Abstract
Natural (NV) and Cobalto60-irradiated (IrV) Crotalus durissus terrificus venom were used to evaluate serum production capacity of sheep and possible hematological and biochemical effects. Freeze-dried venom aliquots were diluted in acidified saline solution (NaCl 150 mM, pH 3.0) and irradiated by a Cobalt 60 source at a dose of 5.54 x 102 Gy/h and a concentration of 2.000 Gy. Twelve sheep were divided into two groups of six animals. One group received irradiated venom (IrV) and the other natural venom (NV). Three antigen doses (venom) were administered at monthly intervals. Blood samples were collected weekly for analysis of serum neutralization potency and capacity, complete blood count (CBC), total plasma protein, fibrinogen, albumin, and globulin. At the end of the experiment, the animals were challenged with a LD50 for sheep and showed no signs of envenoming. The two groups did not present clinical alterations. Results of the total leukocyte count did not present interaction or time factor effect for both groups, but there was a different action between them, with the NV group presenting more cells than the IrV group. The leukocyte increase to 13,000/ml indicates that slight leukocytosis occurred in the week after the first inoculation in the NV group. There was no statistically significant difference between groups in the absolute count of segmented neutrophils, eosinophils, and lymphocytes but there were statistically significant oscillations in values at the different collecting times. The NV group presented an increase in the absolute neutrophil count after the first inoculation that persisted for 5 weeks. In the IrV group, the increase in neutrophils occurred only in the first week returning to normal in the following weeks. The alterations in the neutrophil count are indicative of systemic inflammatory response related to cytokine release; response was more marked in the NV group, showing its greater toxicity.
hematology; gamma radiation; irradiated crotalic venom; crotalic antiserum; ovine; Crotalus durissus terrificus
ORIGINAL PAPER
Hematological changes in sheep inoculated with natural and Cobalt60-irradiated Crotalus durissus terrificus venom (Laurenti, 1768)
Netto D. P.I; Chiacchio S. B.II; Bicudo P. L.II; Alfieri A. A.I; Balarim M. R. S.I; Nascimento N.III
IFaculdade de Medicina Veterinária e Zootecnia, Departamento de Medicina Veterinária Preventiva, Centro de Ciências Agrárias, Universidade Estadual de Londrina, Londrina, Paraná, Brasil
IIFaculdade de Medicina Veterinária e Zootecnia, Universidade Estadual Paulista, Botucatu, São Paulo, Brasil
IIISupervisão de Radiobiologia, Instituto de Pesquisas Energéticas e Nucleares IPEN/CNEM, Universidade de São Paulo, São Paulo, Brasil
Correspondence Correspondence to D. P. Netto Departamento de Medicina Veterinária Preventiva, Centro de Ciências Agrárias Universidade Estadual de Londrina, Campus Universitário 86051-990, Londrina, Paraná, Brasil E-mail: rnetto@uel.br
ABSTRACT
Natural (NV) and Cobalto60-irradiated (IrV) Crotalus durissus terrificus venom were used to evaluate serum production capacity of sheep and possible hematological and biochemical effects. Freeze-dried venom aliquots were diluted in acidified saline solution (NaCl 150 mM, pH 3.0) and irradiated by a Cobalt 60 source at a dose of 5.54 x 102 Gy/h and a concentration of 2.000 Gy. Twelve sheep were divided into two groups of six animals. One group received irradiated venom (IrV) and the other natural venom (NV). Three antigen doses (venom) were administered at monthly intervals. Blood samples were collected weekly for analysis of serum neutralization potency and capacity, complete blood count (CBC), total plasma protein, fibrinogen, albumin, and globulin. At the end of the experiment, the animals were challenged with a LD50 for sheep and showed no signs of envenoming. The two groups did not present clinical alterations. Results of the total leukocyte count did not present interaction or time factor effect for both groups, but there was a different action between them, with the NV group presenting more cells than the IrV group. The leukocyte increase to 13,000/ml indicates that slight leukocytosis occurred in the week after the first inoculation in the NV group. There was no statistically significant difference between groups in the absolute count of segmented neutrophils, eosinophils, and lymphocytes but there were statistically significant oscillations in values at the different collecting times. The NV group presented an increase in the absolute neutrophil count after the first inoculation that persisted for 5 weeks. In the IrV group, the increase in neutrophils occurred only in the first week returning to normal in the following weeks. The alterations in the neutrophil count are indicative of systemic inflammatory response related to cytokine release; response was more marked in the NV group, showing its greater toxicity.
