Abstracts
In the Pantanal, the largest continuous floodplain in the world, the diversity and distribution of anuran amphibians vary in and across distinct subregions and distinct habitats occurring along inundation gradients. Permanent and natural aquatic habitats are relatively scarce in the Pantanal, and occurrence of temporary aquatic habitats varies seasonally, depending on rains. We here present results of evaluations of anuran's species richness and abundance in a seasonally flooded area in the northwestern section of the Pantanal (Fazenda Baía de Pedra, Cáceres municipality, state of Mato Grosso, Brazil), comparing values obtained in 10 plots systematically distributed over 5 km² with those obtained in additional aquatic plots in the study area. Data were obtained in five field trips, from February 2008 to March 2009. In addition to the plots, 36 water bodies (20 permanent and 16 temporary) were also sampled for the presence of anurans. In total, 3,983 individuals from 34 anuran species distributed in five families were recorded: Hylidae (14 species), Leptodactylidae (8), Leiuperidae (6), Microhylidae (4), and Bufonidae (2). Local richness represents 77.3% of the anuran diversity already recorded for the Brazilian Pantanal. The number of species recorded exclusively in systematically distributed terrestrial plots and in water bodies was 28 and 32, respectively. Sampling methods used at Fazenda Baía da Pedra were efficient in determining anuran richness, abundance, composition and distribution. Evaluations of anuran richness and abundance by using permanent sampling plots in the Pantanal may benefit from additional sampling sites, particularly permanent and temporary water bodies.
anura; species rarefaction curve; wetlands; temporary ponds; permanent ponds
A distribuição e a diversidade das espécies de anfíbios anuros no Pantanal, maior planície inundável contínua do mundo, variam dentro e entre as distintas subregiões e os distintos hábitats ao longo do gradiente de inundação. Ambientes aquáticos naturais permanentes são relativamente escassos na planície e a presença de ambientes aquáticos temporários é sazonal, altamente dependente da precipitação. No presente trabalho, apresentamos informações sobre a riqueza e a abundância das espécies de anuros em uma localidade na porção noroeste do Pantanal (Fazenda Baía de Pedra, município de Cáceres, Mato Grosso, Brasil) e comparamos os valores obtidos em dez parcelas sistematicamente distribuídas em 5 km² com parcelas aquáticas adicionais na presente área de estudo. Os dados foram obtidos em cinco campanhas, entre fevereiro de 2008 e março de 2009. Além das parcelas sistematicamente distribuídas, 36 corpos d'água (20 permanentes e 16 temporários) foram também amostrados quanto à presença de anuros. Registramos 3.983 indivíduos pertencentes a 34 espécies de anuros distribuídas em cinco famílias: Hylidae (14), Leptodactylidae (8), Leiuperidae (6), Microhylidae (4) e Bufonidae (2). A riqueza local em espécies representa 77,3 % da riqueza de anuros já registrada para o Pantanal brasileiro. A riqueza detectada exclusivamente nas parcelas foi 28 espécies, enquanto nos corpos d'água, 32. Os métodos de amostragem utilizados na Fazenda Baía de Pedra mostraram-se eficientes na determinação da riqueza, abundância, composição e distribuição das espécies de anuros. Estudos dessa natureza, envolvendo o uso de parcelas sistematicamente distribuídas em áreas do Pantanal, podem ser beneficiados pelo emprego de parcelas adicionais de amostragem constituídas por corpos d'água permanentes e temporários.
