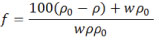Abstract
OBJECTIVES: To evaluate the effect of additives on the water sorption characteristics of Bis-GMA based copolymers and composites containing TEGDMA, CH3Bis-GMA or CF3Bis-GMA. MATERIAL AND METHODS: Fifteen experimental copolymers and corresponding composites were prepared combining Bis-GMA and TEGDMA, CH3Bis-GMA or CF3Bis-GMA, with aldehyde or diketone (24 and 32 mol%) totaling 30 groups. For composites, barium aluminosilicate glass and pyrogenic silica was added to comonomer mixtures. Photopolymerization was effected by 0.2 wt% each of camphorquinone and N,N-dimethyl-p-toluidine. Specimen densities in dry and water saturated conditions were obtained by Archimedes' method. Water sorption and desorption were evaluated in a desorption-sorption-desorption cycle. Water uptake (%WU), water desorption (%WD), equilibrium solubility (ES; µg/mm³), swelling (f) and volume increase (%V) were calculated using appropriate equations. RESULTS: All resins with additives had increased %WU and ES. TEGDMA-containing systems presented higher %WU, %WD, ES, f and %V values, followed by resins based on CH3Bis-GMA and CF3Bis-GMA. CONCLUSIONS: Aldehyde and diketone led to increases in the water sorption characteristics of experimental resins.
Water storage; Solubility; Bis-GMA polymer; Composite resins; Polymerization
ORIGINAL ARTICLES
Water sorption of CH3- and CF3-Bis-GMA based resins with additives
Anuradha PrakkiI; Renato CilliII; Ian Matos VieiraIII; Kristina DudumasIV; José Carlos PereiraV
IDDS, MSc, PhD, Department of Clinical Sciences, Restorative Dentistry, Faculty of Dentistry, University of Toronto, Toronto, Canada
IIDDS, MSc, PhD, Department of Dental Materials and Operative Dentistry, Faculty of Dentistry University of Fortaleza, Fortaleza, CE, Brazil
IIIDDS, MSc, PhD, Department of Restorative Dentistry, Piracicaba School of Dentistry, State University of Campinas, Piracicaba, SP, Brazil
IVUndergraduate student, Department of Clinical Sciences, Restorative Dentistry, Faculty of Dentistry, University of Toronto; Department of Human Biology, University of Toronto, Toronto, Canada
VDDS, PhD Department of Operative Dentistry, Endodontics and Dental Materials, Bauru School of Dentistry, University of São Paulo, Bauru, SP, Brazil
Corresponding address Corresponding address: Anuradha Prakki Restorative Dentistry University of Toronto Faculty of Dentistry Room 354D, 124 - Edward St Toronto - ON - Canada - M5G 1G6 Phone: 416-979-4932 - ext.4389 e-mail: anuradha.prakki@dentistry.utoronto.ca
ABSTRACT
OBJECTIVES: To evaluate the effect of additives on the water sorption characteristics of Bis-GMA based copolymers and composites containing TEGDMA, CH3Bis-GMA or CF3Bis-GMA.
MATERIAL AND METHODS: Fifteen experimental copolymers and corresponding composites were prepared combining Bis-GMA and TEGDMA, CH3Bis-GMA or CF3Bis-GMA, with aldehyde or diketone (24 and 32 mol%) totaling 30 groups. For composites, barium aluminosilicate glass and pyrogenic silica was added to comonomer mixtures. Photopolymerization was effected by 0.2 wt% each of camphorquinone and N,N-dimethyl-p-toluidine. Specimen densities in dry and water saturated conditions were obtained by Archimedes' method. Water sorption and desorption were evaluated in a desorption-sorption-desorption cycle. Water uptake (%WU), water desorption (%WD), equilibrium solubility (ES; µg/mm3), swelling (f) and volume increase (%V) were calculated using appropriate equations.
RESULTS: All resins with additives had increased %WU and ES. TEGDMA-containing systems presented higher %WU, %WD, ES, f and %V values, followed by resins based on CH3Bis-GMA and CF3Bis-GMA.