Key words: hematology, gamma radiation, irradiated crotalic venom, crotalic antiserum, ovine, Crotalus durissus terrificus.
INTRODUCTION
Crotalus durissus terrificus snakes account for 14% of human envenomings in Brazil (17,18). Heterologous sera are used in the treatment of snake envenomings; these sera are usually obtained from venom-hyperimmunized horses. Administration of horse serum-derived F(ab)2 immunoglobulin concentrates constantly causes a type-III hypersensitivity reaction mediated by immune complexes; these are probably due to molecule bivalence and high circulating IgGT titer present in immunized horses (7). According to Fan and França (9), reaction to serotherapy constitutes an immune process where heterologous proteins are released into the organism, a mechanism of hypersensitivity to venom antidote.
The problem of choosing an adequate treatment for horse-serum sensitive patients led to the development of immunization techniques using alternative animals such as rabbits, goats, and sheep, which have shown good results in serum production (6,16,20,21,22,23).
Sjostrom et al. (23) produced antivenoms in sheep and compared them with equine commercial antivenom. Sheep presented tolerance both to Freund's adjuvant and other adjuvants, with no local lesions. High titers of high-affinity circulating antibodies were quickly developed, but IgGT was not detected.
Crotalus durissus terrificus venom consists of highly toxic protein and non-protein compounds. This prevents inoculation at concentrations capable of inducing adequate immune response for the needs of antivenom production (14).
Antigen detoxification or toxoid production requires that the venom loses its toxicity, but at the same time retains maximum immunogenicity (13). The use of electromagnetic or even photon radiation projects an electron and an atom, resulting in the creation of a pair of ions, positive and negative. This phenomenon, called ionization, is the main means by which the energy of ionizing radiation is transferred to biological tissues without producing radioactivity (19). Electromagnetic energy derived from Cobalt60 is used more frequently because this type of energy does not generate heat, and can thus be applied to biological products that are usually more susceptible to relatively high temperatures (10).
Hematological changes in erythrocytes, leukocytes, platelets, coagulation factors (3), and fibrinogen (1,24) are some toxic effects caused by animal venoms in humans. In experiments in rats with a mixture of venoms from different Brazilian snakes, there was a slow and gradual decrease hematocrit values, erythrocyte and lymphocyte counts, and an increase in number of neutrophils and hemoglobin concentration. The number of total leukocytes oscillated, with decreases and increases throughout the experimental period (3).
Cardoso and Mota (4) suggested that the crotoxin fraction of Crotalus durissus terrificus venom had inhibiting effects on the humoral response but did not alter cell response.
The objective of this study was to evaluate cell response in animals inoculated with Cobalt60-irradiated venom from Crotalus durissus terrificus.
MATERIALS AND METHODS
Animals
Twelve one to two-year-old sheep were used in this study, eight females and four males of mixed Ile de France breed, with mean weight of 40 kg. Two groups of six animals were randomly formed, with two males and four females in each group. One group was inoculated with natural venom (NV) and the other with irradiated venom (IrV).
Venom
Venom was obtained from Crotalus durissus terrificus, provided by The Center for the Study of Venoms and Venomous Animals CEVAP Botucatu, São Paulo State, Brazil, freeze dried, divided into two aliquots of 200 mg, and kept at 4°C. One aliquot (IrV) was submitted to gamma rays at the Institute of Energy and Nuclear Research - IPEN, University of São Paulo, SP-Brazil; the other aliquot (NV) was not treated at all.