anura; curva de rarefação de espécies; áreas úmidas; poças temporárias; poças permanentes
ARTICLES
Anuran amphibians' diversity in a northwestern area of the Brazilian Pantanal
Diversidade de anfíbios anuros em uma area na porção noroeste do Pantanal brasileiro
André PansonatoI,* * Corresponding author: André Pansonato, e-mail: andre-pan@hotmail.com ; Tamí MottI,II; Christine StrüssmannI,III
IPrograma de Pós-Graduação em Ecologia e Conservação da Biodiversidade, Instituto de Biociências, Universidade Federal de Mato Grosso UFMT, Av. Fernando Corrêa da Costa, 2367, CEP 78060-900, Cuiabá, MT, Brazil
II Departamento de Biologia e Zoologia, Instituto de Biociências, Universidade Federal de Mato Grosso UFMT, Cuiabá, MT, Brazil
III Departamento de Ciências Básicas e Produção Animal, Faculdade de Agronomia, Medicina Veterinária e Zootecnia, Universidade Federal de Mato Grosso UFMT, Cuiabá, MT, Brazil
ABSTRACT
In the Pantanal, the largest continuous floodplain in the world, the diversity and distribution of anuran amphibians vary in and across distinct subregions and distinct habitats occurring along inundation gradients. Permanent and natural aquatic habitats are relatively scarce in the Pantanal, and occurrence of temporary aquatic habitats varies seasonally, depending on rains. We here present results of evaluations of anuran's species richness and abundance in a seasonally flooded area in the northwestern section of the Pantanal (Fazenda Baía de Pedra, Cáceres municipality, state of Mato Grosso, Brazil), comparing values obtained in 10 plots systematically distributed over 5 km2 with those obtained in additional aquatic plots in the study area. Data were obtained in five field trips, from February 2008 to March 2009. In addition to the plots, 36 water bodies (20 permanent and 16 temporary) were also sampled for the presence of anurans. In total, 3,983 individuals from 34 anuran species distributed in five families were recorded: Hylidae (14 species), Leptodactylidae (8), Leiuperidae (6), Microhylidae (4), and Bufonidae (2). Local richness represents 77.3% of the anuran diversity already recorded for the Brazilian Pantanal. The number of species recorded exclusively in systematically distributed terrestrial plots and in water bodies was 28 and 32, respectively. Sampling methods used at Fazenda Baía da Pedra were efficient in determining anuran richness, abundance, composition and distribution. Evaluations of anuran richness and abundance by using permanent sampling plots in the Pantanal may benefit from additional sampling sites, particularly permanent and temporary water bodies.
Keywords: anura, species rarefaction curve, wetlands, temporary ponds, permanent ponds.
RESUMO
A distribuição e a diversidade das espécies de anfíbios anuros no Pantanal, maior planície inundável contínua do mundo, variam dentro e entre as distintas subregiões e os distintos hábitats ao longo do gradiente de inundação. Ambientes aquáticos naturais permanentes são relativamente escassos na planície e a presença de ambientes aquáticos temporários é sazonal, altamente dependente da precipitação. No presente trabalho, apresentamos informações sobre a riqueza e a abundância das espécies de anuros em uma localidade na porção noroeste do Pantanal (Fazenda Baía de Pedra, município de Cáceres, Mato Grosso, Brasil) e comparamos os valores obtidos em dez parcelas sistematicamente distribuídas em 5 km2 com parcelas aquáticas adicionais na presente área de estudo. Os dados foram obtidos em cinco campanhas, entre fevereiro de 2008 e março de 2009. Além das parcelas sistematicamente distribuídas, 36 corpos d'água (20 permanentes e 16 temporários) foram também amostrados quanto à presença de anuros. Registramos 3.983 indivíduos pertencentes a 34 espécies de anuros distribuídas em cinco famílias: Hylidae (14), Leptodactylidae (8), Leiuperidae (6), Microhylidae (4) e Bufonidae (2). A riqueza local em espécies representa 77,3 % da riqueza de anuros já registrada para o Pantanal brasileiro. A riqueza detectada exclusivamente nas parcelas foi 28 espécies, enquanto nos corpos d'água, 32. Os métodos de amostragem utilizados na Fazenda Baía de Pedra mostraram-se eficientes na determinação da riqueza, abundância, composição e distribuição das espécies de anuros. Estudos dessa natureza, envolvendo o uso de parcelas sistematicamente distribuídas em áreas do Pantanal, podem ser beneficiados pelo emprego de parcelas adicionais de amostragem constituídas por corpos d'água permanentes e temporários.
Palavras-chave: anura, curva de rarefação de espécies, áreas úmidas, poças temporárias, poças permanentes.
Introduction
In the Neotropics, distribution patterns of anuran amphibians may be linked to topographic characteristics, climate, vegetation, as well as to historical factors (Duellman 1999). On a regional scale, anuran amphibians may exhibit distribution patterns dependent mostly on ecological factors, such as altitudinal gradient (Navas 2006), nutrient availability (McDiarmid 1994, Bastazini et al. 2007), and availability of water as rainfall (Duellman 1995), water bodies (Rodrigues et al. 2010), or rivers (Parris & McCarthy 1999).
Diversity patterns and ecological processes in Pantanal, the largest continuous floodplain in the world, are regulated by alternate and recurrent yearly cycles of floods and droughts (Junk et al. 1989, Hamilton et al. 1996, Junk & Wantzen 2004). Species distribution and diversity vary across and within distinct habitats comprising the inundation gradient. These habitats include permanently dry, seasonally flooded, and permanent aquatic areas (Cabido et al. 1996, Nunes da Cunha & Junk 2001, 2010, Silva et al. 2001).
Brazil harbors the highest anuran diversity in the world, with 847 species listed to date (Sociedade... 2011). At least 74 of these have been found in the Brazilian portion of the Upper Paraguay River Basin (UPRB), which includes the Pantanal (45 anuran species recorded) and surrounding plateaus (Strüssmann et al. 2007). Nevertheless, published information on taxonomic composition of anuran faunas inhabiting distinct Pantanal subregions is still scarce (e.g., Strüssmann 2000, 2001, Ávila & Ferreira 2004, Prado et al. 2005, Wang et al. 2005).
