Abstract
Titanium reacts strongly with elements, mainly oxygen at high temperature. The high temperature of titanium laser welding modifies the surface, and may interfere on the metal-ceramic tensile bond strength. OBJECTIVE: The influence of laser welding on the titanium-ceramic bonding has not yet been established. The purpose of this in vitro study was to analyze the influence of laser welding applied to commercially pure titanium (CpTi) substructure on the bond strength of commercial ceramic. The influence of airborne particle abrasion (Al2O3) conditions was also studied. MATERIAL AND METHODS: Forty CpTi cylindrical rods (3 mm x 60 mm) were cast and divided into 2 groups: with laser welding (L) and without laser welding (WL). Each group was divided in 4 subgroups, according to the size of the particles used in airborne particle abrasion: A - Al2O3 (250 µm); B - Al2O3 (180 µm); C - Al2O3 (110 µm); D - Al2O3 (50 µm). Ceramic rings were fused around the CpTi rods. Specimens were invested and their tensile strength was measured at fracture with a universal testing machine at a crosshead speed of 2.0 mm/min and 200 kgf load cell. Statistical analysis was carried out with analysis of variance and compared using the independent t test (p<0.05). RESULTS: Significant differences were found among all subgroups (p<0.05). The highest and the lowest bond strength means were recorded in subgroups WLC (52.62 MPa) and LD (24.02 MPa), respectively. CONCLUSION: Airborne particle abrasion yielded significantly lower bond strength as the Al2O3 particle size decreased. Mechanical retention decreased in the laser-welded specimens, i.e. the metal-ceramic tensile bond strength was lower.
Titanium; Dental porcelain; Tensile strength
Effect of laser welding on the titanium ceramic tensile bond strength
Rodrigo GaloI; Ricardo Faria RibeiroII; Renata Cristina Silveira RodriguesIII; Valéria de Oliveira PagnanoIII; Maria da Glória Chiarello de MattosII
IDDS, MSc, PhD, Department of Prosthodontics and Dental Materials, Ribeirão Preto Dental School, University of São Paulo, Ribeirão Preto, SP, Brazil
IIDDS, MSc, PhD, Full Professor, Department of Prosthodontics and Dental Materials, Ribeirão Preto Dental School, University of São Paulo, Ribeirão Preto, SP, Brazil
IIIDDS, MSc, PhD, Assistant Professor, Department of Prosthodontics and Dental Materials, Ribeirão Preto Dental School, University of São Paulo, Ribeirão Preto, SP, Brazil
Corresponding address Corresponding address: Profa. Dra. Maria da Glória Chiarello de Mattos Departamento de Materiais Dentários e Prótese Faculdade de Odontologia de Ribeirão Preto Universidade de São Paulo Av. do Café s/n, Monte Alegre 14040-904 - Ribeirão Preto, SP - Brasil Fax : +55-16-3602-4780 e-mail: gloria@forp.usp.br
ABSTRACT
Titanium reacts strongly with elements, mainly oxygen at high temperature. The high temperature of titanium laser welding modifies the surface, and may interfere on the metal-ceramic tensile bond strength.
OBJECTIVE: The influence of laser welding on the titanium-ceramic bonding has not yet been established. The purpose of this in vitro study was to analyze the influence of laser welding applied to commercially pure titanium (CpTi) substructure on the bond strength of commercial ceramic. The influence of airborne particle abrasion (Al2O3) conditions was also studied.
MATERIAL AND METHODS: Forty CpTi cylindrical rods (3 mm x 60 mm) were cast and divided into 2 groups: with laser welding (L) and without laser welding (WL). Each group was divided in 4 subgroups, according to the size of the particles used in airborne particle abrasion: A - Al2O3 (250 µm); B - Al2O3 (180 µm); C - Al2O3 (110 µm); D - Al2O3 (50 µm). Ceramic rings were fused around the CpTi rods. Specimens were invested and their tensile strength was measured at fracture with a universal testing machine at a crosshead speed of 2.0 mm/min and 200 kgf load cell. Statistical analysis was carried out with analysis of variance and compared using the independent t test (p<0.05).