Venom Irradiation
The freeze-dried venom aliquot was diluted in acidified saline solution (NaCl 150 mM, pH 3.0) and irradiated by a Cobalt60 source at the dose of 5.54 x 102 Gy/h and a concentration of 2.000 Gy in the presence of O2 at room temperature. After irradiation, the venom was kept at -20°C until use.
Sheep immunization
Both groups received three venom inoculations: the first on the day zero and the others at thirty-day intervals. The NV group received 1.0 mg and the IrV group 2.0 mg doses of venom diluted in 5.0 mL PBS, pH 7.0. Before inoculations, an equal volume of adjuvant was added to the antigens. Freund's complete (Sigma, USA) was used in the first inoculation, Freund's incomplete (Sigma, USA) in the second, and aluminum hydroxide adjuvant in the third. The animals were inoculated subcutaneously (SQ) in the right (5mL) and left (5mL) sub-scapular regions in a total of 10mL of antigen/adjuvant emulsion. On day zero and weekly thereafter, the animals were bled by the jugular vein to obtain total blood for eritrogram, leukogram, fibrinogen, total plasma protein, and albumin biochemical analyses.
All sheep were assessed weekly for inflammatory reactions, hemorrhagic alterations, abscess formation, and lymphonode increase at inoculation sites.
Neutralization and potency of sheep sera
In vivo test
The neutralizing capacity of antibodies induced by NV and IrV was assessed 60 days after the last inoculation. One sheep from each group was challenged intramuscularly with 1 mg/kg (LD50) of NV, as described by Araújo and Belluomini (2). The animals were observed for 48 hours post-inoculation for envenoming signs.
Laboratory testes
Individual blood samples were collected from the jugular vein using disposable needles (21G - 0.8mm) directly into 5.0 mL-vacuum tubes that contained heparin for albumin and total proteins measurements and EDTA (disodic salt of the ethylenediamine tetra-acetic acid at 10%) for CBC. Red blood cells (RBC) and white blood cells (WBC) were counted manually by hemocytometer (12). Hemoglobin concentration was determined by the cyanmethamoglobin method in a spectrophotometer at 540 nm. The packed cell volume (PCV) was determined by the microhematocrit method.
Hematimetric values were obtained according to Wintrobe (1929-32) (11). The differential count was determined by counting 100 cells on blood smears stained by the Dift Quick method (12). Fibrinogen concentration was obtained by heat precipitation (56°C) (12).
Total plasma protein was determined by the biuret method in a spectrophotometer at 545 nm. Albumin was determined by the bromocresol green method in a spectrophotometer at 545 nm wavelength, using ready-to-use commercial reagents. Plasma globulin was obtained from the difference between the total protein and plasma albumin.
Statistical analysis
The variables were studied in both groups (treatments) at 14 different times (CBC and biochemical analysis); analysis of variance with split plots was used (15). The F statistics calculated was significant when p<0.05. Significant tendency was 0.05<p<0.10. The contrasts between pairs of means were studied by calculating the minimum significant difference for p=0.05 by the Tukey test.
RESULTS AND DISCUSSION
At clinical examination, the animals in both groups did not present any local clinical alteration. A small volume increase in the pre-scapular lymphnodes occurred in the two weeks after administrations, but disappeared in the following weeks. Sjostrom et al. (23) also observed that sheep tolerated different adjuvants.
Two sheep challenged with 1 mg/kg of NV, one from each group, did not present any clinical alteration of envenoming, showing that both NV and IrV were efficient in inducing neutralizing antibody production.
The RBC count of NV and IrV-inoculated sheep showed similar results and there was no interaction between groups; there was only a slight time factor effect. Hemoglobin concentration, mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC) remained within reference ranges, behaving statistically similar to RBC count with no interaction and slight treatment effects between groups, with only a time factor effect. PCV values were similar in the two studied groups both for the analyses of time and treatment influence; there was no significant difference between groups. The results only demonstrated stability of eritrogram parameters.
