Abstract
This study investigated the effect of extracts of different composites, glass ionomer cement (GIC)s and compomers on the viability of brine shrimp larvae. Ethanolic extracts of four dental composites (Z-100; Solitaire 2; Filtek P60 and Synergy), a conventional GIC (Ketac-Fil), a resin-modified glass ionomer cement (Vitremer), two compomers (F2000; Dyract AP), and a flowable compomer (Dyract Flow) were prepared from each material. Following evaporation of the ethanol, the extracts were resuspended in distilled water, which was then used to test the effects on the viability of brine shrimp larvae. For the composites, the extract of Synergy was the least toxic (88% viability) followed by the extracts of Solitaire 2, Z100 and P60 (75%, 67.5% and 50% viability, respectively). One-way ANOVA revealed highly significant differences between the resin composite materials (p<0.001). Follow-up comparison between the composite groups by Tukey's pairwise multiple-comparison test (α =0.05) showed that the extract of Synergy was significantly less toxic than the extracts of all the other materials except that of Solitaire 2. The compomers showed 100% lethality, while the percentage of viable larvae for the extracts of Ketac-Fil, and Vitremer were 32.3%, and 37.0%, respectively. One-way ANOVA revealed highly significant differences between the groups of materials (p<0.001). Follow-up comparison between the groups by Tukey's test (α = 0.05) showed that the toxic effect of the extracts of the compomers were significantly greater than that of Ketac-Fil, and Vitremer. The differences in the toxic effects of Vitremer and Ketac-Fil were not statistically significant. In conclusion, the toxicity of composite materials varied according to their chemical composition. Compomers were the most lethal materials to brine shrimp larvae followed by GICs and then composites.
Glass ionomer cements; Compomers; Artemia salina
ORIGINAL ARTICLES
Toxicity Testing of Restorative Dental Materials Using Brine Shrimp Larvae (Artemia salina)
Manar M. MilhemI; Ahmad S. Al-HiyasatII; Homa DarmaniIII
IBSc, MSc, Department of Applied Biology, Faculty of Science, Jordan University of Science and Technology, Irbid, Jordan
IIBDS, MScD, PhD, Department of Restorative Dentistry, Jordan University of Science and Technology, Irbid, Jordan
IIIBSc PhD, Department of Applied Biology, Faculty of Science, Jordan University of Science and Technology, Irbid, Jordan
Corresponding address Corresponding address: Professor Homa Darmani Department of Applied Biology Faculty of Science Jordan University of Science & Technology P.O. Box 3030, Irbid, 22110, Jordan Phone: 962-79-5978834 Fax: 962-2-7278962 e-mail: darmani@just.edu.jo
ABSTRACT
This study investigated the effect of extracts of different composites, glass ionomer cement (GIC)s and compomers on the viability of brine shrimp larvae. Ethanolic extracts of four dental composites (Z-100; Solitaire 2; Filtek P60 and Synergy), a conventional GIC (Ketac-Fil), a resin-modified glass ionomer cement (Vitremer), two compomers (F2000; Dyract AP), and a flowable compomer (Dyract Flow) were prepared from each material. Following evaporation of the ethanol, the extracts were resuspended in distilled water, which was then used to test the effects on the viability of brine shrimp larvae. For the composites, the extract of Synergy was the least toxic (88% viability) followed by the extracts of Solitaire 2, Z100 and P60 (75%, 67.5% and 50% viability, respectively). One-way ANOVA revealed highly significant differences between the resin composite materials (p<0.001). Follow-up comparison between the composite groups by Tukey's pairwise multiple-comparison test (α =0.05) showed that the extract of Synergy was significantly less toxic than the extracts of all the other materials except that of Solitaire 2. The compomers showed 100% lethality, while the percentage of viable larvae for the extracts of Ketac-Fil, and Vitremer were 32.3%, and 37.0%, respectively. One-way ANOVA revealed highly significant differences between the groups of materials (p<0.001). Follow-up comparison between the groups by Tukey's test (α = 0.05) showed that the toxic effect of the extracts of the compomers were significantly greater than that of Ketac-Fil, and Vitremer. The differences in the toxic effects of Vitremer and Ketac-Fil were not statistically significant. In conclusion, the toxicity of composite materials varied according to their chemical composition. Compomers were the most lethal materials to brine shrimp larvae followed by GICs and then composites.
Key words: Glass ionomer cements. Compomers. Artemia salina.