Because biodiversity measures are strongly scale-dependent, data collected at different geographic scales cannot be compared. To conduct biodiversity surveys in a systematic and comparable basis among distinct ecological regions, a standardized sampling procedure had been designed and is now being implemented on a large scale in Brazil by the Biodiversity Research Program (PPBio; see Magnusson et al. 2005). The method consists of sampling 30 plots (250 m each, along the same elevational isocline), uniformly distributed in a grid covering 25 km2. A less expensive approach is to sample smaller subunits ("modules", according Magnusson et al. 2005), composed of only 10 plots uniformly distributed in two, paired, 5 km trails located 1 km apart.
When installed randomly in the landscape, grids or modules may occasionally neither cover adequately all habitat diversity, nor sample habitats used by a particular taxonomic group. This constraint can be overcome by the inclusion of additional plots in habitats of interest. By including riparian plots in PPBio grids in Brazilian Amazonia, Rojas-Ahumada & Menin (2010) and Condrati (2009) obtained better estimates of local anuran richness.
The goals of this study were to: 1) assess species richness and abundance of anuran amphibians in a seasonally flooded area in the northern Pantanal (Fazenda Baía de Pedra, Cáceres municipality, state of Mato Grosso, Brazil), and 2) compare anuran richness and abundance in 10 plots systematically distributed over 5 km2 and in additional aquatic plots in the study area.
Material and Methods
1. Study area
A field study was conducted at Fazenda Baía de Pedra (16º27'59" S and 58º09'09" W, Figure 1), located in the northern Pantanal, in the municipality of Cáceres, Mato Grosso State, Brazil. Climate in the Pantanal floodplain is generally hot and wet ("Aw" in the classification proposed by Köppen 1931 apud Nimer 1979). There are two, well-marked seasons in the year: a dry season, from May to September, and a rainy season, from October to April (Nunes da Cunha & Junk 2004, Junk et al. 2006). Annual rainfalls range from 800 to 1,400 mm, with 80% concentrated from November to March. Mean temperature is 25.8 ºC (Almeida 1998).
Annual hydrological changes in the Pantanal are characterized by four sequential phases: inundation, steady flood, receding, and drought (Prado et al. 1994). Apart from the annual flood cycle, multi-year periods of intense flood and/or severe drought may occur on an irregular basis (Nunes da Cunha & Junk 2001). In fact, our study was conducted during an abnormally dry year, during which floods were negligible due to the low rainfall from May 2008 to March 2009 (Instituto... 2009).
2. Data collection and analysis
Five field expeditions (15 to 20 days each) were made, in February, June, October and December 2008, and February/March 2009. Field data were gathered mainly in plots systematically distributed over 5 km2, following a model developed by the Brazilian government through the Biodiversity Research Program (PPBio; see Magnusson et al. 2005).
In every expedition, 10 plots (250 × 1 m, along the same elevational isocline) were sampled. From the second expedition on, we additionally sampled 36 natural water bodies, located up to 2 km from the area of the permanent sampling module. Twenty of these bodies were permanent ponds, and 16 were temporary ones ("intermittent aquatic systems", according Nunes da Cunha & Junk 2010). These latter bodies were sampled only in the last two field expeditions, after the onset of the rainy season.
Anuran fauna was sampled by using visual searches, bioacoustic records, and captures with pitfall and funnel traps combined with drift fences. Visual and acoustic searches were made from 6:00 PM to 1:00 AM by two observers walking simultaneously along each systematically distributed plot or around the margins of the additionally sampled water bodies.
At the end of each sampling plot, a set of pitfall traps and funnel traps combined with drift fences (see Cechin & Martins 2000) was installed. Originally employed to sample small gymnophthalmid lizards, our pitfalls traps consisted of four 4-l plastic buckets buried in the ground, 10 m apart in a straight line, and connected by a drift fence made of plastic mesh (75 cm of height). A pair of funnel traps was installed on the ground, on each side of the fence, between the first two buckets. Each funnel trap consisted of a cylinder of plastic mesh, 100 cm long, with a funnel (external diameter 96 cm, internal diameter 25 cm) on each extremity (Figure 2).
Traps were visited daily and all anurans trapped were caught and identified. Except for some voucher specimens (collected under permit IBAMA/SISBIO 16.723-1), all other animals were subsequently released nearly 40 m away from traps. Voucher specimens were euthanized, processed using standard procedures (Calleffo 2002), and subsequently incorporated in the Coleção Zoológica de Vertebrados from the Universidade Federal de Mato Grosso (UFMT, Appendix 1 Appendix 1 ). The species list presented herein also includes taxa recorded during occasional encounters.