CONCLUSIONS: Aldehyde and diketone led to increases in the water sorption characteristics of experimental resins.
Key words: Water storage. Solubility. Bis-GMA polymer. Composite resins. Polymerization.
INTRODUCTION
Dilution of Bis-GMA (bisphenol-A-glycidyl dimethacrylate) with the more hydrophilic TEGDMA (triethyleneglycol dimethacrylate) is known to cause adverse effects on the resin matrix due to the increase in water sorption and polymerization shrinkage1,2. To overcome these effects, studies have been directed toward developing low viscosity, more hydrophobic Bis-GMA analogs such as the hydroxyl-free propoxylated Bis-GMA (CH3Bis-GMA) and propoxylated fluorinated Bis-GMA (CF3Bis-GMA), as replacements for TEGDMA in Bis-GMA mixtures6,10. The properties of Bis-GMA copolymers and composites containing these developmental monomers have been evaluated. As TEGDMA is a more flexible monomer with higher degree of conversion, the Young's modulus and flexural strength of Bis-GMA/TEGDMA systems have higher values11.
In an attempt to improve properties of methacrylate resins, aldehyde-propanal (propionaldehyde) or diketone-diacetyl (2,3-butanedione) have been added as potential crosslinking agents with appreciable success12,13,15,16. When these were incorporated into Bis-GMA/Bis-GMA analog systems, a significant increase was noted in the degree of conversion and mechanical properties of a Bis-GMA/CH3Bis-GMA based composite17. Moreover, Bis-GMA/CF3Bis-GMA copolymers had increased glass transition temperature values18. One of the effects of propanal or diacetyl on resin properties maybe related to their reduced viscosities17. Diketone may also function as a free radical photoinitiator23. Furthermore, it has been suggested that the effect of additives on resin properties may also be due to chain transfer reactions14. The possibility should, however, be considered that when unreacted, some of these small molecules may leach from copolymers. Due to the potential impact on the biocompatibility of polymer and to help elucidate the mechanism of action of these molecules, it is appropriate to evaluate their elution into aqueous media. It was the objective of this study to evaluate the influence of propanal or diacetyl on the water sorption characteristics of composites and copolymers based on Bis-GMA diluted with TEGDMA and the Bis-GMA analogs.
MATERIAL AND METHODS
Material
Bis-GMA, TEGDMA, camphorquinone (CQ), hybrid treated fillers (Kerr Corp., Orange, CA, USA), N,N-dimethyl-p-toluidine (DMPT, Alfa Aesar, Ward Hill, MA, USA), propionaldehyde and 2,3-butanedione (Acros Organics, Morris Plains, NJ, USA) were all used as received. The Bis-GMA analogs, CH3Bis-GMA (2,2-bis[4-(2-methacryloxyprop-1-oxy)phenyl]propane) and CF3Bis-GMA (1,1,1,3,3,3-hexafluoro-2,2-bis[4-(2-methacryloxyprop-1-oxy)phenyl]propane), were synthesized, purified and stored according to reported methods7. Synthesized monomers were characterized by 1H-NMR and 13C-NMR, providing spectra that were consistent with the expected products.
Composites and copolymers formulation and specimen preparation
Fifteen experimental resin formulations, with and without fillers, were prepared comprising 30 groups. For control groups containing the resins Bis-GMA/TEGDMA (BT), Bis-GMA/CH3Bis-GMA (BCH3), or Bis-GMA/CF3Bis-GMA (BCF3), the comonomer ratio (mol%) was 25/75. For the other groups containing Bis-GMA/diluent monomers/propanal (A) or diacetyl (D), the comonomer ratios (mol%) were 19/57/24 and 17/51/32 respectively. Resins were activated for visible light polymerization by CQ (0.2 wt%) and DMPT (0.2 wt%). Matrix resins were loaded (60 wt%) with silanized submicron hybrid filler of barium aluminosilicate glass and pyrogenic silica (average particle size of 0.7 µm), using gentle hand mixing. All materials were prepared under safe yellow light. For each group, three disc-shaped specimens were fabricated (7 mm in diameter x 2 mm high) using silicone molds. Unpolymerized material was applied to the molds and covered on top with a polyester matrix strip and a rigid glass microscope slide. Samples were light polymerized (Astralis 5, Ivoclar/Vivadent, Schaan, Liechtenstein; 480 mW/cm2) for 40 s.
