Abstracts
The genus Oecomys Thomas, 1906 is currently composed of 16 species with unclear taxonomy and poorly known geographic limits. O. catherinae Thomas, 1909 is known to occur within the Brazilian Atlantic Forest from the states of Santa Catarina to Pernambuco (where the northernmost previously known specimen of Oecomys in the Atlantic forest was recorded), and along riverine forest into the Cerrado. To gain a greater understanding of its geographical and ecological distribution (mainly in Northeastern Brazil) and of its taxonomic characterization, we provide a short review of karyotypical and morphometrical data from specimens collected within the distribution range of the species. Specimens presented 2n = 60 and AN varying between 62 and 64. A table with external and cranial measurements of the analyzed specimens is provided. In this paper we also report the presence of O. catherinae in the semi-deciduous forests of the state of Paraíba, representing the northernmost records of the species in the Atlantic forest and thereby extending its known geographical limits.
small mammals; new register; Sigmodontinae; cytogenetic; morphometrics
Oecomys Thomas, 1906 é um gênero atualmente composto por 16 espécies reconhecidas que apresentam taxonomia e distribuições geográficas ainda incertas. O. catherinae Thomas, 1906 é a espécie que ocorre ao longo da Floresta Atlântica brasileira dos estados de Santa Catarina a Pernambuco, onde se encontra o registro prévio mais ao norte para Oecomys na Floresta Atlântica, e ao longo de florestas de galeria no Cerrado. Pretendendo esclarecer aspectos relacionados à distribuição geográfica e ecológica de O. catherinae, com ênfase na região nordeste do Brasil, e auxiliar na sua caracterização taxonômica, fornecemos um breve resumo de dados morfométricos e cariotípicos de indivíduos coletados em diversos trabalhos ao longo da área de ocorrência da espécie. Os espécimens apresentaram 2n = 60 e NA variando entre 62 e 64. Uma tabela de medidas corporais externas e cranianas dos indivíduos analisados é fornecida. Também registramos pela primeira vez a presença de O. catherinae nas florestas semi-deciduais do estado da Paraíba, sendo atualmente os registros mais ao norte da espécie na Floresta Atlântica, estendendo os limites geográficos conhecidos para a espécie.
pequenos mamíferos; novo registro; Sigmodontinae; citogenética; morfometria
SHORT COMMUNICATIONS
Distribution of Oecomys catherinae Thomas, 1909 (Rodentia: Cricetidae) in northeastern Brazil with karyotypical and morphometrical notes
Distribuição de Oecomys catherinae Thomas, 1909 (Rodentia: Cricetidae) no nordeste do Brasil com notas morfométricas e cariotípicas
Paulo Henrique AsforaI, * * Corresponding author e-mail: Paulo Henrique Asfora, E-mail: paulo.asfora@gmail.com ; Alexandre Ramlo Torre PalmaII; Diego AstúaIII; Lena GeiseI
IDepartamento de Zoologia, Laboratório de Mastozoologia, Universidade do Estado do Rio de Janeiro - UERJ, Rua São Francisco Xavier 524, CEP 20550-013, Maracanã, Rio de Janeiro, RJ, Brazil
IIDepartamento de Sistemática e Ecologia, Universidade Federal da Paraíba - UFPB,Castelo Branco, João Pessoa, PB, Brazil
IIIDepartamento de Zoologia, Laboratório de Mastozoologia, Universidade Federal de Pernambuco - UFPE, Av. Prof. Moraes Rego s/n, CEP 50670-420, Cidade Universitária, Recife, PE, Brazil
ABSTRACT
The genus Oecomys Thomas, 1906 is currently composed of 16 species with unclear taxonomy and poorly known geographic limits. O. catherinae Thomas, 1909 is known to occur within the Brazilian Atlantic Forest from the states of Santa Catarina to Pernambuco (where the northernmost previously known specimen of Oecomys in the Atlantic forest was recorded), and along riverine forest into the Cerrado. To gain a greater understanding of its geographical and ecological distribution (mainly in Northeastern Brazil) and of its taxonomic characterization, we provide a short review of karyotypical and morphometrical data from specimens collected within the distribution range of the species. Specimens presented 2n = 60 and AN varying between 62 and 64. A table with external and cranial measurements of the analyzed specimens is provided. In this paper we also report the presence of O. catherinae in the semi-deciduous forests of the state of Paraíba, representing the northernmost records of the species in the Atlantic forest and thereby extending its known geographical limits.
Keywords: small mammals, new register, Sigmodontinae, cytogenetic, morphometrics.