The sampling effort employed in active searches (both visual and acoustic) is herein expressed as observer-hours (sum of hours of sampling conducted by each observer). Capture effort for traps is given as the number of container-days (total number of sampling days × total number of containers [buckets + funnels traps]). Sampling effort totaled 409 observer-hours of active searches and 3,520 container-days, during 71 non-consecutive trapping days. Capture rates for each distinct method were obtained by dividing the total number of individuals recorded by the sampling effort.
To estimate local richness based on the number of sampling points (plots plus water bodies), and on the number of sampling days, species rarefaction curves were generated by using first-order Jackknife and Mao (Tau Sobs) estimators, both with 1,000 randomizations. These analyses were conducted using the software EstimateS 7.5.2 (Colwell 2005).
Results and Discussion
We recorded a total of 3,983 individual anuran amphibians representing 34 species, 15 genera, and five families (Table 1). Hylidae and Leptodactylidae were the most abundant families (40 and 23% of the specimens recorded, respectively), following the general pattern observed in other Neotropical sites (e.g., Duellman 1999, Navas 2006) including the Pantanal floodplain and surrounding elevated plateaus (Gordo & Campos 2003, Strüssmann 2000, 2001, Ávila & Ferreira 2004, Prado et al. 2005, Wang et al. 2005, Valério-Brun & Strüssmann 2010). Five species individually represented more than 5% of the total number of specimens recorded throughout the study, and together summed almost 50% of the total abundance: Pseudopaludicola cf. mystacalis, Physalaemus albonotatus, Leptodactylus fuscus, Dendropsophus nanus, and Hypsiboas raniceps. The 29 remaining species individually showed relative abundances of less than 5% (Table 1).
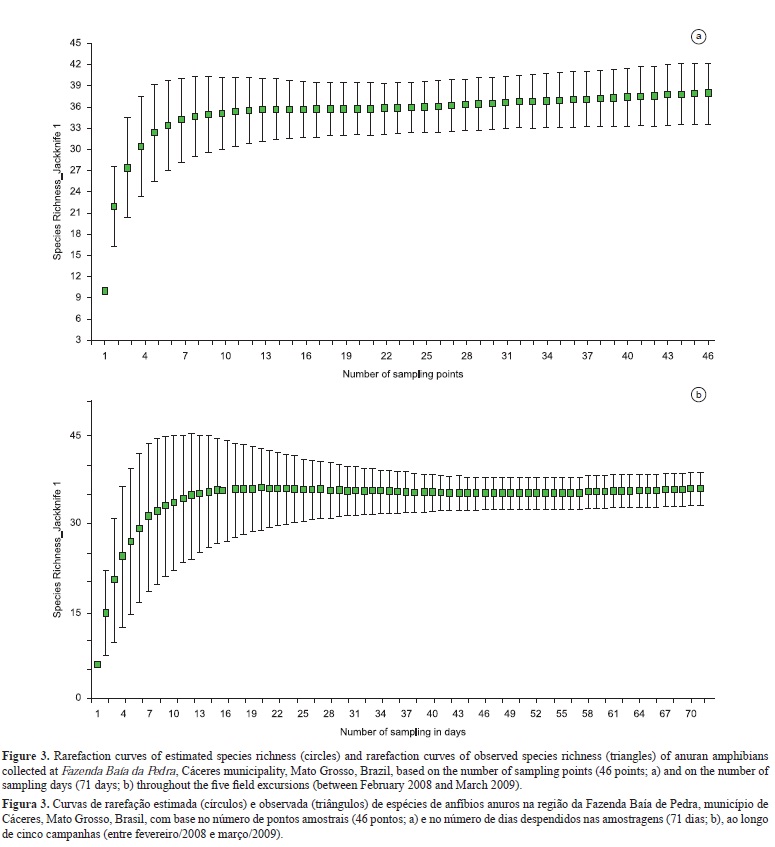
The rarefaction curve based on the number of sampling points estimated a mean number (± standard deviation) of 37.93 ± 2.17 anuran species in the sampling area, and reached an asymptote after 19 sampling points (Figure 3a). When the curve was generated using sampling days, the asymptote was reached after 33 days (Figure 3b).