INTRODUCTION
In the dental clinic, different types of materials are used to restore the patient's dentition. These materials come into direct contact with dentin, periodontium, oral mucosa and body fluids. The biocompatibility of these materials is thus of paramount importance and any adverse reactions due to the leaching of components from these dental materials into the oral environment is a clinical concern.
Dental composite resins are used as filling materials, dentin adhesives, cements or as luting agents for inlays, crowns, veneers and orthodontic brackets12. Glass ionomer cements (GICs) are extensively used as restorative materials, cements and fissure sealants10. Because conventional GICs are brittle and susceptible to wear and dehydration17, researchers have developed hybrid materials that are combinations of conventional GICs and methacrylate resins. Although the cytotoxicity of GICs is reported to be minimal4, individual components of resin materials have been shown to be cytotoxic6. Hybrid materials or the so-called resin modified GICs (RMGICs) and compomers may differ in their toxicity, since the different chemical composition of the materials may result in varying release of potentially cytotoxic products10. Indeed, leachable components from compomers have been characterized as cytotoxic7,20,26, with cytotoxicity ranging from mild to severe21. It has also been reported that leachable components, such as various dental monomers, may cause a wide range of adverse health effects including skin-, eye- and mucous-membrane irritation as well as gastrointestinal complaints15,16.
Cytotoxicity testing is an integral component of the biological evaluation of dental materials and is an essential part of standard screening procedures10. A new screening test for toxicity of dental materials using brine shrimp larvae (Artemia salina) has been reported18. Brine shrimps belong to the phylum Arthropoda, class Crustacea. Their life cycle begins by hatching of dormant cysts where these cysts are inactive but, once in salt water, they become rehydrated and resume their development18. Brine shrimp larvae are commonly used for toxicity assays in pharmacology. These larvae are sensitive to toxic substances. The ratio between dead larvae (no motility) and living larvae (high motility) in comparison to a control without any toxic substances is used to estimate the toxicity of the test substances18.
The aim of this study was to investigate the toxic potential of four composites, two GICs, two compomers and one flowable compomer using the brine shrimp larvae assay.
MATERIAL AND METHODS
Preparation of Material Specimens
Disk-shaped material specimens (5 mm in diameter x 2 mm in thickness) were prepared using a glass mold (ring) with the desirable dimensions. The mold was placed on a glass plate and the material was condensed into the mold from the top. The composite material was delivered directly from its syringe into the ring on the top of the glass plate. The material was pressed using plastic instruments and a myler matrix cellulose strip was then placed onto the ring and pressed on the top surface of the material. The powder and liquid of both Ketac-Fil and Vitremer GICs materials were mixed in the same amounts advised by the manufacturer. Mixing was performed onto the paper pad supplied with the kits using a sterile spatula. The compomer materials were delivered directly from the compules using a special gun and from Dyract Flow's syringe into the glass ring and then pressed and condensed in the ring with plastic instruments. A myler matrix cellulose strip was then placed onto the ring and pressed on the top of the material.
The tip of a light-curing unit (Coltolux4, Coltene/Whaledent, Inc., Mahwah, NJ, USA) was positioned at a distance of 2 mm from the material surface and each material was light activated according to the manufacturers' instruction (Z100 and Solitaire 2: 40 s, Synergy: 30 s and P60: 20 s Vitremer, F2000, Dyract AP and Dyract Flow: 40 s). The light intensity used was 600 mW cm-2. For Ketac-Fil, the material was left to set chemically without light activation, for at least 4 min.
Preparation of Ethanolic Extracts
The 10 discs of each material were placed in 1 mL of 96% ethanol in a glass vial, sonicated for 2 h and thereafter incubated in the ethanol at 37°C for 24 h. Next, the discs were removed from the vials and the ethanol containing any leached components was evaporated using a water bath at 70°C-80°C for 2 h. Sterile distilled water (1 mL) was then added to the vials, which were then sonicated for 30 min to ensure resolubilization of the leached components. The distilled water containing the leached substances was then tested for toxic effects using the brine shrimp larvae assay. As a control, autoclaved Teflon discs were used and treated the same as the material discs.
Brine Shrimp Larvae Assay
Brine shrimp egg hatching
Brine shrimp eggs (Carolina Biological Supply Company, Burlington, NC, USA) were hatched in artificial salt water (25g NaCl/L distilled water) at 20°C and constant illumination. The brine shrimp eggs were incubated in a polypropylene jar with a water height of 1.2 cm. These hatching conditions corresponded to those in the natural environment. After 48 h from hatching, the shrimp larvae were used for the experimental bioassay. At this time, the larvae were still living on their own yolk sac and received no further food during the experimental time.
