Abstract
In spite of considerable efforts to identify effective treatments for urolithiasis, this is a goal yet to be achieved. This review summarizes experimental and clinical data evaluating the effect of the plant Phyllanthus niruri, a plant with worldwide distribution, as a potential agent to prevent and/or to treat urolithiasis The review is based on data from the literature and on the results obtained by our group from either in vivo/in vitro experiments or clinical studies. Phyllanthus niruri has been shown to interfere with many stages of stone formation, reducing crystals aggregation, modifying their structure and composition as well as altering the interaction of the crystals with tubular cells leading to reduced subsequent endocytosis. The clinical beneficial effects of Phyllanthus niruri may be related to ureteral relaxation, helping to eliminate calculi or to clear fragments following lithotripsy, or also to a putative reduction of the excretion of urinary crystallization promoters such as calcium. No adverse renal, cardiovascular, neurological or toxic effects have been detected in either of these studies. Altogether, these studies suggest a preventive effect of Phyllanthus niruri in stone formation or elimination, but still longer-term randomized clinical trials are necessary to confirm its therapeutic properties.
renal; lithiasis; treatment; clinical; Phyllanthus niruri
REVIEW ARTICLE
Phyllanthus niruri as a promising alternative treatment for nephrolithiasis
Mirian A. Boim; Ita P. Heilberg; Nestor Schor
Renal Division, Federal University of Sao Paulo, Unifesp, Sao Paulo, Brazil
CorrespondenceCorrespondence to: Dr. Mirian A. Boim Renal Division, UNIFESP Rua Botucatu, 740 04023-900, São Paulo, SP, Brazil Fax: + 55 11 5904-1684 E-mail: mirian@nefro.epm.br
ABSTRACT
In spite of considerable efforts to identify effective treatments for urolithiasis, this is a goal yet to be achieved. This review summarizes experimental and clinical data evaluating the effect of the plant Phyllanthus niruri, a plant with worldwide distribution, as a potential agent to prevent and/or to treat urolithiasis The review is based on data from the literature and on the results obtained by our group from either in vivo/in vitro experiments or clinical studies. Phyllanthus niruri has been shown to interfere with many stages of stone formation, reducing crystals aggregation, modifying their structure and composition as well as altering the interaction of the crystals with tubular cells leading to reduced subsequent endocytosis. The clinical beneficial effects of Phyllanthus niruri may be related to ureteral relaxation, helping to eliminate calculi or to clear fragments following lithotripsy, or also to a putative reduction of the excretion of urinary crystallization promoters such as calcium. No adverse renal, cardiovascular, neurological or toxic effects have been detected in either of these studies. Altogether, these studies suggest a preventive effect of Phyllanthus niruri in stone formation or elimination, but still longer-term randomized clinical trials are necessary to confirm its therapeutic properties.
Key words: renal; lithiasis; treatment; clinical; Phyllanthus niruri
INTRODUCTION
Urinary stones affect 10-12% of the population in industrialized countries. Their incidence has been increasing over the last years and the age of onset is decreasing. In addition, the recurrence rate is high, more than 50% after 10 years (1,2). Genetic, metabolic, environmental and dietetic factors are involved in the pathogenesis of urolithiasis, all of them propitiating the crystallization of salts inside the renal tubules, further retention and growing to form a stone (3). Given that urine is normally a supersaturated solution, crystalluria is often observed in normal individuals, but if crystals remain apart from each other, they are washed away by urine flow. However, under certain circumstances they bind each other due to chemical and electrical forces triggering the process of aggregation. The crystals or aggregates then attach to the epithelium which allow them to grow further and form the stone (4). Moreover, calcium oxalate (CaOx) crystals, the main constituent of human urinary calculi, may adhere in a specific manner to the plasma membrane of epithelial cells and this process is followed by endocytosis of the crystals resulting in cell damage or death (5,6). Damaged cells exhibit a proliferative response, increase the synthesis of fibrogenic substances promoting additional stimulus for crystal growth (7,8). In addition, dead cells detach from the basement membrane and the cellular debris will adhere to other crystals resulting in a stone nidus. Thus, the cellular pathways involved in endocytosis of CaOx crystals can constitute potential targets for drugs designed for the prophylaxis and/or treatment of urolithiasis. Different substances have been described as modulators of adhesion and/or endocytosis processes (9-11) but none of them seem to be suitable for clinical use. On the other hand, alternative treatments such as the traditional herbal treatments can compliment pharmacotherapies for prevention and/or treatment of urolithiasis with less expense and perhaps fewer side effects, as reviewed by Miyaoka and Monga (12).