The observed richness of anurans in the systematically distributed plots alone (28 species) is 11.3% lower than that recorded for the water bodies (32 species), and 17.7% lower than the total richness (34 species) recorded in this study for the whole region of Fazenda Baía da Pedra. Species recorded exclusively in the plots include the cryptozoic microhylid Chiasmocleis albopunctata and the leptodactylid Leptodactylus cf. diptyx. Species recorded only in the vicinities of water bodies included mostly hylids (Dendropsophus melanargyreus, Dendropsophus minutus, Scinax acuminatus, and Pseudis paradoxa), and the leiuperid Pseudopaludicola sp. (Figure 4). The total richness observed in this study corresponds to 77.3% of the known richness in the Pantanal floodplain (Strüssmann et al. 2007) and is similar to the expected richness (38 species) calculated by the Jackknife estimator.
Species richness in systematically distributed plots was greater in the present study than in another PPBio grid previously installed in an easternmost Pantanal subregion (22 species; Valério-Brun & Strüssmann 2010). In both studies, however, occasional encounters near the plots increased richness estimates, by about 17 and 31%, respectively. This pattern may be related to the differential availability of water bodies across Pantanal landscape. Permanent natural aquatic environments are scarce in the floodplain, man-made water bodies are located near human settlements, and temporary aquatic environments are seasonal and highly dependent on rainfalls. Specimens collected in water bodies at Fazenda Baía da Pedra represented 63% of the total amount of individuals and 94.1% of the species richness recorded in the present study. The sampling of additional plots near water bodies increased the estimates of anuran diversity obtained in systematically distributed plots in the present study, a result also observed in Amazonian sites (Rodrigues et al. 2010, Rojas-Ahumada & Menin 2010, Condrati 2009).
The abundance of individuals in the plots represented 35.7% of the total number of individuals recorded throughout the study. Species that individually represented at least 5% of the total number of individuals recorded in the plots (Table 1) were the leiuperids Pseudopaludicola cf. mystacalis, Physalaemus albonotatus, and the leptodactylids Leptodactylus fuscus, Leptodactylus podicipinus, Leptodactylus elenae, and Leptodactylus chaquensis. Together, they accounted for almost 71.8% of the total number of individuals in the plots.
Species of hylids were also abundant in the water bodies, where 64.3% of the total number of individuals were recorded. Dendropsophus nanus, Leptodactylus fuscus, Pseudopaludicola cf. mystacalis, Hypsiboas raniceps, Physalaemus albonotatus, Pseudis limellum, Eupemphix nattereri, and Leptodactylus chaquensis were the eight most abundant species in the water bodies, summing 58.8% of the total number of individuals. Each of them individually represented between 5-10% of the sample for the water bodies alone, as well as of the sample for the whole area (Table 1).
Distinct sampling methods yield different anuran species composition and relative abundances in the 10 plots surveyed at Fazenda Baía da Pedra. Visual and bioacoustic search efforts in these plots summed 200 observer-hours and returned 913 anurans (4.56 specimens/observer-hour) of 27 species, whereas the rarefaction curve estimated the occurrence of 34 ± 2.62 anuran species. Visual and bioacoustic search efforts in water bodies summed 409 observer‑hours and returned 3,459 anurans (8.45 specimens/observer-hour) of 31 species. Species most frequently recorded during visual and bioacoustic searches were Pseudopaludicola cf. mystacalis (28.5%), Physalaemus albonotatus (18%), Leptodactylus podicipinus (9%), Dendropsophus nanus (7.5%), and Leptodactylus fuscus (7%).
Pitfalls and funnel traps, combined with drift fences, resulted in 477 captures belonging to 18 species, and produced an estimate of 25 ± 2.24 anuran species. The 40 buckets and 40 funnels kept open for 44 days totaled 3,520 container-days and returned a capture rate of 0.13 specimens/container-day. Species most frequently captured in traps were Pseudopaludicola cf. mystacalis (25%), and the two cryptozoic microhylids Elachistocleis matogrosso (9.8%) and Dermatonotus muelleri (9%). The four least-represented species in traps (a single specimen each) were the terrestrial bufonid toad Rhinella schneideri, and the arboreal hylid treefrogs Hypsiboas raniceps, Phylomedusa azurea, and Scinax nasicus.
Richness and abundance of anurans recorded in the present study are roughly the same as those obtained by Valério-Brun & Strüssmann (2010). Although situated nearly at the same latitude of Fazenda Baía de Pedra, the grid area studied by those authors (30 plots; 25 km2) is five times larger than the module studied here (10 plots; 5 km2). In the present study, however, the sampling of water bodies in addition to the systematically distributed plots definitely enhanced estimates of anuran richness and abundance (see also Valério-Brun et al. 2010).