RESUMO
Oecomys Thomas, 1906 é um gênero atualmente composto por 16 espécies reconhecidas que apresentam taxonomia e distribuições geográficas ainda incertas. O. catherinae Thomas, 1906 é a espécie que ocorre ao longo da Floresta Atlântica brasileira dos estados de Santa Catarina a Pernambuco, onde se encontra o registro prévio mais ao norte para Oecomys na Floresta Atlântica, e ao longo de florestas de galeria no Cerrado. Pretendendo esclarecer aspectos relacionados à distribuição geográfica e ecológica de O. catherinae, com ênfase na região nordeste do Brasil, e auxiliar na sua caracterização taxonômica, fornecemos um breve resumo de dados morfométricos e cariotípicos de indivíduos coletados em diversos trabalhos ao longo da área de ocorrência da espécie. Os espécimens apresentaram 2n = 60 e NA variando entre 62 e 64. Uma tabela de medidas corporais externas e cranianas dos indivíduos analisados é fornecida. Também registramos pela primeira vez a presença de O. catherinae nas florestas semi-deciduais do estado da Paraíba, sendo atualmente os registros mais ao norte da espécie na Floresta Atlântica, estendendo os limites geográficos conhecidos para a espécie.
Palavras-chave: pequenos mamíferos, novo registro, Sigmodontinae, citogenética, morfometria.
Introduction
The genus Oecomys Thomas, 1906 is currently composed of 16 species that present an unclear taxonomy and geographic distribution (Musser & Carleton 2005), 12 of which occur in Brazil: O. auyentepui Tate, 1939, O. bicolor (Tomes, 1860), O. catherinae Thomas, 1909, O. cleberi Locks, 1981, O. concolor (Wagner, 1845), O. marmorae (Thomas, 1906), O. paricola (Thomas, 1904), O. rex Thomas, 1910, O. roberti (Thomas, 1904), O. rutilus Anthony, 1921, O. superans Thomas, 1911 and O. trinitatis (J. A. Allen and Chapman, 1893) (see Bonvicino et al. 2008).
Hershkovitz (1960) considered all known Oecomys taxa as junior synonymous of two species, O. concolor (the large-bodied species) or O. bicolor (the small-bodied species). Some papers then refer to Oecomys specimens recorded in the Brazilian Atlantic Forest and Cerrado as O. concolor or O. group concolor (Andrades-Miranda et al. 2001, Andrade & Bonvicino 2003). Langguth et al. (2005) state that the specimens of Oecomys from the state of Pernambuco differ from O. concolor and refer to O. bahiensis (Hershkovitz, 1960) and could be co-specific with the animals sharing the same karyotype recorded further south in the Atlantic Forest, a diploid number (2n) of 60 and an autosomal number (AN) of 62, still called O. concolor in Langguth et al. (2005). Musser & Carleton (2005) treat O. bahiensis as a junior synonym of O. catherinae. So, for the purpose of this paper we considered the specimens of O. bahiensis and the specimens of Oecomys group concolor having a diploid number (2n) of 60 and an autosomal number (AN) of 62 as a synonym of O. catherinae
Cytogenetic analyses performed on several species of Oecomys suggest that this genus presents a karyotypical diversification with a diploid number ranging from 58 to 86, yet some species within this genus present the same diploid number, being differentiated by their autosomal number (see Langguth et al. 2005), (Table 1, for a comprehensive review). Thus, karyotypes can be helpful in revealing the genetic diversity in the genus (Langguth et al. 2005).
According to Musser & Carleton (2005), O. catherinae occurs in the Brazilian Atlantic Forest, from the states of Santa Catarina to Bahia and along riverine forest into the Cerrado and Caatinga regions, with poorly documented limits. Bonvicino et al. (2008) state that O. catherinae occurs from the states of Santa Catarina to Paraíba, citing Costa et al. (2004) - a paper not listed in their references section. In fact, Bonvicino et al. (2008) were referring to Costa et al. (2008), which states that O. catherinae occurs from the states of Santa Catarina to Bahia, and there are no records of the species in Paraíba (C.R. Bonvicino, personal communication). No other reference to the occurrence of O. catherinae in Paraíba state was found in the literature or in the collections of the Museu Nacional, Universidade Federal do Rio de Janeiro (MN), Museu de Zoologia da USP (MZUSP), Universidade Federal de Pernambuco (UFPE) or the Universidade Federal da Paraíba (UFPB) nor in any collection integrating the Species Link (http://www.splink.org.br) or MaNIS network (http://manisnet.org).
Therefore, the northernmost previously known recorded specimen of Oecomys in the Atlantic Forest was collected at Camaragibe, 6 km NE of São Lourenço da Mata (08° 00' 00" S 35° 03' 00" O), in a remnant of the Atlantic Forest of Pernambuco (Oliveira & Langguth 2004, Langguth et al. 2005).