RESULTS: Significant differences were found among all subgroups (p<0.05). The highest and the lowest bond strength means were recorded in subgroups WLC (52.62 MPa) and LD (24.02 MPa), respectively.
CONCLUSION: Airborne particle abrasion yielded significantly lower bond strength as the Al2O3 particle size decreased. Mechanical retention decreased in the laser-welded specimens, i.e. the metal-ceramic tensile bond strength was lower.
Key words: Titanium. Dental porcelain. Tensile strength.
INTRODUCTION
Due to the increasingly widespread use of titanium in implant prosthesis, casting techniques have improved to enhance the fabrication of cast commercially pure titanium (CpTi) frameworks15. Nevertheless, the union of titanium to ceramic in metal-ceramic fixed partial dentures remains problematic because of the lack of a strong bond between ceramic and metal substructures1,9.
The well demonstrated properties of titanium include excellent biocompatibility, high corrosion resistance and similar mechanical properties to those of gold alloys12. On the other hand, in addition to presenting a high melting point and high gas affinity (oxygen, hydrogen, nitrogen and carbon), titanium is highly reactive to investment materials, which can be mainly attributed to the development of casting technology for titanium alloys, such as new casting machines, investment materials and the extensively reported advantages of titanium over other base metal13. The most critical aspects for the use of titanium in metal-ceramic restorations are casting and titanium-ceramic bonding10.
One of the characteristics of soldering titanium is that, in the presence of oxygen, an oxide layer is formed and adheres to titanium surface. While this oxide layer confers corrosion resistance11, it decreases considerably the bond strength12 at the metal-ceramic interface4. This layer is sometimes formed by oxides from the investment that react with titanium surface at high temperatures and interferes with the metal-ceramic bonding11. This might lead to dislodgement of ceramic veneers, staining and pigmentation19, which will undermine the clinical success of the restorations.
Despite the improvements in bonding of esthetic materials to cast metal frameworks, failures may still occur. To overcome these shortcomings, soldering of metal-ceramic crowns has become increasingly more frequent. This procedure reduces the time in the laboratory3 because it is performed directly on the cast model in areas very close to the ceramic without causing damage, fracture or color change, due to the use of techniques that do not affect the metal or alloy structure17. Laser welding is one the currently available options. This soldering technique has been extensively evaluated3 and its reported advantages include: high mechanical strength, minimal zone of heat influence and hence lesser deformation, which allows repairs, and use with almost all dental alloys5.
The purpose of this study was to assess titanium-ceramic tensile bond strength after laser welding and airborne particle abrasion with different particle sizes.
MATERIAL AND METHODS
This study was designed to evaluate the bonding characteristics of commercially pure titanium (CpTi, Tritan, grade 1; Dentaurum, Pfozheim, Germany) to ceramic (Noritake Ti22, Noritake Super Porcelain; Noritake Dental Supply Co Ltd, Nagoya, Japan).
Forty CpTi specimens were prepared. To obtain precise dimensions of the CpTi specimens, brass cylindrical rods (3.0 mm diameter x 60 mm length) were used as patterns and invested in a commercial phosphate bonded investment for titanium (Rematitan Plus; Dentaurum) in casting rings. After investment setting and removal of the brass rods, the casting rings were preheated in a furnace (EDG 7000 3P; EDG Equipamentos e Controles Ltda., São Carlos, SP, Brazil). Thereafter, the casting rings were heated according to the manufacturer's instructions.
Casting of the CpTi specimens was performed in an arc vacuum pressure casting machine (Discovery Plasma Ar; EDG Equipamentos e Controles Ltda.), which produces electric arc melting under vacuum and argon-inert atmosphere, with injection of the alloy into the mold by vacuum pressure. The CpTi rods were divested and cleaned with carbide burs (702L; KG Sorensen Ind. Com. Ltd, Barueri, SP, Brazil) followed by and airborne Al2O3 abrasion, which is a standard procedure recommended by ceramic manufacturers. Airborne particle abrasion was performed with particles approximately 110 µm in size for 4 s at 42 psi (2.95 Kg/cm2) air pressure.