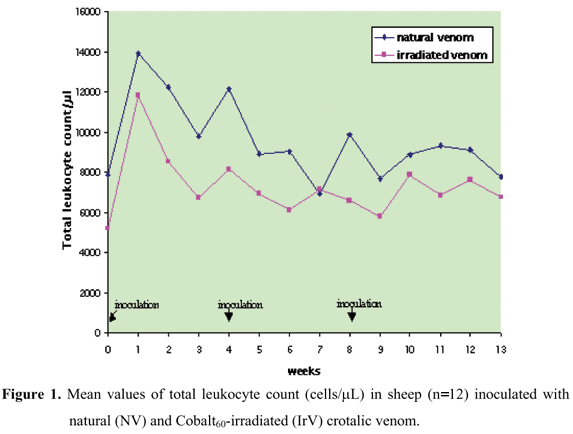
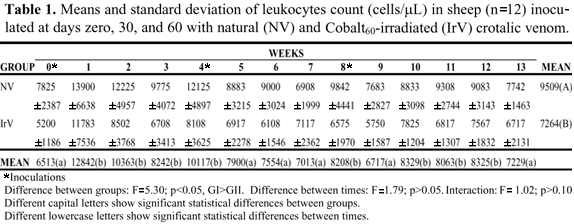
Results of total leukocyte count did not present interaction or time factor effect between groups, but the treatments caused different group action; the NV group had a greater number of cells than the IrV group (Figure 1, Table 1). The increase in leukocyte number to 13,000/mL in adult sheep is indicative of a slight leukocytosis (11) that occurred in the week after the first inoculation in the NV group (Figure 1, Table 1).
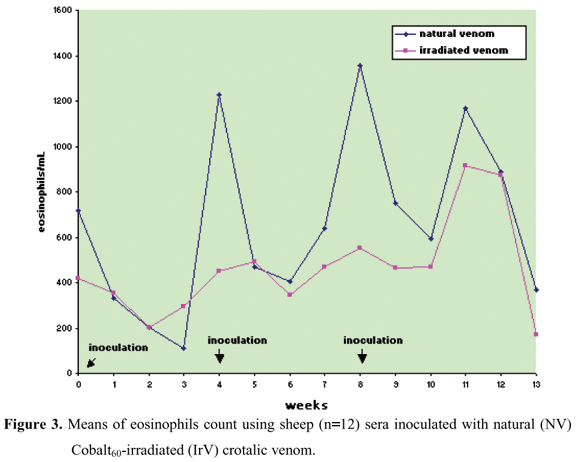
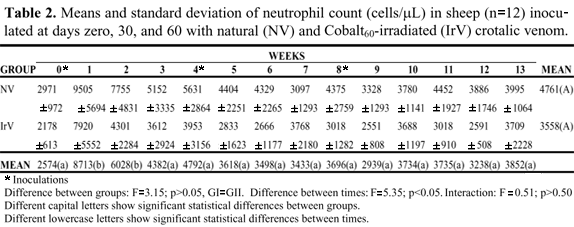
There was no statistically significant difference between the experimental groups for absolute count of segmented neutrophils, eosinophils, and lymphocytes, but there were statistically significant oscillations in values at the different collecting times.
The NV group presented an increase in the absolute neutrophil count after the first inoculation that persisted for 5 weeks. In the IrV group, the increase in neutrophils occurred only in the first week returning to normal in the following weeks (Figures 2 to 4, Table 2 to 4).
The alterations in neutrophil count are indicative of systemic inflammatory response related to cytokine release. Voronov et al. (25) described that total venom induces IL-6 systemic release, suggesting a direct immune response and a quick inversion in the neutrophil:lymphocyte ratio, which indicates that venom can cause cytotoxic activity in addition to previous hematological and immune alterations.
In this experiment, the neutrophil:lymphocyte ratio in both groups presented an inversion, which associated with the increase in lymphocyte number, indicates a response to systemic toxemia (11). This was more persistent in the NV group, showing its greater toxicity (Figures 2 to 4, Tables 2 to 4).