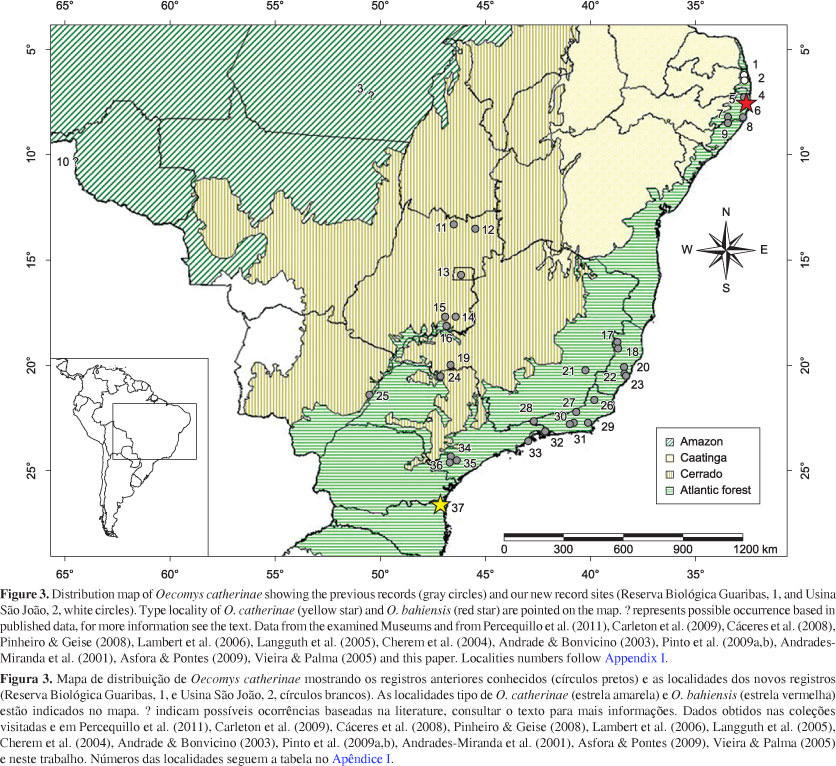
We report here the presence of Oecomys catherinae (Figure 1) in two different localities in the state of Paraíba, Brazil, extending its known distribution in the northeastern Atlantic Forest further north and adding new ecological data about the species. We also present a short review of distributional, morphometrical, and karyological data, and give details on chromosomal variation in the autosomal number from individuals collected across the species distribution range.
Material and Methods
Trapping was carried out at two localities in the state of Paraíba. The first locality is Reserva Biológica Guaribas (06° 44' 59" S, 41° 07' 11" W), within the municipalities of Mamanguape and Rio Tinto. This reserve encompasses three non-contiguous areas: SEMA I (3016 ha), SEMA II (673 ha) and SEMA III (338 ha). The average annual rainfall is 1386 mm/year, with a dry season lasting from September to February (CPTEC/INPE 2009) and average annual temperature 25.5 °C (Hijmans et al. 2005). The vegetation of Reserva Biológica Guaribas is a mosaic of semi-deciduous forests located in valleys, and savannas with sandy soils on the tops of hills (" tabuleiros" vegetation) (Instituto..., 1993, Rodrigues and Silva et al. 2000). The second locality is Usina São João (07° 06' 56" S, 35° 04' 47" W), in the municipality of Cruz do Espírito Santo, with almost 700 ha of forest fragments. The average annual rainfall is 1294 mm/year (Instituto..., 2009) and average annual temperature is 25.0 °C (Hijmans et al. 2005). The vegetation at Usina São João is also a mosaic of savanna and semi-deciduous forest (Instituto..., 1993), similar to Reserva Biológica Guaribas. Both sites ar e included in the Pernambuco Interior Forests ecoregion of the Brazilian Atlantic Forest (Olson et al. 2001).
Three trapping trips in Reserva Biológica Guaribas were carried out, in March 2008, September 2008 and May 2009, lasting 11, 8 and 5 consecutive nights, respectively, totaling 2,978 trap-nights. Two semi-deciduous forest sites and one savanna (Tabuleiro) site were sampled on each field trip. Two trapping trips were carried out in Usina São João, in January and October 2009, lasting 5 consecutive nights each, totaling 1,757 trap-nights. In this locality, one trap line was set in a semi-deciduous forest area (Figure 2), one in the Tabuleiro Forest (low forest on sandy soil), and a third at a Tabuleiro dense savanna site.