Toxic effect of the ethanolic extracts of dental materials
The extracts of restorative dental materials were adjusted to a salt (NaCl) content of 25 g/L. Aliquots of 200 µL of each extract were placed into a 96-well plate, using 5 wells for each material and the control as well. Then, 50 µL of the brine shrimp larvae salt solution (containing 6-17 larvae) were added to each well. After 5 h, the dead shrimp larvae were counted using a stereomicroscope. After 24 h, all larvae in the wells had died and the total number of shrimp larvae per well were counted. The number of living larvae was determined by subtracting the number of the dead larvae after 5 h from the total number of dead larvae in each well.
RESULTS
Figure 1 shows the percentage of viable brine shrimp larvae exposed to the ethanolic extracts of the composite resin material specimens investigated in this study. Synergy extract was the least toxic followed by the extracts of Solitaire 2, Z100 and P60. One-way ANOVA revealed highly significant differences between the resin composite materials (p<0.001). Follow-up comparison between the different composites by Tukey's pairwise multi-comparison test (α =0.05) showed that the extract of Synergy was significantly less toxic than the extracts of all the other materials except that of Solitaire 2, which did not differ significantly. On the other hand the extract of P60 was significantly more toxic than the extract of Synergy. No statistically significant differences were found in toxicity between the extracts of P60 and Z100, and Z100 and Solitaire 2.
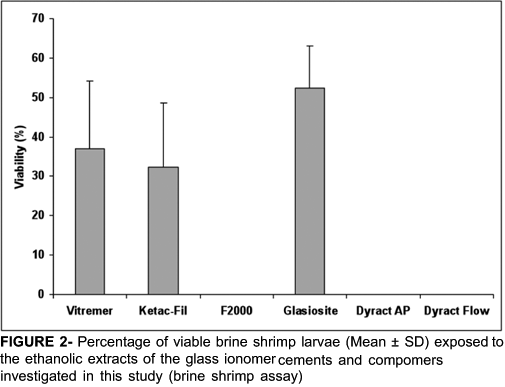
Figure 2 shows the percentage of viable brine shrimp larvae exposed to the ethanolic extracts of glass ionomer and compomer materials investigated in this study. F2000, Dyract AP and Dyract Flow showed 100% lethality. The percentages of viable larvae exposed to the extracts of Ketac-Fil, and Vitremer were 32.3% and 37.0%, respectively. One-way ANOVA revealed highly significant differences between the compomers and GICs (p<0.001). Follow-up comparison between the groups by Tukey's pairwise multi-comparison test (α =0.05) showed that the toxic effect (100% lethality) of the extracts of F2000, Dyract AP and Dyract Flow was significantly greater than that of the GICs. The differences in the toxic effect of Vitremer and Ketac-Fil were not statistically significant.
DISCUSSION
This study was designed to determine the biocompatibility of different dental materials using the brine shrimp larvae assay. Four composite materials with different compositions were tested along with a conventional chemically cured GIC (Ketac-Fil) and a light-cured RMGIC (Vitremer) and two compomers and one flowable compomer. The results of this study support the hypothesis that ethanolic extracts of composites and GICs and compomers are toxic to brine shrimp larvae.
When the extracts of the composite materials were exposed to the brine shrimp larvae, the extract of Synergy showed the least toxicity, followed by the extracts of Solitaire 2, Z100 and P60. The highest percentage of viable brine shrimp larvae was observed after exposure to the extract of Synergy (88%), while the lowest value (50%) was found when the larvae were exposed to extracts of P60, which was thus the most toxic dental material. Interestingly, a previous study of our research group on the cytotoxicity of the same composites using the mitochondrial dehydrogenase activity as an indicator of viability of Balb/C 3T3 fibroblasts5 showed that the ethanolic extracts of these composites exerted much greater levels of cytotoxicity, with percent cell viability ranging from 31% for the extract of Synergy (the most toxic material) to 40% for the extract of Z-100. Although the results of the present study agree with our previous investigation in that Synergy was the least toxic composite, both the ranking and the degree of cytotoxic effect were different when assaying toxicity by the brine shrimp larvae assay and the MTT assay on Balb/C 3T3 fibroblasts. Indeed, in the previous study, fibroblasts exposed to ethanolic extracts of Z100 showed the greatest cell viability (40% viability relative to controls) followed by those exposed to extracts of P60 and Solitaire 2 (38% and 37% viability, respectively). Therefore, the present findings for the toxicity of dental composites to brine shrimp larvae do not exactly match those for the cytotoxicity of dental composites using the MTT assay. It must be kept in mind, however, that the toxic doses for brine shrimp larvae are in the range 10-100 times higher in comparison to cell culture methods22.