We have been evaluating the effects Phyllanthus niruri on several stages of stone formation as well as its potential therapeutic potential in lithiasic patients. Experimental and clinical studies performed by our group and by others have produced interesting and hopeful data concerning the potential therapeutic use of Phyllanthus niruri to treat and/or to prevent stone formation. These data are summarized in this review.
PHYLLANTHUS NIRURI
Phyllanthus niruri, popularly known as “stone-breaker” (“quebra-pedras”) is a plant belonging to the Euphorbiaceae family with a worldwide distribution and it is used in folk Brazilian medicine for patients with urolithiasis (13). More than 50 compounds were identified in the Phyllanthus niruri, including alkaloids, flavanoids, lignans and triterpenes (14). Among these substances, the triterpenes have been found to inhibit the cytotoxicity induced by calcium oxalate (15) as well as to reduce excretion of stone forming constituents (16) and the markers of crystal deposition in the kidneys (17). Moreover, methanol extract from the leaves of Phyllanthus niruri containing substances such as lignans and phyllanthin showed a uricosuric activity in hyperuricemic rats (18). According to Calixto et al. (19) alkaloids extracted from plants of the genus Phyllanthus present an antispasmodic activity leading to smooth muscle relaxation, mostly evidenced in the urinary tract, which would facilitate the elimination of urinary calculi. These data strongly suggest that Phyllanthus niruri may be a potential source of many substances with antilithiasic properties.
Interestingly, given that the maintenance of normal levels of calcium is critical to the function of many plants including plant rigidity, protection, detoxification (heavy metals or oxalic acid), ion balance and even light reflection (20), and because high cellular free calcium concentration may be dangerous for these organisms, higher plants (plants exhibiting a vascular system) developed a very efficient way to neutralize Ca+2 ions, by forming complexes with oxalate (21). Oxalate producing plants, which include many crop plants, accumulate oxalate and as much as 90% of the total calcium in the form of CaOx crystals. Curiously, as shown in Figure-1, refractive CaOx crystals seen in the leaves of Phyllanthus niruri are kept equidistant, do not aggregate hence not forming stones. Therefore, understanding the basic molecular strategies by which some plants are able to package CaOx crystals may provide insights into the potential utilization of these plants to prevent/treat urolithiasis (22).
IN VITRO STUDIES
The effect of the aqueous extract of Phyllanthus niruri on crystallization process of CaOx in human urine has also been investigated in a model of in vitro precipitation of CaOx in human urine (23). Barros et al. (24) observed that the pre-incubation of human urine with Phyllanthus niruri did not inhibit the precipitation of CaOx particles and even more crystals were obtained in Phyllanthus niruri-containing urine, but the crystals were proportionally smaller than those in urine samples without Phyllanthus niruri. Moreover, they observed that after 24 hours, the precipitated crystals formed large agglomerates in untreated urine, but the crystals remained dispersed in urine with Phyllanthus niruri (Figure-2). The authors concluded that Phyllanthus niruri did not decrease the number of crystals but induced a marked reduction of particle size and crystal aggregation. Similar results were obtained by Atmani E and Khan SR (25) employing a different plant species (Herniaria hirsute), which is used in folk medicine in the Mediterranean area for its diuretic properties and to treat kidney stones. Crystalluria is a common event observed even in non-stone forming individuals. CaOx crystals are found in urine under several forms including monohydrate (COM) and dihydrate (COD) forms. Unlike COD, which is predominantly found in normal individuals, COM crystals have higher capacity to aggregate and adhere, and is the main form excreted by the nephrolithiasis patients (26-28). In the model of in vitro precipitation of CaOx using human urine from healthy individuals, Phyllanthus niruri induced an increase in COD forms and reduced the amount of COM crystals, responsible for higher potential risk for stone formation.
Besides CaOx crystallization, it has been shown that the methanol extracts of Phyllanthus niruri showed an inhibitory activity of the enzyme xanthine oxidase in vitro (29) which was attributed to compounds such as flavonoids, polyphenols and tannins (30,31). Therefore, together with its uricosuric property (18), this enzymatic inhibition activity makes Phyllanthus niruri a potential antihyperuricemic agent.