The CpTi rods were assigned to 2 groups (n=20): with laser welding (L) and without laser welding (WL). Laser welding was made with a laser welder (Desktop Laser; Dentaurum; 10 ms impulse duration and 1 Hz frequency), according to manufacturer's instruction. Each group was divided into 4 subgroups (n=5), according to the surface treatment (airborne particle abrasion) prior to ceramic firing: A - 250 µm aluminum oxide particles; B - 180 µm aluminum oxide particles; C - 110 µm aluminum oxide particles; D - 50 µm aluminum oxide particles. To ensure that they were airborne-particle abraded in a single direction, the titanium rods were rotated by an engine at approximately 800 rpm during 10 s, maintaining a distance of 2 mm from the metal handpiece of a modified airborne particle abrasion unit (Bijato; F&F, Araraquara, SP, Brazil).
The area that should receive the ceramic was demarcated by 2 silicone slides (Optosil; Bayer, Leverkusen, Germany) and a 2.5 mm thick polyester spacer (Plexiglass; General Electric, Mt. Vernon, IN, USA)3,8 in order to obtain a standard dentin ceramic for each specimen. In all specimens, ceramic application was performed by a single investigator. After the dentin ceramic firing, excess ceramic was removed by grinding on wet silicon carbide paper. The final dimensions of the ceramic rings around the rods were 6.0 mm diameter x 2.0 mm thickness.
The specimens were individually tested to assess the tensile bond strength at metal-ceramic interface. Two measurements of the diameter of the cylindrical titanium rod (perpendicular to each other) were made at two points: immediately above and immediately below the ceramic ring. In addition, the thickness of the ceramic ring was measured at 4 equally spaced points. The average of these measurements was assumed to be the rod diameter and the ceramic ring thickness, respectively. The following equation (Eq. 1) was used to calculate the metal-ceramic bond area: S=π . Φ. e (1), where: S=metal-ceramic bond area; Φ=rod diameter, and e=ceramic ring thickness.
To determine the metal-ceramic tensile bond strength, the ceramic ring was included in a PVC cylinder with gypsum (Type III; Vigodent S.A Ind. e Com, Rio de Janeiro, RJ, Brazil) and a dental surveyor. After gypsum setting, the PVC rings were removed and the specimens were tested to failure in a universal testing machine (EMIC MEM 2000; EMIC Equipamentos e Sistemas de Ensaio Ltd, São José dos Pinhais, PR, Brazil) at crosshead speed of 2.0 mm/min and 200 kgf load cell6.
The rupture peak load was used to calculate the tensile strength, indicator of metal-ceramic bond strength, using the following equation (Eq. 2): T=F/S x 9.8 MPa (2), where: T=tensile strength; F=critical rupture load; S=metal-ceramic bond area.
Data were subjected to ANOVA and independent t test. All statistical analyses and calculations were undertaken using the SPSS 12.0 for Windows statistical software (SPSS Inc, Chicago, IL, USA) at 5% significance level. In each subgroup, the CpTi rod surface subjected to airborne particle abrasion was examined under scanning electron microscopy (SEM). Representative SEM micrographs were obtained from a different region of each specimen to evaluate the metal with and without laser welding.
RESULTS
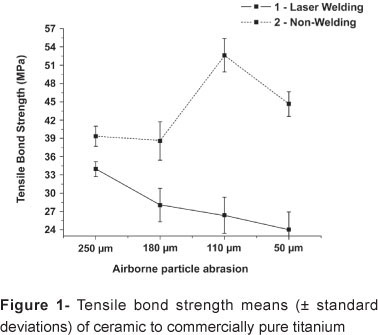
In the laser-welded group, the specimens subjected to airborne particle abrasion with aluminum oxide stream with of different 180, 110 and 50 µm particle size had statistically similar bond strength means (p<0.05) to each other (L) (Figure 1). On the other hand, in the non-welded group, the 110 µm particle size differed significantly (p<0.05), indicating that the bond strength varied with the aluminum oxide particle size.
The SEM micrographs of the surface of non-welded specimens (Figure 2) showed that titanium rods subjected to airborne particle abrasion with larger particles had a more irregular surface, which was beneficial to bond strength. The SEM micrographs of the surface of laser-welded specimens (Figure 3) showed that surface irregularities and bond strength means increased with the increase of Al2O3 particle size.