The two areas, both used for extensive cattle husbandry, harbor 28 species in common. Four species recorded in the Pirizal sampling grid have not been recorded in Baía da Pedra: Hypsiboas aff. geographicus, Hypsiboas punctatus, Physalaemus cuvieri and Leptodactylus sp. (=Adenomera). In contrast, six species recorded in Fazenda Baía de Pedra (Dendropsophus aff. elianeae, Scinax fuscovarius, Leptodactylus bufonius, Leptodactylus mystacinus, Dermatonotus muelleri, and Elachistocleis matogrosso) were not recorded in the Pirizal sampling grid. Of the six species unique to Fazenda Baía, only L. bufonius, a typical inhabitant of Chacoan areas (Souza et al. 2010), may not reach easternmost areas of the Pantanal, such as the Pirizal region.
Studies carried out in Amazonian sites suggested that sampling efforts conducted in small areas (1 to 5 km2) may capture only a small portion of the local diversity, mainly due to spatial heterogeneity Menin et al. 2008). In the more homogeneous Pantanal floodplain, however, smaller modules (instead of complete PPBio grids), which are cheaper to install and check periodically, were found to be efficient in quantifying anuran richness and abundance. This procedure could certainly benefit from the inclusion of additional sampling points, especially permanent or temporary water bodies. Arboreal species such as Dendropsophus melanargyreus and Trachycephalus venulosus, and cryptozoic species such as Dermatonotus muelleri and Chiasmocleis albopunctatus may be detected only when they aggregate for reproduction in a few selected ponds, after heavy rains. Similarly, species highly dependent upon (or strongly associated with) aquatic habitats, such as Pseudis limellum, Pseudis platensis and Hysiboas punctatus, may be absent from permanent sampling plots in areas that periodically dry out.
Acknowledgements
A.P. thanks FAPEMAT (Process 154/2008) for the scholarship; T.M. thanks CAPES/PRODOC for a former fellowship (Process 1024/2006). Authors also thank MCT/CPP for the financial support; IBAMA and ICMBio for collecting permits; Luciano Pinto de Arruda for logistics and permission to conduct fieldwork at Fazenda Baía de Pedra; Débora D. Pinheiro, Érika S. Rodrigues, Marcos A. Gonçalves, Rafaela S. Ribeiro, and Tainá D. Rodrigues for field assistance.
Received 22/12/2010
Revised 06/10/2011
Accepted 16/11/2011
- ALMEIDA, N.N. 1998. Estrutura e dinâmica de uma comunidade de plântulas em uma floresta sazonalmente inundável no Pantanal de Poconé MT. Dissertação de Mestrado, Universidade Federal de Mato Grosso, Cuiabá
- ÁVILA, R.W. & FERREIRA, V.L. 2004. Riqueza e densidade de vocalizações de anuros (Amphibia) em uma área urbana de Corumbá, Mato Grosso do Sul, Brasil. Rev. Bras. de Zool. 21(4):887-892.
- BASTAZINI, C.V., MUNDURUCA, J.F.V., ROCHA, P.L.B. & NAPOLI, M.F. 2007. Which environmental variables better explain changes in anuran community composition? A case study in the Restinga of Mata de São João, Bahia, Brazil. Herpetologica 63(4):459-471. http://dx.doi.org/10.1655/0018-0831(2007)63[459:WEVBEC]2.0.CO;2
- CABIDO, M., ACOSTA, A., DIAZ, S. & GONZALEZ, A.C. 1996. Factores estruturadores en pastizales serranos del centro de Argentina. In Biodiversidad y Funcionamiento de Pestizales y Sabanas em América Latina (G. Sarmiento & M. Cabido eds). Cyted y Cielat, Mérida, p.103-134.
- CALLEFFO, M.E.V. 2002. Anfíbios. In Técnicas de coleta e preparação de vertebrados para fins científicos e didáticos. (P. Auricchio & M.G. Salomão, orgs.). Instituto Pau Brasil de História Natural, Arujá,p.350.
- CECHIN, S.T.Z. & MARTINS, M. 2000. Eficiência de armadilhas de queda (pitfall traps) em amostragem de anfíbios e répteis no Brasil. Rev. Bras. Zool. 17(3):729-740. http://dx.doi.org/10.1590/S0101-81752000000300017
- COLWELL, R.K. 2005. EstimateS: statistical estimation of species richness and shared species from samples. version 7.5.2. http://purl.oclc.org/estimates (ultimo acesso em 15/04/2011).
- CONDRATI, L.H. 2009. Padrões de distribuição e abundância de anuros em áreas ripárias e não ripárias de floresta de terra firme na Reserva Biológia do Uatumã Amazônia Central. Dissertação de Mestrado, Instituto Nacional de Pesquisas da Amazônia, Manaus.
- DUELLMAN, W.E. 1995. Temporal fluctuations in abundance of anuran amphibians in a seasonal amazonian rainforest. J. Herpetol. 29(1):13-21. http://dx.doi.org/10.2307/1565080
- DUELLMAN, W.E. 1999. Distribution patterns of amphibians in South America. In Patterns of distribution of amphibians: a global perspective (W.E. Duellman, ed.) Johns Hopkins, Baltimore, p.255-328.