Specimen volume and density measurements
The diameter and thickness of samples were measured at five points each using a digital caliper (Mitutoyo Corporation, Tokyo, Japan). The average measurements were used to calculate their volume (mm3). The densities of all samples were measured by Archimedes' method under dry (ρ0) and water saturated conditions (ρ) using an analytical balance (AT261 Delta range, Mettler Toledo GmbH, Gieben, Germany).
Water sorption and solubility
Dry specimens were stored in desiccators containing silica gel at 37ºC. After 24 h they were weighed on an analytical balance to 0.00001 g accuracy (AT261 Delta range, Mettler Toledo GmbH, Gieben, Germany). This weighing cycle was repeated until a constant mass (mi) was achieved. The discs were then individually immersed in distilled water at 37ºC. Every 24 h they were removed from the water, carefully wiped with an absorbent paper, weighed and returned to the water. The water uptake was recorded until there was no significant change in weight (ms). The samples were transferred back to the desiccators (37ºC) and a process similar to the one described above was performed for desorption (md). The percentage equilibrium water uptake (%WU) and water desorption (%WD) were calculated using the following equations:
Where, ms represents the weight of the water saturated specimens at equilibrium, and md represents the weight of specimen after desorption at equilibrium. Additionally, the equilibrium solubility (ES; µg/mm3) was calculated as follows:
The fraction of water that contributes to an increase in swelling (f), in dimensionless form, was calculated using the equation shown below8:
Where, f is the fraction of water that contributes to an increase in swelling and ρ0,ρ and w represent the densities of dry specimen, saturated specimen and wt% water, respectively. Using the above mentioned equation the % volume increase (V) was calculated as8:
V = wf ρ0/ρw
Where, ρw represents the density of water.
Descriptive analysis of data8,22 was performed according to the above mentioned equations.
RESULTS
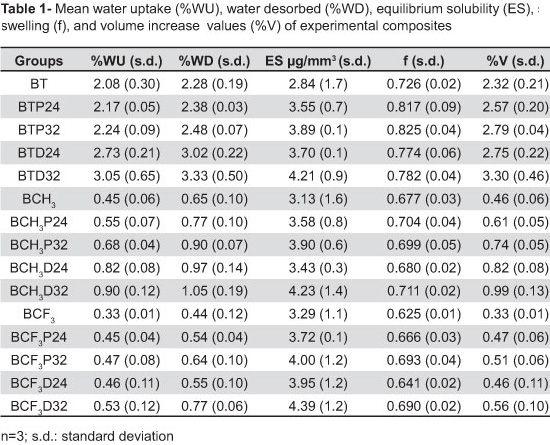
Mean %WU, %WD, ES, f and %V values of tested composites and copolymers, during desorption-sorption-desorption, are shown in Tables 1 and 2 respectively. The results showed that the systems based on Bis-GMA/TEGDMA had the highest %WU (2.08% and 3.65%), followed by the Bis-GMA/CH3Bis-GMA (0.45% and 0.66%) and Bis-GMA/CF3Bis-GMA (0.33% and 0.45%) systems. In the presence of additives all systems had an increase in %WU. Moreover, all evaluated systems had %WD values higher than %WU.
The highest ES values were observed for composites and copolymers of the Bis-GMA/CF3Bis-GMA (3.29 µg/mm3 and 3.69 µg/mm3) system followed by Bis-GMA/CH3Bis-GMA (3.13 µg/mm3 and 3.51 µg/mm3 and) and Bis-GMA/TEGDMA (2.84 µg/mm3 and 3.23 µg/mm3). In the presence of additives, all systems had higher ES, and this was even higher with the incorporation of 32 mol% additives. Composites and copolymers based on TEGDMA had higher f (0.72 and 0.91) and %V (2.32 and 3.65) followed by those based on CH3Bis-GMA (f=0.67 and 0.88; %V=0.46 and 0.67) and CF3Bis-GMA (f=0.62 and 0.81; %V=0.33 and 0.46). Incorporation of additives also led to an increase in values of both composites and copolymers. Overall, the composites presented lower values for water sorption characteristics than did the unfilled counterparts.