Live-traps (Sherman© of three different sizes: height: 10/7.5/10 cm, width: 8/9/11 cm, depth: 30.5/23/38 cm and Tomahawk© height: 15 cm, width: 14 cm, depth: 41 cm) were distributed in points along linear transects formed by 30 trap stations, each 10 m apart from the next. At each station two traps were set; one on the ground and another fixed on tree branches about 2 m above the ground. Traps were baited with a mix of pineapple, banana, sardines, peanut butter and corn flower and were rebaited every other day.
All trapped animals were sexed, measured, weighed, and karyotyped in the field. Specimens were identified to species level by comparison of morphological characters (skin and skull) with descriptions in the literature (Langguth et al. 2005, Bonvicino et al. 2008) and karyological analysis (Andrade & Bonvicino 2003, Langguth et al. 2005). Voucher specimens from the state of Paraíba were collected under IBAMA/ICMBio licenses #11633-2 and # 14734-1, and are currently housed at the Coleção de Mamíferos da Universidade Federal de Pernambuco.
Chromosomes in metaphases from our collected individuals were obtained with in vitro culture (culture of bone marrow grown in Dulbecco´s MEM with 10% fetal bovine serum and colchicine). Conventional coloration with Giemsa 5% was used to count diploid (2n) and autosomal numbers (AN, excluding sexual chromosomes) and to observe variation in chromosome morphology. This analysis was carried out using an optic photomicroscope (Hund Wetzlar - H500 LL HP100). We also summarize karyotipycal data of O. catherinae recorded across the species distribution found in the literature and from our unpublished data to present a review of the actual karyotypical data of the species.
For morphometric analysis we examined specimens of O. catherinae currently housed in MN, UFPE and UFPB as well as individuals collected by us in many localities (Appendix I) during this and previous trips. Following Geise et al. (2008), 20 cranial measurements were obtained with a digital caliper: condylo-incisive length (CIL), breadth of occipital condyles (BOC), length of diastema (LD), length of palatal bridge (LPB), length (LIF) and breadth (BIF) of incisive foramina, length of the maxillary molar row (LM), breadth of M1 (BM1), breadth between the first molars, including both teeth (M1M1), bullar length (BL), height of skull (HS), length of the rostrum (LR), rostrum width (RW), least interorbital length (LIB), internal orbital length (IOL), zygomatic breadth (ZB), breadth of the braincase (BB), breadth of the zygomatic plate (BZP), mandible height (MH) and length (ML). Additionally, head-and-body length (HBL), tail length (TL), Hindfoot length (HF), Ear length (E) and body weight (W) were also analyzed. We performed descriptive statistic analyses of the external and cranial measurements of these specimens including a t-test to detect sexual dimorphism. Age classes were determined according to molar teeth wear (Cerqueira et al. 1989); only adult specimens were included in statistical analyses.
Results and Discussion
We collected 46 individuals of six marsupial species - Caluromys philander Linnaeus, 1758 (n = 2), Didelphis albiventris Lund, 1840 (n = 5), Marmosa murina Linnaeus, 1758 (n = 9), Micoureus demerarae Thomas, 1905 (n = 10), Monodelphis domestica Wagner, 1842 (N = 2) and four rodent species - Akodon cursor Winge, 1887 (n = 12), Necromys lasiurus Lund, 1840 (n = 1), Oecomys catherinae (n = 4) and Oligoryzomys sp. (n = 1).
Two specimens of O. catherinae were trapped in SEMA III, in Reserva Biológica Guaribas, a forest fragment that includes well-preserved late secondary semi-deciduous forest in a natural regeneration process of nearly 60 years (unpublished data). The first specimen was a young male (age class 2) trapped in March 2008 (museum number UFPE1890) and the second specimen was an adult male (museum number UFPE 1889, age class 3) trapped in September 2008. Another two specimens were trapped in October 2009 in Mata do Açude (semi-deciduous forest trap-line), in Usina São João (Figure 2); a male (museum number UFPE1896) and a female (museum number UFPE1897), both adults (age class 4). All were trapped in Sherman® traps set on tree branches at about 2 m off the ground.
These records show the occurrence of O. catherinae in the Atlantic Forest of the state of Paraíba, Brazil, representing the northernmost occurrence of the species and extending its distribution one degree of latitude and 122 km north of the previously known northernmost locality (Langguth et al. 2005). These records also represent an extension of the known ecological limits in northeastern Brazil beyond the coastal rainforests of the state of Pernambuco, in the Pernambuco Coastal Forests ecoregion, into the semi-deciduous forests of the state of Paraíba, part of the Pernambuco Interior Forests ecoregion (Olson et al. 2001) (see Table 1). These new records also place the distributional limits of O. catherinae very close to the northernmost limit of the Brazilian Atlantic Forest, just 110 km further north near the city of Natal (Instituto..., 1993). The records at Guaribas also lie very close (9 km) to the border between the Atlantic Forest and the semi-arid Caatinga shrublands (Instituto..., 1993) (Figure 3).
