Abstracts
Absence of inferior vena cava, caused by aberrant development within the sixth to eighth weeks of gestation, is a rare congenital anomaly. However, it has been recently confirmed as a major risk factor for the development of deep venous thrombosis, especially in young patients. We report a case of inferior vena cava, iliac, femoral and popliteal vein thrombosis in a 16-year-old patient with inferior vena cava agenesis and retroaortic left renal vein.
Inferior vena cava; deep venous thrombosis
A ausência da veia cava inferior, alteração no processo de formação embriológica que ocorre entre a sexta e a oitava semanas de gestação, é uma rara anomalia congênita. Porém, recentemente foi confirmada como sendo um fator de risco importante para o desenvolvimento de trombose venosa profunda, especialmente em jovens. Apresentamos um caso de trombose em veias cava inferior, ilíacas, femorais e poplíteas num jovem de 16 anos com agenesia de um segmento de veia cava infra-renal e veia renal esquerda retroaórtica.
Veia cava inferior; trombose venosa profunda
CASE REPORT
Inferior vena cava malformation and deep venous thrombosis: a risk factor of venous thrombosis in the young
Renan Roque OnziI; Luiz Francisco CostaII; Regis Fernando AngnesI; Luciano Amaral DominguesI; Paulo MoraesIII; Leandro Armani ScaffaroIV; Carolina Mancuzo StapenhorstV
IVascular surgeon, Instituto de Medicina Vascular, Hospital Mãe de Deus, Porto Alegre, RS, Brazil
IIAssociate professor, Vascular Surgery, Universidade Federal do Rio Grande do Sul (UFRGS), Porto Alegre, RS, Brazil. Vascular surgeon, Instituto de Medicina Vascular, Hospital Mãe de Deus, Porto Alegre, RS, Brazil
IIIVascular ultrasonographer, Laboratory of Vascular Medicine, Hospital Mãe de Deus, Porto Alegre, RS, Brazil
IVAngiology and Radiology Service, Hospital Mãe de Deus, Porto Alegre, RS, Brazil
VVascular surgeon, Instituto de Medicina Vascular, Hospital Mãe de Deus, Porto Alegre, RS, Brazil
Correspondence Correspondence: Renan Roque Onzi Rua Carlos de Carvalho, 72 CEP 90630-040 Porto Alegre, RS, Brazil Tel.:(51) 3330.5280 Fax:(51) 3230.2722 Email: renanonzi@cpovo.net
ABSTRACT
Absence of inferior vena cava, caused by aberrant development within the sixth to eighth weeks of gestation, is a rare congenital anomaly. However, it has been recently confirmed as a major risk factor for the development of deep venous thrombosis, especially in young patients. We report a case of inferior vena cava, iliac, femoral and popliteal vein thrombosis in a 16-year-old patient with inferior vena cava agenesis and retroaortic left renal vein.
Keywords: Inferior vena cava, deep venous thrombosis.
Introduction
Venous malformations, especially absence of inferior vena cava (IVC), are unusual and occasional findings during complementary abdomen examinations or as a consequence of acute venous thrombosis of the vena cava.1 Changes in IVC formation process, which occur embryologically between the sixth and tenth weeks of pregnancy, may result in anatomical anomalies. Among 18 previously described variants, the most frequent abnormalities are IVC duplication, retroaortic left renal vein, transposition or circumaortic left IVC and left renal vein.2-4
Some reports5-7 have described deep venous thrombosis (DVT) in patients with venous congenital malformations. Ruggeri et al.8 suggested and Chee et al.9 confirmed that IVC malformations are a major risk factor for DVT. The combination of intense physical activity and anatomical changes in the vena cava, reducing venous flow velocity, may increase possibility of DVT, especially in the young and in athletes.10
We report a case of inferior vena cava, iliac, femoral and popliteal vein thrombosis in a 16-year-old patient with inferior vena cava agenesis and retroaortic left renal vein.
Case report
A male, black 16-year-old was admitted to the emergency room complaining of difficulty in moving his lower limbs for 24 hours. He reported losing 14 kg in 4 months, due to diet and daily physical exercises. He denied other antecedents. On physical examination, he presented increased volume in both lower limbs with mild muscle tenderness in the calf and thigh. A neurological assessment was performed, including lumbar fluid puncture, which was normal. A venous color-flow Doppler ultrasonography of the lower limbs was requested, showing bilateral DVT in the IVC, popliteal and iliac veins. Screening of thrombophilia (S protein, factor V Leiden, homocysteine, lupus anticoagulant, antiphospholipid antibodies) was negative.