DISCUSSION
The systems based on Bis-GMA/TEGDMA had the highest %WU, followed by the Bis-GMA/CH3Bis-GMA and Bis-GMA/CF3Bis-GMA systems. Among the evaluated monomers, the most hydrophilic is TEGDMA, which contains hydrophilic ether linkages5, followed by Bis-GMA with pendant hydroxyl groups that form hydrogen bonds with water molecules. The %WU for their neat monomers was reported to be 3.05% and 6.0%, respectively8. The Bis-GMA analogs had -OH groups replaced by -CH3, and the molecule that contains the fluorinated core group is even more hydrophobic21. These neat monomers were reported to present %WU of 0.58 and 0.18, respectively20.
In addition, it is known that the TEGDMA molecule is more flexible and creates a much denser polymer network than Bis-GMA22 and its analogs. Studies on the structural heterogeneity of polydimethacrylate based networks have reported that the more densely cross-linked the network the more heterogeneous is the structure3,19. This heterogeneity seems to favor the higher %WU of TEGDMA. That is, in such a network, the space created between the polymer clusters is larger and may accommodate a larger quantity of water22. The %WU of Bis-GMA/TEGDMA copolymer has been previously evaluated, and reported to be higher (4.35%) than the present values for similar systems8. Differences in comonomer ratios, type of polymerization and the tertiary amine that had been used, DMAEMA (dimethyl aminoethyl methacrylate) instead of DMPT, may explain the variation in results. In the presence of propionaldehyde or 2,3-butanedione all comonomer systems presented an increase in %WU. This suggests that they are hydrophilic components added to the resins, which is confirmed by that fact that resins that had additives added in the ratio of 32 mol% presented the highest %WU values. In this study, 24 mol% and 32 mol% additive ratios were incorporated into experimental resins because they were shown to promote significant improvements in the mechanical properties14.
For all evaluated systems %WD are higher than %WU values. This is because unreacted monomers or oligomers are expected to be leached together with water. The values of ES correspond to the amount of these unreacted species that are water leachable. During polymerization, they are either trapped inside the microgels between polymer chains and are absorbed into the surrounding network, or they are trapped in micropores. The latter is more susceptible to leaching out22. Components are eluted from resins via diffusion of molecules through the matrix. The extent and rate of elution is dependent upon factors such as the degree of conversion of the polymer network, characteristics of extraction solvent, and size and chemical characteristics of leachable species4. It has been reported that elution of leachable components into water varies between 0.25 and 3.0% of the initial weight of polymerized resin composites4, which is in line with the present findings (0.44% to 3.33%).
According to the present results, Bis-GMA/CF3Bis-GMA systems exhibited the highest ES values, followed by Bis-GMA/CH3Bis-GMA and Bis-GMA/TEGDMA. These values follow the sequence from lower to higher degree of conversion, respectively (i.e., fewer amounts of unreacted monomers) that were previously reported to be (% composites/copolymers): 48.6/54.3 (Bis-GMA/CF3Bis-GMA); 53.3/58.0 (Bis-GMA/CH3Bis-GMA); 65.6/70.3 (Bis-GMA/TEGDMA)17. With the fact that TEGDMA is a more linear and shorter molecule, one can consider that the size and structure of the higher molecular weight analogs may have prevented their systems from leaching to a higher extent. It can also be seen from data that unfilled specimens presented higher %WU and ES than composites. This may be due to the higher content of organic matrix. It is in agreement with other studies that reported that materials with lower filler content i.e., higher resin content, have higher sorption9.