STUDIES IN CULTURED CELLS
Campos and Schor (32) have demonstrated that Phyllanthus niruri exhibited a potent inhibitory effect on CaOx crystal adhesion and/or endocytosis by an immortalized cell line derived from canine kidney (MDCK cells) representative of the medullar collecting duct. This type of inhibitory effect occurred even when high doses (2.5 to 5-fold the upper limit in human urine) of CaOx have been employed and without causing cell toxicity.
EXPERIMENTAL MODELS IN RATS
The effect of Phyllanthus niruri has also been evaluated in experimental models of urolithiasis in rats, mainly those induced by implantation of a calcium oxalate (CaOx) crystal into the bladder (vesical foreign body method). This experimental model of urolithiasis is obtained with no significant metabolic or systemic alterations and the vesical CaOx seed acts as a supporting surface allowing organic and inorganic material to precipitate over the central nidus, mimicking a spontaneous calculus growth. It was initially shown that rats drinking Phyllanthus niruri tea ad libitum, presented decreased rate of stone growth (33). These effects occurred independently of any relevant modification in the urinary excretion of elements known to promote crystallization and stone formation, including calcium, oxalate, uric acid, pH, etc. In order to evaluate if the beneficial effect of Phyllanthus niruri could be mediated by modifications of the inhibitors of stone formation, such as citrate, magnesium and/or glycosaminoglycans, Freitas et al. (34) administered 1.25 mg/mL/day of Phyllanthus niruri for 42 days in rats with vesical CaOx seed. This chronic treatment induced a significant reduction in the calculi growth, in the absence of any modification in the volume diuresis or alterations in the urinary concentration of lithogenic elements including calcium and oxalate. Phyllanthus niruri administration did not modify the urinary excretion of citrate and magnesium, indicating that the putative antilithogenic effect of Phyllanthus niruri was not primarily mediated by modifications in these inhibitors. In contrast, it was observed that Phyllanthus niruri induced a decrease in the urinary excretion of glycosaminoglycans (GAGs) compared with lithiasic animals receiving water. In contrast, the content of GAGs was higher in calculi taken from treated animals suggesting that the inhibitory effect Phyllanthus niruri on crystal growth might have been related to higher incorporation of GAGs into the calculi. The adsorption of these macromolecules into the calculi lead to stones with a predominant intracrystalline amorphous organic matrix. Taken together, these results suggested that Phyllanthus niruri was able to prevent the aggregation of calcium oxalate to the pre-existent crystal without interfering with the incorporation of GAGs into organic matrix. Although the underlying mechanism remains to be clarified, some possible hypotheses can be raised: 1) a neutralization of negative charges of GAGs reduced the negative pole for progressive deposition of cations; 2) active components of the plant could have chelated and/or competed with calcium for binding sites on the crystal surface; 3) effects of Phyllanthus niruri itself on other proteins including Tamm-Horsfall protein, nephrocalcin, osteopontin, prothrombin fragment 1, etc, modulating crystallization, aggregation and calculi growth and 4) Phyllanthus niruri could reduce the crystal adhesion to the tubular epithelium. Essentially, these results suggest that Phyllanthus niruri could interfere with the calculi growth or prevent stone formation rather than dissolving pre-formed stones.
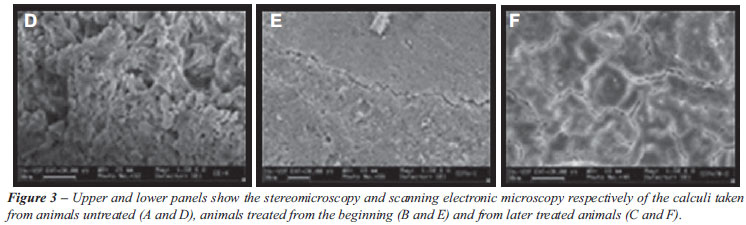
In order to better mimic what is observed in clinical practice, Barros et al. (35) using a vesical foreign body model in rats, compared the efficacy of Phyllanthus niruri treatment started concomitantly with CaOx seed implantation with the same treatment started 30 days after the seed implantation (when the vesical calculus was completely formed). As represented in Figure-3, the early treatment (Figure-3A) caused a significant inhibition in the calculi growth compared with non-treated animals (Figure-3B), as previously observed (34). In contrast, the treatment initiated after the stone formation, did not prevent the calculi to grow further neither propitiated calculi elimination; however it induced drastic changes in the shape and texture of the preformed calculi (Figure-3C). Stones taken from Phyllanthus niruri treated animals were more homogeneous and contained more compact surfaces (Figures-3D and Figure-3E) in contrast to the spicule-shaped surface taken from untreated animals (Figure-3F). This result suggests that Phyllanthus niruri probably interferes with the biomineralization process, by promoting a different interaction between the crystal and the macromolecules of the organic matrix. Although Phyllanthus niruri had not prevented the calculi growth, the treatment resulted in the formation of calculi with smoother surfaces, which could at least, contribute to less painful calculi voiding.
