados e Fundo Estadual de Recursos Hídricos, São Paulo.
- KREBS, C.J. 1999. Ecological methodology. Addison Wesley Longman, New York.
- LOWE-McCONNELL, R.H. 1987. Ecological studies in tropical fish communities. Cambridge University Press, Cambridge.
- MAHON, R. 1984. Divergent structure in fish taxocenes of north temperate stream. Can. J. Fish. Aquat. Sci. 41:330-350.
- MAZZONI, R., FENERICH-VERANI, N. & CARAMASCHI, E.P. 2000. Electrofishing as a sampling technique for coastal stream fish populations and communities in the Southeast of Brazil. Braz. J. Biol. 60:205-216.
- MILLER, G.L. 1984. Seasonal changes in morphological structuring in a guild of benthic stream fishes. Oecologia 63:106-109.
- POWER, M. 1984a. Habitat quality and the distribution of algae-grazing catfish in a Panamanian stream. J. An. Ecol. 53:357-374.
- POWER, M. 1984b. Depth distributions of armored catfish: predator-induced resource avoidance. Ecology 65:523-528.
- RABENI, C.F. & SMALE, M.A. 1995. Effects on siltation on stream fishes and the potential mitigating role of the buffering riparian zone. Hydrobiol. 303:211-219.
- ROSENFELD, J. 2003. Assessing the habitat requirements of stream fishes: an overview and evaluation of different approaches. Trans. Am. Fish. Soc. 132:953-968.
- ROSS, S.T. 1986. Resource partitioning in fish assemblages: a review of field studies. Copeia 1986:352-388.
- ROTH, N.E., SOUTHERLAND, M.T., MERCURIO, G. & VOLSTAD, J.H. 2001. Maryland biological stream survey 2000-2004: Ecological assessment of watersheds sampled in 2000. Prepared by Versar Inc. for the Maryland Department of Natural Resources, Monitoring and Non-Tidal Assessment Division, Annapolis, Maryland, CNWP-MANTA-EA-01-5.
- SMA / IF (Secretaria do Meio Ambiente / Instituto Florestal). 2005. Inventário florestal da vegetação natural do Estado de São Paulo. Imprensa Oficial, São Paulo.
- UIEDA, V.S., BUZZATO, P.& KIKUCHI, R.M. 1997. Partilha de recursos alimentares em peixes em um riacho de serra no sudeste do Brasil. An. Acad. Bras. Cienc. 69:243-252.
- VALENTIN, J.L. 1995. Agrupamento e ordenação. In Tópicos em tratamento de dados biológicos (P.R. Peres-Neto, J.L. Valentin & F.A.S. Fernandez, eds.). Oecologia Brasiliensis, Programa de Pós-Graduação em Ecologia, Instituto de Biologia-UFRJ, Rio de Janeiro, p.27-55.
- WATSON , D.J. & BALON, E.K. 1984. Ecomorphological analysis of fish taxocenes in rainforest streams of northern Borneo. J. Fish Biol. 25:371-384.
- WEBER, C. 2003. Subfamily Hypostominae. In Check list of the freshwater fishes of South and Central America (R.E. Reis, S.O. Kullander & C.J. Ferraris Jr., orgs.). Editora da Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre, p.351-372.
- WICHERT, G.A. & RAPPORT, D.J. 1998. Fish community structure as a measure of degradation and rehabilitation of riparian systems in an agricultural drainage basin. Environ. Manag. 22:425-443.
- WINEMILLER, K.O. 1991. Ecomorphological diversification in lowland freshwater fish assemblages from five biotic regions. Ecol. Monogr. 614:343-365.
- ZAR, J.H. 1999. Biostatistical analysis. Prentice Hall, New Jersey.
Publication Dates
-
Publication in this collection
23 Oct 2006 -
Date of issue
2005
History
-
Accepted
10 Oct 2005 -
Received
05 Apr 2005