DISCUSSION
In Prosthodontics, soldering is used to repair casting voids, and connect retainers and/or pontics in fixed partial dentures6. In addition, it is essential that the solder does not interfere with the mechanical properties and clinical longevity of porcelain fused to metal restorations, nevertheless, few repairs might be required. Results from this study showed higher values of the 110 µm aluminum oxide particles in the non-welded group compared with all of the experimental subgroups, which showed the superiority of ceramic adherence to commercially pure titanium (CpTi) in intermediary irregular surface. In addition, there were significant differences in the titanium-ceramic tensile bond strength between the groups with and without laser welding. These differences may be attributed to the fact that the laser-welded CpTi specimens presented less irregular surfaces and consequently lower bond strength to ceramic.
Using laser welding, Dérand5 (1995) found statistically similar values among the tested surfaces and concluded that welding did not interfere with bond strength. Nevertheless, in the present study, the SEM analysis (Figures 1 and 2) showed that airborne particle abrasion induced significant alterations on CpTi surface, producing more irregularities and defects. The laser-welded specimens had a more regular surface, which confirmed the findings of a previous study that reported that the highest surface roughness increased the tensile bond strength as well as oxide formation during airborne particle abrasion, which improves the ceramic bonding14. However, in the present study, the highest tensile bond strength was recorded with aluminum oxide airborne particle abrasion with 110 and 180 µm.
The lower values obtained with the welded group can be explained by the titanium surface and alterations on the physical and mechanical properties of the titanium that might have occurred after laser welding. The metal area that received the laser welding showed the lowest alterations after airborne-particle abrasion with the aluminum oxide particles. These results indicate that airborne-particle abrasion of titanium after laser welding requires a different technique, involving for example particle size and design, from that normally used for conventional dental alloy7. However, because of titanium reactivity, even when the oxide layer was removed from the surface, re-oxidation of the titanium proceeded immediately20, and this can be a problem for titanium adhesion.
Other possible explanation for these results would be the ability of ceramic to adapt to macroscopic irregularities. According to Oshida, et al.16 (1997), excessive roughness may reduce the contact of ceramic with metal because the viscous ceramic needs to interlock the macroscopic irregularities on the metal surface. Irregularly shaped porosities at the interface may act concentrating stress and reducing bond strength because the stress release might cause metal deformation and subsequent decrease of ceramic bond strength18. Additionally, aluminum oxide particles might contaminate ceramic and disturb its surface integrity and texture, producing unfavorable contact angles and playing an important role on the weakening of the titanium ceramic bond strength17. Furthermore, ultrasonic cleaning of the airborne-particle abraded samples resulted in a slight decrease in the amount of aluminum18. However, based on the variations within the groups the reductions found in the surface are statistically significant.
The findings of this study are consistent with those of a previous investigation10, which found aluminum oxide particles embedded on titanium surface subjected to airborne particle abrasion. Gilbert, et al.8 (1994) reported that the existence of alumina particles embedded on machined titanium surfaces might have contributed to the poor adhesion to ceramic by reducing the titanium area available for ceramic bonding.
Another drawback for bonding is titanium oxidation at high temperatures11. The oxidative behavior of CpTi on the formation of a film between metal and ceramic has been proven dependent on casting temperature and oxidizing atmosphere. Another study that evaluated the bonding at titanium ceramic interface showed that the existence of a certain amount of TiO2 in this region leads to ceramic fractures4. It is also important to realize that even though this is an in vitro study, the clinical implications of the results may be important. Future clinical studies are needed to verify the findings of this study. To better understand titanium-ceramic bonding, further investigations with x-ray photoelectron spectroscopy to determine the oxidation status of titanium, and transmission electron microscopy to closely analyze structure (critical defects) and composition at the titanium-ceramic interfaces are needed.
CONCLUSIONS
This study evaluated the effect of laser welding on the titanium ceramic tensile bond strength and showed higher tensile bond strength in the specimens subjected to aluminum oxide airborne abrasion with intermediate particle size, especially in the non-welded group. The non-welded group had a more consistent titanium ceramic bonding, which might increase the longevity of metal-ceramic restorations.