CLINICAL STUDIES
The initial study (33) addressing the effects of the Phyllanthus niruri administered in the form of tea did not demonstrate any clinical or biochemical adverse effects (cardiovascular, renal, hepatic or neurological) even at high dosage, with excellent tolerability in healthy volunteers. In addition, tea consumption in the same dose by nephrolithiasis patients for a period of 3 months, led to an apparent increased elimination of calculi compared to patients drinking placebo. These results were probably ascribed to the antispasmodic and relaxant effects of Phyllanthus niruri upon ureteral muscle, facilitating calculi voiding.
Subsequently, another study by Nishiura et al. (36), also in our Service, evaluated the effect of a lyophilized 2% aqueous extract of Phyllanthus niruri, in an additional series of 69 lithiasic patients in the form of 450 mg capsules (three times a day) compared with placebo (Chicorium sativum). In this short-term follow-up conducted during a three-months period, no significant differences in calculi voiding and/or pain relief between the groups taking Phyllanthus niruri or the placebo were detected. However, as patients were classified according to the presence of metabolic disturbance, a significant reduction in the mean urinary calcium excretion was observed only among hypercalciuric patients. The phytochemical and pharmacological properties of this plant have been accounted for the action of different substances such as Rutin, beta-amylin, beta-sitosterol, caffeic acid, geranin, quercetin, niruside and repandusinic acid. Although none of these compounds has been shown to have an effect on calciuria to date, such potential beneficial effect of Phyllanthus niruri for hypercalciuric patients needs to be further confirmed in longer-term studies involving a higher number of subjects.
Micali and coworkers (37) observed that patients submitted to extracorporeal shock wave lithotripsy and treated with Phyllanthus niruri during 3 months presented lower incidence of residual stone fragments, mainly those in lower calyceal location compared with non-treated patients. According to these investigators, the efficacy and the lack of side effects of Phyllanthus help improve overall outcomes after extracorporeal shock wave lithotripsy and could be useful as either an alternative or an adjunctive therapy in the treatment of urolithiasis.
Table-1 summarizes the main findings in all studies. Given the experimental characteristic of these studies, except for the clinical ones (Grade of recommendation B), an exact level of evidence is not applicable.
CONCLUSION AND PERSPECTIVES
The experimental studies summarized here suggest that Phyllanthus niruri might interfere with important steps of the calculi formation including crystal aggregation and internalization by the tubular cells, crystal structure and composition. These properties of Phyllanthus niruri may constitute an important advantage in the prevention of lithiasis, inhibiting calculus growth and keeping the crystals dispersed in the urine, with their consequent easier elimination. Although clinical studies are less abundant, available data point to beneficial effects of Phyllanthus by inducing ureteral relaxation, interfering in the excretion of promoters of urinary crystallization such as calcium or helping to clear fragments following lithotripsy. It is important to consider however, that although it is clear that Phyllanthus niruri can interfere with many steps of the stone formation, longer-term clinical studies are necessary to define whether these effects can be translated into real clinical benefit to treat and/or prevent urolithiasis.
CONFLICT OF INTEREST
None declared.
Accepted after revision: May 26, 2010
EDITORIAL COMMENT
The authors provide a nice review on the herb Phyllanthus niruri whose properties appear in fact to be promising assets in stone disease prevention and treatment. However, care must be taken to properly design future clinical studies in a way to provide reliable, consistent and reproducible data. As a major concern, I would point out the definition of accurate dosage and mode of intake. In a recent review on Chinese herbs used for managing stone disease, Miyaoka et al. (1) found several clinical trials demonstrating the likely benefits on stone prevention. However, the lack of standardization on dose and compounds between studies evaluating the same herbs made it extremely difficult to compare them and draw a sustainable conclusion. As a result, although used for hundreds of years with practical evidence of clinical benefits, Chinese herbs still strive to enter the armamentarium of stone therapy as a global consensus.