- GORDO, M. & CAMPOS, Z.M.S. 2003. Listagem dos Anuros da Estação Ecológica Nhumirim e arredores, Pantanal Sul. Embrapa Pantanal, Corumbá. Série Documentos 58.
- HAMILTON, S.K., SIPPEL, S.J. & MELACK, J.M. 1996. Inundation patterns in the Pantanal wetland of South America determined from passive microwave remote sensing. Arch. Hydrobiol. 137(1):1-23.
- INSTITUTO NACIONAL DE METEOROLOGIA - INMET. 2009. http://www.inmet.gov.br (ultimo acesso em 02/06/2009).
» link - JUNK, W.J., BAYLEY, P.B. & SPARKS, R.S. 1989. The flood pulse concept in river floodplain systems. In International Large River Symposium (D.P. Dodge, ed.). Canadian Special Publication of Fisheries and Aquatic Sciences, v.106, p.110-127.
- JUNK, W.J. & WANTZEN, K.M. 2004. The Flood Pulse Concept: new aspects, approaches, and applications - an update. In 2nd International Symposium on the Management of Large Rivers for Fisheries (R.L. Welcomme & T. Petr, eds). Food and Agriculture Organization Regional Office for Asia and the Pacific, Bangkok, p.117-149.
- JUNK, W.J., NUNES DA CUNHA, C., WANTZEN, K.M., PETERMANN, P., STRÜSSMANN, C., MARQUES, M.I. & ADIS, J. 2006. Biodiversity and its conservation in the Pantanal of Mato Grosso, Brazil. Aquat. Sci. 68(3):278-309. http://dx.doi.org/10.1007/s00027-006-0851-4
- MAGNUSSON, W.E., LIMA, A.P., LUIZÃO, R.C., LUIZÃO, F., COSTA, F.R.C., CASTILHO, C.V. & KINUPP, V.F. 2005. RAPELD: uma modificação do método de Gentry para inventários de biodiversidade em sítios para pesquisa ecológica de longa duração. Biota Neotrop. 5(2): http://www.biotaneotropica.org.br/v5n2/pt/abstract?point-of-view+bn01005022005 (ultimo acesso em 20/01/2009).
- MCDIARMID, R.W. 1994. Amphibian diversity and natural history: An overview. In Measuring and Monitoring Biological Diversity: Standard Methods for Amphibians (W.R. Heyer, M.A. Donnelly, R.W. McDiarmid, L.A.C Hayek & M.S. Foster, eds.). Smithsonian Institution, Washington, p.5-15.
- MENIN, M., WALDEZ, F. & LIMA, A.P. 2008. Temporal variation in the abundance and number of species of frogs in 10,000 ha of a forest in Central Amazonia, Brazil. S. Amer. J. Herpetol. 3(1):68-81. http://dx.doi.org/10.2994/1808-9798(2008)3[68:TVITAA]2.0.CO;2
- NIMER, E. 1979. Climatologia do Brasil. Secretaria de Recursos Naturais e Meio Ambiente - SUPREN, Rio de Janeiro.
- NAVAS, C.A. 2006. Patterns of distribution of anurans in high Andean tropical elevations: Insights from integrating biogeography and evolutionary physiology. Int. Comp. Biol. 46(1):82-91. PMid:21672725. http://dx.doi.org/10.1093/icb/icj001
- NUNES DA CUNHA, C. & JUNK, W.J. 2001. Distribution of Wood Plant Communities along the Flood Gradient in the Pantanal of Poconé, Mato Grosso, Brazil. J. Ecol. Environ. 27:63-70.
- NUNES DA CUNHA, C. & JUNK, W.J. 2004. Year-to-year changes in water level drive the invasion of Vochysia divergens in Pantanal grasslands. Appl. Veg. Sci. 7:103-110. http://dx.doi.org/10.1658/1402-2001(2004)007[0103:YCIWLD]2.0.CO;2
- NUNES DA CUNHA, C. & JUNK, W.J. 2010. A preliminary classification of habitats of the Pantanal of Mato Grosso and Mato Grosso do Sul, and its relation to nacional and international wetland classification systems. In The Pantanal: Ecology, biodiversity and sustainable management of a large neotropical seasonal wetland (W.J. Junk, C.J. da Silva, C. Nunes da Cunha & K.M. Wantzen, eds). Pensoft, Sofia, p.127-141.