The present study also showed that the ethanolic extracts of all three compomers were completely lethal to brine shrimp larvae (0% viability). This is in agreement with the findings of Pelka, et al.18, who tested the extracts of some compomer material powders, including Dyract AP, on brine shrimp larvae, and found that the compomers exhibited higher toxic values than the other tested materials. The reasons for this difference could be the high water solubility of the organic matrix components (hydroxyethyl methacrylate: HEMA and TEGDMA) that may leach out of these materials and exert cytotoxic effects.
Previous reports have identified that the release of HEMA from different compomers may be relevant both to the risk of adverse pulpal responses in patients and to the risk of allergy in patients and dental personnel8. Furthermore, the cytotoxic effect of the compomer F-2000 on gingival fibroblasts has been previously reported and has been linked to a strong depletion of cellular glutathione (GSH) that was rapidly-detectable after 1 h of cell treatment23.These results provide evidence that the cytotoxic property of this dental material is associated with depletion of the glutathione level in gingival fibroblasts23.
Glutathione systems constitute one of the main redox systems that repair oxidized and damaged molecules and thus protect cells and muticellular organisms from toxicants and reactive oxygen species. A recent study investigated the toxicity and metabolism of cyanobacterial toxins in the cysts, nauplii and adults of the brine shrimp2. The presence of the phase II detoxication system glutathione S-transferase (sGST) in these stages was shown using different substrates. Indeed, exposure of adult A. salina to the toxins led to an elevation of GST activity in vivo. Thus the death of the brine shrimp larvae exposed to components leached from compomers may be due to depeletion of glutathione levels in this organism which may lead to a general cytotoxic effect induced by depletion of intracellular thiols and thus a loss of protection of larvae against oxidative stress via antioxidative and redox enzymes.
The present study also found that the GICs (Ketac-Fil and Vitremer) were very toxic, though not as much as the compomers, with viability scores of 32% and 37%, respectively. Furthermore, these materials together with compomers are known to release fluoride19,25. Vitremer RMGIC has been reported to release higher amounts of fluoride than conventional GICs11,14. Indeed, the release of fluoride has been suggested to be the cause of toxicity of GICs10. In addition to the cytotoxic effect of fluorides by its inhibition of protein synthesis, mitochondrial function and depletion of cellular ATP9, a minor genotoxic impact on human mucosa and on peripheral lymphocytes has been reported as well13.
Our results also show that the conventional and resin-modified GICs showed greater toxicity on brine shrimp larvae than the composites. These results are in contrast with those of Tai and Chang24, who found that RMGIC was less toxic than composite resin. Furthermore, other studies have also reported that RMGIC preparations have a low cytotoxicity level, which was most severe at early periods and decreased later1,3.
CONCLUSIONS
Based on the obtained results, the following conclusions may be drawn: 1. The compomers were more toxic to brine shrimp larvae than GICs and composites; 2. Exposure to F2000, Dyract AP and Dyract Flow caused 100% lethality of the brine shrimp larvae; 3. GICs were more toxic to brine shrimp larvae than composites.
Received: March 28, 2008
Modification: April 8, 2008
Accepted: April 29, 2008
- 1. Al-Sabek F, Shostad S, Kirkwood KL. Preferential attachment of human gingival fibroblasts to the resin ionomer Geristore. J Endod. 2005;31:205-8.
- 2. Beattie KA, Ressler J, Wiegand C, Krause E, Codd GA, Steinberg CEW, et al. Comparative effects and metabolism of two microcystins and nodularin in the brine shrimp Artemia salina Aquat Toxicol. 2003;62:219-26.
- 3. Bouillaguet S, Assaf Y, Virgillito M, Holz J, Meyer J-M. In vitro cytotoxicity of resin-reinforced glass ionomer cements. Acta Med Dent Helv. 1999;4:14-9.
- 4. Caughman WF, Caughman GB, Dominy WT, Schuster GS. Glass ionomer and composite resin cements: effects on oral cells. J Prosthet Dent. 1990;63:513-21.
- 5. Darmani H, Al-Hiyasat AS, Milhem MM. Cytotoxicity of dental composites and their leached components. Quintessence Int. 2007;38:789-95.
- 6. Geurtsen W. Biocompatibility of resin-modified filling materials. Crit Rev Oral Biol Med. 2000;11:333-55.