REFERENCE
Dr. Ricardo Miyaoka
Division of Urology
University of Campinas, UNICAMP
Campinas, SP, Brazil
E-mail: rmiyaoka@uol.com.br
- 1. Moe OW: Kidney stones: pathophysiology and medical management. Lancet. 2006; 367: 333-44.
- 2. Bartoletti R, Cai T, Mondaini N, Melone F, Travaglini F, Carini M, et al.: Epidemiology and risk factors in urolithiasis. Urol Int. 2007; 79(suppl) 1: 3-7.
- 3. Pak CY, Resnick MI, Preminger GM: Ethnic and geographic diversity of stone disease. Urology. 1997; 50: 504-7.
- 4. Matlaga BR, Coe FL, Evan AP, Lingeman JE: The role of Randall’s plaques in the pathogenesis of calcium stones. J Urol. 2007; 177: 31-8.
- 5. Khaskhali MH, Byer KJ, Khan SR: The effect of calcium on calcium oxalate monohydrate crystal-induced renal epithelial injury. Urol Res. 2009; 37: 1-6.
- 6. Khan SR, Byer KJ, Thamilselvan S, Hackett RL, McCormack WT, Benson NA, et al.: Crystal-cell interaction and apoptosis in oxalate-associated injury of renal epithelial cells. J Am Soc Nephrol. 1999; 10(Suppl) 14: S457-63.
- 7. Lieske JC, Toback FG: Renal cell-urinary crystal interactions. Curr Opin Nephrol Hypertens. 2000; 9: 349-55.
- 8. Lieske JC, Toback FG: Interaction of urinary crystals with renal epithelial cells in the pathogenesis of nephrolithiasis. Semin Nephrol. 1996; 16: 458-73.
- 9. Tsujihata M, Yoshimura K, Tsujikawa K, Tei N, Okuyama A: Fibronectin inhibits endocytosis of calcium oxalate crystals by renal tubular cells. Int J Urol. 2006; 13: 743-6.
- 10. Campos AH, Schor N: Mechanisms involved in calcium oxalate endocytosis by Madin-Darby canine kidney cells. Braz J Med Biol Res. 2000; 33: 111-8.
- 11. Lieske JC, Huang E, Toback FG: Regulation of renal epithelial cell affinity for calcium oxalate monohydrate crystals. Am J Physiol Renal Physiol. 2000; 278: F130-7.
- 12. Miyaoka R, Monga M: Use of traditional Chinese medicine in the management of urinary stone disease. Int Braz J Urol. 2009; 35: 396-405.
- 13. Kieley S, Dwivedi R, Monga M: Ayurvedic medicine and renal calculi. J Endourol. 2008; 22: 1613-6.
- 14. Bagalkotkar G, Sagineedu SR, Saad MS, Stanslas J: Phytochemicals from Phyllanthus niruri Linn. and their pharmacological properties: a review. J Pharm Pharmacol. 2006; 58: 1559-70.
- 15. Malini MM, Lenin M, Varalakshmi P: Protective effect of triterpenes on calcium oxalate crystal-induced peroxidative changes in experimental urolithiasis. Pharmacol Res. 2000; 41: 413-8.
- 16. Vidya L, Lenin M, Varalakshmi P: Evaluation of the effect of triterpenes on urinary risk factors of stone formation in pyridoxine deficient hyperoxaluric rats. Phytother Res. 2002; 16: 514-8.
- 17. Vidya L, Malini MM, Varalakshmi P: Effect of pentacyclic triterpenes on oxalate-induced changes in rat erythrocytes. Pharmacol Res. 2000; 42: 313-6.
- 18. Murugaiyah V, Chan KL: Antihyperuricemic lignans from the leaves of Phyllanthus niruri. Planta Med. 2006; 72: 1262-7.
- 19. Calixto JB, Santos AR, Cechinel Filho V, Yunes RA: A review of the plants of the genus Phyllanthus: their chemistry, pharmacology, and therapeutic potential. Med Res Rev. 1998; 18: 225-58.