- PARRIS, K.M. & MCCARTHY, M.A. 1999. What influences the structure of frog assemblages at forest streams? Aust. J. Ecology 24(5):495-502. http://dx.doi.org/10.1046/j.1442-9993.1999.00989.x
- PRADO, A.L., HECKMAN, C.W. & MARTINS, F.R. 1994. The seasonal succession of biotic communities in wetlands of the tropical wet-and-dry climatic zone: II. The aquatic macrophyte vegetation in the Pantanal of Mato Grosso, Brazil. Hydrobiol. Hydrogr. 79(4):569-589. http://dx.doi.org/10.1002/iroh.19940790407
- PRADO, C.P.D.A., UETANABARO, M. & HADDAD, C.F.B. 2005. Breeding activity patterns, reproductive modes, and habitat use by anurans (Amphibia) in a seasonal environment in the Pantanal, Brazil. Amphibia-Reptilia 26(2):211-221. http://dx.doi.org/10.1163/1568538054253375
- RODRIGUES, D.J., LIMA, A.P., MAGNUSSON, W.E. & COSTA, F.R.C. 2010. Temporary pond availability and tadpole species composition in Central Amazonia. Herpetologica 66(2):113-119. http://dx.doi.org/10.1655/09-020R2.1
- ROJAS-AHUMADA, D.P & MENIN, M. 2010. Composition and abundance of anurans in riparian and non-riparian areas in a forest in Central Amazonia, Brazil. S. Amer. J. Herpetol. 5(2):157-167. http://dx.doi.org/10.2994/057.005.0210
- SILVA, C.J., WATZEN, K.M., NUNES DA CUNHA, C. & MACHADO, F.A. 2001. Biodiversity in the Pantanal wetland, Brasil. In Biodiversity in wetlands: assessment, function and conservation (B. Gopal, W.J. Junk & J.A. Davis, eds). Backhuys, Leiden, p.187-215.
- SOCIEDADE BRASILEIRA DE HIPERTENSÃO - SBH. 2011. Brazilian amphibians. http://www.sbherpetologia.org.br . (ultimo acesso em 14/04/2011).
- SOUZA, F.L., UETANABARO, M., LANDGREF-FILHO, P., PIATTI, L. & PRADO, C.P.A. 2010. Herpetofauna, municipality of Porto Murtinho, Chaco region, state of Mato Grosso do Sul, Brazil. Check List 6(3):470‑475.
- STRÜSSMANN, C. 2000. Herpetofauna. In Fauna silvestre da região do rio Manso, Mato Grosso (C.J.R. Alho, coord.). IBAMA, ELETRONORTE, Brasília, p.153-189.
- STRÜSSMANN, C. 2001. Parque Nacional do Pantanal Matogrossense e entorno. Avaliação Ecológica Rápida. Componente Herpetofauna. IBAMA. http://www.ibama.gov.br/siucweb/unidades/parna/planos_de_manejo/78/index.htm (ultimo acesso em 10/11/2007).
- STRÜSSMANN, C., RIBEIRO, R.A.K., FERREIRA, V.L. & BÉDA, A.F. 2007. Herpetofauna do Pantanal brasileiro. In Herpetologia no Brasil II (L.B. Nascimento & M.E. Oliveira, eds). Sociedade Brasileira de Herpetologia, Belo Horizonte, p.66-84.
- VALÉRIO-BRUN, L.M., PANSONATO, A., SOLINO-CARVALHO, L.A., STRÜSSMANN, C., MOTT, T. & SILVEIRA, R.M.L. 2010. Sapos, rãs e pererecas. In Biodiversidade no Pantanal de Poconé (I.M. Fernandes, C.A., Signor & J. Penha, orgs.). Áttema, Manaus, p.119-136.
- VALÉRIO-BRUN, L.M. & STRÜSSMANN, C. 2010. Riqueza e distribuição espacial de anuros em uma localidade no Pantanal Norte, Mato Grosso, Brasil. In 5 SIMPAN: Simpósio sobre recursos naturais e socioeconômicos do Pantanal. Corumbá. CD-ROM.
- WANG, E., FERREIRA, V.L., HIMMELSTEIN, J. & STRÜSSMANN, C. 2005. Amphibians and reptiles of the southern Pantanal. In Pantanal conservation research initiative (M. Chandler, E. Wang & P. Johansson, eds.). Relatório Anual. Earthwatch, Maynard, p.30-38.
- WELLS, K.D. 1977. The social behaviour of anuran amphibians. Anim. Behav. 25(3):666-693. http://dx.doi.org/10.1016/0003-3472(77)90118-X
Appendix 1

Publication Dates
-
Publication in this collection
25 Apr 2012 -
Date of issue
Dec 2011
History
-
Received
22 Dec 2010 -
Reviewed
06 Oct 2011 -
Accepted
16 Nov 2011