With these new records we suggest that the distribution limits of O. catherinae north from São Francisco river actually reach still further northward and further inland than previously recorded and may closely follow the limits of the Atlantic Forest. No O. catherinae specimens were recorded in the few studies including the " brejos úmidos" (Oliveira & Langguth, 2004; Souza et al. 2004), which are forest enclaves on mesic ridges amidst semi-arid Caatinga.
In the Brazilian Atlantic Forest we found individuals identified as O. catherinae occurring from Joinville, in the state of Santa Catarina, the type locality (Cherem et al. 2004, Carleton et al. 2009), to the state of Espírito Santo (Pinto et al. 2009a, b). However, although Musser & Carleton (2005) and Costa et al. (2008) state that O. catherinae occurs from the states of Santa Catarina to Bahia, the only register of Oecomys collected in the state of Bahia was an individual identified as Oecomys sp. from Una (Pardini 2004). In the states of Pernambuco and Alagoas we found records of individuals identified as O. bahiensis (Langguth et al. 2005, Asfora & Pontes 2009), synonymized as O. catherinae (Musser & Carleton 2005). This apparently disjunct distribution in the Atlantic Forest could be an artifact of insufficient sampling effort in the northeastern region of Brazil, or alternately might suggest that O. bahiensis could be a valid species differing from O. catherinae.
We found several putative records of O. catherinae (or O. concolor presenting 2n = 60 and AN = 62) in the gallery forests in Cerrado biome from the states of Goiás and Minas Gerais and Distrito Federal (Andrades-Miranda 2001, Andrade & Bonvicino 2003, Langguth et al. 2005, Vieira & Palma 2005 and references therein). However we did not examine these specimens and this classification is based only at the karyotypes or the identifications published. No record of Oecomys of any species was found for the Caatinga biome (Freitas et al. 2005, Oliveira et al. 2003).
There is a published record of O. catherinae from Pinkaití Research Station, Ourilândia do Norte, in the southeast of the state of Pará, in a region of the Amazon forest consisting of approximately 70% forest and 30% Cerrado (Lambert et al. 2006). However these identifications were just hypotheses made by Robert Voss (R. Voss, personal communication). Another possible record of O. catherinae in the Amazon biome is from Andrades-Miranda et al. (2001), who found O. concolor presenting 2n = 60, AN = 62, in Guajará-Mirim, in the state of Rondônia. If confirmed these records show another disjunct population of O. catherinae in the north of Brazil and extend the species occurrence to the southern limits of the Amazon forest. Another hypothesis states that more than one species may share the same chromosomal complement, which would make necessary a detailed revision, since we have not observed all specimens or the type of O. catherinae.
External and cranial measurements of the individuals from the state of Paraíba and of all examined specimens are shown in Table 2 and 3. No sexual dimorphism was found in cranial measurements (Table 2) and the only external measurement differing between the sexes was " Ear Length" with a p- = 0.024. Based on their work with O. sydandersoni in Bolivia, Carleton et al. (2009) state that none of the 18 craniodental variables demonstrated significant secondary sexual dimorphism.
The karyotype of the specimens from the state of Paraiba showed a 2n = 60 and an AN = 62 (Figure 4b), matching the already described karyotype for the species (Langguth et al. 2005). Nine karyotyped individuals, from a total of 14, recorded in Cachoeiras de Macacu, Guapimirim and Sumidouro in the Atlantic Forest of Rio de Janeiro state, presented a polymorphism in the autosomal number, with AN = 64 (Table 4). These individuals presented the autosomal complement composed by one pair of large subtelocentric chromosomes, two pairs of small metacentric chromosomes and 26 pairs of large to small acrocentric chromosomes (Figure 4a). The difference between this latter karyotype and the 2n = 60, AN = 62, is the presence of a third small metacentric pair (Figure 4a). Pinheiro & Geise (2008) also reported the karyotype 2n = 60 and AN = 64 from Picinguaba, Ubatuba, in the state of São Paulo. We re-analyzed these karyotypes from Pinheiro & Geise (2008) and confirmed that they showed the same polymorphism as the individuals from the state of Rio de Janeiro.
These data suggest that this polymorphism could be shared by the populations occurring from Cachoeiras de Macacu and Sumidouro, in the state of Rio de Janeiro, to Ubatuba, in the state of São Paulo, inherited from a common ancestor. And it probably could also occur in individuals of O. catherinae collected in the intermediary area of Itatiaia and Mangaratiba, in the state of Rio de Janeiro, (Figure 4a).