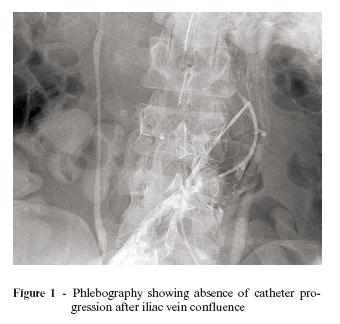
The patient was intravenously heparinized and prepared for thrombolysis. An ultrasound-guided puncture was performed in both popliteal veins with multiperforated 5F catheters. Both catheters progressed easily until the IVC. However, about 5 cm above the iliac vein confluence, the catheters stopped progressing at a site lateral to the IVC wall (Figure 1). Phlebography was performed, confirming a voluminous thrombosis with increased iliac arteries and extensive collateral circulation above those veins, with no contrast in the IVC. Due to the hypothesis of extrinsic IVC compression, thrombolysis was not performed.
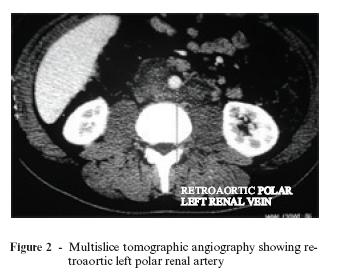
Abdominal tomography was requested, showing absence of a vena cava segment below the right renal vein, at the site where the catheters did not progress during phlebography (Figure 2). The examination was complemented by multislice tomographic angiography (Figure 3), showing common iliac veins forming the IVC to the right, both thrombosed. However, the IVC remained with the azygos vein. This caudal IVC segment was draining to the left polar renal vein with a retroaortic course, forming an arch in the aorta with the right renal vein and presenting preaortic course. In that segment the vena cava was normal, including in the intrahepatic IVC that did not present thrombi. There was no direct communication of IVC thrombosed segment with the intra-hepatic segment.
Because of the diagnosis of IVC malformation and the patient's clinical improvement, even without thrombolysis, we decided to maintain the patient anticoagulated with warfarin and other habitual clinical measures for DVT treatment.
Discussion
IVC anatomical abnormalities are a rare entity caused by developmental disorders occurred between the sixth and 12th gestational week. Infra-hepatic IVC development is a complex process comprehending three pairs of embryogenic veins: posterior cardinal (which forms iliac and confluent veins), subcardinal (which forms the renal and hepatic segment) and the supracardinal (which forms the prerenal segment of the IVC). IVC and renal vein development anomalies have been demonstrated by many authors.2-4,11,12
Prevalence of IVC malformations has been estimated in 0.5%. However, this information could be underestimated, since such anomalies are usually asymptomatic and casually discovered in imaging examinations or abdomen surgery.9,13 Absence of IVC infrarenal segment is an extremely rare anomaly. Previous studies report that only 6% of IVC anomalies occur in the renal or infrarenal segments.14 According to a research carried out by Yun et al.,15 only 21 cases confirmed by imaging examinations had been reported in the literature in English until 2004.
Nowadays, it is possible to identify risk factors in around 80% of DVT. However, over the past years, some isolated reports have drawn attention to another risk factor: anatomical anomalies.9 The cases reported in the literature show an incidence between 5 and 7% of IVC anomalies in patients with DVT and increased incidence in the young and in bilateral DVT.8-10
It is remarkable that IVC abnormalities cannot be detected by phlebography or color-flow Doppler ultrasound, which are choice examinations in a DVT episode.16,17 This could be one of the reasons why such malformations are not diagnosed in patients with DVT. Proper screening in DVT cases in the young reaching the iliac or femoral region should be conducted with computed tomography or magnetic resonance.18
There are no data about the best form of conducting a case of DVT associated with IVC anomalies when there is no other associated risk factor. Prolonged use of anticoagulants in those individuals is not associated with any clinical assessment of evidence.
This case report, associated with other cases published over the past years, confirms that anatomical changes in the IVC are a major risk factor for DVT development, and should always be screened in the young.