- 20. Franceschi V: Calcium oxalate in plants. Trends Plant Sci. 2001; 6: 331.
- 21. Franceschi VR, Nakata PA: Calcium oxalate in plants: formation and function. Annu Rev Plant Biol. 2005; 56: 41-71.
- 22. Ryall RL: The future of stone research: rummagings in the attic, Randall’s plaque, nanobacteria, and lessons from phylogeny. Urol Res. 2008; 36: 77-97.
- 23. Buchholz NP, Kim DS, Grover PK, Dawson CJ, Ryall RL: The effect of warfarin therapy on the charge properties of urinary prothrombin fragment 1 and crystallization of calcium oxalate in undiluted human urine. J Bone Miner Res. 1999; 14: 1003-12.
- 24. Barros ME, Schor N, Boim MA: Effects of an aqueous extract from Phyllantus niruri on calcium oxalate crystallization in vitro. Urol Res. 2003; 30: 374-9.
- 25. Atmani F, Slimani Y, Mimouni M, Aziz M, Hacht B, Ziyyat A: Effect of aqueous extract from Herniaria hirsuta L. on experimentally nephrolithiasic rats. J Ethnopharmacol. 2004; 95: 87-93.
- 26. Dyer R, Nordin BE: Urinary crystals and their relation to stone formation. Nature. 1967; 215: 751-2.
- 27. Sheng X, Ward MD, Wesson JA: Crystal surface adhesion explains the pathological activity of calcium oxalate hydrates in kidney stone formation. J Am Soc Nephrol. 2005; 16: 1904-8.
- 28. Wesson JA, Ward MD: Role of crystal surface adhesion in kidney stone disease. Curr Opin Nephrol Hypertens. 2006; 15: 386-93.
- 29. Murugaiyah V, Chan KL: Mechanisms of antihyperuricemic effect of Phyllanthus niruri and its lignan constituents. J Ethnopharmacol. 2009; 124: 233-9.
- 30. Cos P, Ying L, Calomme M, Hu JP, Cimanga K, Van Poel B, et al.: Structure-activity relationship and classification of flavonoids as inhibitors of xanthine oxidase and superoxide scavengers. J Nat Prod. 1998; 61: 71-6.
- 31. Hatano T, Yasuhara T, Yoshihara R, Agata I, Noro T, Okuda T: Effects of interaction of tannins with co-existing substances. VII. Inhibitory effects of tannins and related polyphenols on xanthine oxidase. Chem Pharm Bull (Tokyo). 1990; 38: 1224-9.
- 32. Campos AH, Schor N: Phyllanthus niruri inhibits calcium oxalate endocytosis by renal tubular cells: its role in urolithiasis. Nephron. 1999; 81: 393-7.
- 33. Santos DR: Produtos Naturais no Tratamento da Nefrolitíase. In: Schor N (ed.), Calculose Renal: Fisiopatologia, Diagnóstico e Tratamento. São Paulo, Ed. Sarvier. 1995; pp. 221-5.
- 34. Freitas AM, Schor N, Boim MA: The effect of Phyllanthus niruri on urinary inhibitors of calcium oxalate crystallization and other factors associated with renal stone formation. BJU Int. 2002; 89: 829-34.
- 35. Barros ME, Lima R, Mercuri LP, Matos JR, Schor N, Boim MA: Effect of extract of Phyllanthus niruri on crystal deposition in experimental urolithiasis. Urol Res. 2006; 34: 351-7.
- 36. Nishiura JL, Campos AH, Boim MA, Heilberg IP, Schor N: Phyllanthus niruri normalizes elevated urinary calcium levels in calcium stone forming (CSF) patients. Urol Res. 2004; 32: 362-6.
- 37. Micali S, Sighinolfi MC, Celia A, De Stefani S, Grande M, Cicero AF, et al.: Can Phyllanthus niruri affect the efficacy of extracorporeal shock wave lithotripsy for renal stones? A randomized, prospective, long-term study. J Urol. 2006; 176: 1020-2.
- 1. Miyaoka R, Monga M: Use of traditional Chinese medicine in the management of urinary stone disease. Int Braz J Urol. 2009; 35: 396-405.
Correspondence to:
Publication Dates
-
Publication in this collection
23 Mar 2011 -
Date of issue
Dec 2010