In the presence of additives, the results show higher solubility for the three evaluated systems. This was shown to be even higher with the incorporation of 32 mol% additive. Considering that additives promote increase in resin conversion it can be assumed that they are being leached to the aqueous media. One of the effects of these agents in the improvement of resin mechanical properties may be related to the fact that, when added to resins, their very low viscosities cause a reduction on the comonomer systems. Propionaldehyde and 2,3-butanedione viscosities were reported to be 0.33 cps and 0.83 cps, respectively18. This would delay the formation of crosslinked networks increasing mobility of reactive species and consequently the conversion. Diketone may additionally function as a free radical photoinitiator as does camphorquinone23. Furthermore, they may act as chain transfer agents reacting with free radicals in a growing polymer by hydrogen abstraction14. This leaves a free radical residing on the attacked species, which may then itself go on to attack methacrylate double bonds. In these reactions, the new small sized radical, consisting of the remaining chain transfer molecule can reach double bonds by diffusion and react with those which, because of steric hindrance, would not have reacted.
The retention of different additive concentrations was evaluated for Bis-GMA/TEGDMA and UEDMA/HEMA systems at an early stage14. For Bis-GMA/TEGDMA, the amount of substance (residual unreacted monomer + additives) that was lost from the specimens varied from 0.56 (baseline) to 2.62 wt% (32 mol% propanal) and from 0.56 to 1.86 wt% (32 mol% diacetyl). The fraction of additives that could evaporate from specimens of resins containing 8-40mol% was 0.18 to 0.46 wt% propanal and 0.13 to 0.30 wt% diacetyl. With a content of up to 24 mol%, there was no significant difference in substance loss between additive-containing materials and baseline condition (without additives). Although material loss measurements were taken for a period of up to 6 months, these values represent estimation over an infinite time interval (M∞), i.e., the maximum material loss possible. It has been stated that the incorporation of propanal/aldehyde into resins without significant increase in M∞ is indicative of the additives having been bound in the polymer to some extent. It was concluded that biocompatibility of resin composites with a content of 24 mol% additives may be similar to that of proprietary composites.
The fraction of uptaken water which contributes to an increase in swelling is f and ranged from 0.62 to 0.82 for composites and from 0.81 to 0.94 for copolymers. The corresponding %V ranged from 0.33 to 3.30 for composites, and from 0.46 to 4.50 for copolymers. Systems based on TEGDMA presented higher f and %V followed by those based on CH3Bis-GMA and CF3Bis-GMA. Incorporation of additives led to an increase in both composite and copolymer values. In general, both results followed the same trend i.e., increased f contributed to higher %V. %WU by dental polymers may affect clinical performance by causing volumetric changes which can induce swelling stresses in the resin and/or peeling stresses in bonding interfaces. It is interesting to note that the provisional significance given to (1 - f) is that it corresponds to the fraction of water accommodated in "microvoids" i.e., without swelling24.
CONCLUSIONS
An increase in %WU of comonomers was observed in the presence of additives, this being higher for the ratio of 32 mol%. Due to hydrophilicity and network characteristics, resins based on TEGDMA presented higher sorption and solubility values, followed by resins based on CH3Bis-GMA and CF3Bis-GMA. Present findings will aid future studies on the biocompatibility of these developmental materials.
ACKNOWLEDGEMENTS
This study was supported by Connaught Start-up award and funds from Faculty of Dentistry, University of Toronto, Canada, and by Fundação de Amparo à Pesquisa do Estado de São Paulo - FAPESP (2008/06170-7), Brazil.
Received: November 18, 2010
Modification: August 04, 2011
Accepted: September 15, 2011
- 1- Anseth KS, Newman SM, Bowman CN. Polymeric dental composites: properties and reaction behavior of multimethacrylate dental restorations. Adv Polym Sci. 1995;122:177-217.
- 2- Braden M. The formulation of composite filling materials. Oper Dent. 1978;3:97-102.
- 3- Elliott JE, Lovell LG, Bowman CN. Primary cyclization in the polymerization of bis-GMA and TEGDMA: a modeling approach to understanding the cure of dental resins. Dent Mater. 2001;17:221-9.
- 4- Ferracane JL. Elution of leachable components from composite. J Oral Rehabil. 1994;21:441-52.