ACKNOWLEDGMENTS
The authors would like to thank CNPq for supporting this research.
Received: August 06, 2009
Modification: March 17, 2010
Accepted: March 26, 2010
- 1- Al Hussaini I, Al Wazzan KA. Effect of surface treatment on bond strength of low-fusing porcelain to commercially pure titanium. J Prosthet Dent. 2005;94:350-6.
- 2- Andrade Tarozzo LS, Chiarello de Mattos M G, Faria Ribeiro R, Semprini M. Comparison of retentive systems for composites used as alternatives to porcelain in fixed partial dentures. J Prosthet Dent. 2003;89:572-8.
- 3- Apotheker H, Nishimura I, Seerattan C. Laser-welded vs soldered nonprecious alloy dental bridges: a comparative study. Lasers Surg Med. 1984;4:207-13.
- 4- Cai Z, Bunce N, Nunn ME, Okabe T. Porcelain adherence to dental cast CP titanium: effects of surface modifications. Biomaterials. 2001;22:979-86.
- 5- Dérand T. Porcelain bond to laser-welded titanium surfaces. Dent Mater. 1995;11:93-5.
- 6- Galindo DF, Ercoli C, Graser GN, Tallents RH, Moss ME. Effect of soldering on metal-porcelain bond strength in repaired porcelain-fused-to-metal castings. J Prosthet Dent. 2001;85:88-94.
- 7- Galo R, Ribeiro RF, Rodrigues RC, Pagnano V O, Mattos M G. Effect of laser welding on the titanium composite tensile bond strength. Braz Dent J. 2009;20:403-9.
- 8- Gilbert JL, Covey DA, Lautenschlager EP. Bond characteristics of porcelain fused to milled titanium. Dent Mater. 1994;10:134-40.
- 9- Giordano RA. Dental ceramic restorative systems. Compend Contin Educ Dent. 1996;17:779-86.
- 10- Hamanaka H, Doi H, Yoneyama T, Okuno O. Dental casting of titanium and Ni-Ti alloys by a new casting machine. J Dent Res. 1989;68:1529-33.
- 11- Hruska AR, Borelli P. Quality criteria for pure titanium casting, laboratory soldering, intraoral welding, and a device to aid in making uncontaminated castings. J Prosthet Dent. 1991;66:561-5.
- 12- Ida K, Togaya T, Tsutsumi S, Suzuki M. Mechanical properties of pure titanium and titanium alloys evaluation for dental casting metals. J Jpn Dent Mater. 1983;2:765-71.
- 13- Kimura H, Horng CJ, Okazaki M, Takahashi J. Oxidation effects on porcelain-titanium interface reactions and bond strength. Dent Mater J. 1990;9:91-9.
- 14- Könönen M, Kivilahti J. Bonding of low-fusing dental porcelain to commercially pure titanium. J Biomed Mater Res. 1994;28:1027-35.
- 15- Okabe T, Hero H. The use of titanium in dentistry. Cells and Materials. 1995;5:211-30.
- 16- Oshida Y, Fung LW, Isikbay SC. Titanium-porcelain system. Part II: Bond strength of fired porcelain on nitrided pure titanium. Biomed Mater Eng. 1997;7:13-34.
- 17- Oshida Y, Reyes MJ. Titanium-porcelain system. Part IV: some mechanistic considerations on porcelain bond strengths. Biomed Mater Eng. 2001;11:137-42.
- 18- Reyes MJ, Oshida Y, Andres CJ, Barco T, Hovijitra S, Brown D. Titanium-porcelain system. Part III: effects of surface modification on bond strengths. Biomed Mater Eng. 2001;11:117-36.
- 19- Taira Y, Imai Y. Primer for bonding resin to metal. Dent Mater. 1995;11:2-6.
- 20- Taira, Y, Matsumura H, Yoshida K, Tanaka T, Atsuta M. Influence of surface oxidation of titanium on adhesion. J Dent. 1998;26:69-73.
Publication Dates
-
Publication in this collection
27 May 2011 -
Date of issue
Aug 2011
History
-
Received
06 Aug 2009 -
Accepted
26 Mar 2010 -
Reviewed
17 Mar 2010