Our work confirms the lack of information about the taxonomic status and geographic distribution of the Oecomys catherinae. We found many uncertain records based mainly in old data of O. concolor, O. aff. concolor or O. group concolor. It shows an urgent need for a taxonomic revision to confirm the identity of these individuals. We showed three apparently disjunctive populations of O. catherinae in: a) the Northern, b) Northeastern and c) Central and Southeastern regions of Brazil, and a chromosomal polymorphism shared by the meta-populations spread among the states of Rio de Janeiro and São Paulo. These data could be a sampling artifact, or O. catherinae as currently recognized could be composed by more than one species.
ACKNOWLEDGEMENTS
Financial support was provided by CNPq (Proc 480877/2007-6 and 553875/2006-0) and FACEPE (APQ-0351-2.04/06). PHA is supported by a fellowship from FAPERJ, LG received research grants from UERJ (Prociencia), CNPq, and CAPES. We thank Mr. Ivaldo Marques for logistic support, the Reserva Biológica Guaribas team for the field assistance, and the Usina São João owners for granting us permission to work there. We are also thankful to the students of the Reserva Biológica Guaribas First Small Mammals Field Course and A.L.C.P. Nascimento, C.E.C. Anacleto, J.A.F. Silva, A.P.A. Júnior and A. B. Sousa for helping with the fieldwork. J. Bloch made the final English revision, and two anonymous reviewers made several suggestions that improved the text.
Recebido em 03/05/2010
Versão reformulada recebida em 05/02/2011
Publicado em 15/04/2011
Apendix I e II
Apendix I - Click to enlarge
Apendix I - Click to enlarge Apendix I - Click to enlarge
Apendix II. Specimens examined: The 52 specimens analyzed by us are deposited in collections from Museu Nacional, Universidade Federal do Rio de Janeiro (MN) and Universidade Federal de Pernambuco (UFPE). Individuals used in cranial analyses are marked with * and individuals kariotyped by us are marked with #. RB-M, FU, FS and SU = field number of Laboratório de Vertebrados, Universidade Federal do Rio de Janeiro; HGB CFVC = field number of Helena de Godoy Bergallo. ALAGOAS: Ibateguara Loc. 9 (UFPE1666*); MINAS GERAIS: Conceição das Alagoas Loc. 19 (MN68206*), Pirapitinga Loc. 21 (MN72737#, MN72738#); PARAÍBA: Cruz do Espírito Santo Loc. 2 (UFPE1896*#, UFPE1897*#), Mamanguape Loc. 1 (UFPE1889*#, UFPE1890#); PERNAMBUCO: Igarassú Loc. 4 (UFPE1892*#), Jaqueira Loc. 7 (UFPE1893*, UFPE1894*, UFPE1891*#), Paudalho Loc. 5 (UFPE1888*#), Rio Formoso Loc. 8 (UFPB4450); RIO DE JANEIRO: Cachoeiras de Macacu Loc. 30 (MN74364#, MN74370*#, MN74374*, MN74371*#, MN74372*#, MN74373), Cambuci Loc. 26 (RB-M-3-51#, RB-M-3-52#), Casimiro de Abreu Loc. 29 (FU16#, FU17#), Guapimirim Loc. 31 (FS04-77, MN74359*#, FS04-27, MN74360*#, MN74361, MN74362*, MN74365, MN74366*#, MN74367#, MN74363*, MN74368#, MN74369*#, FS13-96*), Itatiaia Loc. 28 (HGBCFVC 04), Mangaratiba Loc. 33 (RB-M-58), Sumidouro Loc. 27 (SU63#, SU86#); SÃO PAULO: Ubatuba Loc. 34 (MN74376*#, MN74380*, MN74375*, MN74377*#, MN74378#, MN74379#, MN74381*)
- ANDRADE, A.F. & BONVICINO, C.R. 2003. A new karyological variant of Oecomys (Rodentia: Sigmodontinae) and its phylogenetic relationship based on molecular data. Genome 46:195-203. PMid:12723035. http://dx.doi.org/10.1139/g02-123
- ANDRADES-MIRANDA, J., OLIVEIRA, L.F.B., ZANCHIN, N.I.T. & MATTEVI, M.S. 2001. Chromosomal description of the rodent genera Oecomys and Nectomys from Brazil. Acta Theriol. 46:269-278. http://dx.doi.org/10.1007/BF03192433
- ASFORA, P.H. & PONTES, A.R.M, M.S. 2009. The small mammals of the highly impacted North-eastern Atlantic Forest of Brazil, Pernambuco Endemism Center. Biota Neotrop. 9(1):031-035. ISSN 1676-0603. http://www.biotaneotropica.org.br/v8n4/en/abstract?article+bn00409012009
- BONVICINO, C. R., OLIVEIRA, J.A. & D'ANDREA, P.S. 2008. Guia dos Roedores do Brasil, com chaves para gêneros baseadas em caracteres externos. Centro Pan-Americano de Febre Aftosa - OPAS/OMS, Rio de Janeiro.