- 5- Ferracane JL. Hygroscopic and hydrolytic effects in dental polymer networks. Dent Mater. 2006;22:211-22.
- 6- Kalachandra S. Dimethacrylate monomers and polymers as biomaterials. Polym Sci. 1997;2:95-106.
- 7- Kalachandra S, Taylor DF, Deporter CD, Grubbs HJ, McGrath JE. Polymeric materials for composite matrices in biological environments. Polymer. 1993;34:778-82.
- 8- Kalachandra S, Turner DT. Water sorption of polymethacrylate networks: Bis-GMA/TEGDMA copolymers. J Biomed Mater Res. 1987;21:329-38.
- 9- Oysaed H, Ruyter IE. Water sorption and filler characteristics of composites for use in posterior teeth. J Dent Res. 1986;11:1315-8.
- 10- Pereira SG, Nunes TG, Kalachandra S. Low viscosity dimethacrylate comonomer compositions [Bis-GMA and CH3Bis-GMA] for novel dental composites; analysis of the network by stray-field MRI, solid state NMR and DSC & FTIR. Biomaterials. 2002;23:3799-806.
- 11- Pereira SG, Osorio R, Toledano M, Nunes TG. Evaluation of two Bis-GMA analogues as potential monomer diluents to improve the mechanical properties of light-cured composite resins. Dent Mater. 2005;21:823-30.
- 12- Peutzfeldt A. Quantity of remaining double bonds of diacetyl-containing resins. J Dent Res. 1994;73:511-5.
- 13- Peutzfeldt A. Quantity of remaining double bonds of propanal-containing resins. J Dent Res. 1994;73:1657-62.
- 14- Peutzfeldt A. Retention of propanal and diacetyl in experimental resins. Acta Odontol Scand. 1997;55:94-100.
- 15- Peutzfeldt A, Asmussen E. Influence of aldehydes on selected mechanical properties of resin composites. J Dent Res. 1992;71:1522-4.
- 16- Peutzfeldt A, Asmussen E. Influence of ketones on selected mechanical properties of resin composites. J Dent Res. 1992;71:1847-50.
- 17- Prakki A, Pereira PN, Kalachandra S. Effect of propionaldehyde and 2,3-butanedione additives on the mechanical properties of Bis-GMA analog-based composites. Dent Mater. 2009;25:26-32.
- 18- Prakki A, Tallury P, Mondelli RFL, Kalachandra S. Influence of additives on the properties of Bis-GMA/Bis-GMA analog comonomers and corresponding copolymers. Dent Mater. 2007;23:1199-204.
- 19- Rey L, Duchet J, Galy J, Sautereau H, Vouagner D, Carrion L. Structural heterogeneities and mechanical properties of vinyl/dimethacrylate networks synthesized by thermal free radical polymerisation. Polymer. 2002;43:4375-84.
- 20- Sankarapandian M, Shobha HK, Kalachandra S, McGrath JE, Taylor DF. Characterization of some aromatic dimethacrylates for dental composite applications. J Mater Sci Mater Med. 1997;8:465-8.
- 21- Sankarapandian M, Xu Q, McGrath JE, Taylor DF, Kalachandra S. Influence of resin molecular structure on the processibility and physical behavior of a dental composite matrix. J Adv Mater. 1996;28:59-63.
- 22- Sideridou I, Tserki V, Papanastasiou G. Study of water sorption, solubility and modulus of elasticity of light-cured dimethacrylate-based dental resins. Biomaterials. 2003;24:655-65.
- 23- Sun GJ, Chae KH. Properties of 2,3-butanedione and 1-phenyl-1,2-propanedione as new photosensitizers for visible light cured dental resin composites. Polymer. 2000;41:6205-12.
- 24- Turner DT. Polymethyl methacrylate plus water: sorption kinetics and volumetric changes. Polymer. 1982;23:197-202.
Publication Dates
-
Publication in this collection
27 Sept 2012 -
Date of issue
Aug 2012
History
-
Received
18 Nov 2010 -
Accepted
15 Sept 2011 -
Reviewed
04 Aug 2011