Finally, we would like to reinforce that vascular surgeons and radiologists should be alert to these vascular abnormalities. Radiologists should distinguish between IVC anomalies and other pathological processes of the retroperitoneum. Vascular surgeons need to recognize these anomalies to perform retroperitoneal and transperitoneal surgeries, as well as abdominal aortic surgeries. Similarly, to introduce vena cava filters, the IVC might be duplicated and the procedure could be useless if such anomaly remains unknown.
References
Manuscript received January 19, 2007, accepted April 25, 2007.
- 1. Schneider JG, Eynatten MV, Dugi KA, Duex M, Nawroth PP. Recurrent deep venous thrombosis caused by congenital interruption of the inferior vena cava and heterozygous factor V Leiden mutation. J Intern Med. 2002;252:276-80.
- 2. Moore KL, Persaud TVN. The developing human: clinically "oriented'' embryology. 5th ed. Philadelphia: Saunders; 1993. p. 309-10.
- 3. Kellman GM, Alpern MB, Sandler MA, Craig BM. Computed tomography of vena caval anomalies with embryologic correlation. Radiographics. 1988;8:533-56.
- 4. Larsen WJ. Human embryology. 3rd ed. Philadelphia: Churchill Livingstone; 2001. p. 223-4.
- 5. Saito H, Sano N, Kaneda I, et al. Multisegmental anomaly of the inferior vena cava with thrombosis of the left inferior vena cava. Cardiovasc Intervent Radiol. 1995;18:410-3.
- 6. Zamboni P, Feo C, Pisano L, Galeotti R, Buccoliero F, Liboni A. Deep venous thrombosis in the presence of congenital hypoplasia of the inferior vena cava. J Vasc Investig. 1997;3:100-2.
- 7. Hamoud S, Nitecky S, Engel A, Goldsher D, Hayek T. Hypoplasia of the inferior vena cava with azygous continuation presenting as recurrent leg deep vein thrombosis. Am J Med Sci. 2000;319:414-6.
- 8. Ruggeri M, Tosetto A, Castaman G, Rodeghiero F. Congenital absence of the inferior vena cava: a rare risk factor for idiopathic deep-vein thrombosis. Lancet. 2001;357:441.
- 9. Chee YL, Culligan DJ, Watson HG. Inferior vena cava malformation as a risk factor for deep venous thrombosis in the young. Br J Haematol. 2001;114:878-80.
- 10. Obernosterer A, Aschauer M, Schnedl W, Lipp RW. Anomalies of the inferior vena cava in patients with iliac venous thrombosis. Ann Intern Med. 2002;136:37-41.
- 11. Reis RH, Esenther G. Variations in the pattern of renal vessels and their relation to the type of posterior vena cava in man. Am J Anat. 1959;104:295-318.
- 12. Giordano JM, Trout HH 3rd. Anomalies of the inferior vena cava. J Vasc Surg. 1986;3:924-8.
- 13. Eifert S, Villavicencio JL, Kao TC, Taute BM, Rich NM. Prevalence of deep venous anomalies in congenital vascular malformations of venous predominance. J Vasc Surg. 2000;31:462-71.
- 14. Shah NL, Shanley CJ, Prince MR, Wakefield TW. Deep venous thrombosis complicating a congenital absence of the inferior vena cava. Surgery. 1996;120:891-6.
- 15. Yun SS, Kim JI, Kim KH, et al. Deep venous thrombosis caused by congenital absence of inferior vena cava, combined with hyperhomocysteinemia. Ann Vasc Surg. 2004;18:124-9.
- 16. Prandoni P, Lensing AW, Cogo A, et al. The long-term clinical course of acute deep venous thrombosis. Ann Intern Med. 1996;125:1-7.
- 17. Tiesenhausen K, Amann W, Thalhammer M, Aschauer M. Aplasia of the vena cava inferior as a cause for recurring thrombosis of the lower extremities and pelvic veins. Vasa. 1999;28:289-92.
- 18. Thornton MJ, Ryan R, Varghese JC, Farrell MA, Lucey B, Lee MJ. A three-dimensional gadolinium-enhanced MR venography technique for imaging central veins. AJR Am J Roentgenol. 1999;173:999-1003.
Publication Dates
-
Publication in this collection
20 Sept 2007 -
Date of issue
June 2007
History
-
Accepted
25 Apr 2007 -
Received
19 Jan 2007