- CACERES, N., CARMIGNOTTO, A.P., FISCHER, E. & SANTOS, C.F. 2008. Mammals from Mato Grosso do Sul, Brazil. Check List 4(3):321-335.
- CARLETON, M.D., EMMONS, L.H. & MUSSER, G.C. 2009. A New Species of the Rodent Genus Oecomys (Cricetidae: Sigmodontinae: Oryzomyini) from Eastern Bolivia, with Emended Definitions of O. concolor (Wagner) and O. mamorae (Thomas). Am. Mus. (3661):1-32. http://dx.doi.org/10.1206/612.1
- CERQUEIRA, R., VIEIRA, M.V. & SALLES, L.O. 1989. Habitat and reproduction of Rhipidomys cearanus at São Benedito, Ceará (Rodentia, Cricetidae). Cienc. Cult. 41(10):1009-1013.
- CHEREM, J.J., SIMÕES-LOPES, P.C., ALTHOFF, S. & GRAIPEL, M.E. 2004. Lista dos mamíferos do Estado de Santa Catarina, Sul Do Brasil. Mastozool. Neotrop. 11(2):151-184.
- COSTA, L., BONVICINO, C., WEKSLER, M. & PAGLIA, A. 2008. Oecomys catherinae In IUCN Red List of Threatened Species. (International Union for Conservation of Nature - IUCN). Version 2010.2. www.iucnredlist.org (último acesso em 15/08/2010).
» link - FREITAS, R.R., ROCHA, P.L.B. & SIMÕES-LOPES, P.C. 2005. Habitat structure and small mammals abundances in one semiarid landscape in the Brazilian Caatinga. Rev. Bras. Zool. 22(1):119-129. http://dx.doi.org/10.1590/S0101-81752005000100015
- GEISE, L., BERGALLO, H.G., ESBERARD, C.E.L., ROCHA, C.F.D. & SLUYS, M.V. 2008. The karyotype of Blarinomys breviceps (Mammalia: Rodentia: Cricetidae) with comments on its morphology and some ecological notes. Zootaxa (Online) 1907:47-60.
- HERSHKOVITZ, P. 1960. Mammals of Northern Colombia, Preliminary no.8: arboreal rice rats, a systematic revision of the subgenus Oecomys, genus Oryzomys Proc. U.S. Nat. Mus. 110:535-539.
- HIJMANS, R.J., CAMERON, S.E., PARRA, J.L., JONES, P.G. & JARVIS, A. 2005. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25:1965-1978. http://dx.doi.org/10.1002/joc.1276
- INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA - IBGE. 1993. Mapa de Vegetação do Brasil. IBGE, Brasília.
- INSTITUTO NACIONAL DE PESQUISAS ESPACIAIS - INPE. Centro de Previsões de Tempo e Estudos Climáticos - CPTEC. 2009. Proclima: Programa de monitoramento climático em tempo real da região Nordeste. Balanço hídrico - gráficos/tabela.
- LAMBERT, T.D., MALCOLM, J.R. & ZIMMERMAN, B.L. 2006. Amazonian small mammal abundances in relation to habitat structure and resource abundance. J. Mammal. 87(4):766-776. http://dx.doi.org/10.1644/05-MAMM-A-261R1.1
- LANGGUTH, A., MAIA, V. & MATTEVI, M.S. 2005. Karyology of large size Brazilian species of the genus Oecomys Thomas, 1906 (Rodentia, Muridae, Sigmodontinae). Arq. Mus. Nac. 63(1):183-190.
- MUSSER, G.G. & CARLETON, M.D. 2005. Superfamily Muroidea, Family Cricetidae, Subfamily Sigmodontinae. In Mammal Species of the World (D.E. Wilson & D.M. Reeder, eds). Johns Hopkins University Press, Baltiomre, p. 1086-1186.
- NATIONAL OCEANIC AND ATMOSPHERIC ADMINISTRATION - NOAA & NATIONAL GEOPHYSICAL DATA CENTER - NGDC. 2001. 2-minute Gridded Global Relief Data (ETOPO2). NGDC, Boulde. http://www.ngdc.noaa.gov/mgg/fliers/06mgg01.html (último acesso em 19/04/2010).