This is similar to Costa et al. (5) in Crotalus durissus terrificus venom action in rat erythrocytes and leukocytes. The authors reported initial leukopenia, in the first half hour after inoculation, followed by leukocytosis that returned to normal at the end of the experiment. This variation was accompanied by an increase in the number of segmented neutrophils. A decrease in the globular, erythrocyte, lymphocyte, and eosinophil volumes was also observed. The authors attributed the initial leukopenia to venom action and animal organic reaction to leukocytosis, as there was a concomitant increase in the number of neutrophils.
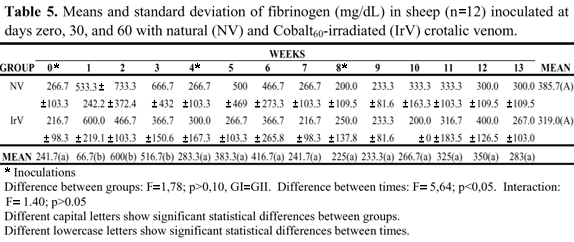
Fibrinogen levels showed statistically significant differences in relation to interaction and treatment effect, and there was only the time factor effect. The NV group showed an increase in fibrinogen after the first inoculation that persisted for 3 weeks, reappearing after the second inoculation, persisting for 2 weeks. In the IrV group, fibrinogen increase occurred for two weeks, returning to normal in the following weeks (Figures 5, Table 5).
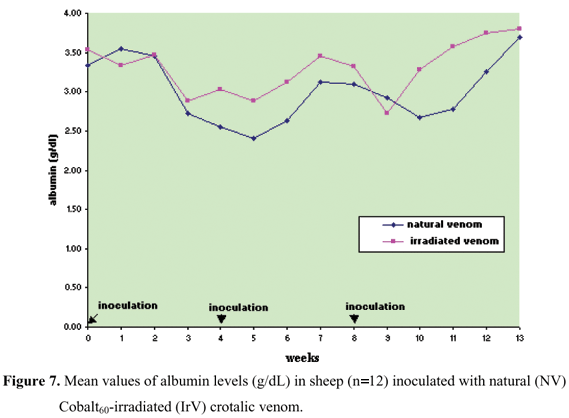
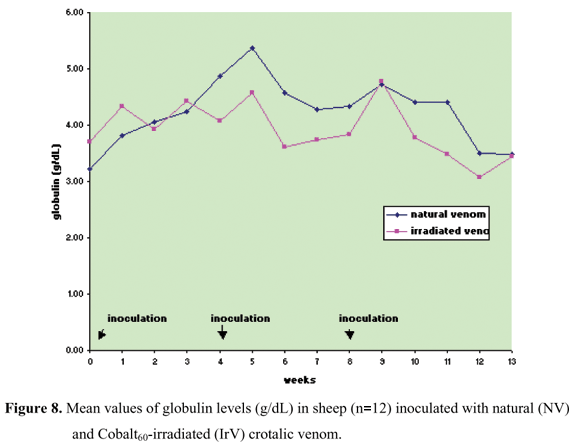
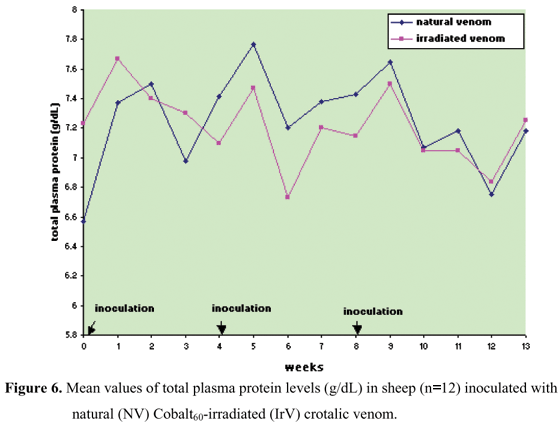
Protein, albumin, and globulin levels did not show any statistically significant differences related to interaction and treatment effect, and there was only the time factor effect (Figures 5 to 8, Table 5 to 8). Total plasma protein values (Figure 6, Table 6) for the IrV group remained normal, except in the first week after the first inoculation. The NV group presented an increase in total plasma protein values in the week after the end of inoculation. Three weeks after inoculation, there was a decrease in serum albumin values in both groups. Only the NV group presented an evident increase in globulin values.