- 7. Geurtsen W, Lehmann F, Spahl W, Leyhausen G. Cytotoxicity of 35 dental resin composite monomers/additives in permanent 3T3 and three human primary fibroblast cultures. J Biomed Mater Res. 1998;41:474-80.
- 8. Hamid A, Okamoto A, Iwaku M, Hume WR. Component release from light-activated glass ionomer and compomer cements. J Oral Rehabil. 1998;25:94-9.
- 9. Jeng JH, Hsieh CC, Lan WH, Chang MC, Lin SK, Hahn LJ, et al. Cytotoxicity of sodium fluoride on human oral mucosal fibroblasts and its mechanisms. Cell Biol Toxicol. 1998;14:383-9.
- 10. Kan KC, Messer LB, Messer HH. Variability in cytotoxicity and fluoride release of resin-modified glass-ionomer cements. J Dent Res. 1997;76:1502-7.
- 11. Kantovitz KR, Pascon FM, Correr GM, Borges AF, Uchoa MN, Puppin-Rontani RM. Inhibition of mineral loss at the enamel/sealant interface of fissures sealed with fluoride- and non-fluoride containing dental materials in vitro. Acta Odontol Scand. 2006;64:376-83.
- 12. Kehe K, Reichl FX, Durner J, Walther U, Hickel R, Forth W. Cytotoxicity of dental composite components and mercury compounds in pulmonary cells. Biomaterials. 2001;22:317-22.
- 13. Kleinsasser NH, Weissacher H, Wallner BC, Kastenbauer ER, Harreus UA. Cytotoxicity and genotoxicity of fluorides in human mucosa and lymphocytes. Laryngorhinootologie. 2001;80:187-90.
- 14. Kosior P, Kaczmarek U. Long term release of fluoride from selected glass-ionomer cements-in vitro study. Ann Acad Med Stetin. 2006;52(Suppl 1):57-60.
- 15. Lonnroth BC, Shahnavaz H. Use of polymer materials in dental clinics, case studies. Swed Dent J. 1997;21:149-59.
- 16. Mathias CGT, Caldwell TM, Maibach HI. Contact dermatitis and gastrointestinal symptoms from hydroxyethylmethylmethacrylate. Brit J Dermatol. 1987;100:447-9.
- 17. Mitra S. Curing reactions of glass ionomer materials. In: Hunt PR, editor. Glass ionomers: the next generation. Proceedings of the Second International Symposium on Glass Ionomers, International Symposia in Dentistry, Philadelphia, PA;1994. p.13-22.
- 18. Pelka M, Danzl C, Distler W, Petschelt A. A new screening test for toxicity testing of dental materials. J Dent. 2000;28:341-5.
- 19. Peng D, Smales RJ, Yip H. In vitro fluoride release from aesthetic restorative materials following recharging with APF gel. Aust Dent J. 2000;45:198-203.
- 20. Ratanasathien S, Wataha JC, Hanks CT, Dennison JB. Cytotoxic interactive effects of dentin bonding components on mouse fibroblasts. J Dent Res. 1995;74:1602-6.
- 21. Sletten GBG, Dahl JE. Cytotoxic effects of extracts of compomers. Acta Odontol Scand. 1999;57:316-22.
- 22. Solis PN, Weight CW, Anderson MM, Gupta MP, Phillipson JD. A microwell cytotoxicity assay using Artemia salina (brine shrimp). Planta Med. 1993;59:250-2.
- 23. Stanislawski L, Soheili-Majd E, Perianin A, Goldberg M. Dental restorative biomaterials induce glutathione depletion in cultured human gingival fibroblast: protective effect of N-acetyl cysteine. J Biomed Mater Res. 2000;51:469-74.
- 24. Tai KW, Chang YC. Cytotoxicity evaluation of perforation repair materials on human periodontal ligament cells in vitro. J Endod. 2000;26:395-7.
- 25. Takahashi K, Emilson CG, Birkhed D. Fluoride release in vitro from various glass-ionomer cements and resin composites after exposure to NaF solutions. Dent Mater. 1993;9:350-4.
- 26. Yoshii E. Cytotoxic effects of acrylates and methacrylates: relationships of monomer structures and cytotoxicity. J Biomed Mater Res. 1997;37:517-24.
Publication Dates
-
Publication in this collection
14 July 2008 -
Date of issue
Aug 2008
History
-
Accepted
29 Apr 2008 -
Reviewed
08 Apr 2008 -
Received
28 Mar 2008