- OLIVEIRA, F.F. & LANGGUTH, A. 2004. Pequenos mamíferos (Didelphimorphia e Rodentia) de Paraíba e Pernambuco. Rev. Nordest. Biol. 18:19-86.
- OLIVEIRA, J.A., P.R. GONÇALVES & C.R. BONVICINO. 2003. Mamíferos da Caatinga. In Ecologia e conservação da Caatinga (I.R. Leal, M. Tabarelli & J.M.C. Silva, eds). Editora Universitária da Universidade Federal de Pernambuco, Recife, p. 275-333.
- OLSON, D.M., DINNERSTEIN, E., WIKRAMANAYAKE, D.E., BURGESS, N.D., POWELL, G.V.N., UNDERWOOD, E.C., D'AMICO, J.A., LOUCKS, C.J., RICKETTS, T.H., KURA, Y., LAMOREUX, J.F., WETTENGEL, W.W., HEDAO, P. & KASEEEM, K.R. 2001. Terrestrial ecoregions of the world: A new map of life on Earth. BioScience 51:933-938. http://dx.doi.org/10.1641/0006-3568(2001)051[0933:TEOTWA]2.0.CO;2
- PARDINI, R. 2004. Effects of forest fragmentation on small mammals in an Atlantic Forest landscape. Biodivers. Conserv. 13:2567-2586. http://dx.doi.org/10.1023/B:BIOC.0000048452.18878.2d
- PERCEQUILLO, A.R., WEKSLER, M. & COSTA, L.P. 2011. A new genus and species of rodent from the Brazilian Atlantic Forest (Rodentia: Cricetidae: Sigmodontinae: Oryzomyini), with comments on oryzomyine biogeography. Zool. J. Linn. Soc-Lond. 161:357-390.
- PINHEIRO, P.S. & GEISE, L. 2008. Non-volant mammals of Picinguaba, Ubatuba, state of São Paulo, southeastern Brazil. Bol. Mus. Biol. Mello Leitão 23:51-59.
- PINTO, I.S., BOTELHO, J.R., LEONORA, L.P., LEITE, Y.L.R. &. LINARDI, P.M. 2009a. Siphonaptera Associated with Wild Mammals From the Central Atlantic Forest Biodiversity Corridor in Southeastern Brazil. J. Med. Entomol. 46(5):1146-1151. http://dx.doi.org/10.1603/033.046.0523
- PINTO, I.S., LOSS, A.C.C., FALQUETO, A. & LEITE, Y.L.R. 2009B. Pequenos mamíferos não voadores em fragmentos de Mata Atlântica e áreas agrícolas em Viana, Espírito Santo, Brasil. Biota Neotrop. 9(3):355-360. http://www.biotaneotropica.org.br/v9n3/pt/abstract?inventory+bn03109032009
- RODRIGUES e SILVA, F.B., SANTOS, J.C.P., SOUZA NETO, N.C., SILVA, A.B., RICHÉ, G.R., TONNEAU, J.P., BRITO, L.T.L., CORREIA, R.C., SILVA, F.H.B.B., SILVA, C.P., LEITE, A.P., OLIVEIRA NETO, M.B., PARAHYBA, R.B.V., ARAÚJO FILHO, J.C., CAVALCANTI, A.C., BURGOS, N. & REIS, R.M.G. 2000. Zoneamento Agroecológico do Nordeste: Diagnóstico e Prognóstico. Embrapa Solos e Embrapa Semi-Árido, Recife.
- SOUZA, M.A.N., LANGGUTH, A. & GIMENEZ, E.A. 2004. Mamíferos dos brejos de altitude da Paraíba e Pernambuco. In Brejos de altitude em Pernambuco e Paraíba: história natural, ecologia e conservação (K.C. Porto, J.J.P. Cabral & M. Tabarelli, eds.). Ministério do Meio Ambiente, Brasília, p. 229-254.
- VIEIRA, E.M. & PALMA, A.R.T. 2005. Pequenos mamíferos do Cerrado: distribuição dos gêneros e estrutura das comunidades nos diferentes habitats. In Cerrado: Ecologia, biodiversidade e conservação (A. Scariot, J.C. Sousa-Filho & J.M. Felfili, orgs). Ministério do Meio Ambiente, Brasília, p. 265-282.
Apendix I - Click to enlarge

Publication Dates
-
Publication in this collection
18 Aug 2011 -
Date of issue
June 2011
History
-
Accepted
15 Apr 2011 -
Reviewed
05 Feb 2011 -
Received
03 May 2010