In the first week after the first inoculation, the NV group presented an increase in fibrinogen concentration above normal limits, which remained above normal for three weeks. This was also observed after the second NV inoculation (Figure 5, Table 5). For the IrV group, the increased fibrinogen values returned to normal in the third week after the first inoculation and did not present any further alteration throughout the experiment. These increases in fibrinogen values are probably due to the acute inflammatory response caused by venom inoculation (Figure 5, Table 5).
Estrada et al. (8) assessed the PCV, hemoglobin concentration, and total serum protein in horses inoculated with Bothrops asper, Lachesis muta, and Crotalus durissus terrificus venoms and obtained slight or decreased changes in values, similar results to those found in this study. The authors reported the increase in total protein concentration and an albumin:globulin ratio inversion during the immunization period.
Jain (11) reported that the increase in total plasma protein level could occur as a response to acute infection due to an increase in the gammaglobulinic fraction.
Thomazini et al. (24) analyzed serum fibrinogen of patients bitten by Crotalus snakes and reported a reduction in the values before antivenom treatment that tended to normalize after 48 hours. These results differ from those found in our experiment, where there was a significant increase in both groups in the three weeks after the first inoculation and decreased in the following weeks.
None of the sheep challenged with a lethal dose (LD50) showed any envenoming signs or clinical alterations in the inoculated animals. The values found for hematological and biochemical variables did not differ statistically between groups, except for leukocyte counts (Figure 1, Table 1), showing an acute inflammatory response. This was more intense in the NV group (Figure 1, Table 1).
The IrV produced less intense hematological and biochemical alterations than the NV. The immunization protocol for both groups was adequate to produce effective sera with lower venom doses than those used in horses. The results of all analyses indicated that sheep are potential animals to be used in anticrotalic serum production.
Received: July 3, 2002
Accepted: November 8, 2002
Published online: February 14, 2004
- 1 AMARAL CFS., REZENDE NA., PEDROSA TMG., SILVA AO., PEDROSO ERP. Afibrinogenemia secundária a acidente crotálico (Crotalus durissus terrificus). Rev. Inst. Med. trop.São Paulo, 1988, 4, 288-92.
- 2 ARAÚJO P., BELLUOMINI HE. Toxicidade de venenos ofídicos I - Sensibilidade específica de animais domésticos e de laboratório. Mem. Inst. Butantan, 1960-62, 30, 133-42.
- 3 BARRAVIERA B., PERAÇOLI MTS. Soroterapia heteróloga. In: BARRAVIERA B. Venenos animais: uma visão integrada. Rio de Janeiro: Editora de Publicações Científicas, 1994: 361-2.
- 4 CARDOSO DF., MOTA I. Effect of Crotalus venom on the humoral and cellular immune response. Toxicon, 1997, 35, 607-12.
- 5 COSTA PI., GARCIA-LIMA E., LAURE CJ. Rattlesnake venom: action upon erythrocytes and leucocytes of rats. Acta Physiol. Pharmacol. Latinoam., 1989, 39, 359-73.
- 6 EGEN N., RUSSEL F., CONSROE P., GERRISH K., DART R.. A new ovine Fab antivenom for North American venomous snakes. Vet. Hum. Toxicol., 1994, 36, 362
- 7 EK N. Serum levels of the immunoglobulins IgG and IgG(T) in horses. Acta Vet. Scand, 1974, 15, 609-19.
- 8 ESTRADA R., CHAVES F., ROBLES A., ROJAS E., SEGURA E., GUTIÉRREZ JM. Valores hematológicos y de enzimas séricas en caballos inoculados com venenos de serpientes para la producción de antivenenos en Costa Rica. Vet. Biol. Trop, 1992, 40, 95-9.
- 9 FAN WF., FRANÇA FOS. Acidente por cobras. In: SCHVARTSMAN S. Plantas venenosas e animais peçonhentos. 2.ed. São Paulo: Sarvier, 1992: 143-8.
- 10 GUIDOLIN R., CORREA A., CICARELLI RMB., PREVIDE E., MORAIS JF., HIGASHI HG. Esterilização de soros e vacinas por radiação gama de Cobalto. Rev. Saúde Pública, 1988, 22, 113-7.
- 11 JAIN NC. Schalm's veterinary hematology 4ed. Philadelphia: Lea & Febiger, 1986 1221p.
- 12 JAIN NC.Essentials of veterinary hematology Philadelphia: Lea & Febiger, 1993 417p.
- 13 KHAN ZH., LARI FA., ALI Z. Preparation of toxoids from the venoms of Pakistan species of snakes (Naja naja, Vipera russelii and Echis carinatus).Jpn. J. Med. Sci. Biol., 1977, 30, 19-23.
- 14 MURATA Y., ROGERO JR. Análise cromatográfica por exclusão em amostras de veneno de cascavel irradiadas com Co60.,Publ. Ipen, 1988, 154, 1-7.
- 15 PIMENTEL GOMES F. Estatística experimental São Paulo: Nobel, 1969 446p.
- 16 RAWAT S., LAING G., SMITH DC., THEAKSTON D., LANDON J. A new antivenom to treat eastern coral snake (Micrurus fulvius fulvius) envenoming. Toxicon, 1994, 32, 185-90
- 17 RESENDE CC., ARAÚJO FAA., SALLENAVE RNUR. Análise epidemiológica dos acidentes ofídicos. Brasil junho de 1986 a dezembro de 1987 Brasília: Ministério da Saúde. Secretaria Nacional de Ações Básicas de Saúde, 1989. 37p.
- 18 RIBEIRO LA. Epidemiology of ophidic accidents. Mem. Inst. Butantan, 1990, Supl.52, 15-6.
- 19 ROGERO JR., NASCIMENTO N. Atenuação da toxicidade de venenos ofídicos por meio da radiação ionizante. Biotecnol. Cienc. Desenvolv,1997, 1, 24-6.
- 20 RUSSEL FE., LAURITZEN L. Antivenins. Trans. R. Soc. trop. Med. Hyg., 1966, 60, 797- 801.
- 21 RUSSEL FE., TIMMERMAN WF., MEADOWS PE. Clinical use of antivenin prepared from goat serum. Toxicon, 1970, 8, 63-5.
- 22 SALAFRANCA ES. Irradiated cobra (Naja naja philippinensis) venom. Int. J. Appl. Radiat. Isot., 1973, 24, 60.
- 23 SJOSTROM L., AL-ABDULLA IH., RAWAT S., SMITH DC., LANDON J. A comparison of ovine and equine antivenoms. Toxicon, 1994, 32, 427-33.
- 24 THOMAZINI IA., IUAN FC., CARVALHO I., HERNANDES DH., AMARAL IF., PEREIRA PCM., BARRAVIERA B. Evaluation of platelet function and of serum fibrinogen levels in patients bitten by snakes of the genus Crotalus Preliminary report. Rev. Inst. Med. Trop. São Paulo, 1991, 3, 219-20.
- 25 VORONOV E., APTE RN., SOFER S. The systemic inflammatory response syndrome related to the release of cytokines following severe envenomation. J. Venom. Anim. Toxins, 1999, 5, 1, 5-33.
Correspondence to
Publication Dates
-
Publication in this collection
02 Apr 2004 -
Date of issue
2004
History
-
Accepted
08 Nov 2002 -
Received
03 July 2002